Abstract
Keywords
Introduction
Chronic noncancer pain (CNCP) and the challenges in treating patients suffering from it are pervasive in primary care. At the peak of opioid prescriptions in 2010, approximately 20% of patients would be prescribed opioid medications at visits for noncancer-related pain. 1 The amount of opioid prescriptions has since plateaued, likely related to growing awareness of the opioid crisis; this eventually led to the US Department of Health and Human Services declaring a nationwide public health emergency in 2017. 2 However, opioid medication for CNCP tripled from 1999 to 2015, and opioid prescriptions per capita increased 7.3% from 2007 to 2012, with opioid prescribing rates increasing more for family practice, general practice, and internal medicine compared with other specialties.2,3 The United States had the largest per-capita consumption of opioid medications in the world between 2013 and 2016. 4 This prolific prescribing has contributed to illicit substance use, but prescription opioids continue to constitute a significant portion of drug overdose deaths with 17 087 deaths in 2016 alone compared with 15 469 due to heroin. 2
Despite this widespread use of opioid medications for CNCP, there is little evidence supporting their efficacy in this role. Systematic reviews have shown no or only modest improvement in functional status and long-term pain control with chronic opioid therapy.5-8 Furthermore, there is growing, albeit weak, evidence that tapering opioid medications is not associated with increased pain and may contribute to increased quality of life.9-11 Currently, there is a lack of high-quality randomized controlled trial evidence regarding the effects of slow tapering of opioid medications when prescribed for CNCP. Despite this, high numbers of practices have required patients to decrease medication doses or discontinue opioid use altogether. From 2009 to 2017, there has been an annual decrease of 9.5% in prescriptions for high dose (>90 MME [morphine milligram equivalent]) opioid medications. 2 The 2016 CDC guideline on prescription of opioid medications for chronic pain suggests a gradual 10% MME per week taper of opioid medications with adjustment based on response. This same guideline notes that this recommendation is based on expert opinion and the current data comparing effectiveness of tapering protocols consists of 2 nonrandomized trials.12,13 Prior to developing a trial to examine the effects of tapering chronic opioid therapy (COT), information regarding its feasibility and patient engagement is needed. The incorporation of patients and stakeholders into initial study design has been identified as a key component in decreasing research waste through incomplete studies or studies that do not address relevant issues for target populations.14,15 Methods to minimize this research inefficiency and increase the likelihood successful study performance have been described in other disease states.15,16 In order to assess the willingness of patients to participate in a randomized and blinded study and improve on the proposed design, our team developed and distributed a survey to a population that included the target population of sufferers of CNCP currently using COT.
Methods
In order to gauge the applicability of the study and interest in participation, the study group solicited patient responses via a self-administered survey. These surveys were distributed in paper form to patients on COT when picking up prescriptions from their primary care provider’s office as well as via an online patient engagement platform managed by the study group’s sponsoring organization (Mayo Clinic Connect). Distribution of the survey took place from June 28, 2019 through August 10, 2019. The online platform consisted of more than 70 000 registered users who self-identify in patient categories with the understanding that they will be able to provide anonymous feedback on topics related to each group. This population included opioid utilizers and nonutilizers that self-identify as belonging to the chronic pain group. This group consisted of individuals suffering from chronic pain as well as family members of patients with chronic pain and other interested individuals. The survey was preceded by a brief description in layman’s terms of the proposed future study with a clear explanation that the survey did not require enrollment in the study and would not be used to identify potential study participants (see the appendix). Therefore, no patient data or identifying information were gathered through this survey. The survey allowed both binary check box and freeform answers for participants to provide feedback that could be incorporated into any future study design. The format of the survey first stratified patients by use of at least 1 month of opioid medication in the past 6 months. They were then asked if they would consider participating in a study such as that described in the introduction and directed to preset binary checkbox and freeform responses based on this answer. Respondents could choose as many or as few options as they desired. Data from electronic surveys was automatically formatted into a spreadsheet for analysis and paper survey results were then manually compiled with the electronic platform data. χ2 analysis was employed to assess for differences between recent opioid utilizers versus nonutilizers using a threshold of P < .05 for statistical significance.
