Abstract
Bundled payments have been proposed as a mechanism for restraining health care spending for total knee arthroplasty (TKA), but empirical data are limited. We used Medicare data to examine variation in payments for TKA during a window extending 30 days before to 90 days after TKA for 167 186 patients who underwent elective primary TKA in 2009. Mean Medicare payment was US$23 656. We found that 2.5% of patients incurred payments of >US$50 000 (0.2% >US$100 000). Payments were lower for men and for non-Hispanic whites but higher for patients with greater comorbidity. Episode-of-care payment for primary TKA varies substantially depending upon patient demographics and comorbidity. To the extent that similar patients tend to be clustered within hospitals, bundled payments could inadvertently cause financial harm to certain health systems while rewarding others.
Introduction
In an effort to slow what is widely viewed as unsustainable spending in the US Medicare program, researchers and policy makers have proposed bundled payments for many high-volume and high-cost procedures. 1,2 Bundled payments would involve a single lump-sum payment from Medicare (or another payor) to an integrated health care delivery system for an entire episode of care. 3,4 The episode of care would typically include all preprocedure, periprocedure, and postprocedure services that a single patient would receive during treatment for a well-circumscribed disease or condition. 3 There is hope that bundled payments would reduce the financial incentives inherent in fee-for-service reimbursement models to “do more,” thereby reducing the overuse of tests, procedures, and services that plague the US health care system for decades. 5 -7
However, the complexity of moving from the current fee-for-service payment model to a bundled payment model is enormous. 8 Currently, hospitals and physicians are largely unaware of the costs of specific tests or procedures. 9,10 This lack of awareness hints at fundamental flaws in how costs are allocated and prices set within health care. 11
At the same time, payors interested in bundled payments are challenged by a lack of concrete studies describing the methods and amounts of money that might be associated with specific diagnoses and conditions (eg, congestive heart failure, cardiac surgery, and joint replacement). Even as Medicare is embarking upon episode-of-care demonstration projects, 12 there are very few peer-reviewed publications describing diagnosis-specific costs and payments using an episode-of-care perspective. 13,14 Without concrete studies describing the methods of calculating bundled payments and the amounts of money that might be expected for a specific diagnosis or condition, payors, administrators, and hospital leadership will be ill-equipped to prepare for coming changes. 13 Moreover, we are aware of few studies that has focused on bundled payments for total knee arthroplasty (TKA), 14,15 despite the fact that TKA is one of the key procedures being evaluated in current Medicare episode-of-care demonstration projects. 12
Our overarching objective was to systematically examine Medicare episode-of-care payments for patients who underwent primary elective TKA. More specifically, we set out to examine the variation in episode-of-care payments and how payments might vary according to patient demographics, burden of comorbidity, and among patients who did and did not experience complications.
Methods
Data
We began our analysis by identifying all fee-for-service Medicare beneficiaries who underwent primary TKA in 2009 using International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) procedure code 81.54 in the Part A (hospital) data files. We verified that all TKA procedures found in the Part A files had a corresponding physician claim in the Medicare carrier files using CPT codes 27437, 27438, 27440, 27441, 27442, 27443, 27445, 27446, and 27447 following protocols that had been developed and published by Singh and Ayub; 16 these algorithms have been shown to be highly sensitive and specific for identification of TKA. The TKA procedures that could not be identified in both the hospital and physician files were excluded because lack of both hospital and physician claims precluded us from accurately calculating complete Medicare payments.
We excluded several patient populations in an effort to identify a homogeneous population of patients who received a single elective primary TKA (see Figure 1). We excluded patients who underwent multiple TKA procedures during the index hospital stay, patients with acute fractures, and patients admitted from the emergency department in accordance with methods used in prior studies of primary TKA. 17,18 We excluded patients who had not been enrolled continuously in both the Medicare Part A and Part B programs for 12 months preceding their TKA and 3 months following their surgery. The requirement for 12 months of enrollment preceding the index surgery was necessary to allow us to be certain that each procedure represented the first TKA and not a readmission for a complication and/or early revision. The 3-month postprocedure enrollment was needed to allow for a full 90-day window for capturing postprocedure Medicare payments. We excluded Medicare enrollees with end-stage renal disease and patients who resided outside the United States as both populations would not be representative of the general TKA population. We also excluded patients for whom Medicare was not listed as the primary payor because in such cases the Medicare payments might not fully capture all payments associated with the TKA procedure.
