Abstract
Background:
Care partners (CPs) are informal or family caregivers who help family or friends manage health care and provide a wide range of assistance. Advance care planning (ACP) supports CPs by clarifying patients’ goals and values.
Methods:
The Meta-LARC Advance Care Planning trial randomized primary care practices to team-based or individual clinician-focused versions of an ACP program to determine if team-based approaches would increase ACP and improve patient outcomes. CPs were included, but not required, in ACP conversations and follow-up; CP outcomes were secondary and exploratory. CPs completed initial, 6-, and 12-month questionnaires, including the Zarit Burden Interview-12, PROMIS-29, and assessed if care matched patient preferences.
Results:
171 CPs participated and completed initial and 6- or 12-month follow-up questionnaires. The PROMIS-29 scale scores were different only for the depression scale at the 6- and 12-month follow-ups: the mean score for CPs in the individual clinician-focused arm was better than the team-based CPs (50.3 vs 49.2, P = .023), though this difference is not considered clinically meaningful. In both team and clinician practices, CPs who rated care more in line with patient’s goals were less likely to report moderate or higher levels of burden (39.4% vs 59.5%, P = .0001) and reported significantly lower levels of anxiety, depression, sleep disturbance, and pain.
Conclusions:
We found no significant differences in most CP outcomes when serious illness conversations with patients in primary care are implemented using a team-based versus individual clinician-focused approach. However, when care matched what was important to patients, CP outcomes were better.
Keywords
Introduction
Care partners (CPs), are ‘informal’ or family caregivers, who provide essential support to individuals with serious chronic conditions and participate in decision-making about tests and treatments for acute illness and chronic conditions, essential needs such as long-term care and housing, and end-of-life care. In 2017, 41 million U.S. CPs provided 34 billion hours of unpaid care valued at $470 billion, 1 allowing frail and seriously ill people to remain in their homes and communities, avoid nursing facility admissions, and maximize quality of life. 2
Essential for CPs’ effectiveness is identifying what is important to the person they help, so they can assure health care and services match patient-defined goals. This is particularly important as CPs may help manage care and be called on to be a surrogate decision maker.3-6 A systematic way to establish patient goals is for clinicians to engage patients in a process of advance care planning (ACP). ACP includes identifying and sharing values and goals and then relating these to preferences for future health care. 7 When a CP is available, they may be included in ACP and keeping CPs apprised of patients’ goals and preferences is an essential part of ACP.
Although, ACP is a best practice, 8 it is not yet routine9-11 and we focused on primary care as an ideal setting for expanding ACP, given that 50.3% of 1 billion office visits occurring in 2019 were with primary care physicians. 12 Chronic conditions were the major reasons for 39% of visits for all ages and 52% for people over 65 years old. 13 Patients and CPs often have longstanding, trust-based relationships with primary care, where patients are cared for through all life stages including serious illness and end-of-life. This provides opportunities for conversations about care preferences when they are more effective, which is before crises or the need for end-of-life decisions. 3 The extended trajectory of chronic illness includes multiple encounters where clinicians can initiate conversations and follow-up to engage patients in ACP. Many CPs coordinate care and accompany patients to appointments, 14 providing opportunities for their inclusion in ACP. CPs who know patients’ preferences are more effective in implementing patient decisions and more confident as substitute decision makers and experience less anxiety, depression, and regret about end-of-life decision making.15,16
However, implementing ACP in under-resourced primary care practices requires efficient approaches. The main hypothesis for the Meta-network Learning and Research Center (Meta-LARC) ACP trial was that team-based approaches to ACP would be more effective in primary care than the traditional approaches that placed responsibility for ACP with a single clinician. The trial compared patient outcomes for team-based to individual clinician-focused versions of a specific evidence-based ACP program (ie, the Serious Illness Care Program [SICP] described in Methods). Given the importance of CPs in serious illness care, CP outcomes were included in secondary and exploratory analyses to (1) compare CPs burden and well-being for team-based and clinician-focused SICP and (2) examine the impact of ACP on CPs regardless of approach.
Methods
We conducted a cluster-randomized trial of ACP for patients with serious illnesses (Clinicaltrials.gov: NCT03577002) in primary care practice-based research networks (PBRNs) belonging to the Meta-LARC consortium: 5 in the U.S. and 2 in Canada. Practices were recruited and randomized equally to team-based or individual clinician-focused versions of ACP. Rationale for a clustered design and the details about randomization, allocation, and implementation are provided in a published protocol. 17 Practices participated in training developed and provided by the trial team, developed workflows, and implemented SICP as assigned. Patients and CPs were enrolled and data collected from February 2019 through February 2021. IRB approval was obtained from the Trial Innovation Network Single IRB at Vanderbilt University Medical Center (IRB #181084) [US sites], the Centre intégré universitaire de santé et de services sociaux de la Capitale-Nationale of Quebec City, Canada (#MP-13-2019-1526) [Quebec sites]; and from the Health Sciences Research Ethics Board of the University of Toronto (protocol #36631) [Ontario sites].