Thematic analysis was performed manually following initial automatic stratification by respondents willing to participate and those expressing unwillingness to participate. A reviewer categorized responses by open coding method utilizing grounded theory to identify specific recurring phrases provided in narrative responses that were relevant to the research question regarding motivation or hesitancy to participate in an opioid tapering study. Given the small data set, the most commonly identified codes were grouped as common themes which were then reviewed by each author to ensure adequate representation of each identified code in narrative responses.
This study was reviewed by the Mayo Clinic Institutional Review Board and determined to constitute minimal risk research under section 45 CFR 46.109, item 1.
Results
The preset responses provided comparable data on common concerns related to opioid therapy and research. A total of 72.1% of all respondents answered positively regarding their willingness to participate in a study such as that proposed. The subset of this group that had recently utilized opioid therapy was higher at 84.0% as shown in Figure 1. Between the 2 groups (those who had utilized chronic opioids in the past 6 months and those who had not), there were similar reasons for participating in the study, including concerns regarding opioid dependence (36.7% and 43.1%, respectively), adding to society’s knowledge of opioid medications (68.4% and 65.5%, respectively), and determining if the respondent would personally receive benefit from opioid medications (39.2% and 36.2%, respectively). The primary difference between groups was that respondents recently on COT felt it was important to be able to withdraw from the study and return to usual care at any time (41.8% for opioid utilizers and 15.5% for nonutilizers, P < .05). Of those that expressed unwillingness to participate, there were no significant between-group differences in reasons given. The most common reason in each group was that respondents did not feel they had enough information to feel comfortable participating. The financial compensation did not appear to be a clear factor in the decision to participate among respondents in either group. There was some small attrition due to respondents that did not select any of the preset responses or provide a freeform comment as represented in Figure 2.

Percentage of respondents indicating they would participate in the described study grouped by whether they had used opioid medications for a 1-month period or more over the past 6 months.

Survey responses compiled from Mayo Clinic Connect Platform and printed surveys. Patient categories were determined by an initial question regarding opioid use for at least 1 month in the past 6 months followed by willingness to participate in the study and selected reasoning options. Numbers in parentheses represent the number of respondents in each group.
The narrative responses from each group did not differ significantly between recent opioid utilizers and nonutilizers. Several themes emerged through code evaluation and these are provided in Figure 3. The first had respondents that felt COT was the only answer to their or their loved ones’ chronic pain and that this study would demonstrate the need to continue these medications long-term. Another focused on anecdotal stories of side effects and dependence with decreasing effectiveness of opioids for pain control. The narrative responses contained many related stories of being suddenly cutoff of opioids by providers that would no longer prescribe them without guidance on what to expect for withdrawal symptoms, safety of discontinuing the medications, or alternative pain management strategies. Related to the recent drive to decrease opioid doses, a group of respondents described being judged or negatively labeled by providers due to their opioid use. In addition to identified themes driving motivation or unwillingness to participate, when respondents were prompted to provide possible ways to improve the study and comment on study design, the majority of responses reflected that the study should include alternative pain management options aimed at mitigating the reasons for opioid use. Underlying this was a prevailing sentiment that respondents desired a better alternative to opioid medications to help manage pain. This was accompanied by several responses with the assumption that pain will worsen as opioid medications are decreased and that no other substitute would be given to treat participants’ pain.
