Abstract
Introduction:
Patient-centered care and assessing patient experience is essential for improving healthcare quality, particularly for individuals with multimorbidity, who require coordinated and personalized care. Despite its importance, no valid and reliable tool is currently available for measuring patient-centered care for the multimorbid population within the Cantonese cultural context, limiting local healthcare evaluations. This study recruited 500 multimorbid participants from a General Outpatient Clinic in Sha Tin, Hong Kong, to validate the translated and culturally adapted patient-centeredness assessment instrument.
Objectives:
This study aimed to adapt and validate the Cantonese version of the Patient-Centered Primary Care instrument, originally developed by Cramm and Nieboer, for use among Hong Kong residents with multimorbidity.
Methods:
The 35-item Cantonese version of the Patient-Centered Primary Care instrument was translated into Cantonese using standard translation procedures. A cross-sectional telephone survey was conducted from August to November 2023, recruiting 500 middle-aged Hong Kong residents with multimorbidity (≥2 chronic conditions). Confirmatory factor analysis was conducted to test the 8-dimensional structure, while reliability (McDonald’s Omega) and validity (discriminant, convergent, and criterion-related) were assessed.
Results:
Confirmatory factor analysis demonstrated marginally acceptable fit indices: SRMR = 0.073, RMSEA = 0.064, CFI = 0.871, and TLI = 0.856. The discriminant validity was supported (HTMT ratios <0.85). The total Omega value for the multidimensional scale was .91, with total Omega values for each dimension ranging from .79 to .95, demonstrating strong reliability. Convergent validity was confirmed via significant correlations with the Patient Enablement Instrument (PEI;
Conclusions:
The Cantonese version of PCPC instrument demonstrates acceptable reliability and validity for assessing patient-centered care in Hong Kong’s multimorbid population. This tool addresses the lack of relevant assessment measures, provides a practical solution for evaluating patient centeredness in this specific clinical context.
Introduction
Multimorbidity, which refers to the simultaneous presence of multiple chronic diseases within an individual, affects 37.2% of the global population and 35% in Asia.1,2 Strongly linked to age, its prevalence is increasing worldwide, particularly in economically advanced regions like Hong Kong, a population with one of the world’s highest life expectancies, 3 resulting in higher rates of multimorbidity.2,4 Over a 9-year period in Hong Kong, characterized by a significant aging population, multimorbidity rates rose from 14.0% in 2009 to 18.9% in 2013, reaching 24.1% by 2018.5,6 The intricate nature of managing multiple chronic conditions presents a formidable obstacle for healthcare infrastructures,7-9 as evidence suggests that individuals with multimorbidity are more susceptible to early death and increased healthcare expenditures,10,11 affecting both physical and mental well-being.12,13 However, it has been observed that robust primary healthcare can mitigate some of the adverse effects associated with multimorbidity. 14
Within the array of primary care frameworks proposed for patients with multimorbidity, the concept of patient-centered care stands out as a critical quality component.15,16 This approach is grounded in recognizing each patient’s uniqueness and specific health needs.17,18 The most recognized framework of patient-centered care was put forth by Mead and Bower, outlining 5 key interconnected elements: a biopsychosocial perspective; “patient-as-person”; a balanced distribution of power and responsibility; a strong therapeutic partnership; and “doctor-as-person.” 19 This framework refines ideas from earlier, broader discussions on the topic, including a detailed qualitative analysis conducted by Harvard School of Medicine in 1993. 20
When it comes to caring for patients with multimorbidity, as opposed to those with a single chronic ailment, there is a need for more thoughtful and distinct approaches in service design, delivery, and the clinician-patient dynamic. 21 This is to ensure a high level of patient-centered care that addresses the complex clinical needs.22,23 Consequently, it is logical for care models targeting multimorbidity to strive for optimal patient-centeredness.24,25 This necessitates the development and refinement of assessment tools to measure the effectiveness of patient-centered care in this context.
Numerous tools have been established and authenticated for the general assessment of patient-centeredness.26-29 However, there was a notable gap in such tools tailored for the multimorbid demographic. This changed in 2018 when Cramm and Nieboer introduced a pioneering instrument within the Dutch community in Tilburg: The Patient-Centered Primary Care (PCPC) Instrument.
30
This tool draws inspiration from 8 distinct facets of patient-centeredness identified in a seminal 1993 qualitative study.