A primary care clinician or team member introduced the study to patients. Patients had to be over 18 years old, community dwelling, and had serious illnesses or conditions that were likely to limit life expectancy to less than 2 years to participate. Patients provided consent for their own participation and were asked if they had a friend or family member who helped with their health care (i.e., a CP). If patients were willing to refer a CP to the study, the CP was contacted and had to provide their own consent to participate.
The Meta-LARC ACP Trial team modified and implemented 2 versions of SICP from Ariadne Labs (www.ariadnelabs.org) with the developers permission. Version (1) Team-based: the care team (clinician and at least one person in a different role: nurse, social worker, etc.) at each practice developed a workflow appropriate for their practice that included how patients would be identified and then invited to engage in ACP and who would initiate the conversation and who would follow-up. Plans included details for hand-offs, communication, and documentation. Version (2) Individual clinician-focused: a primary care clinician (ie, physician, physician assistant, or nurse practitioner) was solely responsible for all aspects of ACP conversations, follow-up, and documentation. Other staff may have been involved in identifying patients appropriate for ACP as part of their normal activities.
Both arms included training and implementation support specific to the SICP version. Training included two 1.5-hour modules, one online and one in-person session with role-play focused on communication skills and use of the Serious Illness Conversation Guide, a structured approach to ACP using patient-tested language.17,18 The Meta-LARC trial team and participating PBRN staff supported implementation using practice facilitation (a model of outreach and collaboration used by PBRNs) to provide technical assistance with patient identification and workflow development and documented progress through regular check-ins and quarterly meetings.
Patients and CPs completed questionnaires after the initial serious illness conversation, then 6 and 12 months later. Questionnaires were completed by telephone, mail, or online. CPs reported their relationship to the patient and whether they lived with the patient. Both CPs and patients identified the patient’s most serious illness/condition and indicated whether the patient had common medical diagnoses. Demographic information was collected for both CPs and patients.
Outcomes collected from CPs included the Short Form Zarit Burden Interview (ZBI-12) measuring how burdened they feel in their caregiving role,19-21 the Patient-Reported Outcome Measurement Information System 29-Item Profile Measure (PROMIS-29) measuring a CP’s overall health-related quality of life,22-27 and the goal-concordant care (GCC) items (treatment that aligns closely with a patient’s stated values, preferences, and goals) designed for this study. The ZBI-12 includes 12 items rated on a 5-point Likert scale (0 = ‘never’ to 4 = ‘nearly always’). Scores were obtained by adding responses together; higher scores indicate greater burden. The PROMIS-29 was designed to collect data on important symptoms and function across conditions and interventions, and CPs were asked to report on their own symptoms and health, including depression and anxiety. Each of the 29 items had 5 answer choices: never, rarely, sometimes, often, and always. GCC was the primary patient outcome for the trial, and the perspectives of CPs were obtained using a parallel version of an overall rating question developed for this trial with patient input. The question asked, ‘How much has the medical care the person you help received in the past 6-months matched what is important to them?’ with response options, 0 = ‘not at all’ to 10 = ‘completely’.
Descriptive statistics were used to summarize characteristics of CP and patients. Because cluster randomization was used at the practice level, P-values were calculated using chi-squared tests as a measure of imbalance between the arms resulting from recruitment of patients and CPs.
For the comparison of CP outcomes by study arm, the analytic sample was limited to CPs with a baseline and at least one follow-up response (i.e., at 6 and/or 12 months after the initial visit). As we did not have an a priori hypothesis that the 2 time points would be different, we examined the estimates adjusted for time point and found they were the same as the unadjusted estimates to the second decimal place. Given this, we combined the 2 time points and used estimators that accounted for the multiple observations from the same care partners. The ZBI-12 score was dichotomized into ‘none to mild’ (0-9) and ‘moderate to high’ (10-48) burden. 28 The PROMIS measure responses were converted to T-scores for each domain, which are standardized scores with a mean of 50 and standard deviation of 10 in a reference population. 26
Odds of CP burden being ‘moderate to high’ compared to ‘none to mild’ were calculated using logistic regression with generalized estimating equations. Mean differences between arms in PROMIS scores, which were not symmetrically distributed, were calculated using linear regression with bootstrapped standard errors where resampling was executed at the CP level. This approach does not include practice-level clustering because of the difficulty of specifying a two-level bootstrap. We compared these results to those obtained from mixed models with robust variance estimators and conclusions were unchanged. Time point was omitted from final models because its coefficient was close to 0 and its effect on the estimates of treatment effect and variance were very small. Adjusted estimates were calculated with the addition of covariate indicator variables for living with the patient and for being a spouse (including ex-spouse and long-term partners), as well as for the CP’s general health, self-rated on a 5-point scale. These covariates were selected a priori for their potential relationship to CP outcomes.