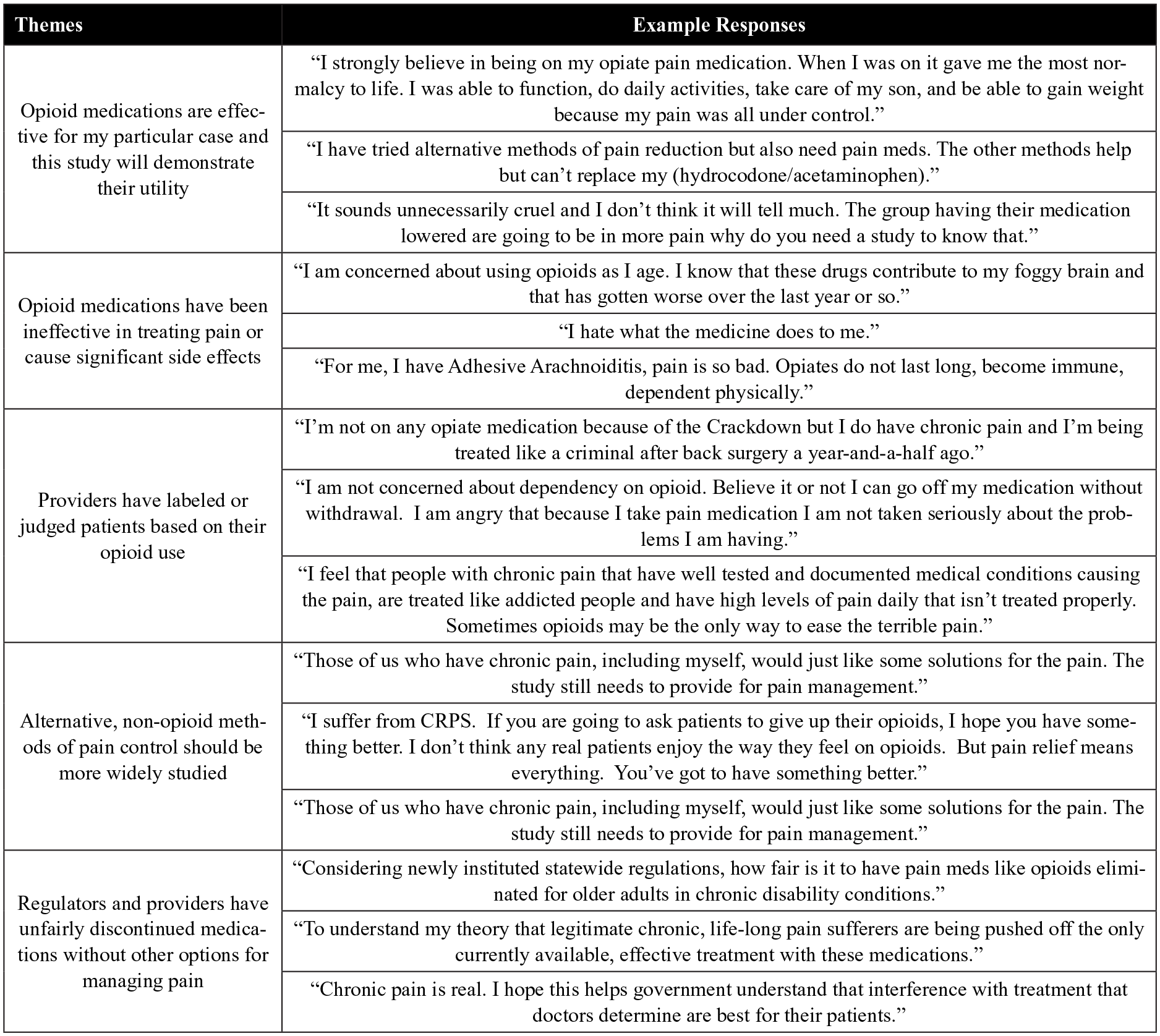
Quotations from survey responses representing each of the themes identified by thematic analysis.
Discussion
An evidence gap exists in the understanding of the effects of tapering opioid medications. The survey results indicate that this lack of knowledge is seen by both prescribers and patients alike. This was reflected by the majority of respondents indicating they would be willing to participate in a study such as that proposed to add to society’s knowledge of opioid medications. The percentage of participants that would be interested in participating in such a study was reassuring for the feasibility of recruitment and retention of patients through study completion. While the opioid-utilizing group had a slightly higher likelihood of participation, the difference between groups was largely explained by the narrative comments that nonopioid users recognized they could not participate in the study. Of note for any future research group, the ability to withdraw from the study and return to usual care at any time was very important to recent utilizers of opioid medications. This was not an unexpected difference for the investigators and underlined the importance of allowing autonomy for each participant in any future trial. This also highlighted the need for close collaboration with the primary care providers at study sites to continue high-quality care, alternative forms of pain management, and follow-up for the duration of the study. Regarding respondents that were unwilling to participate in the proposed study, the primary concern was that there was not enough information provided. Given the brief description of the study, this was an expected finding and emphasized the importance of a clear and comprehensive consent process as well as the meetings with a research coordinator to ensure participants can make fully informed decisions consistent with high-quality, ethical research practices.
