Abstract
Introduction
Interferon-based therapies against the hepatitis C virus had a poor adherence profile. On the other hand, new direct-acting antivirals (DAAs) are orally administered medications, show high efficacy against the hepatitis C virus in addition to a high safety profile. Therefore, adherence to this treatment is expected to improve. Assessment for treatment adherence is mandatory to assess the feasibility of achieving viral hepatitis elimination.
Aim
The study aims to assess the adherence rate and causes of non-adherence in Egyptian hepatitis C patients who received interferon-free treatment regimens.
Methods
Retrospective data analysis for 668 hepatitis C patient’s records from August 2014 to October 2019 was done. Assessment of treatment adherence was done by revising the records and phone calls. However, 172 patients were excluded due to the absence of contact data. Rest of patients (n = 496) was categorized into 2 groups: Adherent (n = 432) and non-adherent (n = 64). For whom comparative analysis was done.
Results
The adherent group (87%) achieved 100 % sustained virological response after 12 weeks (SVR 12). Non-adherence was reported in 12.9% of patients. Low awareness was the main cause of non-adherence (43.75%). BMI was the only significant risk factor for poor adherence (P = .04). Other Patient demographics, clinical, and laboratory data didn’t show any significant differences between both groups.
Conclusion
Interferon-free regimens are tolerable. Raising awareness is mandatory for proper treatment adherence and, subsequently, good clinical outcomes.
Introduction
Hepatitis C virus (HCV) is a global healthcare problem. Hepatitis C is a viral infection with marked hepatic and extra-hepatic manifestations. Not only is hepatitis C a common cause of cirrhosis, but it is also a significant risk factor for hepatocellular carcinoma and one of the leading indications for liver transplantation. Moreover, its extra-hepatic manifestations include varied expressions of glomerulonephritis, cryoglobulinemia, and lymphoma. 1
Many people with chronic hepatitis are unaware of their disease status until they develop symptoms of advanced disease. Delays in identifying disease status can increase the likelihood of virus transmission to others, limited treatment options, and worsened patient outcomes. Despite the increasing public awareness of viral hepatitis in the past 2 decades, significant knowledge gaps remain even in the highest risk populations. 2
Globally, there are about 71 million patients infected with HCV. Since the introduction of sofosbuvir in 2014, HCV treatment has become more accessible and tolerable. 3 Previous interferon (IFN) based regimens were fairly intolerable for their side effects and long therapy duration. Therefore, treatment stoppage cases have been reported in many cases. 4
Adherence to therapy is defined by the World Health Organization (WHO) as “the degree to which the person’s behavior corresponds with the agreed recommendations from a health care provider”. 5 Adherence is vital for a good clinical outcome. Many personal and drug-related factors could lead to non-adherence. There are 2 main types of non-adherence: Primary, which occurs before the initiation of therapy, and secondary, which occur after starting treatment. 6 Several Egyptian studies have addressed different screening, investigations, and treatment methods of Hepatitis C in Egypt and even the cure rate. But till now, to our knowledge, no studies have addressed the causes and barriers of non-adherence to treatment, especially during the Egyptian National Program that launched in October 2018.7,8
We aim to assess patients’ adherence to IFN-free HCV medication in the Egyptian National Program to treat HCV and identify risk factors for non-continuation of therapy.
Patients and Methods
This retrospective descriptive study was done in Cairo university hospitals in the period from March to July 2019. Records of 668 adult HCV patients who started treatment for HCV were retrieved from the medical records from August 2014 to October 2019. The retrieved data included demographic data, medical history and comorbidities, assessment of hepatic condition either clinically or through imaging and laboratory investigations, treatment outcome, and adherence profile.
Assessment of adherence was done by revising patients’ records and phone calls to assess the sustained virological response after 12 weeks (SVR 12) of all patients and asking patients who didn’t complete treatment for causes of non-adherence. A toal of 172 patients were excluded due to the absence of contact data. Rest of patients (n = 496) was categorized into 2 groups: Adherent (n = 432) and non-adherent (n = 64). For whom comparative analysis was done to identify risk factors for non-adherence.
Ethical Consideration
Ethical approvals were obtained from the Research Ethics Committee at Cairo University, the Viral Hepatitis Center at Kasr Alainy hospital, and the National Committee for Control of Viral Hepatitis in the Ministry of Health. Oral consent was taken from the patients at the start of each phone call.
