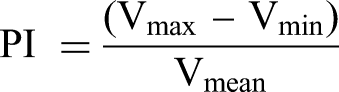Abstract
Background
The study objective was to determine the impact of cardiopulmonary bypass perfusion modalities on cerebral hemodynamics and clinical outcomes in congenital cardiac surgery patients stratified by acyanotic versus cyanotic heart disease.
Methods
A total of 159 pediatric (age <18 years) cardiac surgery patients were prospectively randomized to pulsatile or nonpulsatile cardiopulmonary bypass and stratified by type of congenital heart disease: acyanotic versus cyanotic. Intraoperative cerebral gaseous microemboli counts and middle cerebral artery pulsatility index were assessed. Organ injury was quantified by Pediatric Logistic Organ Dysfunction-2 (PELOD-2) score at 24, 48, and 72 h postoperatively. Additional outcomes included Pediatric Risk of Mortality-3 score, vasoactive–inotropic score, duration of mechanical ventilation, intensive care and hospital length of stay, and 180-day mortality. Heterogenous variance linear models (ie, ANOVA and mixed models) and χ2 tests were used to compare groups for continuous and categorical variables, respectively.
Results
Within congenital heart disease subgroups, patients randomized to nonpulsatile versus pulsatile bypass had similar preoperative and operative characteristics. While the intraoperative pulsatility index was higher in the pulsatile subset of both acyanotic and cyanotic groups (P < .05), regional cerebral oxygen saturation, mean arterial pressure, and gaseous microemboli counts were similar. Postoperative PELOD-2 scores decreased at similar rates in the acyanotic and cyanotic subgroups regardless of the perfusion modality utilized. There were also no significant between-group differences in the additional postoperative outcomes by perfusion modality in either acyanotic or cyanotic groups.
Conclusions
Despite patients undergoing pulsatile cardiopulmonary bypass experiencing a more physiologic pulsatility index in both acyanotic and cyanotic groups, no significant differences in cerebral hemodynamics or clinical outcomes were appreciated.
Keywords
Introduction
Since the start of cardiopulmonary bypass (CPB) use for cardiac surgery in the 1950s, the search for methods to improve patient outcomes continues and remains a pressing need, especially for patients undergoing congenital heart disease (CHD) surgery. 1 Children undergoing surgery for CHD represent a unique cohort, with distinct risk profiles based on the nature of their congenital heart defects. A traditional dichotomy is the distinction between cyanotic and acyanotic heart disease, based on the presence or absence of intra-/extracardiac right-to-left shunting which causes chronic hypoxemia. Despite significant advances in preoperative, operative, and postoperative care in the last several decades, children with cyanotic heart disease continue to have greater mortality risk, 2 greater risk of vital end organ injury,3‐5 and have worse long-term neurodevelopmental outcomes6,7 compared with children with acyanotic heart disease. Chronic hypoxemia is known to alter cardiomyocyte oxidative metabolism, as well as the expression of genes related to the modulation of inflammation, apoptosis, and myocardial contractility, making patients with cyanotic CHD more vulnerable to cellular injury and ventricular dysfunction following CPB. 8 The question remains of whether further advances in perfusion modalities can improve patient outcomes, particularly in the subset of children with cyanotic heart disease. To date, aside from studies regarding the avoidance of hyperoxia during CPB, 8 there have been few attempts at identifying customized CPB strategies to specifically benefit the cyanotic CHD population.
Of interest, pulsatile perfusion during CPB has been demonstrated to have multiple features that could, in theory, provide benefit to children undergoing CHD surgery, but data regarding its effect in this specific patient cohort are currently lacking. What is known from studies in other patient populations and animal models is that pulsatile perfusion might provide greater cerebral perfusion9‐12 and is associated with lower levels of cerebral injury modulators compared with nonpulsatile perfusion methods. 13 Pulsatile perfusion has also been associated with improved myocardial and renal perfusion.12,14 However, concerns have arisen regarding the potential for increased hemolysis, 15 reduced capture of gaseous microemboli (GME), 16 and the introduction of additional circuit complexity. 17
Our group recently conducted a prospective randomized clinical trial to assess the effects of pulsatile versus nonpulsatile CPB modalities on cerebral hemodynamics, vital organ injury (as quantified by the Pediatric Logistic Organ Dysfunction-2 Score, PELOD-2), and clinical outcomes in children undergoing CPB for CHD surgery. 18 In that study, no difference in PELOD-2 scores or clinical outcomes was identified in a cohort of children undergoing congenital heart surgery based on perfusion modality. However, the question of the impact on specific subpopulations was not addressed. In the present analysis, we hypothesized that children with cyanotic heart disease may see greater benefit from pulsatile perfusion given their greater overall risk profile. That is, that children with cyanotic heart disease in whom pulsatile perfusion was used would have better markers of cerebral hemodynamics, less vital organ injury, and better clinical outcomes, compared with those with cyanotic heart disease who undergo traditional nonpulsatile perfusion modality.
Patients and Methods
Study Design and Randomization Protocol
After parental informed consent, eligible subjects were enrolled at Penn State Health Children's Hospital (Penn State College of Medicine IRB Study ID: PRAMS019299-A, last approval: 12/04/2023; NCT00862407). To be eligible for the study, patients were less than 18 years old at the time of enrollment and were scheduled to undergo cardiac surgery requiring CPB for CHD. Additionally, patients utilizing only 8F to 14F aortic CPB cannulas were included in the study to create more homogeneous groups for comparison. Patients meeting the inclusion criteria were randomized to undergo CPB using either pulsatile or nonpulsatile perfusion settings. Details explaining the randomization process are presented in Supplemental Figure 1 and Supplementary Materials section. For the present analysis, patients were stratified by type of heart disease (acyanotic vs cyanotic), and each group was studied to determine the impact of perfusion-modality settings (nonpulsatile vs pulsatile). All methods were carried out in accordance with relevant guidelines and regulations. Penn State College of Medicine IRB approved all experimental protocols.