The narrative comments revealed prevailing underlying sentiments that many providers encounter in practice. Many patients and their loved ones have had poor outcomes related to chronic opioid therapy, escalating doses, or sudden discontinuation. These stories reflect the current lack of evidence and study-based guidance for the use of chronic opioid therapy in providers’ current work environments. With the growing body of evidence for alternative forms of management for chronic pain, the respondents’ desire for incorporation of opioid-sparing pain treatment aligns well with current guideline-based models of care and the importance of trials targeted at nonopioid treatments of CNCP. For a future opioid tapering study, results of our survey emphasize the need to maintain ongoing alternative pain management treatment with the participants’ primary care provider, counseling on side effects and withdrawal, contingencies for increased pain or withdrawal symptoms, and multidisciplinary treatment team involvement throughout any study targeted at examining opioid medications and chronic pain treatment. This allows for recognition of the need for research into alternative modalities for pain, but simultaneously emphasizes that the proposed study is targeted at the effects of slow tapering of opioids. To avoid confounders, standard of care with alternative forms of pain treatment should continue for the duration of any future study. This study was limited due to the short duration of administration of the survey, the use of a population of convenience, and the limited data on respondents. Had demographic data been collected on participants, additional variables may have been identified for a more in depth understanding of motivations regarding decisions to participate in a tapering trial. Currently, these survey data add to the understanding of what topics are most important to patients on COT as well as their concerns with the current practice changes surrounding the prescription of opioids. This creates opportunities for further study including a possible dedicated focus group to gather further qualitative data, an expanded Likert scale quantitative survey reaching a larger population, incorporation of additional patient demographic data to identify characteristics that may influence perceptions of opioid prescribing practices, as well as development of a high-quality trial aimed at determining the effects of slow tapering of opioid medications on quality of life, mood symptoms, pain, and function.
Conclusion
Given the information gathered with this survey, there appears to be patient support for future research into the effects of tapering opioid medications. Any future study could expect adequate community and patient participation while addressing the patient concerns outlined in the narrative survey results.
Footnotes
Appendix
Dear Patient,
Family Medicine physicians at Mayo Clinic Rochester and the Mayo Clinic Health System propose to conduct a study with people who are currently on chronic opioid therapy for non-cancer pain. (Opioids are strong pain medications such as OxyContin, Vicodin, morphine, Tramadol, etc) Before we recruit participants to the study, we want to know what you think of our study proposal. We invite you to take part in this anonymous survey to help us define and develop the study to make it meaningful for people managing pain.
Acknowledgements
We wish to thank Julie Maxson, CCRP, for her endless support and facilitation for the duration of the study as well as nursing staff at all collection locations for their efforts in conducting the study. We also wish to thank Colleen Young, Community Director for Mayo Clinic Connect and Marquita Davis, Mayo Office of Patient Experience for their assistance in the development and distribution of the surveys on Mayo Clinic Connect. Finally, a special thanks to all the survey participants who took the time to complete this survey. Without their participation, this study would not have been possible.
Author Contributions
All the authors participated in the study concept and design, analysis and interpretation of data, drafting and revising the paper, and have seen and approved the final version of the manuscript. JJ, BL, and TW conceived of the study concept and participated in design; provided administrative, technical, and material support; had full oversight of the study conduct during data collection; had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; participated in review and interpretation of study results; and also drafted the manuscript and participated in critical revision of the manuscript for important intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by our institution.
Ethical Approval
In accordance with the Declaration of Helsinki, this study was reviewed and determined to be exempt (ID 19-004703) by the Mayo Clinic Institutional Review Board (IRB). The IRB-approved passive consent was obtained for all study participants prior to study participation. This study was reviewed by expedited process and determined to be minimal risk research for Institutional Review Board Approval under 45 CFR 46.109f, item 1. The oral contact cover letter (which served as the passive consent) and the survey were both reviewed by the IRB and noted. Passive consent was obtained from all study participants prior to study initiation.
