Abstract
Background:
Proton pump inhibitors (PPIs) are frequently prescribed in primary care, often for durations that exceed guideline recommendations. We evaluated whether a digital point‑of‑prescription educational intervention based on the updated March 2023 Deutsche Gesellschaft für Gastroenterologie, Verdauungs- und Stoffwechselkrankheiten (DGVS) GERD guideline could influence the prescribing patterns of primary care physicians (PCPs).
Methods:
We conducted a 12‑month retrospective cohort study using anonymized prescription data from PCPs in Germany. Intervention physicians (n = 886) received EMR‑integrated pop‑up messages with guideline‑based recommendations and optional links to further content on the “Praxiswelt” (Practice World) educational platform. A matched control group (n = 886) received no messages. Physician engagement (click rates), PPI prescription volume, pack size, and use of ICD‑10 diagnosis codes were compared pre‑ (Q2-Q4 2022) and post‑guideline (Q2-Q4 2023). Analyses were descriptive.
Results:
A total of 31 629 pop‑ups were displayed (mean 37.7 per physician); of these, 32.7% generated positive interactions, 0.4% negative, and 66.9% no interaction. PPI prescription volume changed by −2.8% in the intervention group versus +0.7% in controls. Most prescriptions exceeded the recommended 8‑week duration (91%-100%). ICD‑10 coding rates remained low (missing codes: intervention 75.2% pre-guideline vs 76.7% post-guideline; controls 90.0% vs 88.1%).
Conclusion:
Digital pop‑up education at the point of prescription produced only minimal changes in prescribing volume and coding behavior. The limited effect may reflect alert fatigue, passive content, and single‑channel delivery. Future strategies should combine point‑of‑prescription alerts with interactive decision support, audit and feedback, and system‑level changes to improve both adherence and documentation.
Introduction
Proton pump inhibitors (PPIs) are among the most widely prescribed medications globally and are used by nearly 25% of adults to manage the symptoms of gastroesophageal reflux disease (GERD) or related conditions. 1 Although short-term use is generally safe, growing evidence indicates potential adverse effects with prolonged use.2 -5 In Germany, retrospective data show substantial discrepancies between guideline recommendations and real-world PPI prescribing, particularly in the prescription of high doses and long-term therapy without appropriate indications. 6 Similar patterns have also been observed internationally; for example, a U.S. study found that only half of patients on long-term PPI therapy had a valid clinical indication. 7
The updated DGVS GERD treatment guideline (March 2023) emphasizes a stepwise approach starting with alternatives such as antacids, alginates, or H2-receptor antagonists before initiating PPI therapy and limiting therapy to the lowest effective dose for no longer than 4 to 8 weeks in most patients. 8 Comparable deprescribing recommendations have been issued by the American Gastroenterological Association (AGA) 9 and also form part of the relevant Canadian guidelines. 10
Despite these recommendations, overprescription remains widespread in both primary and secondary care.11 -13 Physician prescribing patterns may be shaped by habit, perceived patient expectations, and workflow constraints. 13 Well-designed point-of-care interventions could help reduce inappropriate prescribing.
The objective of this study was to assess whether real-time educational pop-up messages integrated into the EMR could modify PCP prescribing patterns in line with the updated DGVS guidelines.
Methods
Study Design and Objectives
We conducted a 12‑month non‑interventional retrospective cohort study to assess the impact of EMR‑integrated educational pop‑ups on PCP prescribing behavior. The primary objective was to evaluate changes in PPI prescription volume, PPI pack size (duration of therapy), and ICD‑10 diagnosis documentation rates.
The secondary objective was to assess physician engagement with the educational content as measured by click rates on pop‑up messages. Outcomes for the intervention group were compared with those for a matched control group.
Intervention
The intervention consisted of short, non-commercial pop-up messages delivered through the WICOM Assist module within the physician’s EMR. Each message summarized a key recommendation from the March 2023 DGVS GERD guideline and included a hyperlink to related educational articles on “Praxiswelt,” an independent, physician-oriented educational website. Messages could be clicked (“positive interaction”), dismissed permanently (“negative interaction”), or closed without action (“no interaction”).
Messages were tailored to the patient context based on pre-defined inclusion criteria (e.g., age, sex, diagnosis, therapy history). The new DGVS guideline was introduced in the first quarter of 2023, after which content was refreshed quarterly. Messages were triggered only when relevant to the patient record being reviewed.