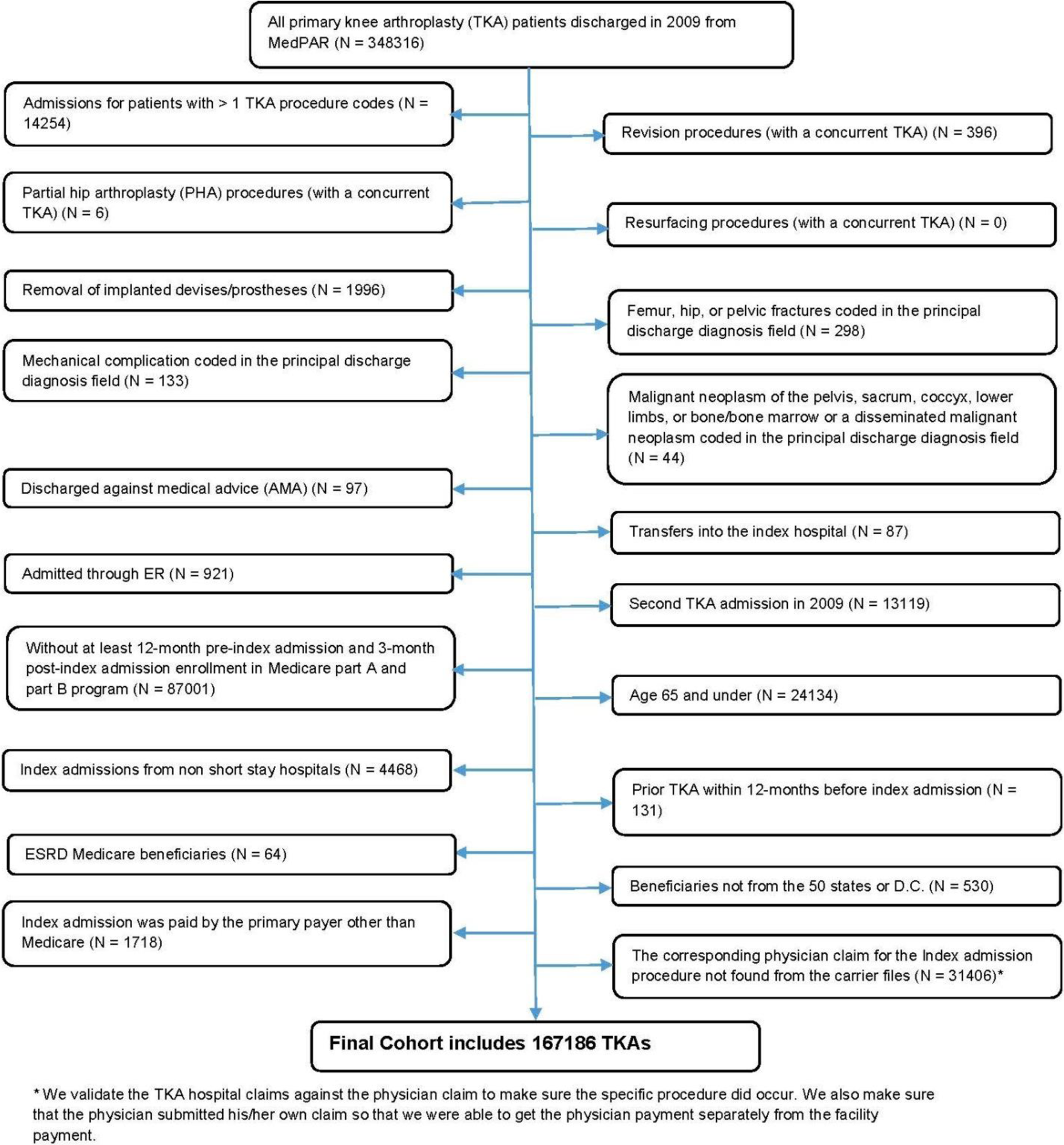
Flowchart for generation of analytic cohort.
For each patient in our final analytic cohort, we created a linked longitudinal record extending from 30 days prior to admission until 90 days following surgery; thus, the total episode of care for each patient extended for a full 120 days in accordance with current proposals for bundled payments. 2,4,8 We linked 5 separate data files (Table 1) to comprehensively capture all payments made by Centers for Medicare and Medicaid Services (CMS) during the study period. When used in combination, these files allow for the capture of all inpatient, outpatient, and postacute care. Additional details about the coding processes and procedures used in our calculations are available by request.