Anesthesia and Perfusion
Isoflurane, pancuronium, and fentanyl were utilized for general anesthesia. A median sternotomy was used as the primary approach in all surgeries. The CPB circuit comprises a Maquet-HL-20 heart-lung machine (Maquet Cardiopulmonary, Getinge Group), Capiox hollow-fiber membrane oxygenator (RX05/RX15, Terumo Cardiovascular Systems Corporation), Capiox pediatric 32-mm arterial filter (CXAF02, Terumo Cardiovascular Systems Corporation), cardioplegia set (66483-01, Terumo Cardiovascular Systems Corporation), Minntech hemoconcentrator (HemoCor HPH400TS, Medivators, Inc.), polyvinyl chloride tubing (LivaNova Smart Perfusion Pack), Stockert heater-cooler system (LivaNova), and CPB aortic cannulas selected from the DLP series (Medtronic), Sarns Tender Flow series (Terumo Cardiovascular Systems), or Fem-Flex II series (Edwards Lifesciences Corp). The CPB circuit was primed with 400 mL of Plasmalyte-A, 50 mL of 25% Human Albumin, 15 mEq NaHCO3, and 1000 U of Heparin. Approximately 120 to 180 mL of priming solution was removed, which was used as additional volume as needed during the surgery, and 250 mL of packed red blood cells were added. Hematocrit goal during CPB was set to ≥26% for all patients. Additionally, a 0.5 g/kg dose of mannitol was administered after the initiation of CPB. Initiation of vasoactives is based on the transesophageal echocardiogram, visualization of the myocardial contractility, or congenital heart lesion. Milrinone is often used. Additional augmentation of myocardial function or vasomotor tone is through the use of epinephrine. Mechanical ventilation goals are targeted for a normal pH and PO2 required for the type of congenital heart defect noted. All patients underwent slow continuous ultrafiltration as well as modified ultrafiltration. All patients receive protamine post bypass. The dose is predicated on the heparin used and ACT. All patients receive furosemide upon successful separation from CPB.
Patients in the pulsatile perfusion group were placed on CPB at the following settings: 10% of the base flow, 20% of the pump head start point, and 80% of the pump head stop target. The pump rate was set based on patient weight: >15 kg, 90 beats/min; 7 to 15 kg, 100 beats/min; and <7 kg, 120 beats/min.
Pediatric Intensive Care Unit Protocols
Most patients are sedated with fentanyl and/or dexmedetomidine. Mechanical ventilation is performed with a tidal volume of 10 mL/kg and minute ventilation adjusted to a normal pH or pH necessary to balance Qp/Qs in shunted patients. The oxygen and PEEP are titrated to maintain a P02 appropriate for the defect being palliated or repaired. Patients at risk or with pulmonary hypertension have an alkalotic pH and receive nitric oxide. Vasoactives consist of milrinone, epinephrine, and nitroprusside. Vasoactives, which are initiated in the operating room, are continued postoperatively. Nitroprusside is often titrated to maintain an age appropriate mean arterial pressure (MAP) at the 50th percentile. All patients receive continuous cerebral oximetry, electrocardiogram, arterial blood pressure, central venous pressure, end tidal, and oxygen saturation monitoring. Serial lactate levels and mixed venous oxygen saturations are obtained based on the defect. The titration and weaning of inotropes and/or volume are dependent on the clinical examination, perfusion, urine output, and hemodynamic and oximetric variables continuously or intermittently monitored. Weaning of vasoactive usually occurs on the first postoperative day. The administration of packed red blood cells is dependent on maintaining a hematocrit of >40% in cyanotic patients and >30% in acyanotic patients who are hypotensive, hypovolemic, or persistently acidotic. Platelets and fresh frozen plasma are given for active bleeding.
Intraoperative Monitoring of Hemodynamics and Oxygen Saturation
Transcranial doppler
To measure cerebral blood flow velocities, pulsatility index, and detect the presence of GME counts during CPB, Transcranial Doppler (TCD) ultrasound (Pioneer TC8080; Nicolet Biomedical Inc) was utilized. One transcranial doppler (TCD) sensor was attached to the patient's right temporal window above the zygoma to insonate the right middle cerebral artery (MCA). Additionally, a second sensor was placed on the CPB circuit tubing before the aortic cannula. Using a 2-MHz pulsed-waved transducer, simultaneous M-mode and spectrogram readings were generated. The insonation depth was set between 25 and 50 nm. Pulsatility index is defined as the difference between the maximum systolic (Vmax) and the minimum diastolic (Vmin) blood flow velocity divided by the mean blood flow velocity (Vmean).
10
Near-infrared spectroscopy
Quantification of regional cerebral oxygen saturation (rSO2) was performed via the INVOS 5100B near-infrared spectroscopy (NIRS) monitor (Somanetics). Near-infrared spectroscopy is a noninvasive method of measuring real-time changes in the percentage of oxyhemoglobin to total hemoglobin. Pediatric SomaSensors (Somanetics) were attached to the forehead below the hairline to collect NIRS data.
