Abstract
Introduction:
Advances in oncologic therapy have extended survival among women of reproductive age, repositioning fertility and family-building as core survivorship concerns. Population-level data on post-diagnosis pregnancy and real-world fertility-care utilization across cancers remain limited. This study evaluated post-diagnosis pregnancy and fertility-care utilization among reproductive-aged women (18-45 years) with incident invasive cancer and examined variation by age, parity, race, and neighborhood social vulnerability.
Methods:
We conducted a retrospective cohort study using 2 large U.S. datasets. The primary cohort was drawn from IBM MarketScan (2013-2018). A validation cohort was assembled using Epic Cosmos (2018-2021), an electronic health record-based patient registry. Women aged 18 to 45 years with incident invasive cancer were matched to cancer-free controls. The main exposure was incident cancer diagnosis. The primary outcome was subsequent pregnancy after diagnosis, with secondary outcomes including fertility evaluation, fertility testing, and fertility preservation. Hazard ratios (HRs) and 95% confidence intervals (CIs) measured associations, including subgroup analyses by parity, age, race, and social vulnerability.
Results:
In the MarketScan cohort, pregnancy occurred in 103 (2.3%) survivors versus 388 (4.5%) controls (HR = 0.47, 95% CI = 0.38-0.59). Survivors had higher rates of fertility evaluation (42 [0.9%] vs 25 [0.3%]; HR = 3.00, 95% CI = 1.81-4.97), fertility testing (1759 [39.7%] vs 1031 [11.9%]; HR = 4.23, 95% CI = 3.91-4.58), and fertility preservation (34 [0.8%] vs 25 [0.3%]; HR = 2.36, 95% CI = 1.39-4.00), although absolute preservation rates remained low. Associations were strongest among nulliparous women (pregnancy HR = 0.50, 95% CI = 0.37-0.69). Increasing age reduced pregnancy (HR = 0.75, 95% CI = 0.73-0.78) and preservation (HR = 0.86, 95% CI = 0.80-0.92). No racial or neighborhood social vulnerability trends were identified. Results were replicated in the Cosmos validation cohort, with consistently higher fertility evaluation (HR = 7.47), testing (HR = 5.54), and preservation (HR = 6.56) after cancer diagnosis.
Conclusions:
Among reproductive-aged women, cancer diagnosis was associated with fewer and delayed pregnancies and persistently low absolute uptake of fertility-preserving care, particularly among nulliparous and older patients. These findings highlight the need for early, standardized onco-fertility counseling, automatic pre-treatment referral for patients undergoing gonadotoxic therapy, and policies designed to mitigate barriers to fertility care.
Introduction
Cancer among women of reproductive age represents an important and growing public health concern in the United States. A spectrum of malignancies can occur during the reproductive years, including breast, endometrial, gastrointestinal, lung, neuroendocrine, ovarian, thyroid, and genitourinary cancers. 1 Breast cancer remains the most commonly diagnosed invasive malignancy in this population; according to U.S. Cancer Statistics registry data, 27 136 new breast cancer cases were reported among women younger than 45 years in the United States in 2022. 2 Improvements in early detection and cancer treatment have substantially improved cancer outcomes among young women; for example, CDC data indicate that 92% of women with breast cancer were alive 5 years after diagnosis without death from breast cancer. 3 As survival improves, a growing population of reproductive-aged cancer survivors must navigate the potential reproductive consequences of cancer therapy. 4 At the same time, broader demographic trends, including delayed childbearing and the age-related decline in fecundity beginning in the early 30s, mean that time spent undergoing cancer treatment or recovery may further narrow the reproductive window. 5
Young women who survive cancer have substantially lower post-treatment pregnancy rates compared with their peers; this reduction likely reflects both treatment-related gonadotoxicity and delays in attempting conception during cancer therapy and recovery.6-8 Importantly, emerging evidence suggests that pregnancy after cancer does not worsen maternal survival or increase recurrence risk, and many survivors can ultimately achieve healthy pregnancies, even following hormone-sensitive malignancies such as breast cancer. 9 Despite clear clinical guidelines and advances in fertility preservation technologies, uptake of fertility counseling and preservation services among reproductive-aged women with cancer remains low.10,11 Disparities in access to fertility preservation further exacerbate this gap: younger patients in their 20s are more likely to receive fertility preservation than those in their 30s and 40s, while racial and socioeconomic inequities continue to limit access to fertility services among minority and disadvantaged populations. 11 Collectively, these findings highlight a persistent gap between guideline recommendations and real-world practice and underscore the need to improve oncofertility counseling and access to fertility preservation before cancer treatment. 12
Knowledge regarding fertility outcomes after cancer among young women remains limited, with few studies comprehensively examining multiple fertility-related outcomes across diverse cancer types.13,14 This study evaluated post-diagnosis pregnancy and fertility-care utilization among reproductive-aged women (18-45 years) with incident invasive cancer and examined variation by age, parity, race, and neighborhood social vulnerability using 2 large national databases. By quantifying both pregnancy incidence and real-world uptake of fertility services across cancer types, we sought to characterize reproductive trajectories after cancer diagnosis and identify gaps in oncofertility care delivery. In this framework, incident invasive cancer was treated as exposure, with post-diagnosis pregnancy as the primary reproductive outcome and fertility-care utilization as related care-process outcomes.
