Abstract
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD), a preventable chronic health condition, is the fourth leading cause of death globally 1 and costs the US health care system more than $50 billion annually. 2 Proper diagnosis, with spirometry testing, is essential to improving the morbidity and mortality associated with this preventable disease.1,3 According to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines, spirometry testing is recommended in patients older than 40 years who present with dyspnea, chronic cough, chronic sputum production, history of exposure to risk factors, or family history of COPD. 1
Patients with undiagnosed COPD are most likely to encounter the health care system in the primary care setting. In one study, among patients who were 40 years or older, had a smoking history of 20+ pack-years, and visited their primary care provider (PCP) for any reason, COPD was identified via spirometry testing in 1 of every 5 adults (20.7%). 4 Multiple studies have demonstrated that the majority of patients with COPD (67% to 80%) are undiagnosed.4-6 Results from these studies show that there is a high prevalence of COPD in high-risk patients and a high rate of COPD underdiagnosis. Underdiagnosis increases the risk of hospitalizations, pneumonia, disease progression, and mortality.4-6
Alternatively, without spirometry patients may be mis/overdiagnosed, leading to unwarranted medication burden, resultant costs to patients and the health care system, and increased mortality risk.1,4,6,7 Accurate COPD diagnosis can lead to improved evidenced-based medication regimens, improvement in lung function, better exercise capacity, reduced exacerbations, and enhanced quality of life.1,6,7
Recognizing the high prevalence of COPD and misdiagnosis, pharmacists at PrimaryOne Health (P1H) began offering spirometry testing in January 2016 at 2 clinic sites. Providers are able to refer patients in need of spirometry for in-house testing. Previous research has shown that pharmacists are capable of providing quality spirometry testing that meets published standards. 8 Previous research conducted at P1H showed having a spirometry service within the primary care setting helps to increase the overall number of referrals ordered and completed. 9 However, there is no data to determine if high-risk patients are being appropriately screened. This study aims to determine the effect of proactive pharmacist identification of high-risk patients eligible for diagnostic spirometry screening within a federally qualified health center (FQHC) on the percentage of appropriate spirometry referrals ordered and completed.
Methods
This is a prospective study performed at P1H from August 29, 2017 to May 30, 2018. This study was in collaboration with The Ohio State University College of Pharmacy and was approved by its institutional review board (2017H0217).
Setting
P1H is an FQHC and recognized as a level 3 patient-centered medical home by the National Committee for Quality Assurance since 2012. P1H serves more than 48,000 unique individuals in culturally and socioeconomically diverse areas throughout central Ohio and offers comprehensive, interprofessional primary care services. The 2 clinic sites chosen for this study were the sites that offered spirometry services at the time.
Study Population
PCPs who provided verbal consent were included if they practiced at either the intervention or control sites on 2 specific weekdays during the prospective intervention period (August 29, 2017 to November 21, 2017). Study days were chosen based on the study pharmacist’s availability to be present at the intervention clinic for the proactive screening recommendations. At both sites, patients were considered high-risk and eligible for spirometry screening if they were (1) active tobacco smokers, (2) aged 40 years or older, and (3) present for their scheduled office visit with the PCP. On chart review, patients were excluded if they (1) possessed any relative or absolute contraindications for pulmonary function testing (PFT) in the primary care setting10-17 (see the appendix), (2) had a diagnosis of COPD in the chart with confirmatory PFT results, or (3) had spirometry testing in the past 3 years.
Intervention
At the intervention site, a list of patients who met the first 2 inclusion criteria and were scheduled for a PCP appointment on a designated intervention day was generated from the electronic medical record (EMR) the day prior. Each patient’s chart was reviewed for exclusion criteria and to confirm inclusion criteria. The pharmacist recommended a spirometry screening to the PCP prior to identified patients’ appointments. Providers were given the choice of either an individual patient message within the EMR or a list of all eligible patients handed to them at the beginning of the day.
The control site was representative of usual care so there was no proactive identification or screening recommendations provided. For data collection purposes, the same patient list was generated retrospectively for those patients who presented to their PCP appointments at the control site.