Both TCD and NIRS data were collected at the following times: baseline (preincision), on bypass before cross-clamp, and on bypass (after cross-clamp) at 5, 20, 40, and 60 min.
Postoperative Outcomes
Pediatric Logistic Organ Dysfunction-2 score
Postoperative vital organ injury was quantified using PELOD-2 score,19,20 which was calculated at 24, 48, and 72 h postoperatively for all patients remaining in the intensive care unit (CU) at the collection time. The score is derived using ten variables from five major organ systems, including neurologic, cardiovascular, renal, respiratory, and hematologic: Glasgow Coma Scale, pupillary reaction, lactatemia, systemic MAP, serum creatinine, ratio of PaO2 to fraction of inspired oxygen, PaCO2, presence of invasive ventilation, white blood cell count, and platelet count.19,20 A higher PELOD-2 score implies more organ injury and corresponds to higher mortality.
Pediatric Risk of Mortality-3 score
The Pediatric Risk of Mortality (PRISM) 3 score 21 was utilized to estimate mortality risk in children admitted to the ICU based on 17 physiologic variables: systolic blood pressure, temperature, Glasgow Coma Scale, heart rate, number of pupils that are >3 mm and fixed, and arterial pH, total CO2, PaCO2, PaO2, glucose, potassium, creatinine, urea, white blood cell count, prothrombin time, partial thromboplastin time, and platelet count within the first 12 h of admission to the ICU. A higher PRISM-3 score is associated with higher mortality risk.
Vasoactive–Inotropic score
The Vasoactive–Inotropic score (VIS) 22 quantifies the cardiovascular support required by patients and was assessed at 24 and 48 h after surgery. The score is calculated using the following equation:
VIS = Dopamine dose (µg/kg/min) + Dobutamine dose (µg/kg/min) + 100 × Epinephrine dose (µg/kg/min) + 10 × Milrinone dose (µg/kg/min) + 10,000 × Vasopressin dose (units/kg/min) + 100 × Norepinephrine dose (µg/kg/min)
Additional outcomes included intubation duration, ICU length of stay, hospital length of stay, and mortality within 180 days.
Statistical Methods
Descriptive statistics are reported as mean ± standard deviation (SD) for continuous variables and as frequencies (ie, n) and percentages for categorical variables. Chi-square tests, or Fisher exact tests if expected counts were small, were used to compare groups for categorical characteristics (eg, sex). Heterogenous variance ANOVA models having fixed factors of patient status (ie, cyanotic, acyanotic), pulsatility mode (ie, pulsatile, nonpulsatile), and their interaction were fit to continuous characteristics collected at one point in time (eg, aortic cross-clamp time). Linear mixed models having a random subject effect and fixed effects for patient status, pulsatility mode, time, and their interactions were fit to continuous characteristics collected at multiple points in time (eg, vasoactive-inotropic score [VIS] score at 24 and 48 h). The mixed models used a first-order ante-dependence variance–covariance matrix to account for the within-subject, as well as the between-subject, variability inherent in data collected at multiple time points (ie, repeated measures). Contrasts were constructed from the ANOVA and mixed models to test hypotheses of interest (ie, cyanotic vs acyanotic, as well as pulsatile vs nonpulsatile within cyanotic and within acyanotic patients). Residual diagnostics were used to assess ANOVA and linear mixed model fit, as well as underlying parametric model assumptions (eg, quantile–quantile plots to assess normality). All hypothesis tests were two-sided and all analyses were performed using SAS software, version 9.4 (SAS Institute Inc., Cary, NC).
Results
Preoperative and Operative Characteristics
Of the 159 randomized patients included in this study (n = 83 in the pulsatile [P] group and n = 76 in the nonpulsatile [NP] group), 115 were classified as having acyanotic heart disease (n = 60 in the P group vs n = 55 in the NP group), and 44 as having cyanotic heart disease (n = 23 in the P group vs n = 21 in the NP group). When patients within each CHD group were compared based on pulsatile versus nonpulsatile perfusion modes, the subgroups were noted to be similar in age, weight, height, STAT mortality score, aortic cross-clamp time, CPB time, CPB temperature, pump flow index, MAP, CPB arterial line pressure, slow continuous ultrafiltration volume, modified ultrafiltration volume, urine output during CPB, and plasma free hemoglobin (Table 1). When compared to patients in the acyanotic group, patients in the cyanotic group were more likely to be male, were younger, had higher STAT mortality scores, longer aortic cross-clamp and CPB times, and demonstrated a greater degree of hemolysis, as expected (Table 1).
Preoperative and Operative Characteristics of Pulsatile Versus Nonpulsatile Cohorts for Both Acyanotic and Cyanotic Patients (Mean ± SD).