Methods
Data Source and Study Population
We conducted a retrospective cohort study using 2 large national data sources. The primary analytic cohort was derived from the IBM MarketScan Commercial Claims and Encounters database to identify reproductive-aged women (18-45 years) with an incident diagnosis of invasive cancer between January 1, 2013, and December 31, 2018, using International Classification of Diseases (ICD) codes. Access to the IBM MarketScan Commercial Claims and Encounters Database was obtained through a paid institutional license. All data access and use were governed by a formal data use agreement outlining requirements for data security, privacy protection, and permitted research activities. The analytic dataset used in this study was extracted by the research team in accordance with the terms of this agreement. MarketScan captures medical and pharmacy claims from a national convenience sample of commercially insured individuals across the U.S. 15 Participation in the database is based on insurer contribution rather than population-based sampling, resulting in a large but non-random national convenience sample of commercially insured individuals. 15
A valid cancer diagnosis was defined as ≥2 claims for the same malignancy recorded at least 30 days apart, with no record of that cancer in the preceding year, as noted previously.16-18 Eligible cancers included breast, endometrial, gastrointestinal malignancies (ie, hepatobiliary, colorectal, pancreatic, gastric, esophageal, and small intestine), lung, neuroendocrine, ovarian, thyroid, and genitourinary (ie, bladder and renal). 19 We included multiple invasive cancer types diagnosed during the reproductive years to enhance generalizability and to reflect real-world oncologic practice, recognizing that fertility implications and treatment exposures vary across malignancies but collectively shape reproductive outcomes at the population level. 20 The analytic cohort was restricted to women who maintained continuous insurance enrollment for ≥12 months before and ≥36 months after the index diagnosis, had no record of prior sterilizing procedures (eg, hysterectomy, bilateral oophorectomy, or tubal ligation), and had no evidence of pregnancy or delivery within 6 months of diagnosis. Analyses were further limited to couples in which both partners were enrolled for ≥12 months before diagnosis to ensure complete baseline information and avoid bias from incomplete pre-diagnosis data. Because fertility-related services in claims data may involve both partners and may be billed under either individual, restricting to enrolled couples minimized incomplete ascertainment.
A validation cohort was constructed using Epic Cosmos data between 2018 and 2021, a multi-institutional electronic health record platform aggregating data from more than 190 U.S. health systems. Epic Cosmos is a multi-institutional electronic health record platform aggregating de-identified data from more than 300 million patients across participating U.S. health systems. Queries for the validation cohort were performed by the study investigators using SQL within the Epic Cosmos research environment. Access to Epic Cosmos was obtained through institutional participation in the Epic Cosmos research platform, which allows approved investigators to perform aggregate queries on de-identified electronic health record data from participating health systems. Data access is governed by institutional permissions and platform data-use policies. The Epic Cosmos cohort was used to replicate fertility-care utilization findings observed in MarketScan and to examine variation by race and neighborhood social vulnerability, variables not fully available in the claims dataset. Similar inclusion and exclusion criteria were applied, except for insurance enrollment requirements, since Cosmos does not capture enrollment data. The study years differed from the MarketScan cohort because Epic Cosmos data became available for research queries beginning in 2018 and expanded during subsequent years. Women aged 18 to 45 years were selected to reflect the conventional reproductive age range and to focus on individuals for whom fertility preservation and post-treatment pregnancy are clinically relevant considerations. The study followed the STROBE reporting guidelines. The project was deemed exempt from the Institutional Review Board because it involved only de-identified secondary data.
Covariates and Outcomes of Interest
Demographic, socioeconomic, and clinical covariates included patient age (ie, continuous; categorized as 18-30, 31-35, 36-40, and 41-45 years), 21 sex (ie, female), comorbidity burden, employment status (ie, full-time, part-time, or other), employment role (ie, whether the individual was the primary insurance beneficiary or a dependent spouse covered under the employee’s insurance plan in the MarketScan database), insurance type (ie, preferred provider organization [PPO], health maintenance organization [HMO], point-of-service [POS], comprehensive, consumer-directed/high-deductible health plan [CDHP/HDHP], or other), residential area (ie, urban, rural), geographic region (ie, Northeast, North Central, South, and West), and index year. Reproductive covariates included nulliparity, mean and minimum age of child, and number of children (ie, 0, 1, 2, and ≥3). Comorbidity burden was assessed using the Charlson Comorbidity Index (CCI), a validated weighted score of 17 chronic medical conditions, categorized as ≤2 or >2. 22 Scores were calculated using ICD-10 codes based on previously validated algorithms. 23 The index date was defined as the first recorded diagnosis of an invasive malignancy during the study period. Incident cancer cases were identified using malignant ICD-9/ICD-10 codes and required ≥2 claims for the same malignancy recorded at least 30 days apart, with no record of that malignancy in the preceding year (Supplemental Table 1). Follow-up time was defined as the duration from the index date to the earliest of outcome occurrence, insurance disenrollment, or end of the study period. In the validation cohort, follow-up was defined as time from index date to last recorded encounter or end of study period. Cancer-related covariates included cancer site (ie, breast, endometrial, gastrointestinal [hepatobiliary, colorectal, pancreatic, gastric, esophageal, and small intestine], lung, neuroendocrine, ovarian, thyroid, genitourinary [renal and bladder]), and receipt of cancer-directed therapy (ie, surgery, chemotherapy, and/or radiotherapy).