List of Data Files Used in Calculating Bundled Payments.

Analyses
First, we explored the demographic characteristics (ie, age, sex, and race) and comorbidities of our cohort of TKA recipients. Comorbidities were identified by mapping specific ICD-9-CM codes to Condition Categories (CC) using methods developed by CMS. 19 Comorbidities were identified using both Part A and Part B data files beginning 12 months prior to the TKA procedure and continuing through hospital discharge.
Second, we examined the mean, median, and range in Medicare payments for the preoperative, perioperative, and postoperative periods and total payments over the entire 120-day episode of care. The preoperative window included the time extending from 30 days prior to admission up to and including the day immediately prior to the TKA hospital admission. The perioperative window involved all Medicare payments during the index hospitalization for TKA. The postoperative window involved all payments occurring between the time of discharge after TKA until 90 days after the date of surgery; patients who had a postoperative hospital stay of 90 days or longer, there would be no postdischarge payments but exceedingly large in-hospital payments reflecting the complicated postoperative course.
Third, we examined how payment varied when patients were stratified by age, sex, race, and number of comorbid conditions; the number of comorbid conditions for each patient was determined by counting the number of CC that a given patient had. These stratified analyses were designed to provide insight into how hospitals might benefit or be disadvantaged depending upon their particular patient population.
Finally, we examined how episode-of-care payments varied among patients who did and did not experience surgical complications. We identified 7 individual complications that have been identified previously in studies of TKA using administrative data; complications were identified using ICD-9-CM codes and algorithms that we and others have used previously. 20 -22 Complications included acute myocardial infarction, pneumonia, sepsis, surgical site bleeding, pulmonary embolism, mechanical complications, and wound infection. Complications were identified during the index admission or readmission within 90 days of surgery. We compared episode-of-care costs for patients who did and did not experience one or more complications. We then conducted more detailed analyses to examine episode-of-care costs for each of the individual complications. Additional details of statistical coding are available by request from the authors.
