Abstract
Background
Peripheral artery disease (PAD) is associated with cardiovascular risk in type 2 diabetes mellitus (DM). The ankle-brachial index (ABI) is used for diagnosis of PAD.
Objectives
Establish the prevalence and incidence rate for PAD and determine the associated factors and survival time for the development of PAD.
Methods
Retrospective cross-sectional cohort study (follow up: 10 years) in 578 DM patients with at least 1 ABI measurement in a primary level of care diabetes clinic. Data was collected from clinical records. Sociodemographic and laboratory variables were analyzed determining its association (mean difference and bivariate logistic regression). Survival was calculated through life tables and Kaplan-Meier analysis.
Results
The prevalence of PAD was 13.98%. The incidence rate through the time of follow up was 23.38 per 1000 person-year (95% CI: 19.91-27.26). The group that developed PAD showed higher glycated hemoglobin levels (
Conclusion
Half of a DM patient’s population in primary level of care will develop PAD in the third decade of disease. There are identifiable risk factors for PAD development in DM in the primary level of care such as low GFR, female sex, and use of metformin plus insulin.
Keywords
Introduction
Peripheral artery disease (PAD) has classically been defined as the obstruction of the lower extremity arteries by atherosclerotic plaques. 1 Although this concept is widely accepted, peripheral arteries can also be affected by arterial calcification (AC). Thus, the term lower extremity artery disease (LEAD) will be used to include both, PAD and AC, for the purpose of this investigation.2,3 LEAD has been associated with cardiovascular events in patients with type 2 diabetes mellitus (DM). These events have included coronary heart disease, stroke and other atherosclerotic related causes of death. 3 For PAD, risk factors include the presence of diabetes mellitus, hypertension, previous history of cardiovascular disease, smoking, and hypercholesterolemia. 4
The American Diabetes Association (ADA) suggests PAD screening every 5 years in DM patients older than 50 years with normal ankle-brachial index (ABI) and patients younger than 50 years with risk factors.1,5 The ABI is a simple and non-invasive method that allows diagnosis of PAD. Although other methods exist for assessing PAD (like toe-brachial index) the ABI method calculated using a sphygmomanometer and vascular Doppler is widely available in primary level of care. 6 This method has a sensitivity of 69% to 95% and specificity from 83% to 93%, when there is an obstruction greater than 50%.1,3,7 The prevalence of altered ABI in patients with diabetes is variable, ranging from 18.8% in Spain to 52.5% in Nigeria.8,9 In DM hyperglycemia and hyperinsulinemia produce inflammation, reducing vasodilation and favoring coagulation, vascular proliferation, and inflammatory markers in the endothelium. These factors facilitate leukocitary migration to this site and their transformation into foam cells, developing atheromatosis which ends up in arteriopathy (macrovascular disease). 1
The objectives of the present research are to determine the associated factors and survival time for PAD in DM patients. In addition, it aims to establish the incidence rate and prevalence for PAD and the prevalence of patients with LEAD in a Latin population with DM in the primary level of care. With these objectives we expect to establish risk factors for PAD and a baseline prevalence and survival time for LEAD, AC, and PAD in Latin patients with DM.
Methods
Setting, Population, and Data Recovery
This retrospective cross-sectional cohort study, worked with DM diagnosed patients treated in the Diabetes Clinic of primary level of care in Chimbacalle (Quito, Ecuador), from 2007 to 2017. The data for this study was obtained from the clinical records of 674 participants in year 2018. The minimum sample size for an alpha of 0.05 and a statistical power of 0.80, was estimated in 465 patients. The final sample size was 578 patients who voluntarily agreed to participate in this study (Figure 1).

Flow Diagram of Elegible Participants in the Study.
All patients who fulfilled the following parameters were included in the study: DM diagnosis (in accordance with criteria stated by ADA)10,11; at least 1 ABI measurement during the study period (3 measurements every 30 months starting from 2012); a minimum of a year of baseline biological parameters and laboratory tests (twice a year; total cholesterol—TC, high density lipoproteins—cHDL, low density lipoproteins—cLDL, triglycerides—Tg, glycated hemoglobin—HbA1c, urea, creatinine, and uric acid) prior to the first ABI measurement; being older than 18 years and having signed a written consent. The exclusion criteria were: type 1 diabetes mellitus diagnosis, incomplete data in clinical records, previous revascularization, pregnancy, no documented ABI, lower limb amputation, previous diagnosis of LEAD, and the ones who did not sign a written consent.