Social Vulnerability Index (SVI), race/ethnicity (ie, White, Black, Asian, Other), and marital status (ie, married, other [single, divorced, separated, or widowed]), were available only in the Cosmos validation cohort. Residential area was categorized as urban versus rural in the MarketScan cohort and as metropolitan versus non-metropolitan in the Cosmos cohort, according to dataset-specific classifications. The SVI, a CDC-developed, census tract-level composite measure incorporating 18 socioeconomic and demographic indicators from the American Community Survey across 4 domains (socioeconomic status, household composition, minority status/language, and housing/transportation); in this study, SVI was categorized into tertiles (low, medium, and high vulnerability).24,25
The primary outcome was post-diagnosis pregnancy, defined using validated diagnosis and procedure codes (ICD-9/ICD-10) capturing all pregnancy outcomes (including live birth, stillbirth, spontaneous abortion, induced termination, and ectopic pregnancy), as described previously.26,27 Secondary outcomes included fertility-related measures, such as (1) fertility evaluation, defined as consultation or referral to a reproductive specialist; (2) fertility testing, including laboratory or imaging assessments of reproductive function; and (3) fertility preservation, such as oocyte or embryo cryopreservation (Supplemental Table 2). 7
Statistical Analyses
Categorical variables were summarized as frequencies with proportions (%), and continuous variables were summarized as medians with interquartile ranges (IQRs). Group comparisons were performed using χ² tests for categorical variables and Kruskal-Wallis tests for continuous variables. Cox proportional hazards analysis was used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for the association between cancer status and time-to-event outcomes, including pregnancy, fertility evaluation, fertility testing, and fertility preservation. Logistic regression was used for binary outcomes when appropriate. Multivariable Cox models were fitted separately for each cohort; in the primary analytic cohort, models were adjusted for CCI, insurance plan type, geographic region, and rurality. In the validation cohort, models were adjusted for race/ethnicity, marital status, geographic region, and CCI. Sensitivity analyses stratified models by number of children, SVI tertiles, age categories, and racial groups. All tests were 2-sided, and P < .05 was considered statistically significant. Analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC).
Results
Baseline Characteristics
Among 13 112 women in the analytic cohort, 33.8% (n = 4428) had cancer diagnosis and 66.2% (n = 8684) were cancer-free controls (Table 1). Median index age was 42 years (IQR = 39-44); most women were employed full-time (n = 11 295; 86.1%), resided in urban areas (n = 11 265; 85.9%), and were nulliparous (n = 9792; 74.7%). Among parous women, the median number of children was 2 (IQR = 1-2), with most women having 1 child (n = 4492; 34.3%) or 2 (n = 4070; 31.0%). Among women with cancer, breast cancer was most common (n = 2718; 61.4%), followed by thyroid cancer (n = 912; 20.6%) and gastrointestinal cancers (n = 304; 6.9%; Supplemental Table 3). The median follow-up was 61.7 months (IQR = 47.3-80.9) and was shorter for women with cancer (59.0 months [46.1-77.4]) compared with controls (63.1 months [48.4-82.7]; P < .001; Table 1). More than half of women with cancer underwent surgery (n = 2491; 56.3%), while 69.8% (n = 3089) and 38.5% (n = 1706) received chemotherapy and radiotherapy, respectively (Supplemental Table 3). Moreover, women with cancer diagnosis were more likely to have CCI > 2 (2.5%, n = 109 vs. 0.1% n = 13; P < .001) and were more often enrolled in PPO insurance plans (56.7%, n = 2512 vs. 41.6%, n = 3615; P < .001) than cancer-free patients (Table 1).
Baseline Characteristics of the Study Cohort by Cancer Status.