Usual care provided at the patient’s visit with the pharmacist includes the spirometry test, a comprehensive evaluation, diagnosis in consultation with a provider, and medication recommendations and education on inhaler devices from the pharmacist. Patients also have the opportunity to be seen for smoking cessation during or after the visit, if interested. Pharmacists schedule follow-up appointments with the patient to assess improvement in COPD symptoms based on medication changes, if appropriate.
Data Collection
At both sites, a chart review was completed 6 months after the intervention dates to determine if referrals were ordered and tests were completed for patients who qualified. Data collected included patient demographics, COPD diagnosis prior to appointment, referring provider type (physician vs nurse practitioner), referral status (ordered vs not ordered), location of referral ordered (internal vs external), spirometry screening test status (completed internally, externally, or not completed within 6 months), and spirometry result interpretation (evidence of normal function, COPD, or a different lung condition). If a patient presented for spirometry at the intervention site and the results indicated a diagnosis of COPD, the following data were gathered if the patient provided written consent: the patient’s symptoms of COPD—via the COPD Assessment Test (CAT) questionnaire 18 —and the history of any respiratory-related emergency department and hospital visits within the 12 months prior. Exacerbation information was self-reported during the visit and the study investigator retrospectively reviewed patient’s chart for evidence.
Statistical Analysis