Our exploratory analysis of the cross-sectional relationship between CP rating of goal concordance of patient care and CP outcomes was based on our assumption that higher GCC for patients would contribute to better CP outcomes. We calculated the mean differences in outcomes, using unadjusted models as described above, after splitting observations into 2 groups: CPs that reported (1) high GCC (rated 9 or 10) and (2) lower ratings (rated 0-8), a cut point pre-specified in the main trial analysis for patient responses to the similar question. Up to 3 responses per CP could be included (initial, 6, and 12 months). No adjustments were made to P values for multiplicity. 29
Results
Forty primary care practices referred patients to the study. Almost one-third (253, 31.5%) of the 802 patients who participated in the trial identified a CP and allowed us to contact this CP, then 213 CPs consented and enrolled. One hundred seventy-one (171) CPs provided sufficient data on initial questionnaires for analyses. Of these, 135 completed 6-month and 117 completed 12-month follow-ups. The study flow diagram in Figure 1 documents participation and reasons for loss to follow-up. Figure 2, an upset plot, shows how many CPs completed each combination of questionnaires. The number of unique CPs and responses included are specified for each analysis in the tables.

Study flow diagram.
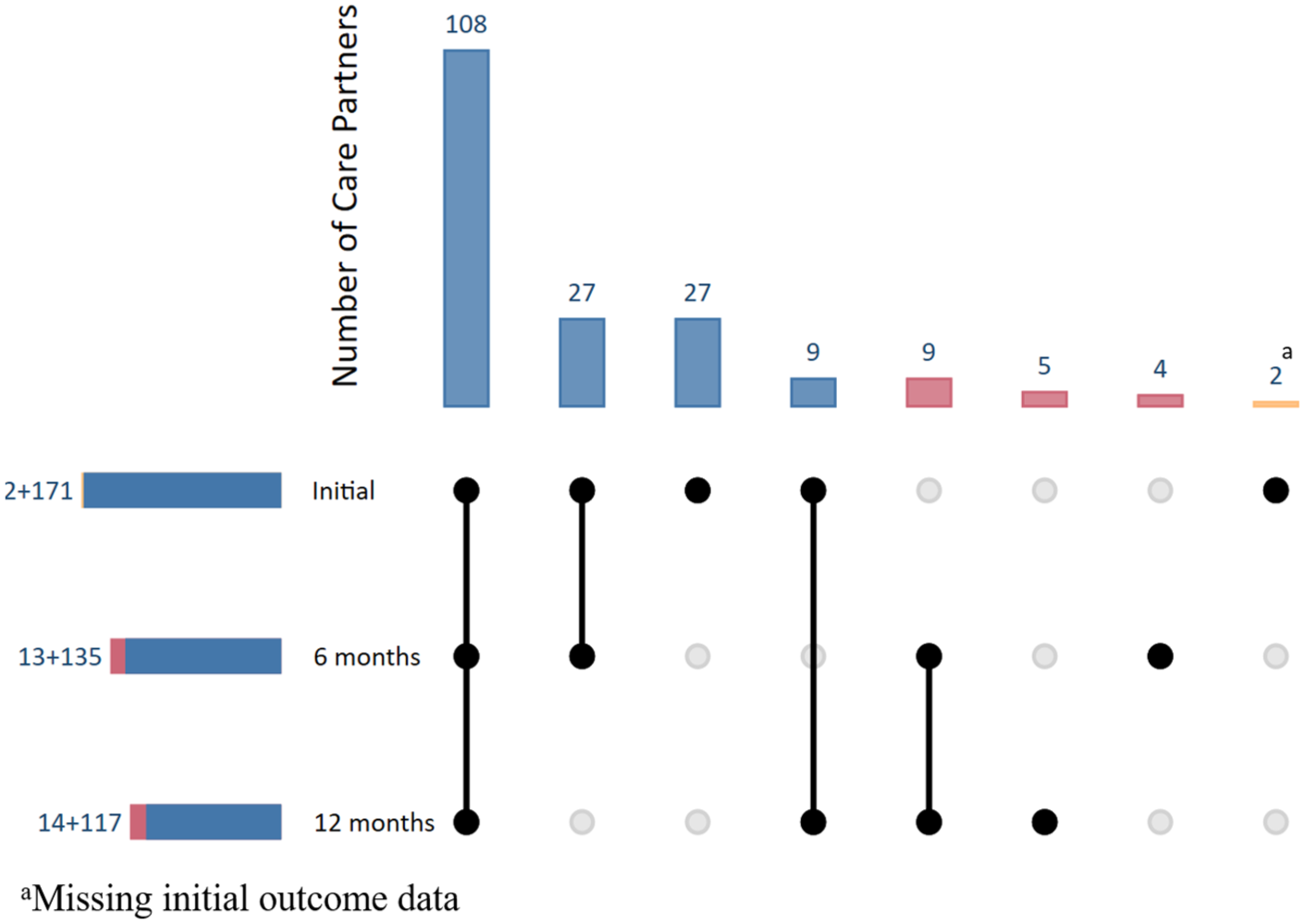
Upset plot.