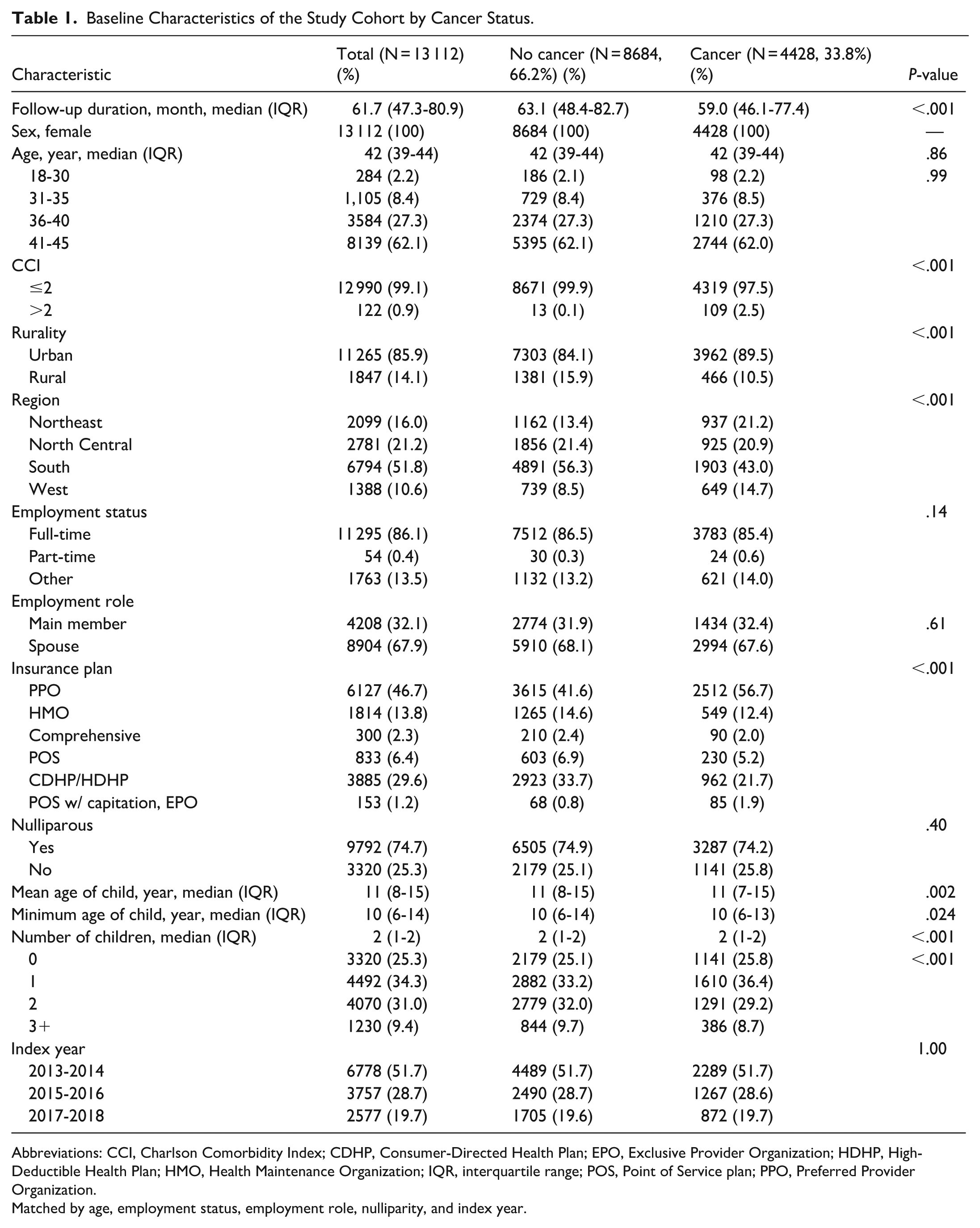
Abbreviations: CCI, Charlson Comorbidity Index; CDHP, Consumer-Directed Health Plan; EPO, Exclusive Provider Organization; HDHP, High-Deductible Health Plan; HMO, Health Maintenance Organization; IQR, interquartile range; POS, Point of Service plan; PPO, Preferred Provider Organization.
Matched by age, employment status, employment role, nulliparity, and index year.
Pregnancy and Fertility Outcomes
A total of 491 (3.7%) women experienced a post-diagnosis pregnancy after the index date. Pregnancy occurred in 2.3% (n = 103) of women with cancer compared with 4.5% (n = 388) of cancer-free controls (P < .001), as demonstrated in the cumulative incidence curves (Figure 1). Fertility evaluation was performed in 0.9% (n = 42) of women with cancer compared with 0.3% (n = 25) of cancer-free controls (P < .001; Figure 2). Fertility testing was substantially higher among women with cancer (39.7%, n = 1759 vs 11.9%, n = 1031; P < .001; Figure 3), as was fertility preservation (0.8%, n = 34 vs 0.3%, n = 25; P < .001; Figure 4; Table 2).

Cumulative incidence of post-diagnosis pregnancy among women with and without cancer.
