Abstract
Keywords
Introduction
In the United States, over 10 million adults have osteoporosis and over 43 million are at significant risk due to low bone mineral density. 1 Osteoporotic fractures contribute to significant morbidity and mortality and with the aging population, it is estimated to carry an annual economic burden of over $25 billion by 2025. 2 Medication adherence, also termed compliance, is a significant issue for the health care system and a growing area of research. 3 It is well established that adherence to therapies for the prevention and treatment of osteoporosis is poor, especially for oral bisphosphonates, 4 the first-line treatment for osteoporosis.5,6. Adherence rates less than 50% in the first year are reported by multiple studies.4,7 In postmenopausal females, low adherence is associated with increased fracture risk.8,9
The Veterans Health Administration (VA) operates the largest integrated health care system in the United States. The VA pharmacy and clinical providers operate in a shared electronic medical record (EMR) allowing an opportunity to promote medication adherence. For example, a prescription fill is generated automatically with the majority delivered via mail when a provider orders a medication during a patient visit. In evaluating adherence to statin therapy in the VA system, adherence in the VA was better than reports from existing metanalyses for non-VA populations. 10 Medication possession ratio (MPR), an established method to measure adherence, uses prescription fills to determine the portion of a treatment interval that a patient has a medication. Using MPR, a study in 200 male Veterans found recommended adherence (≥80%) to alendronate for 1 year and 2 years to be 59% and 54%, respectively, 11 which is modestly better than reports in non-VA population. This study did not report on clinical outcomes related to adherence.
In a cohort of Veterans receiving care at the VA St Louis Health Care System (VASTLHCS), we aimed to determine several endpoints: (a) adherence to once weekly oral alendronate for prevention and treatment of osteoporosis with measurements of MPR and persistence, 12 (b) predictors of adherence, and (c) the effect of adherence on clinical outcome measures, including change in bone mineral density (BMD) and incident fracture.
Methods
Patients
The study was approved by the institutional review board of the VASTLHCS, Saint Louis, Missouri. Using administrative data from the US Department of Veterans Affairs, we identified patients who had received an incident (new fill status and database demonstrated no prior prescription in the 1 year preceding) oral bisphosphonate prescription from October 1, 2002 to September 30, 2013 through a clinical provider at the VASTLHCS and had no history of other therapies for osteoporosis (prescription of zoledronic acid, pamidronate, teriparatide, or denosumab). The date of first bisphosphonate (index prescription) was denoted as T0 (n = 2447; (Supplementary Figure S1 available online). Additionally, patients were selected on being male aged ≥50 years or female aged ≥45 years but <90 years and having at least 2 years of follow-up from T0. Subjects were excluded if they had a diagnosis of Paget’s disease (n = 33), osteonecrosis of the jaw (n = 0), and if oral bisphosphonate was detailed in EMR as an allergy or adverse event (n = 28). We included only weekly alendronate (excluded risedronate) with index script for at least 4 weeks supporting the prescriber planned for chronic therapy (n = 987). Last, inclusion required that in the year preceding the index prescription there was an identifiable reason for chronic osteoporosis therapy (index BMD T-score ≤−1 at total hip or lumbar spine, diagnosis of prostate cancer, evidence of a fracture, diagnosis of osteoporosis or osteopenia) yielding a final analytic cohort of 913 patients. Weekly oral alendronate was a formulary medication during this time period with monthly patient cost ~$5 to $11 per month.