Patient characteristics were analyzed to compare the intervention and control sites using Student’s t tests or Fisher’s exact tests as appropriate. Fisher’s exact test was used to analyze the difference in number of referrals ordered and tests completed between the intervention and control sites.
Results
Across the intervention and control sites, 251 patients were identified on the reports (n = 171 vs n = 80, respectively), with 61 patients being excluded (n = 46 vs n = 15, respectively), see Figure 1. The number of patients eligible for spirometry screening at the intervention and control sites was 190 (n = 125 vs n = 65, respectively). Baseline characteristics were comparable between intervention and control sites except for differences in language and race (Table 1). All patients were active tobacco smokers at both the corresponding PCP and spirometry appointments, except 2 patients at the intervention site who quit within the past 1 to 2 months prior to spirometry appointment (no statistical difference).

Patient algorithm.
Patient Demographics.
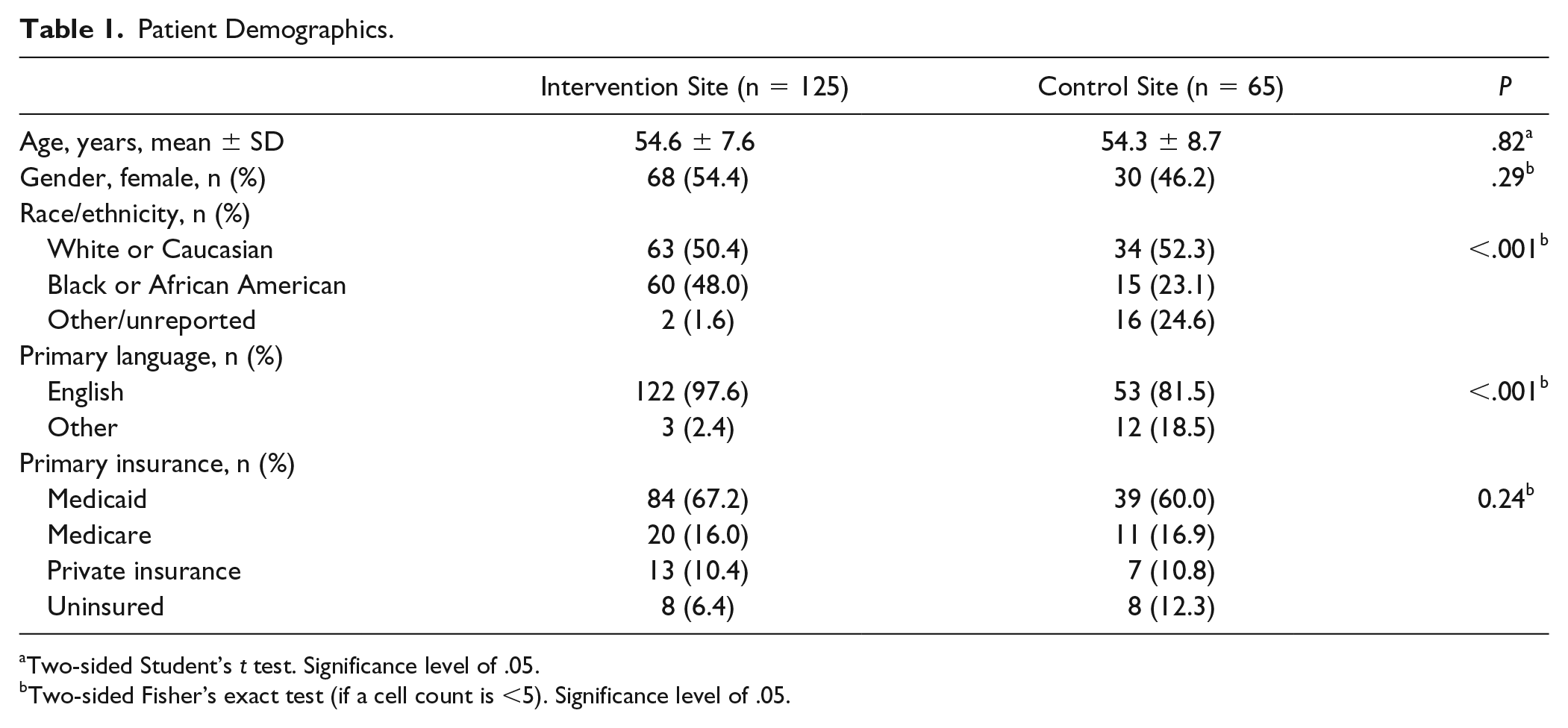
Two-sided Student’s t test. Significance level of .05.
Two-sided Fisher’s exact test (if a cell count is <5). Significance level of .05.
Among eligible patients, the rate of referrals ordered was significantly higher at the intervention site (47.2% vs 7.7%, P < .001), Figure 2. Among patients who qualified, completion of spirometry testing was significantly higher in the intervention site compared with control (23.2% vs 3.1%, P < .001), Table 2.

Spirometry referral rate in high-risk patients with and without pharmacist intervention.
Spirometry Screening Test Completion Rate.
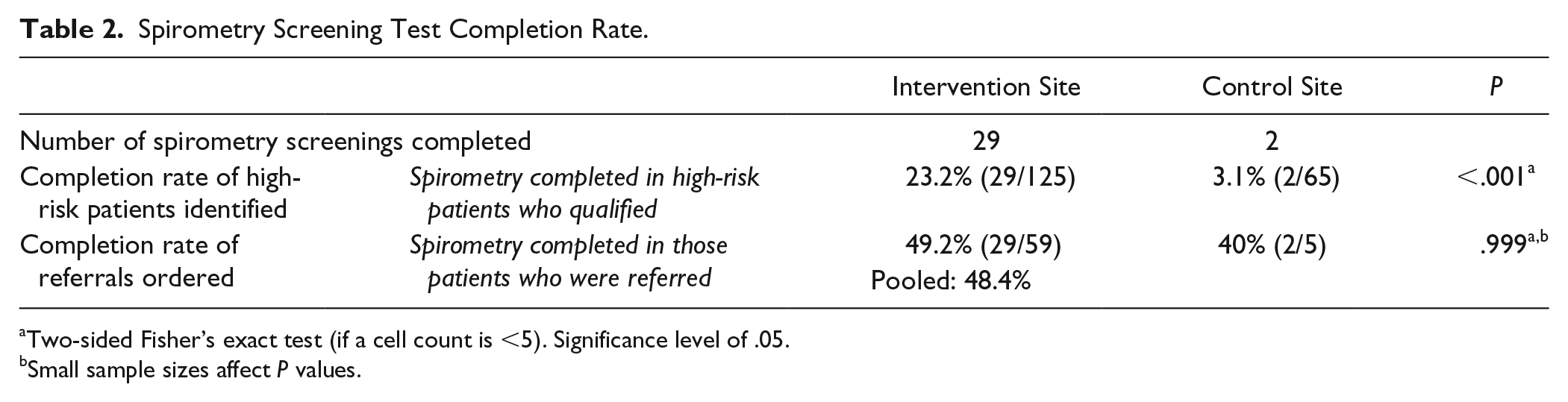
Two-sided Fisher’s exact test (if a cell count is <5). Significance level of .05.