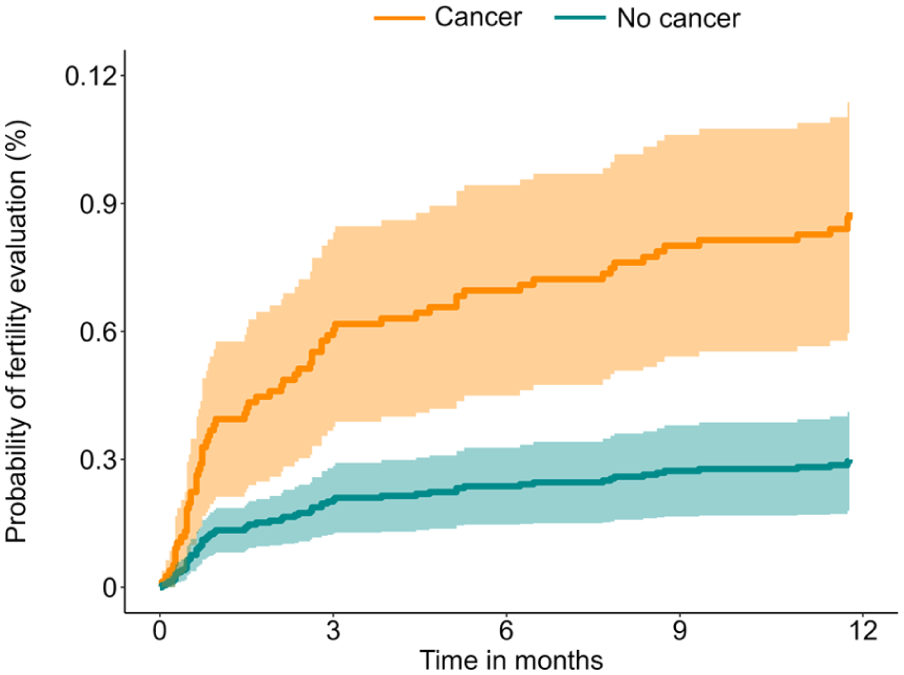
Cumulative incidence of fertility evaluation after index date among women with and without cancer.

Cumulative incidence of fertility testing after index date among women with and without cancer.

Cumulative incidence of fertility preservation after index date among women with and without cancer.
Pregnancy and Fertility Outcomes by Cancer Status in the Study Cohort.

Matched by age, employment status, employment role, nulliparity, and index year.
Following matching based on age, employment status, employment role, nulliparity, and index year, and further adjustment for relevant covariates, cancer was associated with 53% lower likelihood of pregnancy (aHR = 0.47, 95% CI = 0.38-0.59). Women with cancer had threefold higher rate of fertility evaluation (HR = 3.00, 95% CI = 1.81-4.97), over fourfold higher likelihood of fertility testing (HR = 4.23, 95% CI = 3.91-4.58), and over twofold higher rate of fertility preservation (HR = 2.36, 95% CI = 1.39-4.00; Table 3).
Adjusted Hazard Ratio for Pregnancy and Fertility Outcomes by Cancer Status in the Study Cohort.
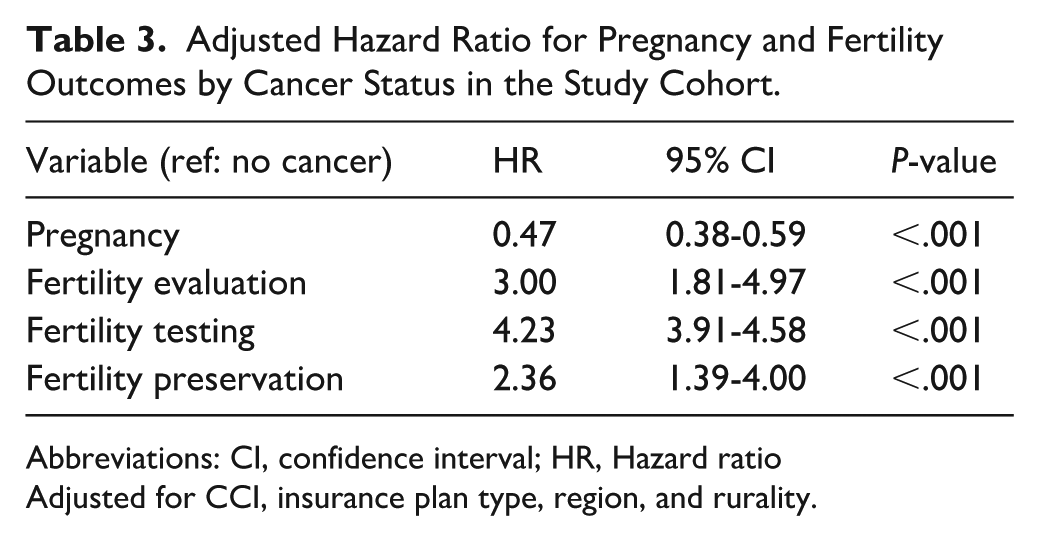
Abbreviations: CI, confidence interval; HR, Hazard ratio
Adjusted for CCI, insurance plan type, region, and rurality.