Table 1 provides descriptive characteristics of the 171 CPs who completed initial questionnaires and demographic questions, 95 (56%) were spouses/significant others and 119 (70%) lived with the patients. One hundred twenty-eight (76%) were female, 103 (61%) were aged 55 to 74 years, and 42 (25%) were 75 years old or older. One hundred nine (64%) had some college education or were college graduates. One hundred fifty-two (91%) were White and non-Hispanic. Twenty-seven (17%) CPs reported their own health as fair or poor. Characteristics of CPs associated with practices randomized to the team and clinician approaches were similar. Table 1 also includes characteristics of the patients enrolled in the study that these CPs helped.
Care Partner and Patient Characteristics.
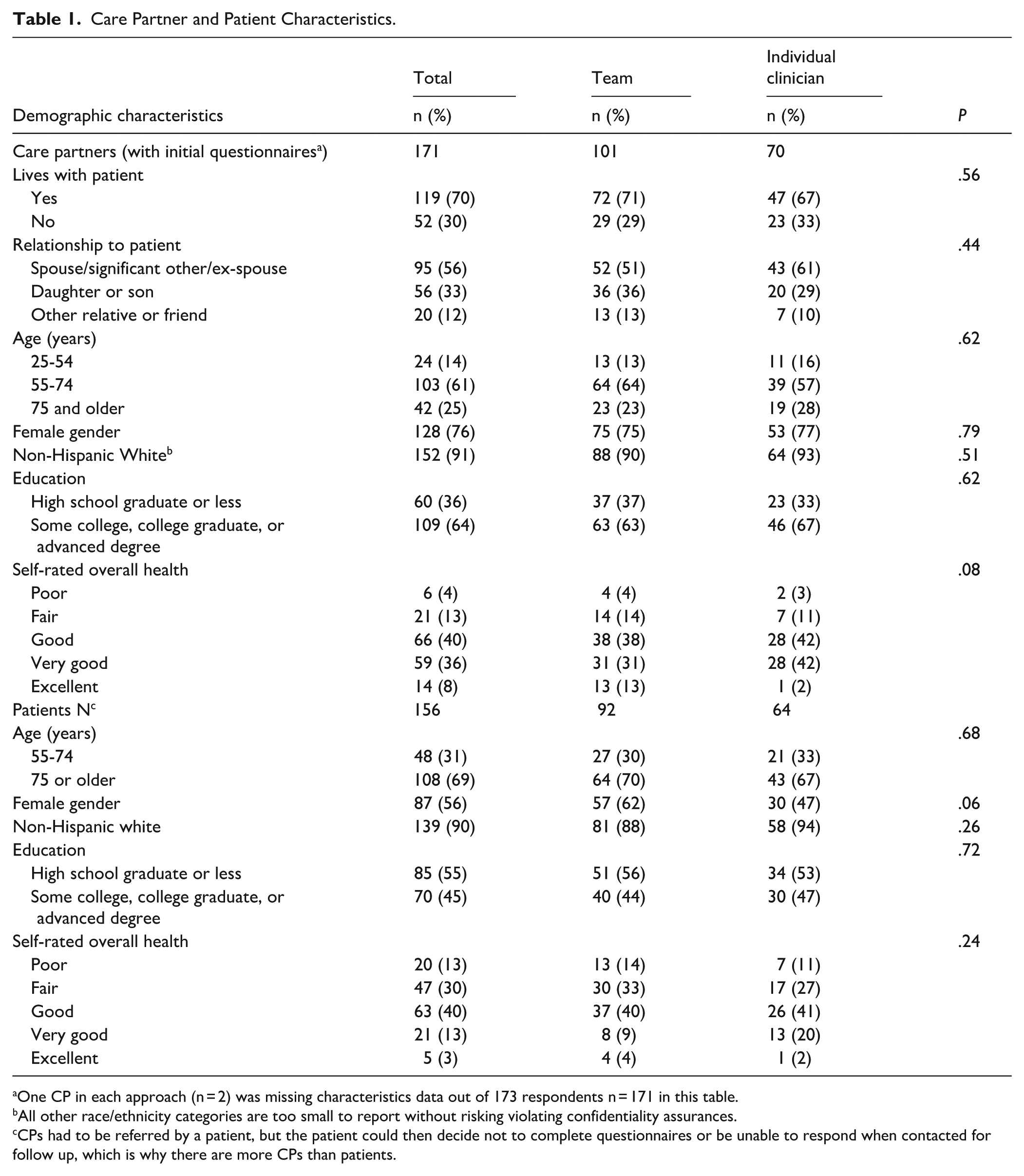
One CP in each approach (n = 2) was missing characteristics data out of 173 respondents n = 171 in this table.
All other race/ethnicity categories are too small to report without risking violating confidentiality assurances.
CPs had to be referred by a patient, but the patient could then decide not to complete questionnaires or be unable to respond when contacted for follow up, which is why there are more CPs than patients.