Statistical Analysis
Data were coded and entered in Microsoft Excel® and then exported for analysis to The Statistical Package for the Social Sciences (IBM SPSS®) (version22). Data were statistically described in terms of Mean ± Standard Deviation (±SD) or Frequencies (Number of cases) and percentages when appropriate. An exact test was used instead when the expected frequency is less than 5. For comparing categorical data, Chi-square (χ2) test was performed. Logistic regression analysis was done to determine significant predictors for compliant patients. A P-value less than .05 was considered statistically significant, and a P-value less than .01 was considered highly significant.
Results
The adherent group (87%) achieved 100 % SVR 12. Demographic analysis of the studied groups revealed a higher “non-significant” mean age of non-adherent group versus adherent group (50.44 ± 12.39 vs 48.7 ± 13.56). There was no statistical difference between both groups regarding gender distribution and service availability (Table 1). The non-adherent group had a significantly higher BMI than the adherent group (P = .04). There was no significant difference between both groups regarding treatment history, type, and duration (Table 2). Liver function tests and liver imaging by the US didn’t show significant differences between both groups (Table 3). The main causes for non-adherence were: Low awareness (43.75%), Unaffordable cost of follow-up laboratories (17.1%), especially for those who missed the date for the free follow-up laboratories, and development of complications (12.5%). There are 3 mortality cases that are not related to medication (Figure 1).
Demographic Features of the Study Groups.

SD: standard deviation.
Clinical Characteristics of the Study Groups.
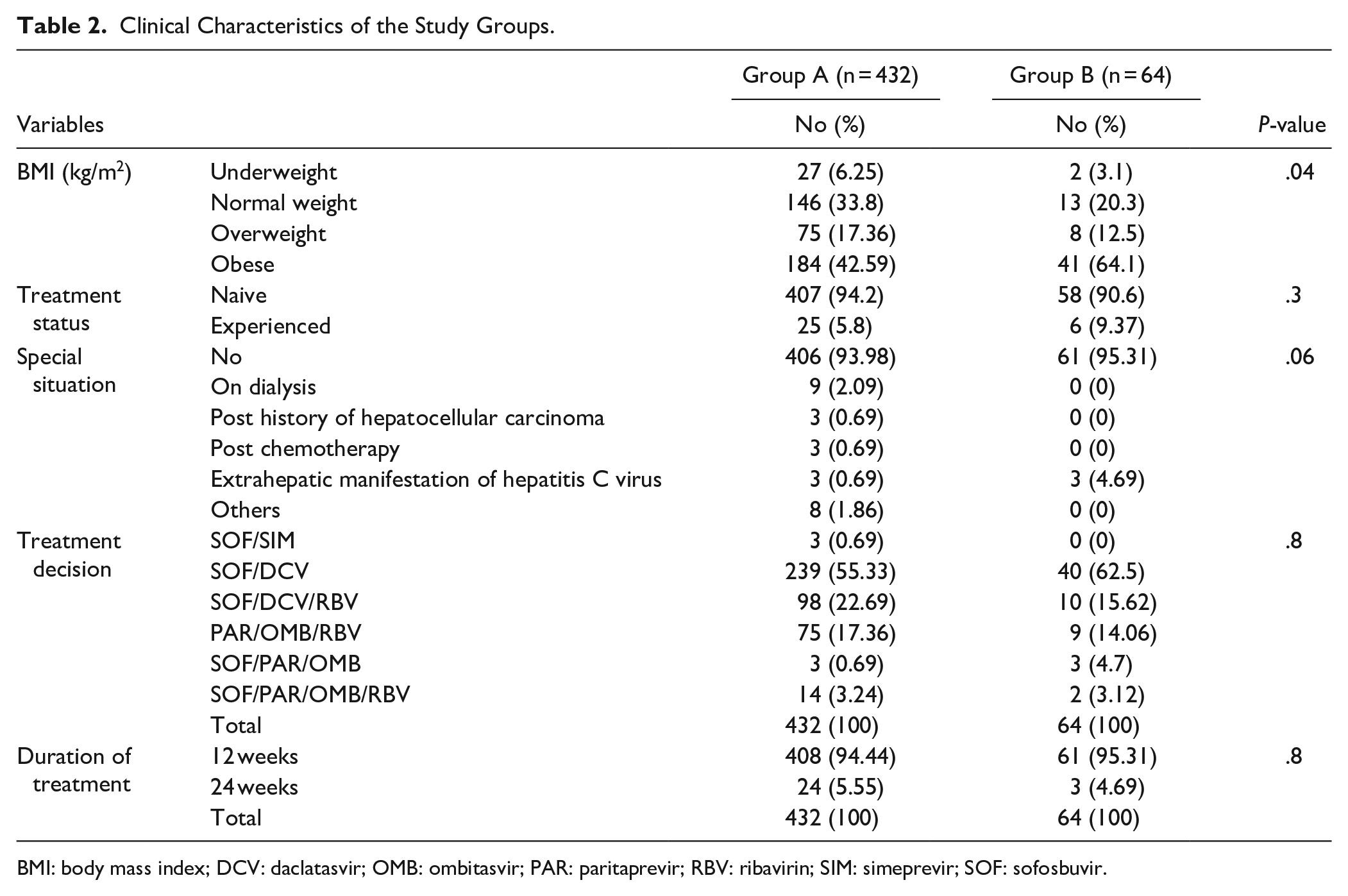
BMI: body mass index; DCV: daclatasvir; OMB: ombitasvir; PAR: paritaprevir; RBV: ribavirin; SIM: simeprevir; SOF: sofosbuvir.
Laboratory and Ultrasonographic Findings in Study Groups.

ALT: alanine aminotransferase; AST: aspartate transaminase; US: ultrasound.
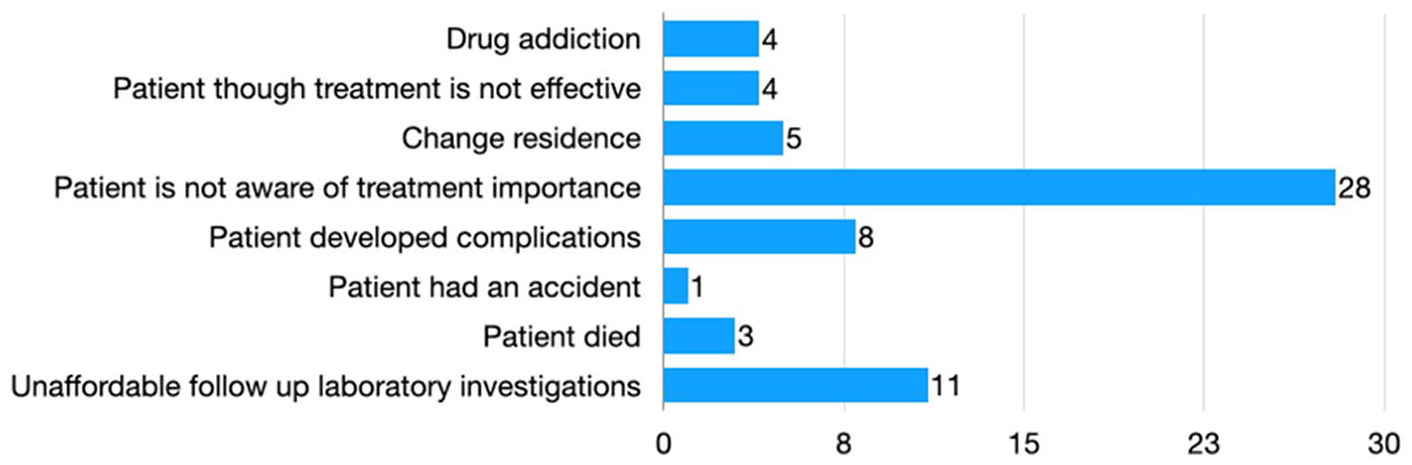
Causes of non-adherence among study participants.
Discussion
In 2016, WHO had set a goal of eliminating viral hepatitis by 2030. 9 Therefore, in October 2018, Egypt launched an extensive national campaign to screen and treat HCV targeting 62.5 million adult Egyptian citizens. More than 49 million people were screened, and the overall HCV seroprevalence in Egypt was 4.61%. 8 Mass treatment is one of the main pillars of elimination. Many follow-up studies have been done to ensure the efficiency and quality of the treatment program. Adherence to therapy is an essential factor of treatment success, and it’s an important indicator of the quality of the treatment program.