Parity-stratified models showed consistent reductions in pregnancy rates; compared with cancer-free controls, nulliparous women had 50% lower likelihood (HR = 0.50; 95% CI = 0.37-0.69) of pregnancy, while women with 1 (HR = 0.47, 95% CI = 0.32-0.69), or ≥2 children (HR = 0.41, 95% CI = 0.25-0.60) had 53% and 59% decreased likelihood of pregnancy, respectively. The association cancer diagnosis and fertility evaluation were most pronounced among nulliparae (HR = 3.78, 95% CI = 2.10-6.79), similarly to fertility preservation (HR = 2.94, 95% CI = 1.59-5.41). Following cancer diagnosis, fertility testing rose across all parity strata, with the greatest likelihood observed among nulliparae (HR = 2.26, 95% CI = 1.83-2.80), followed by women with 1 child (HR = 2.18, 95% CI = 1.81-2.63) and those with ≥2 (HR = 2.09, 95% CI = 1.74-2.50; Table 4).
Adjusted Hazard Ratio for Pregnancy and Fertility Outcomes by Cancer Status in the Study Cohort, Stratified by Number of Children.
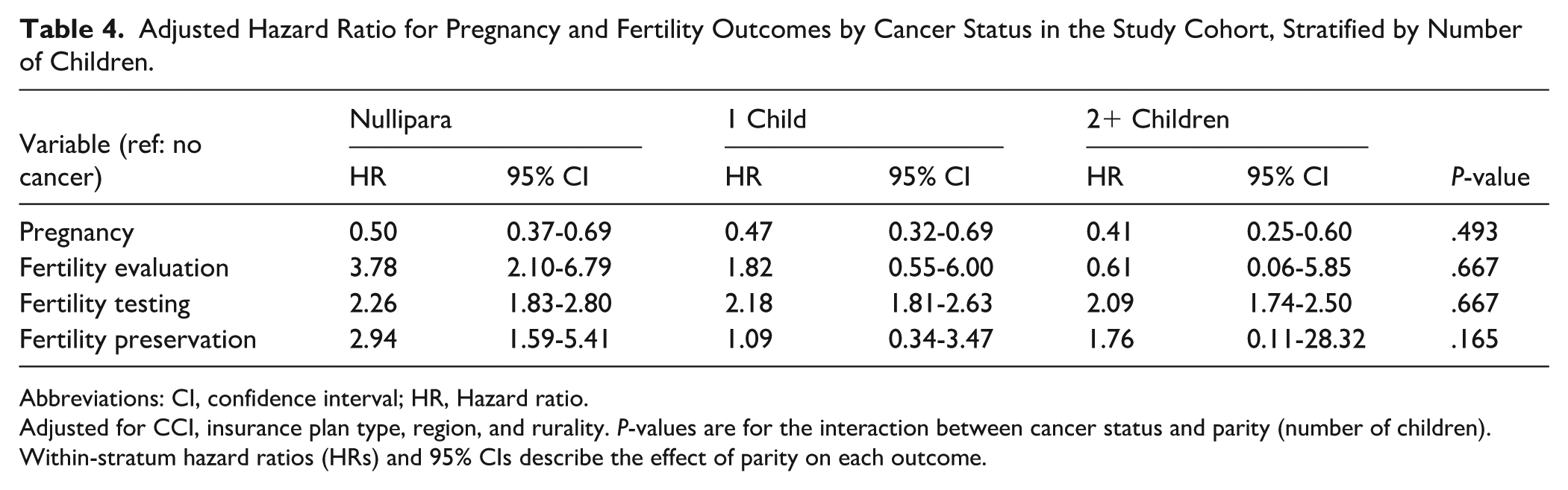
Abbreviations: CI, confidence interval; HR, Hazard ratio.
Adjusted for CCI, insurance plan type, region, and rurality. P-values are for the interaction between cancer status and parity (number of children). Within-stratum hazard ratios (HRs) and 95% CIs describe the effect of parity on each outcome.
Increasing age was associated with a lower likelihood of pregnancy in both cancer (HR = 0.75, 95% CI = 0.73-0.78) and cancer-free groups (HR = 0.74, 95% CI = 0.73-0.76), while also reducing the probability of fertility evaluation (cancer: HR = 0.81, 95% CI = 0.77-0.86; cancer-free: HR = 0.85, 95% CI = 0.79-0.91) and preservation (cancer: HR = 0.86, 95% CI = 0.80-0.92; HR = 0.81, 95% CI = 0.76-0.86). In multivariable analyses, age was not associated with fertility testing in either women with cancer or cancer-free controls (p>0.05; Table 5).
Adjusted Hazard Ratio for association between age and Pregnancy and Fertility Outcomes, Stratified by Cancer Status.