Data Sources
We used Department of VA databases, including inpatient and outpatient medical SAS data sets (that include utilization data related to all encounters within the VA system) to ascertain detailed patient demographic characteristics and comorbidity information.13-17 The VA Pharmacy Benefits Management (PBM) dataset provided information on all prescriptions. The VA Vital Status file provided demographic characteristics and death.13-17 Dual-energy x-ray absorptiometry (DXA) scan BMD measurements and T-scores for lumbar spine and total proximal femur (total hip) as reported by the interpreting radiologist were manually extracted from medical records. T-scores greater than 2.5 at either site with corresponding BMD and subsequent site specific data were excluded. 18 This occurred only in the lumbar spine in approximately 20 subjects, the majority of whom had excessive degenerative artifacts. Subjects with BMD percent change between 2 consecutive scans >15% were reviewed for technical or artifact-related issues that would preclude accurate interpretation. Five subjects had BMD measurements excluded due to excessive interval change attributed to poor positioning, vertebral fractures, surgery, and excessive degenerative artifacts.
Measures of Adherence
Adherence was assessed through the MPR and persistence. As an initial medication dispense occurs with the provider’s prescription in the VA EMR, this study investigated secondary adherence. Calculation of MPR was days of supply (number of pills multiplied by 7) divided by the observation period. This was then multiplied by 100 to obtain percent 12 ; maximum allowed MPR was 100%. MPR was assessed as binary, where it was categorized as <80% or ≥80% with MPR <80% as reference due to established outcomes. 19 Persistence was defined as refills without a permissible gap of 60 days. This gap length is consistent with prior research on persistence for osteoporosis pharmacotherapy. 20 One- and 2-year MPR and persistence were calculated over 1- and cumulative 2-year periods. MPR used in the measure of association with BMD change per year was calculated from time of index prescription to time of last follow-up DXA scan within 3 years of the index prescription.
Outcomes
Change in BMD was calculated as percent change per year and was completed for both the total hip and the lumbar spine (LS). Date of the first DXA scan was set to the index prescription date. The index DXA scan was required to be obtained between 12 months prior or less than 6 months after the index prescription. A follow-up scan was required to be at minimum 12 months from index prescription. The percent change in BMD was taken from the index DXA scan to the last available DXA scan within 3 years of the index prescription date. Occurrence of incident fracture was assessed as time until fracture from index prescription in subjects who had no history of fracture at time of T0.
Covariates
Covariates included age, gender, acid suppression prescription, oral glucocorticoid prescription, polypharmacy which were assessed at time of T0. Acid suppression therapy included a prescription for either a histamine type 2 receptor antagonist (H2 antagonist) or proton pump inhibitor (PPI). Oral glucocorticoid prescription was defined as prednisone ≥5 mg daily or equivalent for at least 3 months in the 6 months preceding T0. Additional covariates included diagnoses of rheumatoid arthritis (RA), baseline fracture, tobacco use, alcohol abuse. Last, Charlson-Deyo Comorbidity Index (Deyo score) 21 and lowest T-score from index DXA scan, were included as covariates. Race was categorized as black or white as only 3 patients were another race. Comorbidities were assigned based on relevant ICD-9-CM (International Classification of Diseases, Ninth Revision, Clinical Modification) codes in the VA Medical SAS data sets.13-17). Lowest T-score was the value at either site (total hip or LS) from the index DXA scan. Polypharmacy was defined as 10 or more unique drug type prescriptions.
DXA Scan
BMD was measured by DXA on a single machine, Hologic Delphi-A sn#70532A, between January 2002 to January 2015. The scans were completed and interpreted by two ARRT (American Registry of Radiologic Technologists) credentialled technologists and 2 American board-certified nuclear medicine physicians, all 4 who were certified by the International Society of Clinical Densitometry (ISCD) and were consistent through the years of the study. The device was calibrated daily using a phantom of the LS and the coefficient of variation was 0.29%. Reports included BMD measurements (gm/cm2) at the femoral neck, total proximal femur, LS, and distal 1/3 forearm. This study focused on the measurements for total proximal femur (total hip) and LS.