All P values are 2-tailed, with P values less than .05 deemed statistically significant. All statistical analyses were performed using SAS 9.2 (SAS Institute Inc., Cary, North Carolina). This project was approved by the University of Iowa Institutional Review Board.
Results
Our study cohort consisted of 167 186 patients who underwent unilateral elective TKA in 2009. The demographic characteristics and prevalence of key comorbid conditions are described in Table 2. Mean age of our population was approximately 75 and 65% were women; as expected, 100% of our sample (patients undergoing TKA) had osteoarthritis.
Demographic Characteristics and Comorbidity of Patient Population.
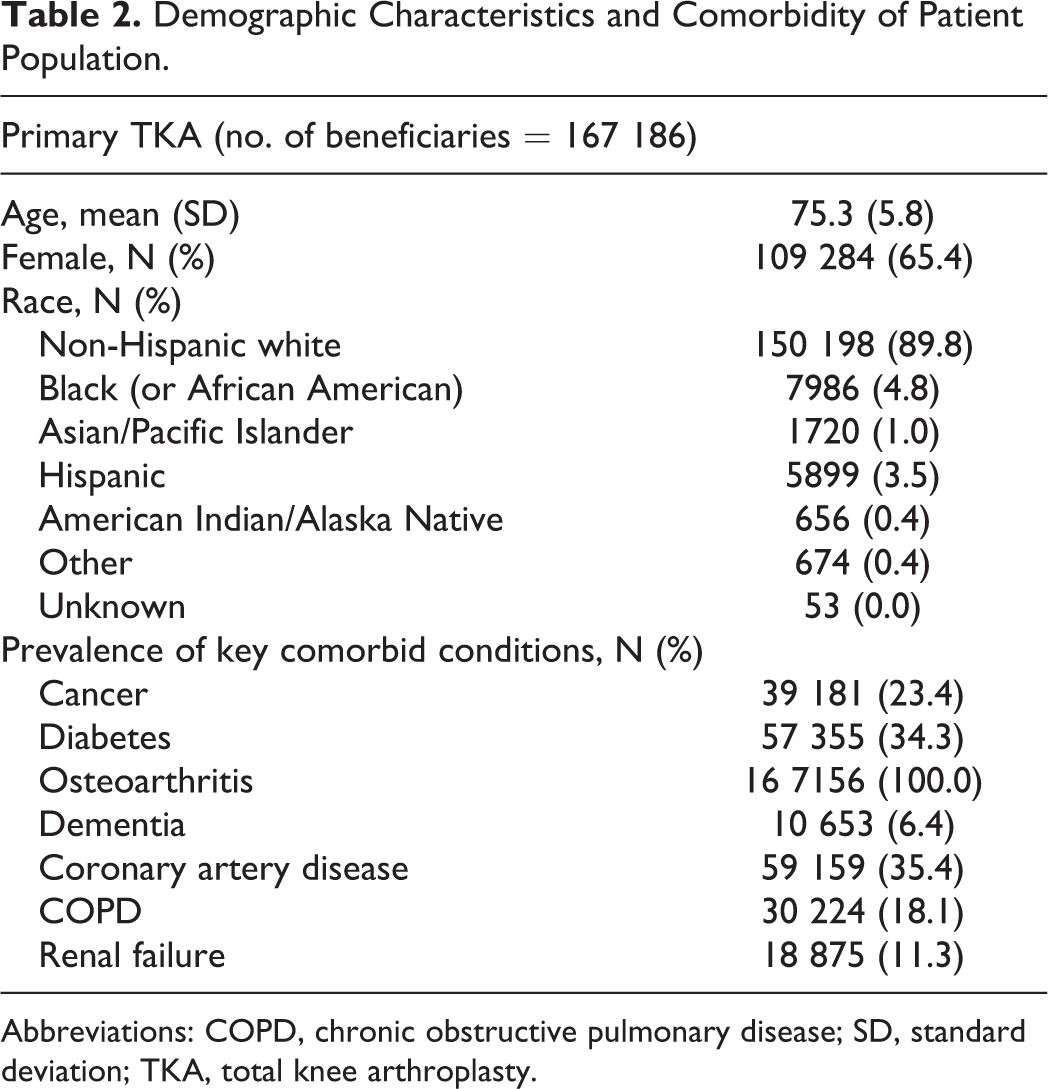
Abbreviations: COPD, chronic obstructive pulmonary disease; SD, standard deviation; TKA, total knee arthroplasty.
Mean Medicare payments for the preoperative, intraoperative, and postdischarge periods were US$701, US$13 847, and US$9116, respectively (Table 3); mean total payment for TKA was US$23 656 and median was US$20 835. We found that 2.5% of patients (N = 4173) incurred Medicare payments of >US$50 000, 0.2% of patients (N = 291) incurred Medicare payments of >US$100 000, and 0.01% of patients (N = 22) incurred payments of >US$200 000. The range in total payments was more than US$320 000.
Medicare Payments.

Abbreviations: IQR, interquartile range; std, standard deviation; TKA, total knee arthroplasty.
aDefined as 30 days prior to the index admission date (exclusive).
bDefined as the entire duration of the index admission during which TKA was performed (from admission until discharge).
cDefined as the time period after discharge from the index admission extending up to 90 days after the TKA procedure. Patients who were hospitalized for 90 days or more during index admission would have no postdischarge costs. Patients who died during the index admission did not incur any postdischarge costs.
dTotal = preoperative + perioperative + postdischarge payment amount.
Medicare payment for TKA among key patient subgroups is displayed in Table 4. Total payments increased significantly with increasing patient age, and payments were approximately US$1400 higher for women compared to men (P < .0001). Medicare payments were approximately US$4000 lower for non-Hispanic whites when compared to other race/ethnicity groups. The mean total Medicare payments during the episode-of-care surrounding TKA increased substantially with increasing number of comorbidities from US$19 633 for patients with 2 or less comorbidities to US$27 578 for patients with 5 or more comorbidities (Table 4; P < .0001).
Payment According to Patient Age and Comorbidity Burden.