In this diabetes clinic, laboratory exams and medication (antihypertensive, lipid-lowering, glucose-lowering, and others) are free. The lipid-lowering drugs are given to all patients with a diagnosis of dyslipidemia and continued even if target values are achieved after treatment with these drugs. When being diagnosed with DM all patients are prescribed with acetyl salicylic acid, unless contraindications are present. Additionally, patients have an active participation in the diabetes club including educational talks every 2 weeks and physical activity 3 times per week.
Measurements
ABI was determined using a vascular Doppler (SONOTRAX, Ultrasonic Pocket Doppler version 1.5, manufactured in Germany) and a sphygmomanometer (RIESTER, Big Ben), both calibrated. The ABI was measured by a single operator during the time of follow up. Arterial pressure was assessed while the patient was lying supine. To determine systolic blood pressure (SBP) the brachial and dorsal pedal or posterior tibial arterie pulse were located. Posteriorly the sphygmomanometer was inflated 20 mmHg over the level on which the doppler signal was absent, and deflated until the doppler signal was heard.3,12 The process was repeated in the 4 limbs. The ABI estimation was manually calculated using the following formula:
Statistical Analysis
Data was collected on Microsoft Excel (Microsoft Office, version 2010). The statistical analysis was performed with SPSS Statistical Analysis Software (IBM SPSS, version 23). Patients were divided into 2 groups according to ABI: with PAD and without PAD. For categorical variables, proportions were calculated; whereas, for numerical variables, measures of central tendency were used. For comparison between the 2 groups by sex and PAD status, the mean difference (
For survival analysis, the survival time for the development of PAD was the interval between the index time (diagnosis of DM) and occurrence of the event of interest (diagnosis of PAD or end of follow up). End of follow up was defined as the date of loss of follow up of the patient or December 31st, 2017 when the study ended. Life tables were used to estimate the survival time for developing PAD and Kaplan-Meier to assess the accumulative probability for the presence of PAD using Cox proportional hazards model. The analysis was controlled for the following covariables: sex, family history of cardiovascular disease, smoking history, HTN, Body Mass Index (BMI, >or equal 25), altered electrocardiogram (EKG), age at DM diagnosis (<65 years), GFR (<60 mL/min/m2), TC (>200 mg/dL), cHDL (<40 mg/dL in males and <50 mg/dL in females), cLDL (>100 mg/dL), Tg (>150 mg/dL), atherogenic index (low vs moderate and high risk), HbA1c (>7%), and medication (treatment with: metformin, metformin plus glibenclamide, insulin, metformin plus insulin, other treatments). Binary logistic regression was made through the enter method, with an emphasis on factors that were statistically significant in the univariate analysis. Statistically significant analyses were assessed as a
Ethics
The study was approved by the Bioethics Committee at the Central University of Ecuador as part of one of the Biomedicine Investigation Institute projects at the same institution (resolution no. 279-CE-UCE-2015).
Results
Out of the 578 patients included in the study, 452 (78.2%) were female and 472 (99%) considered themselves Latin. The mean age was 65.99 years (SD: 12.03; range: 47.8-96.15) and the mean years of disease was 13.11 (SD: 7.43; range: 1-43). There was a difference between sexes for BMI, TC, cHDL, uric acid, ABI, and smoking history (Table 1). The LEAD prevalence in 2017 was 15.27% (71/465 patients) and the PAD prevalence was 13.98% (65/465 patients), 7% (7/100 patients) in males and 15.89% (58/365 patients) in females. During the 10 years follow up there were 11 (1.9%) patients with AC and 158 (27.3%) patients with PAD; in total 169 (29.2%) patients developed LEAD. The incidence rate was 23.38 (95% CI: 19.91-27.26) cases of PAD per 1000 persons-year. Between the group with PAD and without PAD the mean difference for years of disease, age of diagnosis of PAD, HbA1c, and GFR were statistically significant. The risk association between the 2 groups with binary variables was found on female sex (OR: 2.50, 95% CI: 1.48-4.24,
Clinical and Metabolic Characteristics of Type 2 Diabetes Mellitus Patients for General Population and According to Sex.