Statistical Analysis
Means and standard deviations are presented for normally distributed continuous variables. Medians and interquartile ranges are presented for nonnormally distributed continuous variables and counts and percentages are presented for categorical variables. Logistic regression was used in the assessment of predictors for MPR and persistence; odds ratios (OR), a measure of the odds of an outcome compared to not having an outcome, and 95% confidence intervals (CI) are presented. An OR greater than 1 indicates an increased odds of the outcome in the exposed compared with the nonexposed. In the analyses of categorized MPR, MPR <80% was treated as the referent category. Covariates were chosen based on clinical relevance to medication adherence and osteoporosis outcomes; models were adjusted for gender, age, baseline fracture, lowest T-score, tobacco use, alcohol abuse, acid suppression prescription, glucocorticoid prescription, diagnosis RA, polypharmacy, Deyo score, and in males only diagnosis of prostate cancer. Linear regression was used in the analysis assessing the association of MPR with BMD change; the regression coefficients (β) and P values are presented. MPR was assessed in two forms, as a binary variable comparing MPR ≥80% versus MPR <80%, with the latter as the reference category, and as continuous variable, assessed for every 10% change. BMD models were adjusted for age, tobacco use, alcohol abuse, acid suppression prescription, glucocorticoid prescription, and diagnosis of RA as considered relevant to change in BMD. Because of limited sample size, separate male-only data were not included for this aim. We also investigated time until incident fracture in Veterans by adherence group amongst those without baseline fracture. Secondary prevention was not investigated due to the difficulties in clearly identifying subsequent events in database research. Kaplan-Meier curves were generated, and results of the log-rank test for difference in strata are presented. Cox proportional hazard models were built, and the proportional hazard assumption was checked via time-interaction terms and log negative log plots and was met. Models for year 1 MPR as a predictor of incident fracture were adjusted for age, tobacco use, alcohol abuse, acid suppression prescription, glucocorticoid prescription, and diagnosis of rheumatoid arthritis as considered relevant to risk of incident fracture. Hazard ratios (HRs) and 95% CI are presented. All analyses, unless otherwise indicated, were repeated in the subsample of male veterans to allow for additional adjustment by prostate cancer. In regression analyses, a 95% CI of an OR or HR that does not include unity was considered statistically significant. In all analyses a P value of .05 or less was considered statistically significant. Data are presented for the whole cohort and then the significant relationships for males only. All analyses were performed using SAS Enterprise Guide version 7.1.
Results
The analysis included 913 VASTLHCS patients with an identifiable reason for chronic oral bisphosphonate therapy at time of T0 (Supplementary Figure S1). For the entire cohort the mean MPRs were 68% and 59% for year 1 and cumulative 2 years, respectively. Table 1 details the baseline characteristics of the groups with 52% being low adherence (year 1 MPR <80%) and 48% being high adherence (year 1 MPR ≥80%). Included in the low adherence group are 125 patients who only received the index prescription (13.7%). The majority of the high adherence group maintained desired compliance in year 2 as the group’s cumulative MPR for the 2 years was 86%. Concordantly, 92% of the high MPR group had desired persistence (no gap in medication fill of 60 or more days) in year 1 but had a decline for the 2 years cumulatively to 62%. The cohort’s mean age was 68 years and 23% were female. The 2 groups had similar age, gender, and race distribution. Baseline fracture diagnosis occurred in 98 patients and was more frequent in the low-adherence group (61 vs 37, P = .042). An index DXA scan was available in 744 patients (low MPR n = 389, high MPR n = 355) and lowest T-scores were similar for both groups. A diagnosis of prostate cancer occurred in 21% of males and was more prevalent in the high adherence group (60 vs 86, P = .004). Tobacco use (115 vs 53, P < .001) and alcohol abuse (37 vs 14, P = .004) were more prevalent in the low adherence group. Diagnosis of RA, polypharmacy, and baseline acid suppression or glucocorticoid therapy were similar in both groups.
Baseline Characteristics for Low- and High-Adherence Groups (n = 913) Defined by Year 1 MPR. a

Abbreviation: MPR, medication possession ratio.
Details captured at time of index script or within 12 months prior.
Prostate cancer analyzed in males only, n = 706.