Small sample sizes affect P values.
Of all the patients who completed spirometry testing across both sites (n = 31), the impact of the spirometry findings on COPD diagnosis can be seen in Figure 3. Most notably, 29% (9 cases) resulted in a correction of an inappropriate/misdiagnosis of COPD and 6% (2) resulted in a new diagnosis of COPD.

Impact of the spirometry findings on chronic obstructive pulmonary disease (COPD) diagnosis (n = 31).
Of the 4 patients at the intervention site who had spirometry results diagnostic of COPD, their CAT scores were 23, 23, 19, and 11. For the 12-month time period prior to the visit day, 50% (2/4) self-reported a respiratory-related emergency department visit and 25% (1/4) self-reported a respiratory-related hospitalization.
Discussion
At the intervention site there was a significantly higher percentage of appropriate referrals ordered by PCPs for high-risk patients (47.2% vs 7.7%) and a significantly higher percentage of appropriate spirometry screening tests completed by eligible high-risk patients (23.2% vs 3.1%). These increased rates of spirometry completion are of clinical significance as it leads to proper diagnosis of COPD, which is essential to improving morbidity and mortality.1,6,7
The results demonstrate the importance of spirometry for proper diagnosis of COPD, as almost 30% of the completed tests corrected an inappropriate/misdiagnosis of COPD. Correcting the diagnosis is expected to reduce unnecessary inhaler burden and decrease costs to the patient and health care system. About 6% of cases resulted in a new COPD diagnosis and another 10% of cases provided gold-standard diagnostic confirmation and staging of symptomatically diagnosed COPD. All of these patients had CAT scores >10, which means they met criteria for at least GOLD treatment group B, requiring a maintenance bronchodilator inhaler. This finding highlights that utilizing a pharmacist as the provider for this service creates efficiency; by the end of the spirometry visit, pharmacists had evidenced-based medication recommendations ready for the PCP, which streamlined the process for getting patients on the appropriate regimens.
When pharmacists provide the spirometry test, the patient’s “rest” time between the maneuver trials can be optimized for discussions on smoking cessation, symptom assessments, medication use/adherence/tolerance, COPD care plans, vaccine recommendations, inhaler education, and more.19,20 In our study, all patients received brief smoking cessation counseling from the pharmacist during their spirometry visit; this included an offer for future follow-up and/or enrollment into a free smoking cessation program. The goal was to proactively prevent disease development in those without COPD (42% of our cases) and prevent further disease progression in those with COPD. Pharmacists are highly trained and prepared to step into the role to ask the right clinical questions and clinically investigate respiratory-related history based on emerging PFT trial results throughout the test. The pharmacists’ ability to optimize the time with the patient to accomplish more than just the traditional lung function test is indispensable and creates efficiencies in the care process.
There are several factors that may explain why the recommendation acceptance rate in the intervention clinic was 47.2%. First, providers may not have had time to initiate a discussion about spirometry and COPD risk with the patient. Second, providers may have determined that the test was not necessary after collecting more information from the patient. Third, each provider was able to choose their preference on how they received recommendations each morning. Two providers wanted a hardcopy list, which may have made it easier for the paper to be set aside or lost in the shuffle. The other method was individual patient messages within the EMR, but these messages were not marked with “high priority” and may not have been seen by the provider until after the patient appointment.
The rate of test completion among patients with a referral ordered showed no significant difference (49.2% vs 40%) between intervention and control sites. We would not expect those rates to differ as both sites offered pulmonary function testing prior to this study. It is worthwhile to note that the overall completion rate when pooling both sites together was 48.4%, which is lower than expected when compared with a previous study in the same clinics that demonstrated a 70.0% completion rate. 9 The likely explanation for the difference in completion rates is that the patients in the previous study were referred to spirometry via a trigger such as a patient symptom complaint or PCP’s detection of wheezing on exam; whereas patients in this study were recommended for screening to the PCP irrespective of presence of symptoms as long as they met high-risk criteria. Our study patients may have been less motivated to seek care if they did not have active breathing concerns, were resistant to smoking cessation, or felt a PFT was unnecessary to complete. Our study did not specifically explore these reasons or follow-up with patients who did not complete spirometry, but this is certainly an opportunity for future exploration.