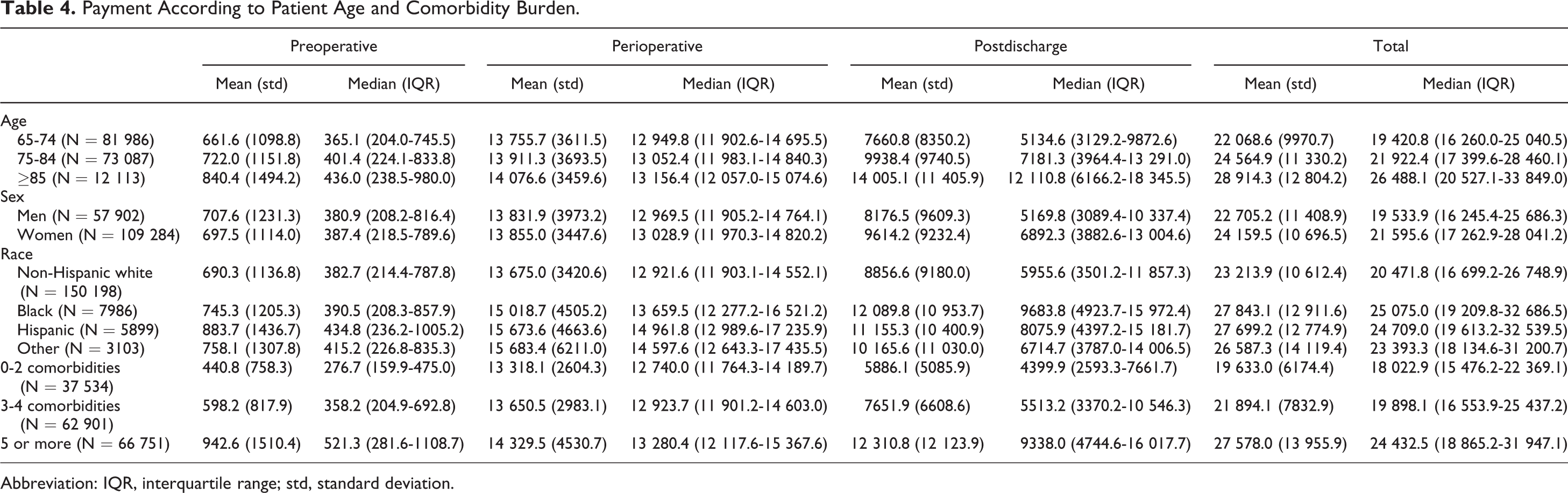
Abbreviation: IQR, interquartile range; std, standard deviation.
We found that 3.7% of all patients with primary TKA experienced one or more complications resulting in hospital readmission within 90 days of surgery (Table 5). Interestingly, patients who experienced complications incurred higher Medicare payments in the perioperative and postoperative periods but also in the preoperative period. From the perspective of medicare patients who experienced one-or-more complications cost an average of US$39 000 as compared to US$ 23 000 for patients who experienced no complications (P < .001). Among complications, the costs for acute myocardial infarction (US$45 000) and infection (US$51 000) were notably higher than that for other complications such as pneumonia (US$30 000) and pulmonary embolism (US$34 000).
Payments for Patients Who Did and Did Not Experience Complications.

Abbreviation: IQR, interquartile range; std, standard deviation.
Discussion
We used a combination of linked files from the US Medicare program to determine the average total payments for primary elective TKA. We found that approximately 3% of payments were incurred during the preoperative period, 59% during the index hospitalization, and 38% during the postoperative period. We also found that while the mean total payment for TKA was approximately US$24 000, only 0.2% of TKA procedures resulted in payments of US$100 000 or more. Finally, we demonstrate that payments tended to be higher for certain easily identified patient populations including those who were older and those with more comorbid illness, suggesting the potential importance of risk adjustment when determining actual bundled payment amounts. In aggregate, our results highlight many of the complexities of implementing a bundled payment system even for an extremely refined and standardized procedure such as elective primary TKA.
A number of our findings warrant additional discussion. First, our finding that Medicare payments were higher for several patient populations (women, older patients, and patients with greater comorbid illness) is not unexpected; our findings are likely a reflection of both higher complication rates for TKA in these subgroups and less social support necessitating greater reliance on home health services. 23 -26 The implications of higher complication rates (and costs) for certain populations could have profound implications under a switch to bundled payments. To the extent that patients with similar sociodemographics (and risk for complications) cluster within hospitals, 27 -29 certain hospitals could benefit by having a relatively low-risk TKA population and see profits from TKA soar; alternatively, other hospitals with a predominantly higher risk TKA population could see TKA as financially detrimental based upon geographic location and catchment area alone. 30 There is certainly reason to be concerned that episode-of-care payments could provide additional incentive for hospitals and physicians to select lower risk patients while avoiding higher risk patients. 31 Of course if bundled payment amounts were adjusted to reflect each hospital’s particular patient mix, such problems could be alleviated to some degree.