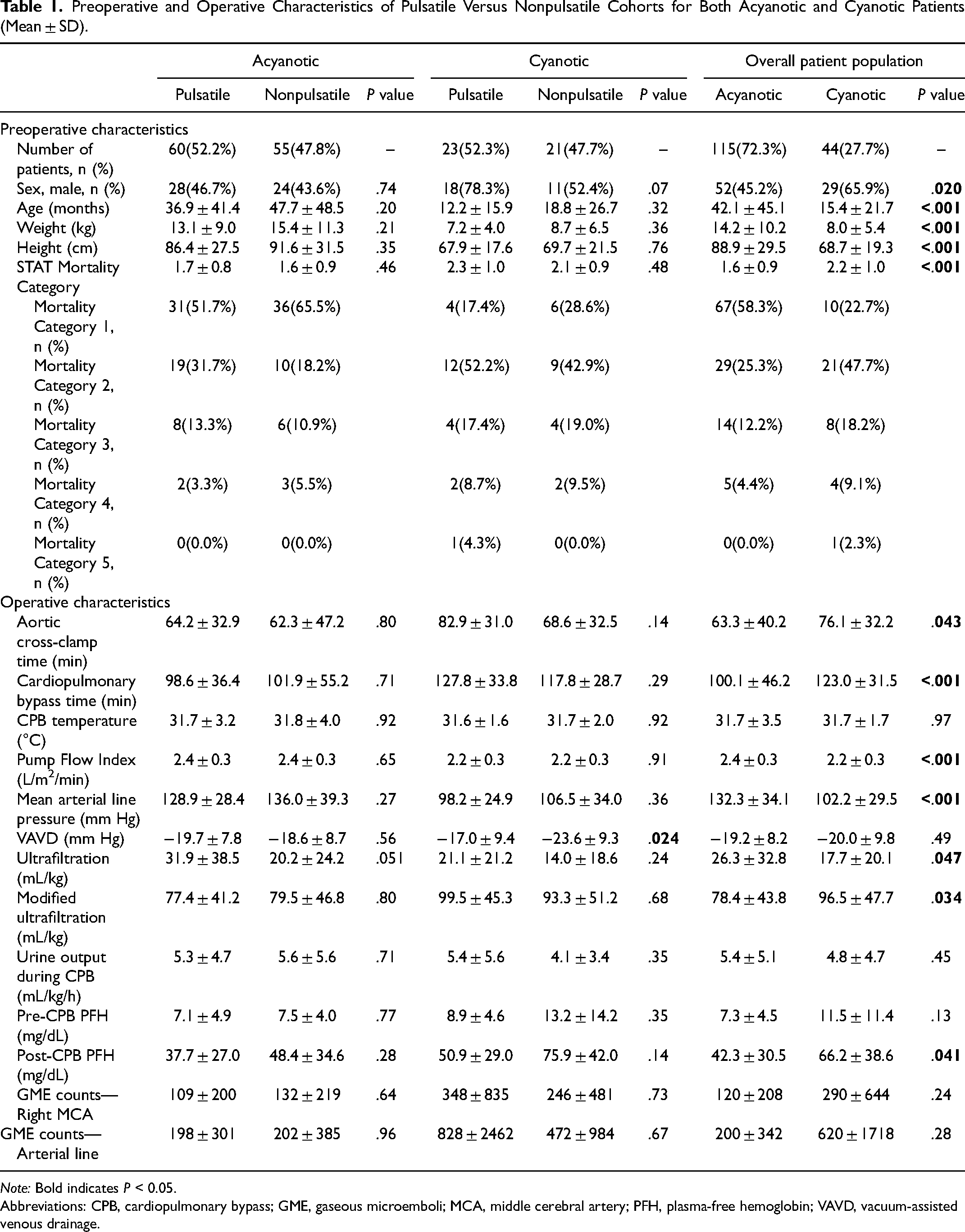
Note: Bold indicates P < 0.05.
Abbreviations: CPB, cardiopulmonary bypass; GME, gaseous microemboli; MCA, middle cerebral artery; PFH, plasma-free hemoglobin; VAVD, vacuum-assisted venous drainage.
Pulsatility Index and GME Counts
Pulsatility index values at the right MCA and by CPB arterial line were consistently higher in the pulsatile group for both acyanotic and cyanotic patients at all aortic cross-clamp time points (Figure 1A and B). The CPB arterial line pulsatility index was higher in the pulsatile group compared with the nonpulsatile group in both CHD groups under all experimental conditions (P < .05) (Figure 1B). Even under “nonpulsatile” flow, a measurable degree of pulsatility (PI > 0.5) was observed by CPB arterial line (Figure 1B). However, there was no difference in CPB arterial line pulsatility index between acyanotic versus cyanotic groups at any time point. The number of GME counts to the right MCA and in the CPB arterial line was similar between perfusion modes in both acyanotic and cyanotic patients (Table 1). Furthermore, no differences in overall GME counts were noted between the acyanotic versus cyanotic patients.