Abbreviations: CI, confidence interval; HR, Hazard ratio.
Adjusted for CCI, insurance plan type, region, and rurality. P-values represent the significance of the interaction between cancer status and age for each outcome. Within-stratum hazard ratios (HRs) and 95% CIs describe the effect of age on each outcome among individuals with and without cancer.
Validation Cohort: Baseline Characteristics
Among 74 447 women in the validation cohort, following matching based on age and index year, 25.0% (n = 18 615) had a cancer diagnosis and 75.0% (n = 55 832) were cancer-free controls. Median age was 39 years (IQR = 34-39) and most participants were White (n = 50 858; 68.3%), married (n = 37 135; 49.9%), and resided in metropolitan areas (n = 66 004; 88.7%). Of note, women with cancer were more likely to present with CCI >2 (11.3%, n = 2105 vs. 0.8%, n = 474) and live in the South (39.9%, n = 7423 vs. 33.9%, n = 18 926; both P < .001, Supplemental Table 4). Among patients with cancer, breast cancer was most prevalent (n = 9495; 51.0%), followed by thyroid cancer (n = 3530; 19.0%) and gastrointestinal cancers (n = 2552; 13.7%; Supplemental Table 5).
Validation Cohort: Pregnancy and Fertility Outcomes
All fertility-related outcomes were more frequent among patients with cancer, including fertility evaluation (2.2%, n = 413 vs. 0.3%, n = 184), fertility testing (34.5%, n = 6414 vs. 7.2%, n = 4018), and fertility preservation (2.5%, n = 468 vs. 0.4%, n = 238; all P < .001; Supplemental Table 4). After adjusting for race, area, region, comorbidity, and marital status, cancer remained associated with fertility evaluation (HR = 7.47, 95% CI = 6.26-8.92), fertility testing (HR = 5.54, 95% CI = 5.32-5.77), and fertility preservation (HR = 6.56, 95% CI = 6.00-7.70; Supplemental Table 6). In the validation cohort, no consistent trends with fertility outcomes were observed across racial subgroups (Supplemental Table 7). Interestingly, cancer diagnosis was associated with fertility outcomes across all SVI strata, with a linear gradient evident only for fertility testing (low SVI: HR = 7.34, 95% CI = 6.80-7.91; medium SVI: HR = 5.63, 95% CI = 5.23-6.05; high SVI: 4.38, 95% CI = 4.10-4.68); however, only modest variation in effect size across SVI strata was observed for fertility evaluation and preservation in the validation cohort (Supplemental Tables 8 and 9). Similar to the primary analytic cohort, nulliparous women with cancer had higher rates of fertility evaluation (10.2%, n = 36 vs. 3.4%; n = 31), preservation (11.1%, n = 39 vs. 4.4%, n = 40), and testing (46.0%, n = 162 vs. 12.6%, n = 115) compared with cancer-free controls, with testing also elevated among women with 1 child (43.4%, n = 449 vs. 7.8%, n = 264) and ≥2 children (41.8%, n = 785 vs. 7.7%, n = 538; all P < .001; Supplemental Table 10). Furthermore, fertility testing demonstrated a modest inverse gradient with age; differences between women with cancer and cancer-free controls narrowed from 43.0% vs 8.2% at 18 to 30 years to 29.6% vs 6.5% at 41 to 45 years, respectively. In addition, relative differences were largest at 18 to 30 years (HR = 6.50, 95% CI = 5.91-7.16) and decreased slightly by 41 to 45 years (HR = 5.05, 95% CI = 4.72-5.40; Supplemental Tables 11 and 12).
Discussion
Advances in oncologic therapy have extended survival among women of reproductive age, however this progress comes at the cost of treatment-related compromise of reproductive health and fertility. 28 Importantly, young cancer survivors face the dual burden of therapy and a narrowing reproductive horizon, yet few studies have assessed pregnancy outcomes and fertility-care utilization across cancer types.7,29,30 Using 2 large, distinct, national databases, we evaluated both post-diagnosis pregnancy trends, and fertility-care use among reproductive-aged women. Two consistent patterns emerged; first, pregnancy following cancer was less likely and occurred after longer time intervals compared with cancer-free controls; second, although survivors had relatively higher fertility service utilization, overall uptake remained low. We included a broad range of malignancies diagnosed during reproductive years to capture population-level fertility trajectories across heterogeneous cancer types. 31 Nulliparous women were the most likely to seek fertility care, while pregnancy likelihood decreased with increasing age and parity. Together, these findings provide robust, population-level evidence on how cancer reshapes reproductive potential, while highlighting the importance of early counseling and streamlined access to fertility-preserving options.