Further characterization between the groups for other diagnosis and baseline exposures that potentially affect bone health was completed and are included in the supplement (Supplementary Table S1). Numerous pharmacologic contributors to bone health were reviewed and the only significant difference was that the high-adherence group was prescribed statin therapy more often (185 vs 211, P = .003). The calcium and vitamin D supplementation details provided in the Supplementary Table S1 are for patients receiving prescription fills from the VASTLHCS and was similar for both groups.
Predictors of Adherence
Table 2 details predictors of year 1 MPR in 744 patients who had complete data, including a baseline DXA scan. While baseline fracture, tobacco use, and alcohol abuse were associated with lower odds of an MPR ≥80% in unadjusted models, only tobacco use was associated with lower odds [OR 0.45, 95% CI: (0.30, 0.67)] of MPR ≥80% in adjusted models. The relationship of tobacco use was similar in males only (n = 559) [0.44 (0.27, 0.70)], and a diagnosis of prostate cancer was associated with twice the odds of high adherence [2.04 (1.23, 3.39)] after adjustment for other variables. Other measures did not demonstrate evidence of relationship in the adjusted analysis. Consistent with findings for MPR, in the adjusted model tobacco use also predicted lower odds of persistence in the entire cohort [OR 0.41, 95% CI: (0.27, 0.62)] and in males [0.37(0.23, 0.60)], while the diagnosis of prostate cancer in males was associated with higher odds of persistence [1.73 (1.05, 2.85)] (Supplementary Table S2). In adjusted analyses, no other covariates were associated with persistence in the whole cohort nor males only.
Predictors of Adherence Measured by MPR in 744 Patients (Males n = 599) With Baseline DXA Scan. a

Abbreviations: MPR, medication possession ratio; DXA, dual-energy x-ray absorptiometry; OR, odds ratio; CI, confidence interval.
Detailed unadjusted and adjusted analysis for binary MPR (with reference category MPR <80%).
Boldfaced values indicate statistical significance (P ≤ .05).
Age is analyzed by 10-year increments.
Predictors of Changes in BMD
Adherence, estimated by MPR, to weekly alendronate was investigated for its relationship to change in BMD. Only 239 patients had follow-up scans (males n = 155). The median interval from baseline DXA scan to T0 was 0.03 (interquartile range: 0.01-0.19) years and the median interval to follow-up scan was 2.00 (1.48-2.38) years from index prescription. For the low- and high-adherence groups, the mean annual change in BMD at the total hip was −0.25% ± 3.70% and 1.10% ± 4.8%, respectively, and for LS 1.67% ± 3.10% and 3.07% ± 6.54%, respectively. The analysis summarized in Table 3 utilized MPR over the interval from the T0 to the follow-up DXA scan and the annualized percent changes in BMD at the total hip (n = 239) and LS (n = 233).