Care Partner Outcomes: Team Versus Individual Clinician
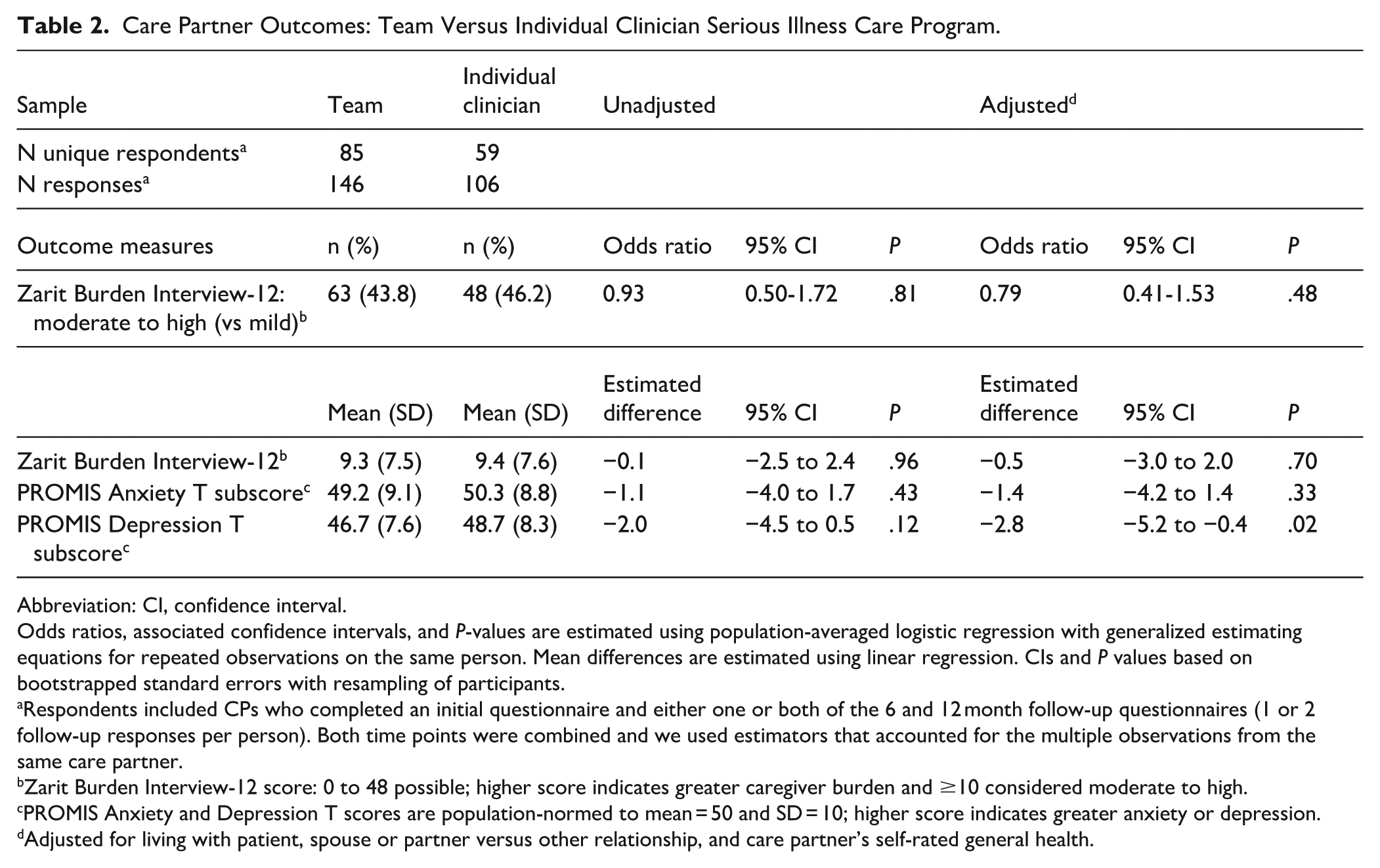
Almost half of the CPs reported moderate to high levels of burden on the ZBI-12 (44% in the team and 46% in the clinician arm) at 6- and 12-month follow-up. After adjusting for self-rated general health and whether the CP lived with the patient at the time of enrollment, the difference in burden remains not statistically significant (adjusted OR 0.79, 95% CI 0.41-1.53; Table 2).
Care Partner Outcomes: Team Versus Individual Clinician Serious Illness Care Program.
Abbreviation: CI, confidence interval.
Odds ratios, associated confidence intervals, and P-values are estimated using population-averaged logistic regression with generalized estimating equations for repeated observations on the same person. Mean differences are estimated using linear regression. CIs and P values based on bootstrapped standard errors with resampling of participants.
Respondents included CPs who completed an initial questionnaire and either one or both of the 6 and 12 month follow-up questionnaires (1 or 2 follow-up responses per person). Both time points were combined and we used estimators that accounted for the multiple observations from the same care partner.
Zarit Burden Interview-12 score: 0 to 48 possible; higher score indicates greater caregiver burden and ≥10 considered moderate to high.