Study Population
Physicians: The intervention group included 886 PCPs actively using WICOM Assist. The control group comprised 886 matched PCPs from the IQVIA One Panel who did not use WICOM Assist. Matching was performed on the basis of practice size, prescribing habits, and geographic region. All data were anonymized prior to analysis.
Patients: Inclusion and exclusion criteria were derived from recent GERD and PPI deprescribing guidelines and grouped into 5 cohorts: pregnancy, uncomplicated reflux, diagnosed GERD, diagnosed non‑erosive reflux disease (NERD), and PPI non‑responders. Table 1 provides the full criteria.
Inclusion and Exclusion Criteria for Patients Used for the Educational Messages for Physicians.

Ethical Considerations
The local German ethics committee (Landesärztekammer Baden-Württemberg) confirmed that formal ethical approval was not required for this study as it involved the analysis of anonymized prescription data that did not allow for the identification of patients or healthcare professionals. This research was classified as belonging to the “Other Studies” category and was not subject to the German Medicinal Product Act (AMG) or Medical Devices Act (MPG). All data handling procedures complied with German data protection regulations and institutional standards.
Study Period
Prescription and coding data were extracted from anonymized EMR records for Q2-Q4 2022 (pre‑guideline period) and Q2-Q4 2023 (post‑guideline period). Physician engagement was calculated as the proportion of positive interactions with respect to the total pop‑up messages displayed.
Statistical Approach
Given the real-world observational nature of this study and the data format limitations to which it was subject, analyses were primarily descriptive. No formal hypothesis testing was conducted, and results should be interpreted as exploratory findings rather than definitive causal relationships.
Results
Physician Engagement with Educational Messages
Table 2 presents the distribution and engagement patterns for educational messages across patient cohorts in 2023. A total of 31 629 messages were displayed to 886 physicians, averaging 37.7 messages per physician over the study period.
Number and Type of Messages per Physician in 2023 (n = 886 physicians).

Engagement patterns gave rise to the following observations: 32.7% positive interactions (physicians clicked for additional information), 0.4% negative interactions (physicians opted out of future messages), and 66.9% no interaction (physicians closed messages without engaging).
The diagnosed GERD cohort generated the highest message volume (26.2 messages per physician), followed by diagnosed NERD (7.4 messages per physician) and uncomplicated reflux (3.9 messages per physician). The PPI non-responder and pregnancy cohorts had minimal message volumes (0.2 and 0.02 messages per physician respectively).
Notably, engagement rates varied by cohort complexity: PPI non-responder patients showed the highest positive interaction rate (47.3%), followed by uncomplicated reflux (45.0%) and diagnosed NERD (39.1%), suggesting that physicians were more likely to seek additional information for complex or straightforward cases.
PPI Prescription Volume
Figure 1 shows the total number of PPI prescriptions in both groups across the study period. The intervention group showed a minimal decrease from 337,809 prescriptions in Q2-Q4 2022 to 328 478 in Q2-Q4 2023 (−2.8%). By contrast, the control group showed a slight increase from 337 756 to 339 960 prescriptions (+0.7%) over the same period.
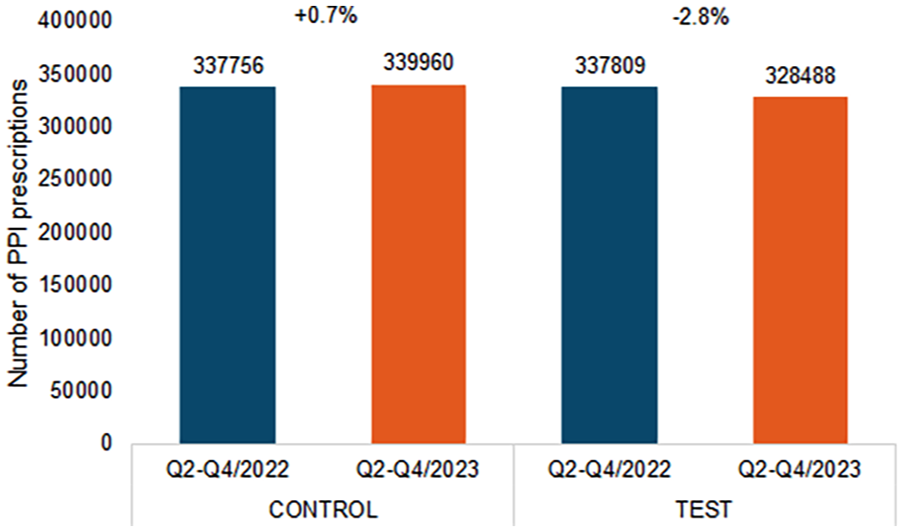
Total number of PPI prescriptions in the intervention and control groups in Q2 to Q4 of 2022 and Q2 to Q4 of 2023.