Second, although our analysis provides important information about the variation in Medicare payments for primary TKA under the current fee-for-service model, our results say little about how a bundled payment amount should be set. From the standpoint of hospitals, physicians, and other organizations in the business of providing medical care, it would obviously be highly desirable if the bundled payment amounts were set at the highest level possible; for example, at the current (2009) mean payment amount of US$23 655. Although setting a bundled payment for TKA at US$23 655 would accomplish the desired goal of providing the health care delivery system with incentive to improve efficiency by reducing readmissions or unnecessary imaging procedures, setting payment at the current fee-for-service level would not result in any net savings to the Medicare program itself. If the true goal of bundled payments is to restrain or even reduce Medicare spending to insure the long-term sustainability of the Medicare program and the financial solvency of the United States, it might be reasonable to set the mean bundled payment price and a level modestly below the mean fee-for-service price (eg, US$23 000) to actually reduce spending. Determining the actual level of payment for each diagnosis and/or condition is likely to be challenging from both administrative and political perspective. Moreover, as Weeks et al point out in a recent article, bundled payments do nothing to restrain spending growth that might result from increased procedure volumes. 13
Third, it is important to comment on the challenge that physicians, health systems, and accountable care organizations will have in adapting to a bundled payment model. A number of recently published studies have provided insight into the difficulties that hospitals and physicians are having in understanding their own internal costs and prices. 9 -11 CMS has begun to release large quantities of Medicare data to provider groups in hopes that greater access to data will enhance knowledge and understanding among the provider community. In turn, there is an assumption that improved understanding of utilization and cost data will assist providers in improving efficiency. We are skeptical about such assumptions. As demonstrated in our current analysis, analyzing Medicare data and computing bundled payment costs is time consuming and complicated and likely beyond the expertise of most health care delivery systems. Adapting to a bundled payment model is likely to have an extremely steep learning curve for most provider groups.
Where might cost savings come from? In the acute care setting, there is ample evidence of tremendous variation in the prices that organizations pay for implants. 32 We would guess that movement toward bundled payments will continue to increase pressure on hospitals to drive down costs of implants, and, in-turn, hospitals will increasingly demand price concessions from implant vendors. 33 We suspect that such pressure will be successful and that implant prices will decline without substantial evidence that newer devices generate improved patient outcomes. Cost saving opportunities will also be found in unnecessary preoperative diagnostic testing (eg, electrocardiograms, echocardiograms, and pulmonary function testing) for low-risk patients. 34,35 Cost savings will also come from reductions in inpatient rehabilitation and also utilization of home-based health care services. 36,37
Fourth, it is important to discuss our work in light of prior studies. Our team recently published an analysis evaluating variation in Medicare payments to hospitals for TKA 17 ; we found mean hospital payment of US$13 464 for TKA. However, our prior analysis used only Medicare Part A data and thus did not capture any of the preoperative or postoperative costs associated with TKA that are included in this article. Bozic and colleagues evaluated bundled payments for total joint arthroplasty (combined TKA and total hip arthroplasty [THA]) in California, finding mean payment of approximately US$26 000 for primary procedures; their analysis was limited to a single academic medical center and did not separate TKA from THA. 15
Our study has several limitations that merit brief mention. First, our analysis was limited to elective primary TKA in the Medicare population, so extrapolation to the non-Medicare population or other conditions that might be considered for bundled payments such as congestive heart failure should be done with caution. Second, we did not include prescription drug costs in our analysis—inclusion of these costs would have increased the bundled payment amount modestly. Third, our study cohort was deliberately limited to patients with primary elective TKA; it is nearly certain that episode-of-care payments in certain high-risk subgroups (eg, patients with acute fractures) would be higher still.
In summary, our analysis provides concrete estimates of aggregate bundled payment amounts related to primary elective TKA and illustrates how historical Medicare payments vary across key patient subgroups. Our results hint at the complexities of implanting a bundled payment system and both the strengths and the weaknesses of such an approach.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PC is supported by a K24 award from NIAMS (AR062133). This work is also funded in part by R01 AG033035 from NIA at the NIH.