PROMIS Anxiety and Depression T scores are population-normed to mean = 50 and SD = 10; higher score indicates greater anxiety or depression.
Adjusted for living with patient, spouse or partner versus other relationship, and care partner’s self-rated general health.
Scores for anxiety and depression from PROMIS-29 were close to population means near 50 and no significant difference between the groups. For anxiety, the means (SD) were 49.2 (9.1) in the team arm and 50.3 (8.8) in the clinician arm; for depression means were 46.7 (7.6) for the team and 48.7 (8.3) for the clinician arm. We estimated unadjusted differences and then adjusted for CP self-rated baseline health, relationship to patient, and living with the patient. After adjustment, the 2.8-point difference in the depression mean was statistically significant, favoring the clinician group; however, this difference is not considered clinically relevant. 30 Results remained not significantly different for burden and anxiety.
Care Partner Outcomes: Association With Goal-Concordant Care
We assessed the association of CP outcomes with their ratings of the extent health care for the patient they help was concordant with the patient’s goals (ie, CPs GCC rating). For this analysis, we included questionnaires from any of the 3 time points for all CPs who completed the initial questionnaire and provided both GCC ratings and CP outcomes.
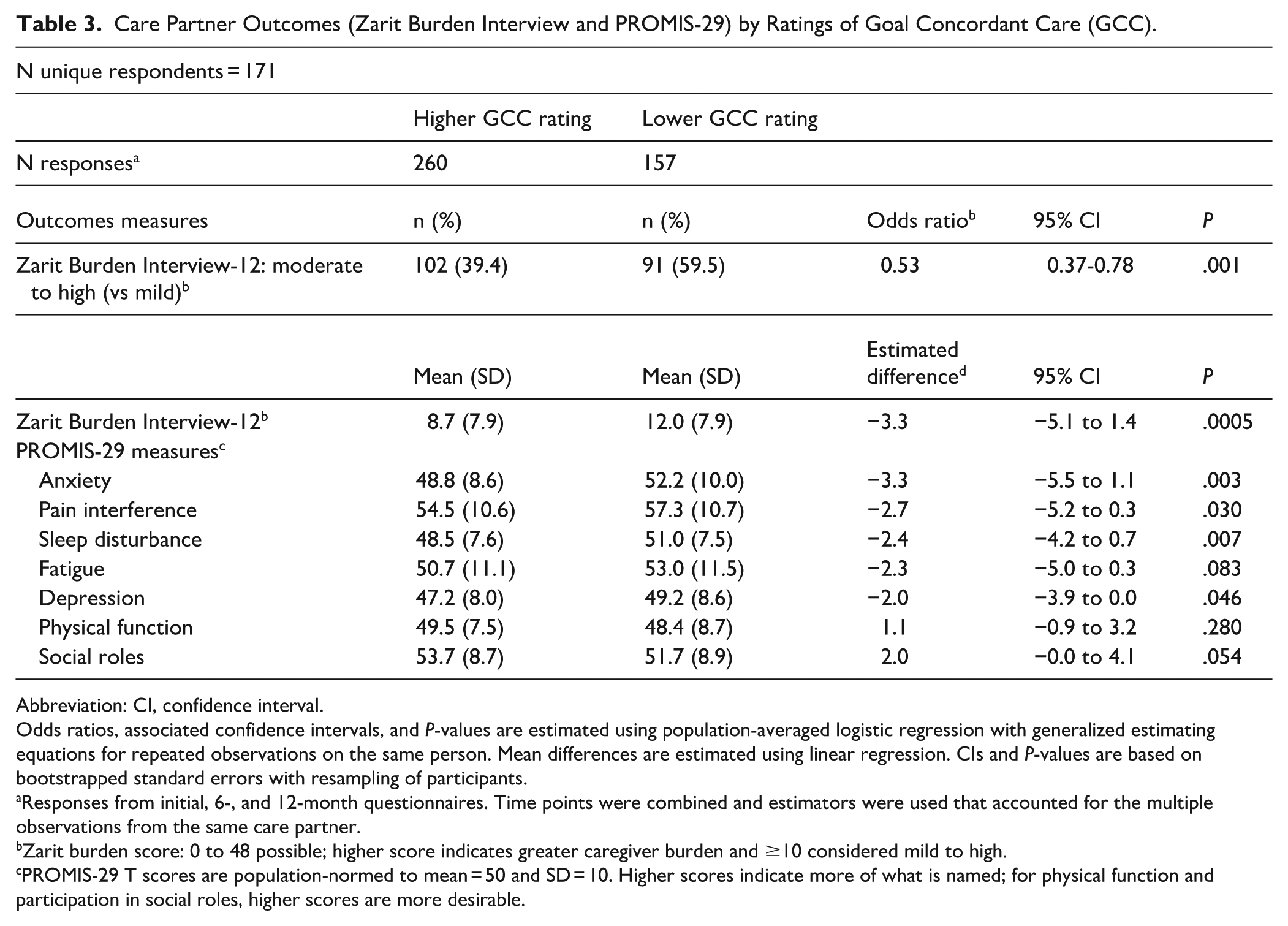
Table 3 compares ZBI-12 and PROMIS-29 outcomes for CPs who rated GCC higher versus lower. Higher GCC ratings were associated with better CP outcomes (Table 3). Fewer CPs who rated care more in line with patient’s goals than CPs who rated care less in line with patient’s goals (39.4% vs 59.5%, P = .0001) reported moderate to high levels of burden on the ZBI-12. On PROMIS-29 measures, CPs who rated patient care as highly goal-concordant reported significantly lower mean scores for anxiety, depression, sleep disturbance, and pain. There were no significant differences in reports of social roles, physical function, and fatigue.