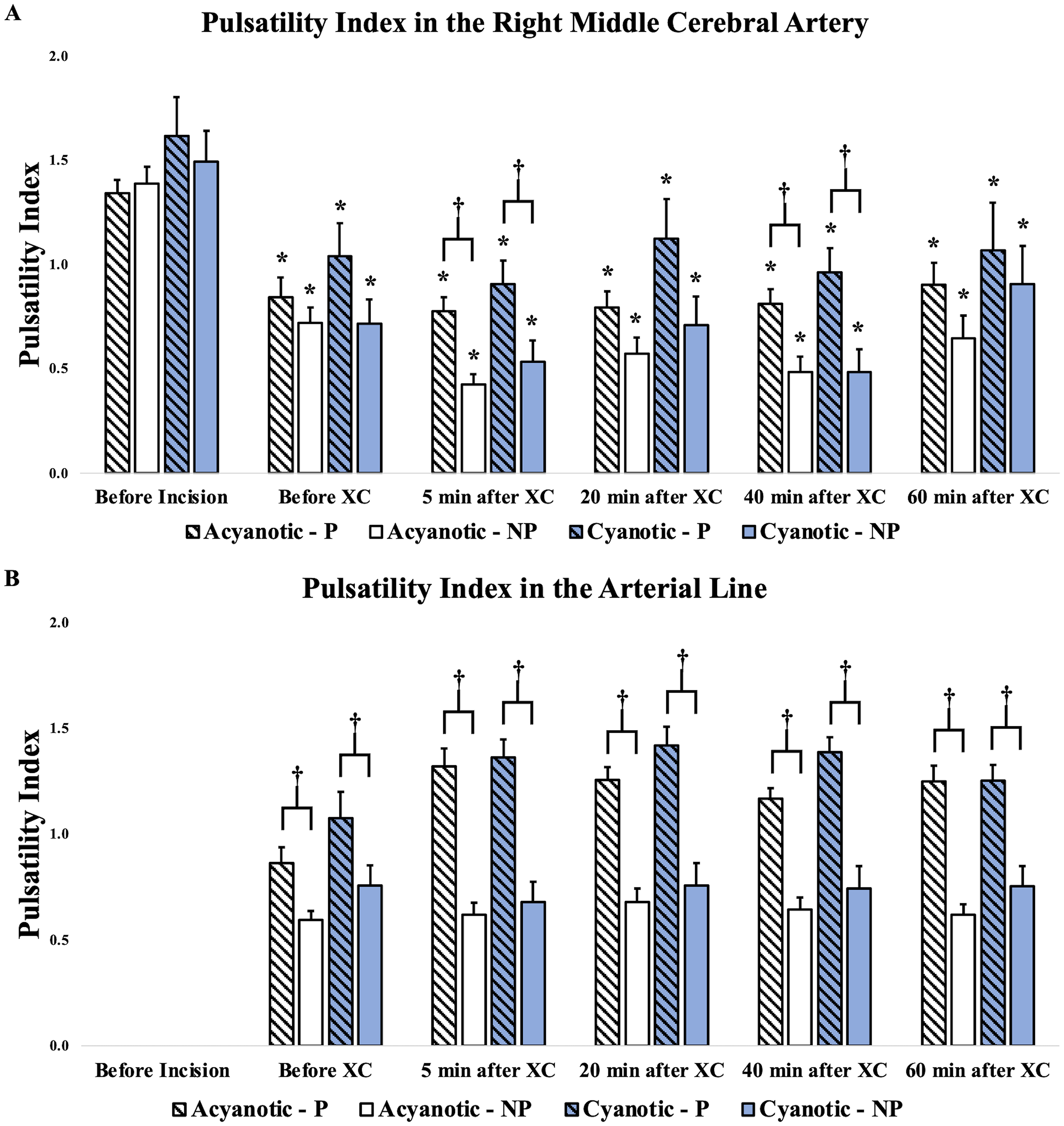
Pulsatility index at the right middle cerebral artery and in the CPB circuit arterial line for pulsatile versus nonpulsatile cohorts stratified by acyanotic versus cyanotic heart disease. *P < .001, comparison of PI at the given timepoint with its respective baseline (before incision) value, †P < .05, comparison of PI between NP and P modality at a given timepoint. Abbreviations: NP, nonpulsatile; P, pulsatile; XC, cross-clamp.
Regional Cerebral Oxygen Saturation and MAP
For both CHD groups and perfusion modality subgroups, rSO2 measurements were lower at all CPB timepoints compared with baseline values (Figure 2). Similarly, patients’ MAP values were lower compared with baseline values at all CPB timepoints in both CHD groups and perfusion modality subgroups (Figure 2). No differences in regional cerebral oxygen saturation or MAP were noted between perfusion modes or between acyanotic and cyanotic patients (Figure 2).
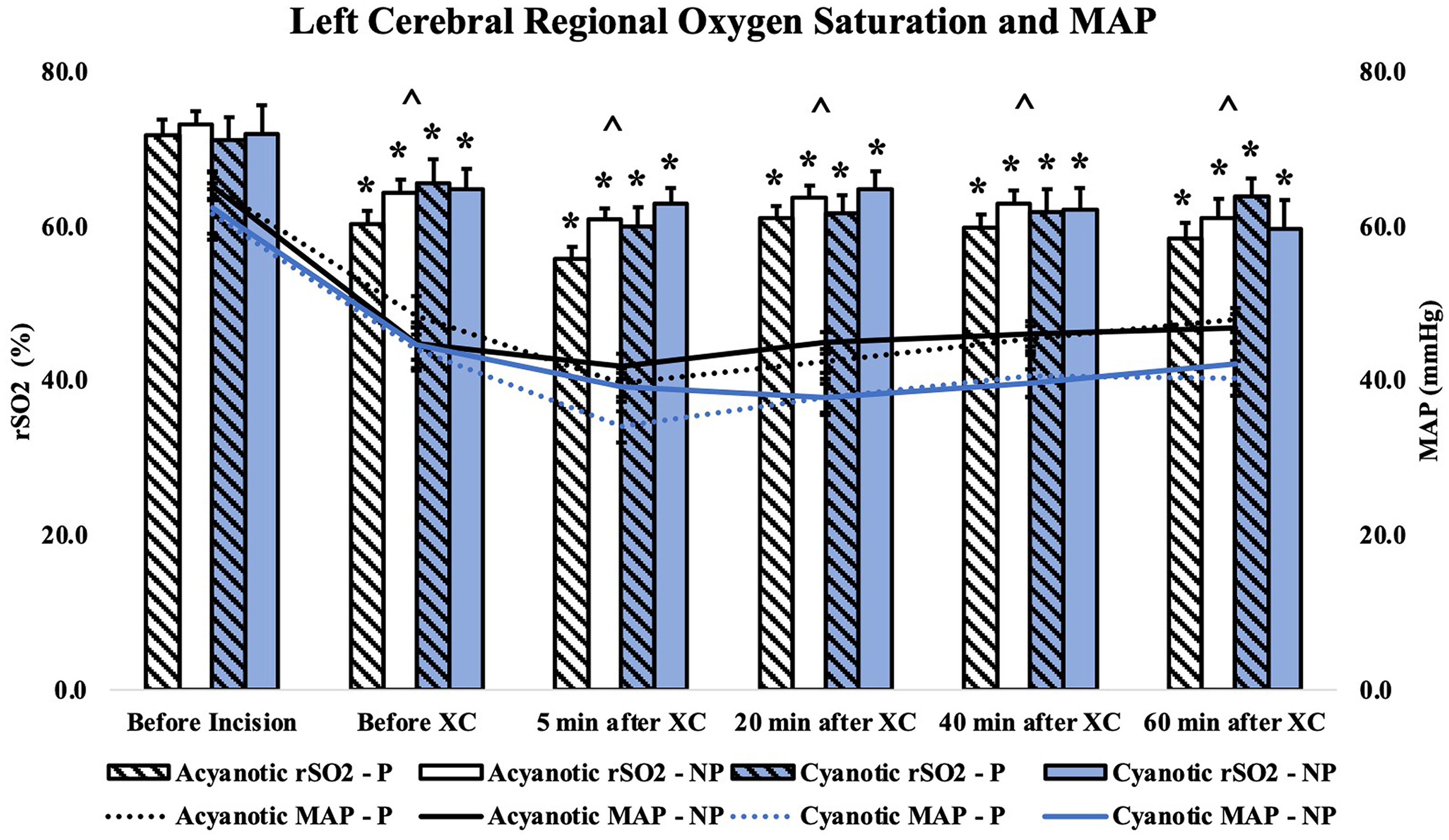