One limitation of this study is that we did not track the reasons why referrals were not ordered by PCPs for patients who were identified on the report. This could be affected by the lack of a standardized process for how providers received the list of patients each morning, as discussed earlier. Second, patient populations between the 2 sites differed in terms of race/ethnicity and language; however, we do not feel this had any clinically significant impact on our results. Third, there are multiple ways staff can document a patient’s tobacco use within the EMR chart, and to the best of our knowledge, the reporting software pulled the majority (if not all) the items. However, it is possible that the software could have failed to catch some patients on the reports. Fourth, the intervention site providers are different than the control site providers, and differences in training and experience could affect care habits. Lastly, 13% of spirometry cases had no interpretable information available due to poor quality results/technique (or no reproducible maneuvers). This is consistent with existing literature that reports rates of 10% to 21% for patients unable to perform spirometry of good quality.21-24
An important take-away from this study is the actual time invested each day to proactively identify patients prior to the recommendations took longer than anticipated. If a similar process to our study’s procedures is adopted by other primary care settings, we recommend a more simplified workflow via efficient reporting software or within the EMR functionality that is more accurate in identifying patient eligibility criteria and would minimize the preparation time required to integrate this approach in a practical manner.
Because of the success of pharmacist involvement in spirometry at our institution, collaborative practice (consult) agreements between pharmacists and physicians were implemented for tobacco dependence and COPD disease state management postspirometric diagnosis. Additionally, P1H expanded spirometry to four additional clinic sites to expand patient access to this important service.
Conclusion
Pharmacists proactively identifying high-risk patients that qualify for diagnostic spirometry screenings within a FQHC significantly increases the rate of appropriate referrals that PCPs order and the rate of appropriate spirometry tests completed by high-risk patients. Early diagnosis is crucial in a population of high-risk adult smokers and spirometry results can open the door to a smoking cessation discussion in those with and without COPD. Pharmacists in the primary care setting can aid the PCP in the management of COPD through significantly increased spirometry referrals and therapeutic clinical assessments and recommendations. They also can provide face-to-face motivational counseling for smoking cessation, inhaler education, and vaccine recommendations. Proactive screening for and subsequent spirometry testing of high-risk patient populations represents an emerging role for pharmacists in primary care and could be adopted in other primary care settings.
Footnotes
Appendix
Exclusion Criteria for Diagnostic Spirometry Screening. a

| Active | • Pregnancy • Hemoptysis/hematemesis/oral bleeding (or open sores) • Unstable angina • Physical limitations (eg, broken jaw) |
• Inability to follow directions (eg, cognitive impairment/ learning disability) • Management by a pulmonary specialist |
| Active and/or history of | • Aneurysm (aortic—thoracic/cerebral/abdominal) • Stroke/CVA/TIA “mini stroke” (ischemic or hemorrhagic) |
• Pneumothorax • Pulmonary embolism • Throat or oral cancer |
| Recent (within past 1 month) | • Myocardial infarction • Concussion |
|
| Recent surgery (within past 6 weeks) | • Thoracic/abdominal • Brain/neuro • Cardiac |
• Eye/ophthalmic • Sinus/middle ear |
| Infection | Active and/or history of within past 2 weeks: • Lower respiratory tract infection • Middle ear infection |
Active: • Tuberculosis (TB) • Hepatitis (except A) • HIV/AIDS |
Abbreviations: CVA, cerebrovascular accident; TIA, transient ischemic attack.
Acknowledgements
Authors would like to thank Dr Stuart Beatty and Dr Andrew Faiella for their help in reviewing the manuscript and Junan Li for providing biostatistical support.
Authors’ Note
At the time of this project, Dr Whitner and Dr Mueller were PGY2 Pharmacy Residents at The Ohio State University College of Pharmacy and PrimaryOne Health.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JBW and LAM declare that they have no conflicts of interest. ASV reports receiving honoraria from Theravance Pharmaceuticals for participation on an advisory board outside the scope of the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