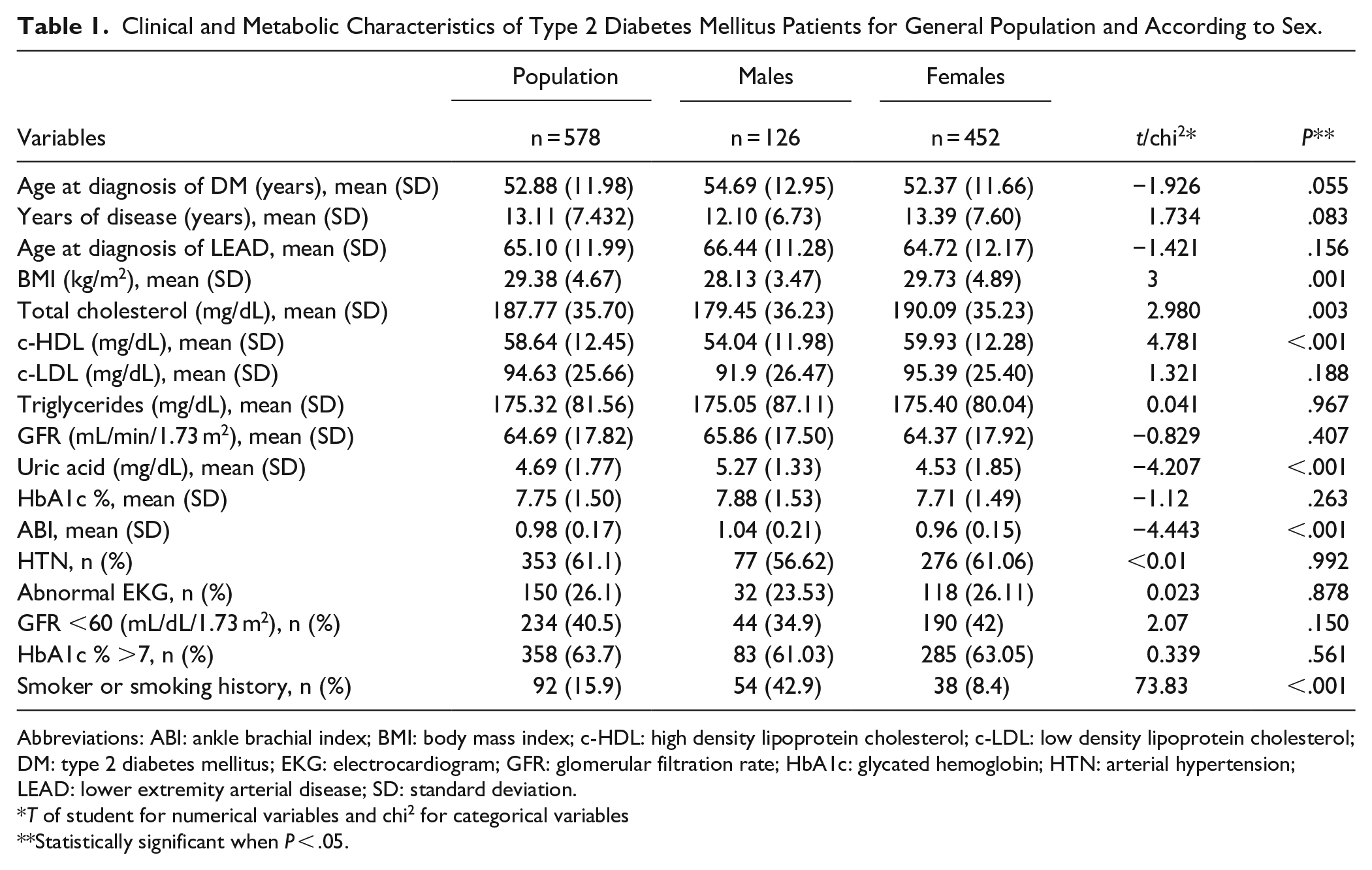
Abbreviations: ABI: ankle brachial index; BMI: body mass index; c-HDL: high density lipoprotein cholesterol; c-LDL: low density lipoprotein cholesterol; DM: type 2 diabetes mellitus; EKG: electrocardiogram; GFR: glomerular filtration rate; HbA1c: glycated hemoglobin; HTN: arterial hypertension; LEAD: lower extremity arterial disease; SD: standard deviation.