While this represents a small difference between groups, overall prescription volumes remained largely stable, indicating that the educational intervention had a minimal impact on PPI prescribing behavior in general.
PPI Treatment Duration
Table 3 reveals that the vast majority of PPI prescriptions exceeded the DGVS recommended 8-week treatment duration for both uncomplicated reflux and diagnosed GERD. For uncomplicated reflux, 91%-92% of prescriptions provided more than 8 weeks of therapy, with an average supply of 95-96 days. For diagnosed GERD, 100% of prescriptions exceeded 8 weeks, with an average supply of 100 days.
Proportion of Prescriptions Exceeding the Recommended 8-week Duration and Average Number of PPI days’ Supply (n = 886 physicians).
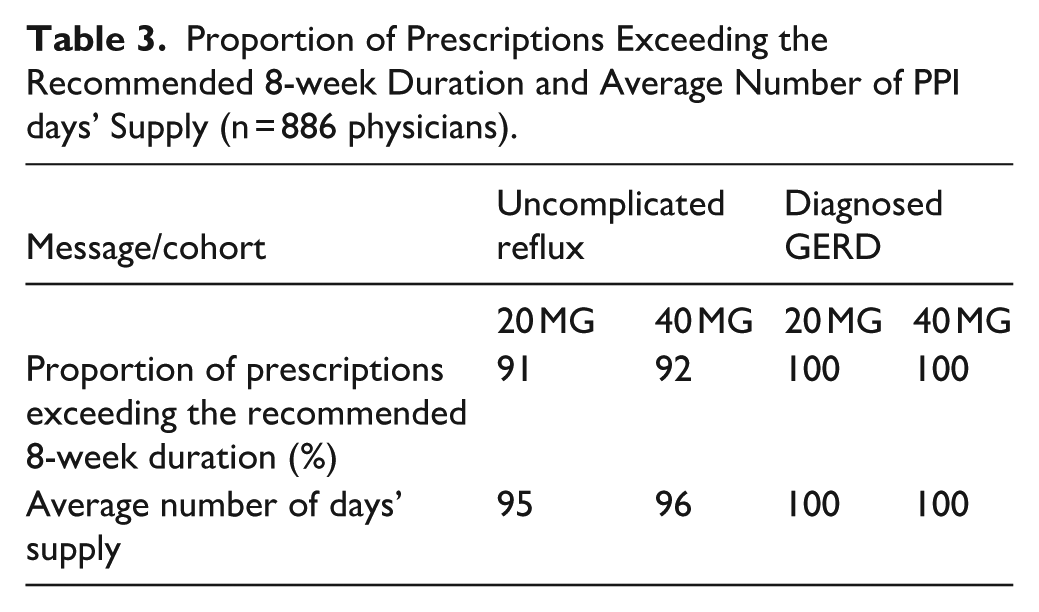
These findings indicate that physicians continued prescribing PPIs for extended durations despite guideline recommendations, suggesting that the educational intervention had a limited impact on treatment duration decisions.
Diagnostic Coding Practices
Figure 2 illustrates the distribution of ICD-10 diagnostic codes used with PPI prescriptions. Missing diagnostic codes remained persistently high in both groups throughout the study period. In the intervention group, missing codes changed minimally from 75.2% in Q2-Q4 2022 to 76.7% in Q2-Q4 2023. The control group showed a slight improvement from 90.0% to 88.1% for missing codes over the same period.