Epidemiologic studies consistently demonstrate reduced post-diagnosis pregnancy rates among young female cancer survivors compared with women in the general population, estimating a 40-60% lower likelihood of conception compared with women in the general population. 7 In our analysis, only 2.3% of survivors conceived within 3 years of diagnosis versus 4.5% of controls, representing a ~53% relative reduction in post-diagnosis pregnancy likelihood. The observed reduction likely reflects both gonadotoxic effects of systemic therapies and delays imposed by cancer treatment and surveillance during a narrowing reproductive window. 32 Despite survivors being 3 to 4 times more likely than controls to undergo fertility evaluation or testing, absolute utilization of fertility preservation remained very low, with only ~0.8% pursuing egg or embryo freezing. In contrast to prior studies reporting racial and socioeconomic disparities in fertility-care access, our cohorts demonstrated relatively uniform uptake across racial and community vulnerability strata. 33 This difference may reflect the privately insured composition of our primary cohort.
Historically, prior studies have documented racial and socioeconomic disparities in fertility counseling and preservation.34,35 However, in our study, fertility-service uptake appeared relatively uniform across racial groups and community vulnerability strata. Instead, age and parity emerged as the strongest predictors of fertility-service engagement, with nulliparous and younger women more likely to pursue consultation or preservation. These findings suggest that baseline family-building status and reproductive urgency may outweigh social or demographic factors within an insured population, 36 and may also reflect the privately insured composition of the primary cohort. 29
Our findings reinforce current guideline recommendations that reproductive-aged patients should be counseled regarding infertility risks and offered fertility-preservation options prior to treatment initiation. 37 Although survivors demonstrated higher relative utilization of fertility services—including up to a fivefold increase in testing in the validation cohort—absolute uptake remained low. Integrating pre-treatment fertility consultation into routine oncology workflows, alongside policies that expand coverage and reduce logistical barriers, may help close this gap and better align practice with established recommendations. 38
This study has several strengths, including the use of complementary national data sources. MarketScan enabled evaluation of both post-diagnosis pregnancy incidence and fertility-care utilization, while Epic Cosmos served as an external validation cohort for fertility-service patterns. The large sample sizes and time-stamped claims enhanced outcome ascertainment, and matched comparators strengthened interpretation of pregnancy differences. Cosmos additionally allowed assessment of racial and neighborhood-level variation not available in claims data. There are, however, certain limitations. MarketScan lacks mortality; requiring ≥3 years of continuous enrollment post-diagnosis enriches for survivors and may underestimate adverse post-diagnosis outcomes (survivorship bias). Its generalizability is limited to commercially insured populations, and granular oncologic (eg, stage) and sociodemographic details are sparse. Cosmos does not capture continuous enrollment; services obtained outside Epic systems are unobserved, likely biasing fertility-care uptake downward. Parity/gravidity are incompletely captured in Cosmos—typically only among patients with a documented pregnancy—constraining parity-specific analyses of service use. We did not distinguish between pregnancy outcomes (eg, live birth, miscarriage, or termination), as our objective was to assess overall post-diagnosis pregnancy incidence rather than outcome-specific rates. Further, across both platforms, code-based algorithms may miss early pregnancy losses or services paid out-of-pocket/outside billing workflows, and unmeasured factors (eg, pregnancy intentions, contraception, partnership/marital status, and employer fertility benefits) introduce residual confounding. To this end, fertility-service estimates are conservative in Cosmos and MarketScan findings reflect a survivor cohort; nonetheless, convergent signals across 2 distinct national datasets support the robustness and clinical relevance of our conclusions.
In conclusion, cancer during the reproductive years was associated with a lower likelihood of pregnancy and delayed time to conception. Survivors were more likely to engage with fertility-preservation care compared with controls, yet absolute uptake remained low. These findings provide rare population-level insights into reproductive trajectories and fertility-care pathways after cancer and underscore the importance of early, coordinated oncology-reproductive care to optimize care delivery and improve reproductive outcomes for this vulnerable population.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319261437902 – Supplemental material for Impact of Cancer on Pregnancy and Fertility Service Use in Reproductive-Aged Women
Supplemental material, sj-docx-1-jpc-10.1177_21501319261437902 for Impact of Cancer on Pregnancy and Fertility Service Use in Reproductive-Aged Women by Azza Sarfraz, Selamawit Woldesenbet, Odysseas P. Chatzipanagiotou, Areesh Mevawalla, Qaidar Alizai, Rida Ejaz, Abdullah Altaf, Mujtaba Khalil, Zayed Rashid and Timothy M. Pawlik in Journal of Primary Care & Community Health
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This study used 2 proprietary data sources: (1) Epic Cosmos, a multi-institutional, de-identified electronic health record platform, and (2) the IBM MarketScan Commercial Database. Access to Epic Cosmos is restricted and governed by institutional agreements and data-use policies with Epic Systems Corporation. IBM MarketScan data are licensed and subject to contractual restrictions. Neither dataset is publicly available; qualified researchers may request access through their institutions and the respective data custodians, contingent on appropriate permissions, licensing, and data-use approvals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