Statistically significant when
Clinical and Metabolic Characteristics According to Peripheral Artery Disease Status in Type 2 Diabetes Mellitus Patients.

Abbreviations: ABI: ankle brachial index; BMI: body mass index; c-HDL: high density lipoprotein cholesterol; c-LDL: low density lipoprotein cholesterol; DM: type 2 diabetes mellitus; GFR: glomerular filtration rate; HbA1c: glycated hemoglobin; PAD: peripheral artery disease; SD: standard deviation.
Statistically significant when
Last control of 2017.
The estimated mean time for developing AC was 40.28 (95% CI: 38.47-42.08) years, for PAD 23.81 (95% CI: 22.01-25.61) years and for LEAD 22.94 years (95% CI: 21.25-24.63). Life tables by 10-year periods revealed that 50% of patients would develop PAD in 26.97 (95% CI: 26.89-27.05) years of disease. The Hazard Ratio was of 1.857 (95% CI: 1.853-1.861) for the first decade of disease, 2.52 (95% CI: 2.513-2.527) for the second decade, 3.83 (95% CI: 3.812-3.847) for patients in the third decade of disease and 10.53 (95% CI: 10.471-10.582) in the fourth decade of disease (Figure 2A).
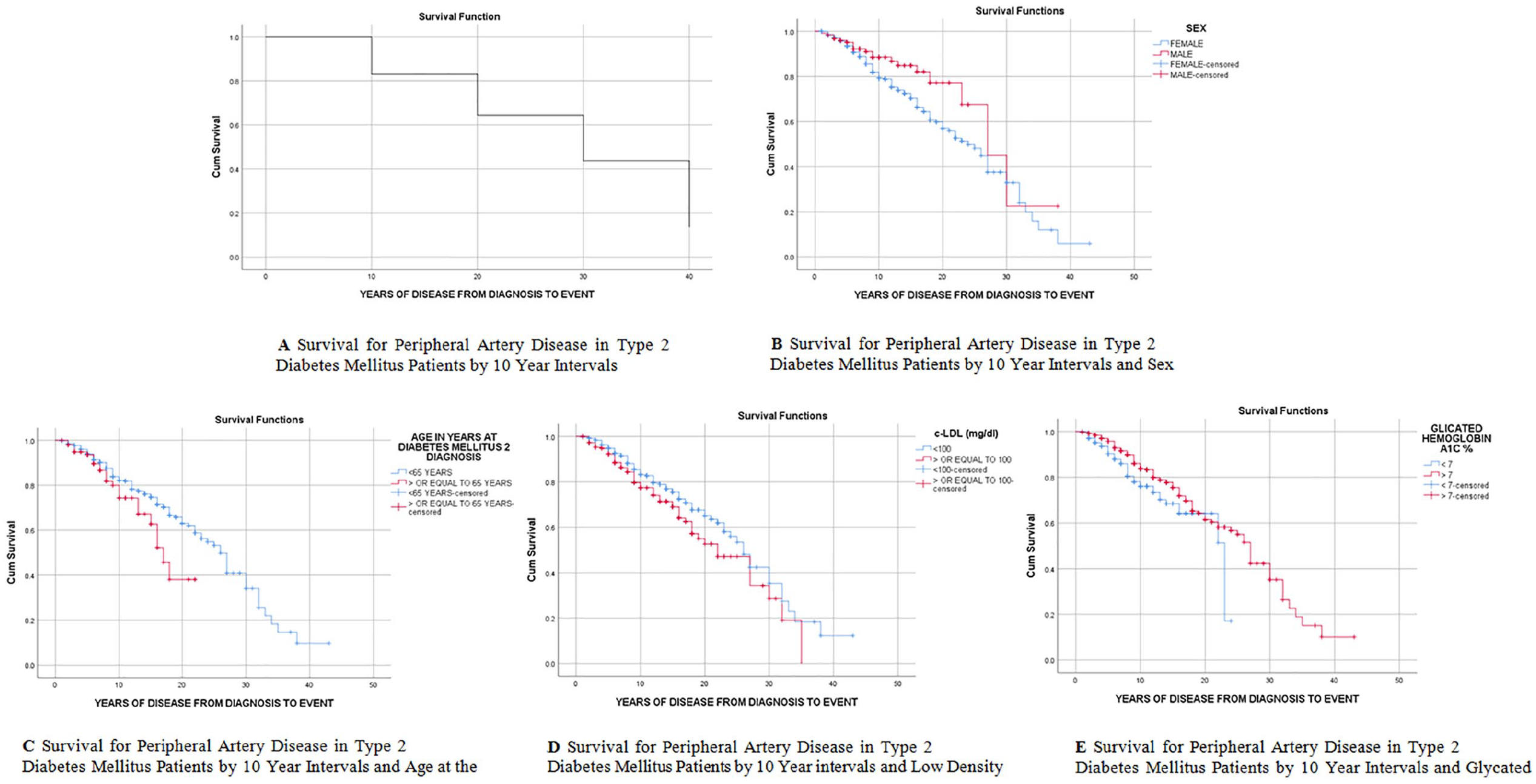
Survival curves for peripheral atherosclerosis by different variables.
Table 3 and Figure 2B-E show the mean number of years with the disease for developing PAD and the survival curves for different variables. The significant results were sex, age at diagnosis of DM, cLDL, and HbA1c. There was no association with the rest of the variables.
Survival Years for Peripheral Artery Disease According to Different Variables in Type 2 Diabetes Mellitus Patients.

Abbreviations: c-LDL: low density lipoprotein cholesterol; DM: type 2 diabetes mellitus; HbA1c: glycated hemoglobin.
Statistically significant when
The output model of logistic regression showed the variables related to PAD were: female sex (Expβ: 2.5, 95% CI: 1.51-4.38,
Discussion
The main findings in this work have shown that the prevalence of PAD for a Latin population with diabetes in primary level of care is of 13.98%. In a 10-year follow up 27.3% will develop PAD, with an incidence rate of 23.38 cases of PAD per 1000 persons-year. The survival time for PAD for 50% of the DM population will be 26.97 years of disease. Variables associated with a lower survival time for PAD where: female sex, age of diagnosis of DM >65 years, cLDL >100 mg/dL, and HbA1c <7%. Finally the regression model demonstrated relation of PAD with: female sex, treatment with insulin plus metformin, and GFR <60 mL/min/m2.
In absence of other LEAD and PAD prevalence studies in DM population in Ecuador, it is not possible to compare and be sure what this result represents in this population. However, the found LEAD prevalence (15%) doubles the one found in a DM population in China, but it is lower than the one found in Spain.8,20,21 The estimated PAD prevalence in the general population from 65 to 69 years is 9.91% for females and 6.74% for males, in middle income countries; these values are lower than