20
Comprises 36 items, these facets include “respect for patients,” “values, preferences, and expressed needs,” “provision of information and education,” “access to care,” “emotional support to relieve fear and anxiety,” “involvement of family and friends,” “continuity and secure transition between health care settings,” “physical comfort,” and “coordination of care.” Each facet is represented by 3 to 7 specific statements, rated on a 5-point Likert scale, such as “My medical information is accurately transferred with referrals.” The instrument demonstrated high reliability (Cronbach’s alpha = .89 for the entire scale, and ranged from .72 to .92 for each dimension) and significant validity (correlating with the Satisfaction with In-patient Stroke Care (SASC) scale at .53,
Assessing patient experiences is now acknowledged as a critical component in evaluating the quality of healthcare services, alongside factors like efficacy and safety. 34 To address this, the Hong Kong Hospital Authority has implemented the Patient Experience Survey (PES) for better understanding patients’ experiences and feelings who received healthcare services and launch continuous service improvement. 35 However, the 3 validated PES instruments currently used by Hong Kong’s Hospital Authority,36-38 mainly examined patient experience instruments for inpatient and specialist outpatient settings in Hong Kong, which may not be entirely suitable for primary care settings. Though the locally validated Person-Centered Primary Care Measure (PCPCM) from Tse et al, 39 assesses general patient-centeredness, it lacks specificity for monitoring complex multimorbid care needs. Consequently, the multidimensional PCPC instrument remains essential to address critical gaps, particularly in assessing actionable domains integrating physical, emotional, and social needs. This study will first translate the PCPC instrument into Cantonese and adapt it to Hong Kong’s culture to suit the local primary care system. Then, the psychometric properties of the Cantonese version of Patient-Centered Primary Care (Can-PCPC), including its reliability and validity, will be verified using responses collected from patients with multimorbidity in Hong Kong primary care settings. Critically, participants must be fluent Cantonese speakers, aged 40 years or older, diagnosed with more than 2 chronic conditions, and have received primary care for chronic conditions for at least 12 months since their first visit. Consequently, the Can-PCPC developed from this proposed project may serve as a supplemental tool for monitoring patient experience.
Methods
Translation and Cultural Adaption
The Cantonese version of PCPC was developed using standard translation procedures. 40 Two independent professional translators conducted forward- and back-translation to create an initial draft of the Cantonese version. Then, the initial draft was reviewed and discussed by an expert panel comprising 3 physicians and 1 nurse practicing in family doctor clinics, 1 epidemiologist expert in population health statistics, 1 professor expert in primary healthcare settings. Six experts rated the degree of content validity of each of the items using the 4-point scale (1 = irrelevant, 2 = somewhat relevant, 3 = quite relevant, and 4 = highly relevant) and provided comments for modification if the wording or content of the items was inappropriate or inaccurate.
We conducted 2 rounds of the expert panel to identify discrepancies. Feedback from the first round was gathered to address the dispute items and refine the translation. In this stage, to address linguistic accuracy, conceptual equivalence, and cultural relevance. This involved: (1) Localizing language to natural Cantonese (eg, “
A revised translated version was then formed for further evaluation. During the second expert panel, this revised instrument achieved a content validity index (CVI) above or equal to 0.83 for all items. Then, using the revised version, we conducted the cognitive debriefing in 10 patients who met the following inclusion criteria to assess the clarity and interpretation of each item, resulting the final version of the Cantonese Patient-Centered Primary Care (Can-PCPC; Supplemental Appendix eTables 1 and 2) for the subsequent psychometric evaluations.
Study Design and Data Collection
We conducted a cross-sectional telephone survey at a general outpatient clinic (GOPC) in Sha Tin, Hong Kong, from August to November 2023, using a convenience sampling strategy. GOPC staff first identified eligible patients with clinically diagnosed multimorbidity (≥2 chronic diseases) based on study criteria and medical records. After obtaining verbal consent during clinical visits, staff provided patients’ contact information to the research team for recruitment. All procedures followed approved ethics documentation, ensuring patient confidentiality. Survey responses were collected via 15- to 20-min phone calls conducted at participants’ convenience, eliminating scheduling and location constraints. All telephone interviewers received standardized training on protocols and questionnaire administration prior to data collection. To ensure accuracy and consistency, we implemented a multi-step process: (1) Protocol-specific training for all research staff; (2) Daily automated data checks using custom R scripts for value range errors; and (3) Coordinator verification comparing entered data against original records with manual outlier detection. This approach minimized errors and preserved data integrity. The inclusion criteria were as follows: (i) aged 40 years or older; (ii) received primary care from the clinic for their chronic conditions for at least 12 months (since the first visit for the same conditions); (iii) diagnosed with at least 2 or more chronic conditions (ie, multimorbidity); and (iv) proficient in Cantonese. These criteria ensured a culturally and clinically appropriate sample for validating the instrument within Hong Kong’s primary care context. The inclusion criteria targeting adults aged ≥40 years was established because Hong Kong’s multimorbidity epidemiology shows a negligible burden in those under 50 years: only 0.9% of people with 2 chronic conditions and 0.1% with 3 condition were <50 years, with no cases of ≥4 conditions in this age group7. This pattern justifies focusing on ≥40 years to capture the earliest at-risk transition phase while excluding populations irrelevant to local multimorbidity. Furthermore, since patients visiting Hong Kong’s GOPCs are typically older, this age criterion enhances recruitment feasibility by targeting the clinic’s predominant demographic. While the second criteria requiring more than 12 months since first visit ensures participants have sufficient time for managing clinic visits. A total of 990 telephone survey attempts were made, 291 calls resulted in non-response, 166 individuals declined participation, and 533 respondents provided informed consent (with 500 completing full interviews). Consequently, the overall response rate was calculated to be 50.5%. Although 533 individuals provided consent, the recruitment was stopped once the pre-defined sample size of 500 participants was achieved. Participants were asked to complete a structured questionnaire comprising socioeconomic and demographic items, the Can-PCPC, and the Patient Enablement Instrument (PEI). 42
Among 500 enrolled telephone survey participants, 409 provided complete answers, while 91 responded with “Not Applicable.” All 500 responses were included in the final analysis. Number and the proportion of the “Not Applicable” answers are presented in Table 2. Additionally, a follow-up survey was conducted with 50 out of the 500 participants initially recruited who provided full answers with no “Not Applicable” responses, 2 weeks after the main round of the survey. Participants received $50 supermarket coupons as incentives in each round survey.