The definition of adherence varies; it can be defined as completion of at least 80% of treatment or taking all prescribed medications in addition to attending all follow-up visits. 4 Taking all prescribed medications was the definition used in this study. 10 There is a direct relationship between adherence to therapy and achieving SVR. 11 Non-adherence could emerge because of many factors. All factors need proper evaluation and management.
Physical and cognitive dysfunction is considered the main drive for non-adherence in elderly patients. 12 The mean age of the non-adherent group was “non-significantly” higher than the adherent group in our study. Specially designed educational programs need to be implemented for elderly patients and use dose monitoring techniques such as modified directly observed therapy (DOT) approach for follow-up of these patients. 13 Obesity also has been identified as a risk factor for non-adherence to HCV treatment in our study. Previous studies found an inverse relationship between obesity and adherence to certain medications, such as oral anti-diabetic medications. 14 A condition that may be attributed to the high prevalence of depression among obese people. 14 Additional personal factors for non-adherence such as injecting drugs, alcohol consumption, and illiteracy are reported in many studies; however, these risk factors were not significant in our study.
One of the major barriers to adherence to the treatment in our study was a lack of awareness among the non-adherent group. The effect of the educational interventions diagnosed with HCV is crucial to increase the adherence rate to the treatment. 15 Type of medication and route of administration can also affect drug adherence. In the past, higher levels of non-adherence were reported during treating HCV using pegylated interferon and ribavirin. A German study reported that 23% of HCV patients showed poor compliance during treatment using interferon with or without ribavirin. 16 IFN-free DAA regimens show higher tolerability than IFN-based regimens due to easy administration (oral vs subcutaneous injection), low pill burden (2 vs 5-6 pills per day), shorter duration (12 or 24 vs 48 weeks), High SVR rates, and low adverse events profile. IFN and ribavirin have many adverse events such as bony pains, jaundice, and anemia, besides psychiatric complications. 4 Our study participants used IFN-free DAA regimens. Only 12.9% of patients showed poor compliance. No statistically significant differences were noticed between different IFN-free regimens.
Treatment cost is considered an important factor in adherence to HCV treatment. 17 Free dispensing of medications in the Egyptian program leads to better adherence to therapy. The cost of laboratory investigations was one of the barriers to the completion of treatment. As the patient had to pay this cost if he missed the free scheduled laboratory investigations. Advanced liver disease (Child-Pugh class: C) is an exclusion criterion to start DAA therapy according to the national committee’s guidelines for control of viral hepatitis. Therefore, fibrosis state by the US, liver transaminases (ALT and AST), and liver synthetic functions (Albumin and INR) didn’t show a significant change between both groups.
Assessing a patient’s adherence is not an easy task. Many methods have been developed for that reason. 11 Patient self-reporting is the easiest and simplest way for assessment; however, it’s not accurate and depends on the patient’s cooperation, which is not suitable in the elderly or in patients with poor educational level or poor mental state. It’s the most common method of assessment of adherence in the Egyptian program. 18 Pill count and pharmacy refills are other ways to assess adherence that have been used in many studies.19,20 New techniques such as microelectronic monitoring have been proposed to evaluate adherence, but it’s not widely used in HCV management. 21
The system of care is also responsible for proper adherence to therapy. In 2019, an American study found that treatment adherence and success rates are higher when using specialized HCV clinics than general hepatology clinics. 22 Using specialized clinics to treat viral hepatitis was a cornerstone of the success of the Egyptian program for HCV elimination. More than 100 treatment centers were capable of treating more than 1 million patients. They provide easy access to care. 23 Additionally, these centers are responsible for raising awareness and health education. About 43.75% of non-adherent patients were because of their low level of awareness, which necessitates a more focus on health education. Health education should be multidisciplinary. Cooperation between a hepatologist, family physician, and psychiatrist is needed for a good outcome.
The main weakness of the study is the potential bias related to recall problems when contacting patients who completed treatment years ago. Besides, many patients were excluded from the analysis due to the absence of contact data; however, they did not influence the results.
Conclusion
Overall, DAAs showed high efficacy, tolerability, and better adherence profile versus Interferon-based therapies in the Egyptian HCV patients. Low awareness was the main cause of non-adherence in patients receiving DAAs. Raising awareness is mandatory for proper treatment adherence and, subsequently, good clinical outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