the ones reported in this study.22-24 This may be explained because the DM population is at higher risk of PAD than general population.21,24,25 The DM prevalence in this research is higher, although comparable, than the ones found in United States, Spain or Colombia but lower than in other Latin American populations.20,26-29 Hence, we should consider the PAD risk variability even between Latino subgroups and genetic differences related to ABI results.30,31
A study on 48.607 male health professionals with DM in United States didn’t find a difference for PAD survival between more or less than 10 years of disease. However, they recognized a lower PAD survival in DM population versus the population without it. Furthermore, they reported the highest risk for PAD was in patients with more than 25 years of disease. 32 Similarly, our actual work found a median for PAD survival of 27 years, with the highest risk in the fourth decade of disease. In addition, the risk for developing PAD was correlated with females, GFR <60/mL/m2 and treatment with metformin plus insulin. Other authors support these results in Scotland and Thailand.33,34 According to other research, the risk also presents in the opposite direction, with the presence of PAD adding 3 times the risk of developing early diabetic nephropathy independent of albuminuria. 35
Our investigation shows a difference between the survival time for: sex, age at diagnosis of DM, cLDL, and HbA1c. 34 It stands out that patients with HbA1c less than 7% have a lower survival time for PAD than their counterparts. No studies were found supporting the lower survival for PAD in patients with good glycemic control. It is known that HbA1c levels relate with the presence of peripheral neuropathy and AC. The lower survival with good glycemic control may be explained by the higher incidence of complications in patients with bad glycemic control and the lower liability of ABI in the presence of complications. 36 Other researchers found that patients with HbA1c levels 7%, or higher, had a greater decline on ABI in a 10-year follow up; expecting a lower survival time for PAD in patients with bad glycemic control. 37 Our study found the opposite, but the mean difference between groups showed a higher HbA1c for PAD group. Therefore, it is not clear whether bad glycemic control diminishes survival time for PAD, although the previously mentioned evidence tends to support this fact. 37 There are few investigations trying to assess the influence of different variables in the probability of developing PAD. A multivariate regression model in a Spanish DM population found an association of PAD with age and the presence of retinopathy. 38 However, there was no evidence of association neither with diagnosis age nor with any other variable reported in our model. The absence of HbA1c in the binary logistic regression model shows that HbA1c is independently related to PAD, but not when it is analyzed with other variables.