MPR as a Predictor of Yearly Percent Change in BMD by Site (n = 239). a
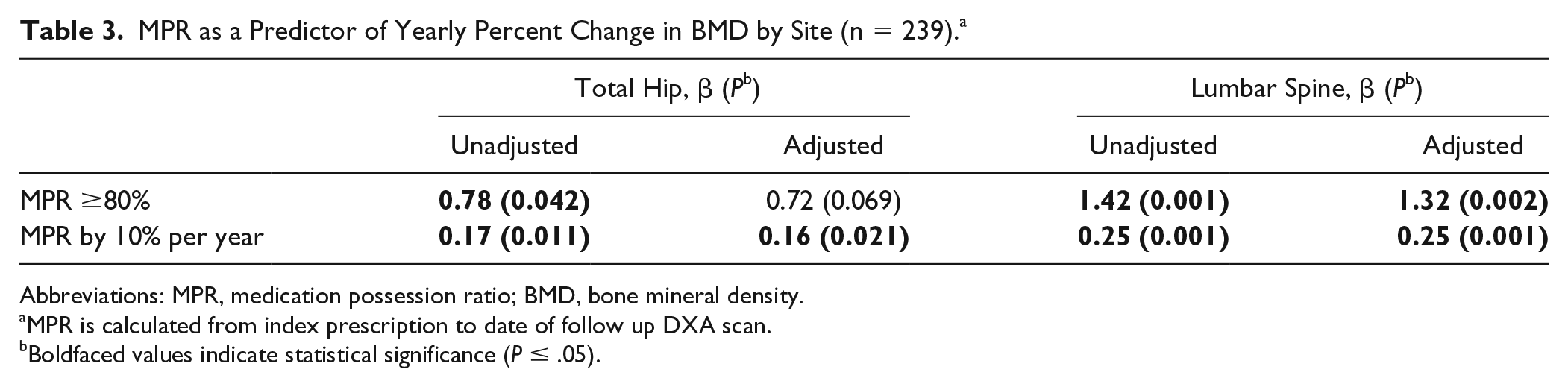
Abbreviations: MPR, medication possession ratio; BMD, bone mineral density.
MPR is calculated from index prescription to date of follow up DXA scan.
Boldfaced values indicate statistical significance (P ≤ .05).
MPR ≥80% predicted annual increases in BMD for both total hip and LS, 0.78% (P = .042) and 1.42% (P < .001) per year, respectively. In the adjusted analysis, MPR ≥80% only remained associated with BMD increase per year at the LS, 1.32% (P = .002) at the LS. The analysis was also run with 10% increment and in this adjusted analysis, evidence of relationships remained for both the total hip (0.16%, P = .021) and LS (0.25%, P = .001). None of the covariates were associated with yearly percent change in BMD (data not shown).
Incident Fracture
We identified pertinent clinical predictors of incident fracture in the 815 patients who did not have a baseline diagnosis of fracture (Table 4). There were 109 patients with incident fracture with a median follow-up of 4.6 (2.9-7.1) years. Incident fracture rates were similar by gender such that 82 (13%) of the 626 males and 27 (14%) of the 189 females developed a fracture. Kaplan-Meier curve of incident fracture during follow-up are presented for the whole cohort (Figure 1) and for males (Supplementary Figure S1) by MPR strata, with evidence of differences in time until incident fracture by MPR in both. In the whole cohort, MPR ≥80% predicted lower risk for fracture [HR: 0.65; 95% CI: (0.44, 0.96)] while acid suppression therapy, tobacco use, and alcohol abuse were associated with increased risk of fracture; however, after adjustment, evidence of an association between MPR and incidence fracture did not persist [HR: 0.70; 95% CI: (0.47, 1.03)]. Only baseline prescription for acid suppression therapy demonstrated a robust relationship with incident fracture [HR: 1.72; 95% CI: (1.17, 2.52)] after adjustment for all covariates. In the adjusted analyses in males, MPR ≥80% was associated with a risk reduction for incident fracture [HR: 0.60; 95% CI: (0.38, 0.95)] compared with MPR<80%, and similar to what was observed in the whole cohort, acid suppression therapy was associated with increased fracture risk [HR: 1.63; 95% CI: (1.04, 2.55)].
Year 1 MPR as Predictor of Time Until Incident Fracture in Patients Without a Baseline Fracture (n = 815) Calculated for Low and High Adherence.

Abbreviations: MPR, medication possession ratio; HR, hazard ratio; CI, confidence interval.
Boldfaced values indicate statistical significance (P ≤ .05).

Kaplan-Meier curves of incident fracture by year 1 medication possession ratio (MPR) strata in patients without baseline fracture (n = 815).