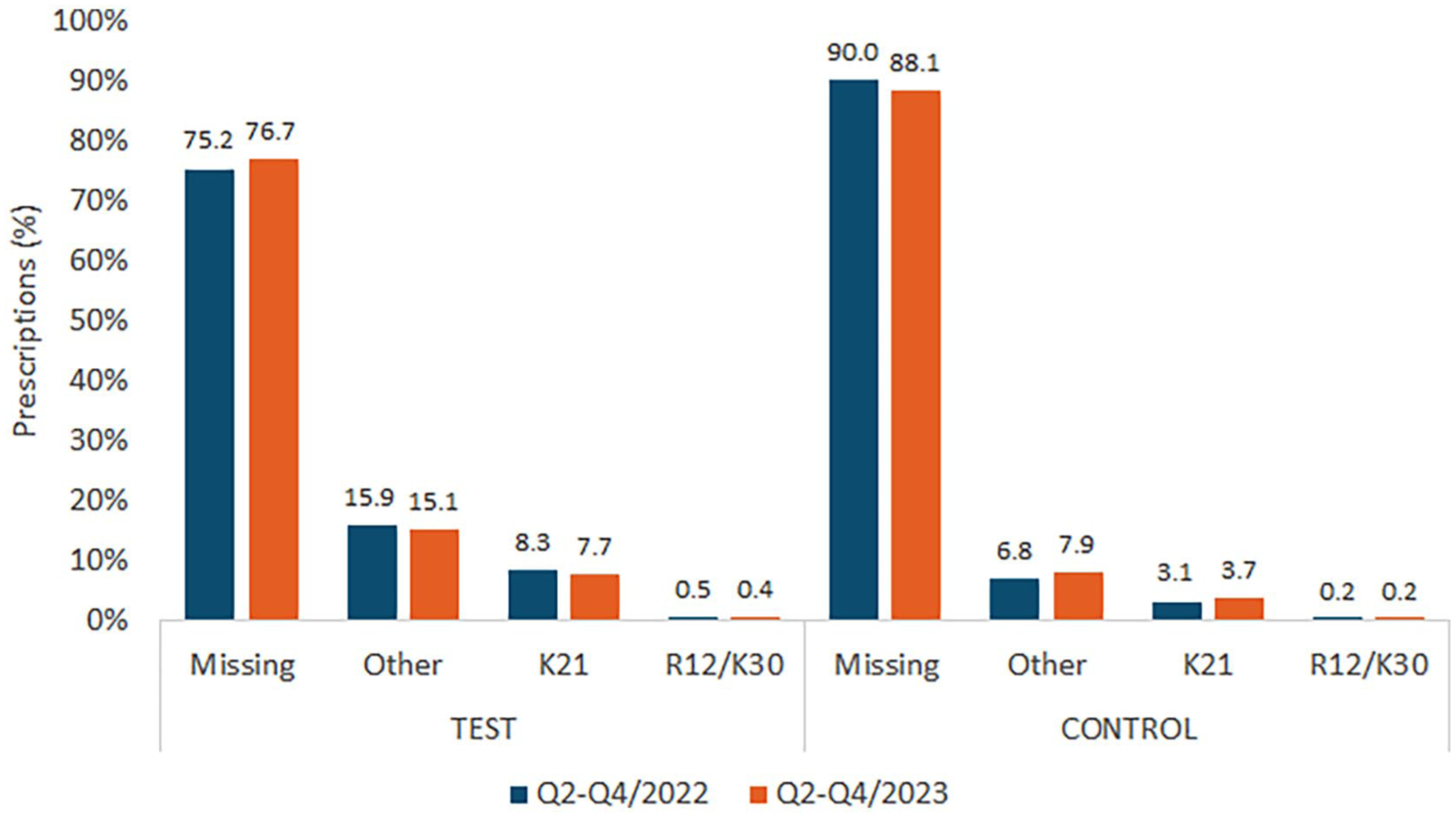
Distribution of ICD-10 diagnosis codes used in the intervention and control groups for prescriptions in Q2 to Q4 of 2022 and Q2 to Q4 of 2023.
The utilization of specific codes (K21 and “other” diagnoses) showed minimal changes in both groups, indicating that the educational intervention did not substantially improve diagnostic documentation practices.
Discussion
Principal Findings
This real-world study evaluated the effectiveness of EMR-based educational messages in modifying PPI prescription practices among German primary care physicians. The intervention resulted in minimal changes across all measured outcomes: a small decrease in prescription volume (-2.8% vs +0.7% in controls), persistent long-term prescription exceeding guideline recommendations, and continued poor diagnostic coding practices.
Limited Intervention Effectiveness
The minimal impact of our educational intervention aligns with growing evidence that simple, passive educational approaches have limited effectiveness in changing established prescribing behaviors. Several factors may explain these findings, including physician engagement patterns, behavioral Inertia, and system-level barriers.
While one-third of messages received positive interactions, two-thirds were ignored, suggesting that pop-up messages may be perceived as workflow interruptions rather than valuable educational tools. The low negative interaction rate (0.4%) indicates that physicians were not actively rejecting the content, but rather passively dismissing it.
Van Gestel et al 13 identified habit and routine as key drivers of PPI prescribing patterns. Our findings suggest that brief educational messages may be insufficient to overcome established prescribing habits, particularly for medications with perceived low-risk profiles such as PPIs.