Regional cerebral oxygen saturation and mean arterial pressure for pulsatile versus nonpulsatile cohorts stratified by acyanotic versus cyanotic heart disease. MAP, mean arterial pressure. *P < .001, comparison of rSO2 at the given timepoint with its respective baseline (before incision) value, ^P < .001, comparison of MAP at a given timepoint with its respective baseline (before incision) value for all groups. Abbreviations: NP, nonpulsatile; P, pulsatile, XC, cross-clamp.
Postoperative Outcomes
Pediatric Logistic Organ Dysfunction-2 scores decreased over time in all groups. However, no differences were detected in PELOD-2 scores at any time point between pulsatile and nonpulsatile perfusion modes in either acyanotic or cyanotic patients (Figure 3). In the overall patient population, the PRISM-3 score was significantly higher in the cyanotic group compared with the acyanotic group (7.5 ± 3.5 vs 5.6 ± 2.5, P = .002). No differences were noted in PRISM-3 scores, VIS scores at 24 h, intubation time, ICU length of stay, hospital length of stay, and 180-day mortality rate between pulsatile and nonpulsatile perfusion modes in either acyanotic or cyanotic patients (Table 2). However, pulsatile perfusion was associated with higher VIS scores at 48 h in both acyanotic patients (P = 1.7 ± 3.4 vs NP = 0.6 ± 1.5, P = .022) and cyanotic patients (P = 2.4 ± 2.6 vs NP = 0.5 ± 1.0, P = .012), with the greatest bimodality difference seen in cyanotic patients (Table 2).
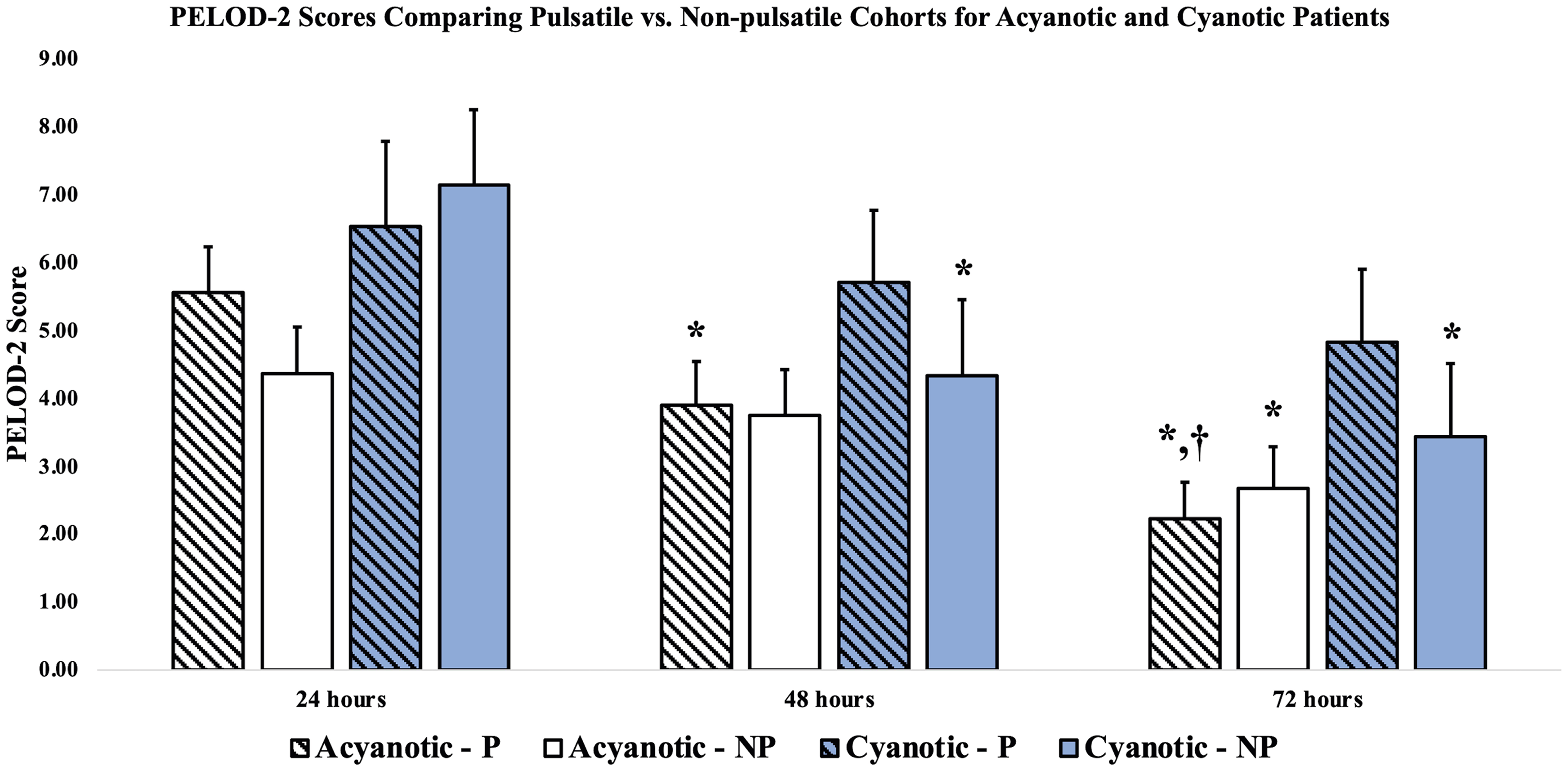
PELOD-2 scores for pulsatile versus nonpulsatile cohorts stratified by acyanotic versus cyanotic heart disease. PELOD, Pediatric Logistic Organ Dysfunction. *P < .05, comparison of PELOD-2 Score at the given timepoint with its respective value at 24 h postop, †P < .05, comparison of PELOD-2 Score at the given timepoint with its respective value at 48 h postop.
Postoperative Characteristics of Pulsatile Versus Nonpulsatile Cohorts for Both Acyanotic and Cyanotic Patients (Mean ± SD).
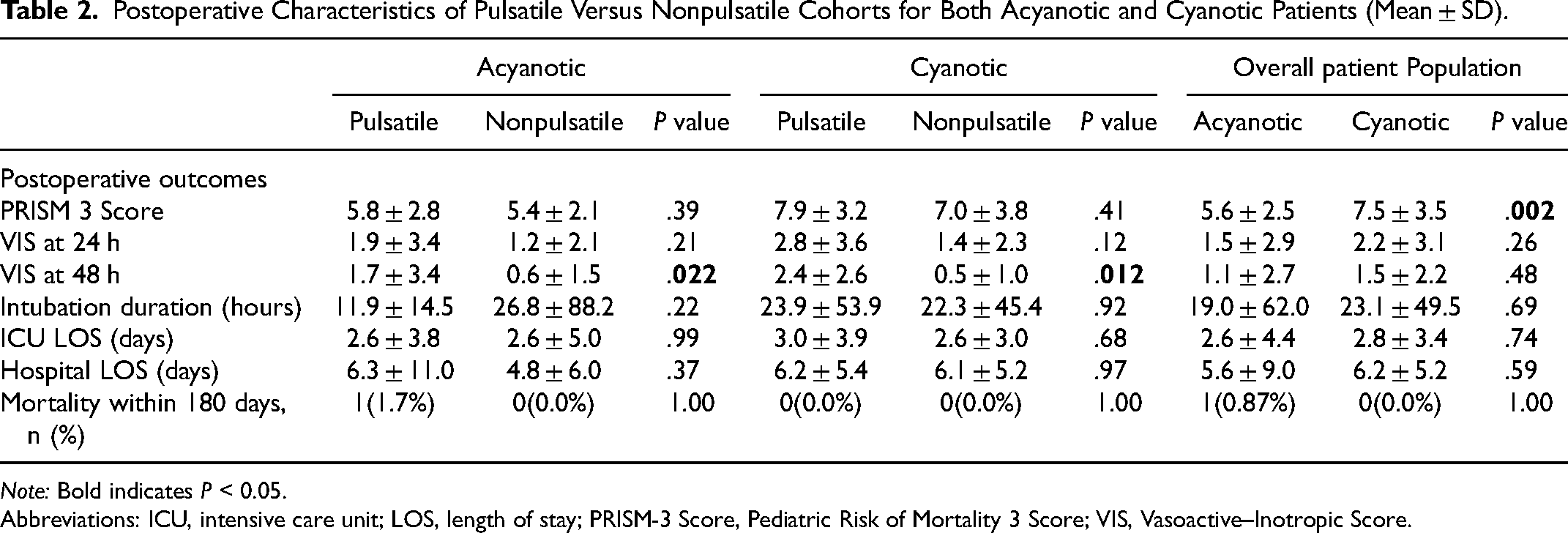
Note: Bold indicates P < 0.05.
Abbreviations: ICU, intensive care unit; LOS, length of stay; PRISM-3 Score, Pediatric Risk of Mortality 3 Score; VIS, Vasoactive–Inotropic Score.