Discussion
Despite an integrated health care system, only 48% of Veterans in year 1 of therapy were adherent to oral bisphosphonate, the most commonly used agent for prevention and treatment of osteoporosis. 6 Of the patients who were adherent in year 1, most continued to be adherent into the second year of treatment (86% cumulative). We found that compared with Veterans who were nonadherent in the first year, those who were adherent had a 30% reduced risk of incident fracture over 10 years. The reduced risk was 40% in males, similar to reported in clinical trials. 22 As it is more difficult to distinguish a secondary event from a primary event with database research, we focused on primary prevention in this analysis. The fracture prevention rate may have been higher if we had also investigated secondary prevention as alendronate reduces subsequent fractures greater than initial fractures. 23
In this investigation, criteria were selected to establish a cohort that was planned for chronic oral bisphosphonate treatment for osteoporosis and to limit issues related to a historical cohort established from an electronic database. Once weekly alendronate was selected as it is the most common osteoporosis treatment prescribed in the VA and has better adherence compared to daily dosing. 24 To focus on medication-use patterns of VA patients rather than anticipated reasons for discontinuation of therapy, we excluded patients whose VA medical chart detailed an allergy or adverse event with oral bisphosphonates, but this was a <3% of our cohort. To be included, a patient required at least 2 years of follow-up, a time period sufficient to detect outcome differences from drug effect and to limit patients with a baseline level of comorbidity likely to impair adherence. Despite this approach, 52% of the cohort was nonadherent (MPR<80%) in year 1 consistent with multiple other studies.11,25,26 This demonstrates oral bisphosphonate adherence presents a specific challenge for the treatment of osteoporosis. Similar to our findings, others have reported that low adherence to oral bisphosphonates contributes to poor BMD response 26 and increased fractures. 25 Current treatment guidelines detail that patients with such events should be evaluated as possible “nonresponders” and be assessed for adherence. 6 However, these guidelines do not include recommendations to modify treatment based on individualized measurements of adherence. The VA’s specific guidance for osteoporosis recommends that for low adherence (<80%) to oral bisphosphonates to “institute local measures to improve adherence” after 2 years 5 ; however, reliable methods to improve adherence are lacking. 4
Nearly 20% of our cohort was diagnosed as tobacco users which was associated with 65% lower odds of being adherent. In another study of male Veterans, Hansen et al 11 reported that a diagnosis of tobacco use doubled the odds of being nonadherent. Tobacco use is an independent risk factor for osteoporotic fractures, including hip fractures. 27 Furthermore, tobacco use contributes to increased risk of fracture and poor BMD response during treatment with an oral bisphosphonate. 28 In our adjusted model, there was a trend toward tobacco use increasing the odds of developing an incident fracture [OR 1.55 (0.97, 2.48)]. We also found that baseline fracture predicted low adherence [OR 0.64 (0.41, 0.98)], but this was not maintained in our adjusted analyses. Others have reported baseline fracture to be associated with poor adherence in postmenopausal females and increased osteoporosis related events. 9 Overall, the current data provide further support that tobacco users and those with baseline fracture are especially at risk of low adherence and poorer osteoporosis outcomes.
Our findings increase the evidence that early adherence should influence clinical decision making for osteoporosis treatment. A review of published interventions to promote adherence to osteoporosis treatment found many interventions were not effective and the ones that reported improved adherence were expensive and/or lower quality studies. 4 With this in mind, low-adherence patients should be considered for osteoporosis therapies that are known to have better adherence, including denosumab 29 and zoledronic acid. 30 This is especially relevant for high fracture risks patients such as those with baseline fracture or tobacco users. Our patients with low adherence during year 1 had poorer BMD response and increased fracture long-term. In a post hoc analysis of the alendronate Fracture Intervention Trial in postmenopausal females, the time to benefit for fracture prevention was 11 months for all participants and 8 and 19 months for those aged ≥70 years and <70 years, respectively. 31 In our cohort, adherent was defined as ≥80% as other studies support this to be the most beneficial level. At adherence rates between 50% and 80%, the clinical benefit is reduced and at rates <50% most benefit is lost.25,26 Considering these defined levels of benefit by adherence and defined time to benefit, we propose providers to access adherence to oral bisphosphonates between 6 and 12 months of therapy. This does require that providers have tools to evaluate adherence and systems-based approaches would support providers. Within the VA EMR the dates of medication fills completed by the VA are readily available allowing the opportunity to intervene based on adherence.