The persistently high rates of missing diagnostic codes (>75% in both groups) suggest that documentation challenges may stem from EMR usability issues, time constraints, or workflow pressures rather than knowledge gaps alone.
Treatment Duration Patterns
The finding that 91% to 100% of PPI prescriptions exceeded the recommended 8-week durations, with average supplies amounting to 95-100 days, highlights a significant gap between guidelines and practice. This pattern persisted despite educational messaging, suggesting that treatment duration decisions may be influenced by factors other than guideline awareness, such as patient expectations and preferences, convenience of longer prescriptions, concern about symptom recurrence, and lack of systematic review processes for ongoing prescriptions.
Diagnostic Coding Challenges
The minimal improvement in diagnostic coding practices indicates that educational interventions alone may be insufficient to address documentation deficiencies. The persistently high rates of missing codes suggest structural barriers including EMR workflow limitations, time pressure during consultations, lack of automated coding support, and insufficient emphasis on the importance of coding in medical education.
Implications for Future Interventions
Our findings suggest that more comprehensive, multi-faceted approaches may be necessary to achieve meaningful changes in prescribing behavior:
First, future interventions should incorporate case-based learning, decision support tools requiring physician input, and personalized feedback on prescribing patterns.
Second, addressing structural barriers through improved EMR interfaces, automated coding suggestions, and workflow optimization may be more effective than education alone.
Third, single-point educational interventions may require reinforcement through ongoing professional development, peer feedback, and quality improvement initiatives.
Study Strengths and Limitations
This study provides valuable real-world evidence using a large, representative sample of German primary care physicians. The matched design and extended observation period enhance the validity of its findings. The analysis of both prescribing patterns and physician engagement provides valuable insights into intervention mechanisms.
However, the study is also subject to several important limitations that must be acknowledged. The lack of randomization and statistical hypothesis testing limits causal inferences. External factors could not be controlled, and the descriptive nature of the analyses weakens conclusions about intervention effectiveness. Furthermore, the study did not assess clinical outcomes, meaning that the impact of prescription changes on patient health remains unknown. Moreover, the analysis was based on the assumption that dosage was 1 tablet per day, but some patients may use twice-daily regimens, potentially affecting duration calculations. Additionally, the aggregated nature of the data available precluded advanced statistical analyses, including significance testing and multivariable modeling that could have provided stronger evidence with respect to intervention effects. Finally, the 12-month observation period may be insufficient to detect longer-term behavioral changes that could emerge with continued exposure to educational content.
Clinical and research implications
Despite its limited immediate impact, this study contributes important insights for clinical practice and research. Healthcare systems should recognize that simple educational interventions alone are insufficient to change complex prescribing behaviors. Multi-modal approaches addressing knowledge, workflow, and system-level barriers are likely necessary for meaningful improvement.
Future studies should incorporate randomized designs, longer follow-up periods, patient outcome measures, and more sophisticated behavioral change frameworks. Understanding the mechanisms underlying prescribing behavior change remains critical for developing effective interventions.
Conclusion
Educational pop-ups delivered at the point of prescription resulted in minimal changes to PPI prescribing practices and diagnostic coding behavior among German primary care physicians. While the intervention demonstrated feasibility and achieved modest physician engagement, the limited impact suggests that comprehensive, multi-faceted educational approaches addressing system-level barriers may be necessary to achieve meaningful guideline adherence and optimize PPI prescribing practices.
Footnotes
Acknowledgements
None
Authors Note
Karel Kostev is now affiliated to Philipps-University, University Hospital, Marburg, Germany.
Ethical Considerations
German law allows the use of anonymous electronic medical records for research purposes under certain conditions. According to this legislation, it is not necessary to obtain informed consent from patients or approval from a medical ethics committee for this type of observational study that contains no directly identifiable data. Therefore, no waiver of ethical approval was obtained from an Institutional Review Board (IRB) or ethics committee. The authors had no access to any identifying information at any moment during the analysis of the data.
Author Contributions
Joshua Wray, Fergus deGrey-Warter, Erzsebet Horvath, Monika Haering, Patricia Aluko, Kate Plehhova conceptualized the study and critically reviewed the manuscript. Karel Kostev was a major contributor to writing the manuscript. All authors have read and approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Reckitt Benckiser Healthcare Limited, Hull, United Kingdom
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joshua Wray, Fergus deGrey-Warter, Erzsebet Horvath, Monika Haering, Patricia Aluko, Kate Plehhova are employees of Reckitt Benckiser. Karel Kostev is employee of IQVIA.