Care Partner Outcomes (Zarit Burden Interview and PROMIS-29) by Ratings of Goal Concordant Care (GCC).
Abbreviation: CI, confidence interval.
Odds ratios, associated confidence intervals, and P-values are estimated using population-averaged logistic regression with generalized estimating equations for repeated observations on the same person. Mean differences are estimated using linear regression. CIs and P-values are based on bootstrapped standard errors with resampling of participants.
Responses from initial, 6-, and 12-month questionnaires. Time points were combined and estimators were used that accounted for the multiple observations from the same care partner.
Zarit burden score: 0 to 48 possible; higher score indicates greater caregiver burden and ≥10 considered mild to high.
PROMIS-29 T scores are population-normed to mean = 50 and SD = 10. Higher scores indicate more of what is named; for physical function and participation in social roles, higher scores are more desirable.
Discussion
The Meta-LARC ACP trial was based on the experience that most serious illnesses are diagnosed months or years before death and there are common trajectories: (1) steady and predictable decline; (2) unpredictable decline with repeating exacerbation; and (3) slow and prolonged decline with frailty.31,32 As serious illnesses progress, tests and treatments can become increasingly burdensome and invasive; some medical care may no longer support what matters most to patients; and the number and complexity of health-related decisions and tasks often increases for both patients and care partners.
Our research assumes that it is possible for healthcare to be a positive force, even as patient life expectancy is limited and CPs face care coordination challenges. Evaluations of care navigator programs, care management, hospice, and palliative care have demonstrated that the last years, months, and days of life can be meaningful, high quality, and comfortable.33-37 Research assessing programs designed to support CPs through education, coaching, respite, care management, and social services have reduced CP burden, improved CP physical and emotional health, minimized regret and conflict, and prevented or delayed withdrawal from the CP role.38-40
The objective of the Meta-LARC ACP Trial was to contribute to knowledge about the effectiveness of different approaches to ACP, with the goal of moving ACP to earlier in the serious illness trajectory before crises and need for end-of-life care decisions, and to help make ACP for people with serious illness routine in primary care. Understanding the impact of ACP using serious illness conversations on CPs contributes to this effort; however, CPs were not required to participate and were not the focus of the trial.
These analyses have limitations. First, assessing the impact on CPs was not the main objective of the trial and CPs were not included for all patients, they are not a random sample, and they may differ from those CPs who did not participate or who were not referred to the study by the patient they help, limiting the generalizability of our findings. Second, our data about CPs involvement in ACP and knowledge of the patient is limited. It does not include information about whether or how frequently CPs participated in discussions with the primary care clinician or team and patient or only received information from the patient. Third, differences in CPs’ outcomes may exist, but our measures may not capture this difference. While they are validated measures of burden and self-reported outcomes, they were not designed to measure increases in CP confidence or decreases in regret expected with ACP. It is also possible we did not follow patients and CPs long enough for serious illness conversations to influence care and burden. Fourth, our assessment of CPs’ perceptions of GCC and outcomes is based on cross-sectional measurements, which means that we can say that GCC ratings are associated with CP outcomes, but we cannot verify the direction of the effect. Our assumption is that CPs who perceive that care matches patient goals will have better outcomes, specifically, less burden, anxiety, and depression. But causality could be in the other direction; that is, it could be people who are less burdened, anxious, or depressed may be more likely to rate patient care as goal concordant.
Despite these limitations, our analyses indicate that team-based and individual clinician-focused SICP approaches yield comparable outcomes for CPs. This suggests that the structure of the intervention—whether delivered by a coordinated team or a single clinician—may be less influential than previously assumed. What does appear to matter, however, is CPs’ perceptions that the care provided to the patient aligns with the patient goals. When CPs believe that the care the patient receives is truly goal-concordant, their own outcomes improve. These findings carry meaningful implications for practice, policy, and research. Clinically, they highlight the need to prioritize conversations and workflows that make patient goals visible, actionable, and consistently integrated into care decisions. From a policy perspective, they point to the value of systems that support goal‑concordant care—such as documentation standards, communication pathways, and accountability measures. For researchers, they suggest that future work should examine not only intervention delivery models but also the mechanisms through which perceptions of goal alignment shape CP experiences and outcomes.