The actual guidelines and evidence for PAD management recommend lipid-lowering drugs in all patients with PAD. These drugs reduce atheromatous plaque formation (therefore the PAD and cardiovascular risk) and lower limb amputation in DM patients.2,6,39-41 In the study population, all individuals with dyslipidemia diagnosis are receiving lipid-lowering drugs, although they are in target values. This may explain why there was not relevant association between lipid profile and PAD. Even when smoking has been recognized as a risk factor for PAD, this factor was not determining for PAD development in the study’s population.32,34 However, we should bear in mind that smoking prevalence in Latino and Ecuadorian population (even Hispanic in North America) is lower than in other locations.42,43
The strengths of this study are the longitudinal design and that it comprehends a real clinical population of DM patients in the primary level of care. In addition, these patients receive free health services including medication and exams. We consider the data to be reliable given the continuity of the data registry by the same physician. Although it is not an objective of this study, the authors highlight that during the 10 years of follow up there were no amputations or revascularizations of lower limbs needed in the study population.
The study limitations include a lack of randomization and a volunteer bias. This has led to a higher population of females and higher risk of smoking in men. Other limitations include a finite population of DM with control of their comorbidities (HTN, hypothyroidism, and others) and a high proportion of Latinos. Due to these facts, we consider that the results may only be extrapolated to specialized centers for DM patients but no to the general population. An important limitation is the use of only 1 method for ABI detection that could be affected by the concomitant presence of complications which are not assessed in this investigation.
The results point out that the PAD risk is higher in females, longer time of disease, higher HbA1c, GFR <60 mL/min/m2 and treatment with metformin plus insulin. In patients with these characteristics the PAD detection should be emphasized. DM complications develop in older adults, as is shown by the highest risk of PAD in the fourth decade of disease. This highlights the importance of the proper management of older adults with DM complications. Although it seems in a Latino population smoking is not a determinant factor for PAD presence, more studies are needed to assure this fact. We have not found many studies that allow the comparison of PAD survival in populations with DM; more data is needed in this field.
Conclusion
In a DM population with strict metabolic control that assists to a diabetes specialized clinic in the primary level of care, half will present PAD in approximately 27 years of disease with the highest risk in the fourth decade of disease. The population’s PAD prevalence is comparable to that of high-income countries. In addition, sociodemographic and clinical factors that will facilitate the detection of patients with higher risk of PAD are female sex, age of diagnosis <65 years, longer time with the disease, higher HbA1c, GFR <60 mL/min/m2 and treatment with metformin plus insulin
Footnotes
Acknowledgements
The authors thank the staff and the patients of Chimbacalle Medical Centre for their valuable contributions and trust through all these years.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors whose names are listed immediately below certify they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliation, knowledge or beliefs) in the subject matter or materials discussed in this manuscript. Author names: Barrera-Guarderas Francisco, Carrasco-Tenezaca Felipe, and De la Torre-Cisneros Katherine.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