Comment
In this prospective, randomized clinical study, we observed a significant difference in pulsatility index between patients undergoing pulsatile versus nonpulsatile CPB, indicating that patients randomized to these two perfusion modalities did indeed experience distinct hemodynamic conditions. Despite the different hemodynamic profiles experienced, we found no effect of perfusion modality on the duration of mechanical ventilation, ICU, or hospital LOS, even considering cyanotic versus acyanotic CHD. Patients in both CHD groups had an improvement in PELOD-2 scores over time, indicating clinical improvement. However, perfusion modality did not impact the extent of PELOD-2 score improvement. Reassuringly, within a given CHD group, perfusion modality also did not significantly affect GME counts, regional cerebral oxygen saturation, the extent of hemolysis,nor markers of end-organ perfusion. Only one mortality was observed in our study, so the effect of perfusion modality on mortality could not be accurately assessed. The intraoperative use of TCD and NIRS monitoring facilitated this direct comparison of physiologic parameters and risk profiles and would be a valuable component of future perfusion modality studies.
Notably, we did observe that in both CHD groups, patients in the pulsatile subgroups experienced a slower improvement in VIS, with a higher VIS at 48 h postoperatively compared with their nonpulsatile counterparts. Interestingly, the cyanotic heart disease patients appeared more significantly affected. Of note, the ICU teams were not aware of the perfusion modality utilized in the operating room, which we believe reduces the likelihood of systematic bias in postoperative care on the basis of perfusion modality. The validity and physiologic basis for this vasoactive requirement difference is not yet clear, but could suggest a differential effect of pulsatile CPB that is augmented by underlying cyanotic CHD. Given what is known regarding the effects of chronic hypoxemia on cardiomyocyte oxidative metabolism, augmented post-CPB inflammation, and impairment of myocardial contractility, 8 this finding would warrant close attention if corroborated in subsequent studies.
Our study also confirmed that roller pumps could safely generate a different quality of pulsatility in the MCA and CPB circuitry under different perfusion modalities. Specifically, pulsatile flow settings, in terms of base flow, pump-head start point, pump-head stop target, and pump frequency, directly impacted the quality of the pulsatility index. We selected the current pulsatile flow parameters based on our in vitro evaluations and pilot clinical trials.10,23 We used the same CPB machine (HL-20) for all study conditions, so pulsatile flow did not incur additional costs.
As previously noted, we found that even the traditional, “nonpulsatile” CPB modality did not deliver a truly fixed flow rate, with minor fluctuations still present. As such, it is unknown whether these minor fluctuations contributed to the inability to identify between-group clinical differences, even though quantifiable differences in pulsatility index were observed. To best ensure distinct hemodynamic profiles between groups, the multidisciplinary team at Penn State Pediatric Cardiovascular Research Center has done extensive in vitro23‐26 and in vivo9,10,24‐26 studies to optimize each of the CPB circuit components, including blood pump, oxygenator, and CPB aortic cannulae. We have documented the ability to generate measurably distinct pulsatility using an aortic cannula as small as 8F in an optimized circuitry for neonatal cardiac patients. 27 To better isolate the effect of perfusion modality on outcomes, in our current prospective randomized clinical trial and previous retrospective trial of 284 pediatric cardiac patients (n = 152 for nonpulsatile and n = 132 for pulsatile groups), MAPs were kept similar between pulsatile and nonpulsatile groups. 27 Likewise, intraoperative oxygenation was maintained similarly in patients regardless of perfusion modality or CHD type.
Another limitation of this study may be sample size, especially for subgroups taking pulsatile status into account. With 115 acyanotic patients and 44 cyanotic patients, the study had 80% power to detect a standardized effect size (ie, difference in means divided by the SD) of 0.5 using a two-sided test and a significance level of 0.05. However, as the sample size is rather small in some subgroups, there is an increased probability of a type II error. Thus, we cannot rule out that statistically insignificant results are not clinically relevant due to the possibility of low power.
In summary, although we found that pulsatile CPB leads to greater arterial pulsatility than nonpulsatile modalities and was not overtly associated with a greater risk of short-term adverse effects compared with nonpulsatile CPB, we did not find any greater improvement in PELOD-2 scores nor clinical outcomes in patients with either cyanotic or acyanotic CHD. That said, we feel that our study's unique finding of different vasoactive requirements postoperatively might warrant further investigation. For example, future studies could evaluate whether CPB perfusion modalities have differential effects on vascular tone, vasoactive medication responsiveness, myocardial recovery, or post-CPB inflammation, potentially identifying microvascular or cellular changes that we were unable to assess in our study. Based on our current results, we feel that pulsatile perfusion remains a viable option for use in the CHD population, but further research is warranted to further elucidate the effects of different CPB perfusion modality parameters, the macro-hemodynamic as well as possible microvascular effects of these modifications, as well as both short- and long-term clinical outcomes—importantly, taking into account CHD patients with a range of physiologic profiles.
Supplemental Material
sj-docx-1-pch-10.1177_21501351241288835 - Supplemental material for Randomized Trial of Pulsatile and Nonpulsatile Flow in Cyanotic and Acyanotic Congenital Heart Surgery
Supplemental material, sj-docx-1-pch-10.1177_21501351241288835 for Randomized Trial of Pulsatile and Nonpulsatile Flow in Cyanotic and Acyanotic Congenital Heart Surgery by Krishna Patel, Tracie K. Lin, Joseph B. Clark, Gary D. Ceneviva, Jason R. Imundo, Debra Spear, Allen R. Kunselman, Neal J. Thomas, John L. Myers and Akif Ündar in World Journal for Pediatric and Congenital Heart Surgery
Footnotes
Acknowledgments
This project was supported by seed funds from the Pediatric Cardiovascular Research Center at Penn State Health Children's Hospital and Penn State College of Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.