CP involvement in ACP could not only contribute to the future care of the patient, but our results suggest this may be protective for CPs, potentially reducing negative impacts of caregiving. With patient agreement, this supports including CPs in ACP early and often. Medicare allows billing clinicians to engage CPs in ACP, whether the patient can participate or not. Policies could be developed that encourage the use of these billing codes, such as removing co-pays or expanding the clinicians and staff that can contribute to the billable time. Future research should consider collecting more CP data in studies of serious illness conversations or designing studies to specifically examine the impact of ACP on CPs.
Conclusion
We analyzed CP data from a cluster randomized trial of ACP conducted in primary care practices. We found no significant differences in CP outcomes based on whether the practice was assigned to the team-based or individual clinician-focused version of ACP. However, CPs who reported higher GCC for the person they help reported better outcomes including lower burden, anxiety, and depression. These findings suggest that ACP, regardless of how it is implemented, can have a positive impact on CPs.
Footnotes
Acknowledgements
The Meta-LARC ACP Trial Teams includes the named authors and the following people, who served as university investigators, PBRN and clinic leadership, research staff, patient and family advisors, and consultants. Individuals are listed by their university (PBPRN) affiliation. Duke University School of Medicine (PCRC): B. Angeloe Burch Sr., PhD, DD, ThD; Rowena J. Dolor, MD, MHS; Azalea Kim, MD; Kimberly Leathers; Jared Lowe, MD; Jessica E. Ma, MD. Oregon Health & Science University (ORPRN): Angela Combe, MS; David A. Dorr, MD, MS; Lyle J. Fagnan, MD; Elizabeth Fernley, MPH; Shigeko (Seiko) Izumi, PhD, RN, FPNC; Kylie Lanman, BA; Jodi Lapidus, PhD; Susan Lowe; Sean Rice, PhD. Université Laval (QPBRN): Patrick M. Archambault, MD, MSc, FRCPC; Sabrina Guay-Bélanger, PhD, MSc; Georgina Suélène Dofara, MS; Souleymane Gadio, PhD; France Légaré, MD, PhD, C.Q., B.Sc. Arch, MSc, CCMF, FCMF; Jean-Sebastien Paquette, MD, MSc, FRCPC. University of Colorado (SNOCAP): Taryn Bogdewiecz, BA; Ken Dailey; Cat Halliwell, BA; Donald E. Nease, Jr., MD; David Nowels, MD, MPH; Meredith K. Warman, MPH. University of Iowa (IRENE): Gail Drey, BA; Kate Hanrahan, MD, MME, FAAFP; Peter Kim, MD, MPH; Barcey T. Levy, PhD, MD; Olga Petrova, PhD; Keith Provin, MBA; Megan Schmidt, MPH. University of Toronto (UTOPIAN): Michelle Greiver, MD, MSc, CCFP, FCFP; Judy Katz, BSc. Gurnoor Kaur Brar, MPH; Sharon E. Straus, MD, MSc, FRCPC, CM; Kirsten Wentlandt, MD, PhD, MHs, FCFP (PC), FRCPC. University of Wisconsin-Madison (WREN): Deb Constien, BS; Shelbey Hagen, BS; Mary F. Henningfield, PhD; Lisa Sampson, MBA; Sarina Schrager, MD. Institute for Patient and Family-Centered Care: Deborah Dokken; Mary M. Minniti, CPHQ. At-Large: Jacqueline D. Alikhaani, B.A.; Kathy Kastner; James Pantelas; MBA.
Ethical Considerations
IRB approval was obtained from the Trial Innovation Network Single IRB at Vanderbilt University Medical Center (IRB #181084) for US sites, the Centre intégré universitaire de santé et de services sociaux de la Capitale-Nationale of Quebec City, Canada (#MP-13-2019-1526) for Quebec sites; and from the Health Sciences Research Ethics Board of the University of Toronto (protocol #36631) for Ontario sites.
Author Contributions
Concept and Design: Totten, Daly, Michaels, Ramsey. Acquisition, analysis, and interpretation: Totten, Daly, Michaels, Ramsey, Howard, Bumatay, Rogash. Statistical Analysis: Ramsey, Rogash, Totten, Daly. Administrative, technical, or material support: Howard, Bumatay.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this article was funded through a Patient-Centered Outcomes Research Institute® (PCORI®) Award (PLC-1609-36277). The use of REDCap electronic data capture tools in this trial was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under award number UL1TR002369.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
For US sites, a limited data set is available through the PCORI data repository. For Canadian sites, accessing data requires an ethics application submitted to the appropriate Research Ethics Board (REB) due to provincial laws and regulations. In Quebec, applications should be submitted to the Research Ethics Board of the centre intégré universitaire de santé et de services sociaux (CIUSSS) de la Capitale-Nationale in Quebec City. For Ontario, applications should be submitted to the Health Sciences Research Ethics board of the University of Toronto.
Trial Registration
Sponsors Role
The funding organizations had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. This article reports analysis from the Meta-LARC ACP Trial.