In our cohort, acid suppression therapy (prescription for H2 antagonist or PPI) was associated with 63% to 72% increased risk of being diagnosed with an incident fracture over a median follow up of approximately 5 years. Multiple studies have reported that treatment with a PPI occurs with increased fracture risk. In the current study, 83% of the acid suppression prescriptions were for PPIs. There is conflicting evidence regarding the impact of concurrent PPI on the benefits of oral bisphosphonate therapy with data showing significant reduction in the antifracture efficacy 32 and others showing no effect. In a meta-analysis with over 57 000 patients by Yang et al, 33 concurrent PPI with oral bisphosphonate increased fracture risk [OR 1.52 (1.05, 2.19), P = .025]; however, there was considerable heterogeneity and the magnitude of risk varied by population. Multiple mechanisms have been proposed for the reduction of the efficacy of oral bisphosphonate with PPIs, including interfering in the activity of osteoclasts 34 and limiting the calcium absorption. On the contrary, increasing gastric pH doubled bioavailability of alendronate; however, this was carried out with a H2 antagonist infusion. 35
There are certain points that need to be considered when interpreting the current study. As with other retrospective studies only association and not causation can be inferred, and residual confounding remains a possibility. Our cohort was primarily male, older, and white, so results may not be generalizable to other populations. In this specific database analysis, it is possible some patients received osteoporosis medications from a non-VA setting and that was not captured. Non-VA medications can be recorded in the VA EMR, but the data are less reliable and were not included. In the analysis with MPR as a predictor of annual BMD change our sample size was limited as only 32% of the patients had follow-up DXA scans. Desired adherence did occur with improvement in BMD but to a lesser degree that other studies have detailed. 26 Baseline statin prescription was significantly more frequent in the high adherence group but was not included as a covariate due to mixed literature on impact of statins and osteoporosis outcomes 36 Estrogen therapy was not included as a covariate; however, the study was in an era when postmenopausal estrogen use in the VA, similar to private sector, was especially low. 37 Last, calcium and vitamin D intake were not included in the analyses as many patients purchase over the counter medications outside the VA and this is less accurately captured in the EMR.
In the general population, 80% of patients with osteoporosis are female; however, in our cohort the demographics are reversed, 80% male and 20% female. We aimed to add to the limited osteoporosis literature in males but did not chose to only investigate males as we wanted a representation of the VA population, which we achieved. We found that low adherence occurred with reduced improvements in BMD and increased risk for long-term incident fracture. The VA EMR details medication fills allowing providers to assess adherence. We propose a more proactive approach to evaluate oral bisphosphonate adherence at 6 to 12 months from initiation. With the growing literature detailing consistently high rates of nonadherence with oral bisphosphonates, poor outcomes with low adherence, and no high-quality data for an effective intervention to improve adherence, clinical decision making that considers individualized measures of adherence to oral bisphosphonates would be expected to improve outcomes, especially in high-risk patients.
Supplemental Material
Supplement_9.27.19_CLEAN – Supplemental material for Predictors and Outcomes Associated With Adherence to Weekly Alendronate in US Military Veterans: Clinical Research Study
Supplemental material, Supplement_9.27.19_CLEAN for Predictors and Outcomes Associated With Adherence to Weekly Alendronate in US Military Veterans: Clinical Research Study by Sarah Bou Malham, Benjamin Bowe, Sumon K. Sen, Rong Mei Zhang, Barbara Sterkel and Julia P. Dunn in Journal of Primary Care & Community Health
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RMZ was supported by NIDDK of the National Institutes of Health under award number T32 DK007120. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, the US Department of Veterans Affairs, or the United States Government.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