Measures
Patient-Centered Primary Care Score
Patient-centeredness is a biopsychosocial approach focused on delivering respectful and individualized care, which emphasizes encouraging individuals to actively participate in their care while ensuring their needs and preferences are met. 43 Following the standard translation procedure and cultural adaptation of the 36-item Patient-Centered Primary Care (PCPC) instrument, the translated Cantonese version comprises 35 items organized into 8 factors, aligning with the institute’s 8 principles of patient-centered care: (1) Patients’ Preferences (7 items); (2) Physical Comfort (5 items); (3) Coordination of Care (4 items); (4) Continuity and Transition (4 items); (5) Emotional Support (4 items); (6) Access to Care (4 items); (7) Information and Education (4 items); and (8) Family and Friends (3 items). Responses were measured on a 5-point Likert scale, with options ranging from 1 (totally disagree) to 5 (totally agree). Mean PCPC scores were calculated as the average of all 35 items (range = 1-5), where higher average score indicates that the medical care the individual received is perceived to be more patient centered.
Patient Enablement Instrument Score
This study also assessed patient enablement using the Chinese version of the Patient Enablement Instrument (PEI), which measures the effectiveness of primary care consultations, 42 was included to assess the criterion-related validity. The PEI, originally developed by Howie et al, 44 evaluates patients’ sense of empowerment following consultations with their primary care clinicians. This instrument captures a direct, patient-reported outcome of successful patient-centered interactions and represents the core objective of the patient-centered care. The Chinese version, adapted through translation and cultural validation, consists of 6 items: (1) able to cope with life, (2) able to understand your illness, (3) able to cope with your illness, (4) able to maintain your health, (5) confident about your health, and (6) able to help yourself. Responses are scored as 2 for “much better/much more,” 1 for “better/more,” and 0 for “same or less,” resulting in a total PEI score ranging from 0 to 12. The Chinese PEI score is calculated by taking the mean of applicable item scores and multiplying by 6. According to Lam et al, the Chinese version of the PEI demonstrated strong acceptability (response rate = 83%), and reliability (Cronbach’s alpha = .84) among Chinese patients attending public primary care clinics in Hong Kong. Given its established validation in Hong Kong and conceptual alignment with the Can-PCPC, the PEI was selected to measure criterion-related validity.
Number of Chronic Conditions and Number of Medical Specialties the Subjects Receive Care From
The criteria-related validity of the Cantonese version of the PCPC was assessed by examining its correlations with 2 external criteria to further evaluate its effectiveness. First, the PCPC scores were correlated with the number of chronic conditions of each individual. This was based on the assumption that patients with a higher number of chronic diseases may require more comprehensive care from the primary care settings, 45 then this would be reflected in their PCPC scores. Second, the association between PCPC scores and the number of medical specialties subjects received care from was investigated. This criterion was included to assess whether individuals seeking care from multiple specialties, which is correspond with more complex healthcare needs, would score differently on the PCPC, potentially reflecting the tool’s sensitivity.
The number of chronic diseases and medical specialties for each participant was operationally defined based on 43 chronic conditions across 15 categories, as adopted in previous local multimorbidity studies. Chronic disease conditions among primary care patients from GOPCs were confirmed using both medical records from the public clinical management system and patients’ self-reports. These categories include: metabolic diseases; cancer; cardiovascular diseases; respiratory diseases; hepatobiliary conditions; gastrointestinal disorders; musculoskeletal and connective tissue diseases; genitourinary conditions; ear, nose, and throat disorders; visual system diseases; dermatological conditions; hematological disorders; neurological diseases; mental health conditions; and other chronic conditions. 46 An experienced nursing staff applied a standardized process to extract and clean participants’ medical histories of participants, resulting in : (1) individual chronic condition lists; (2) total chronic disease counts; and (3) specialist clinical utilization.
Statistical Analysis
The demographics were presented as number (percentage). The PCPC scores were displayed as mean ± SD (Standard deviation) and 1-way ANOVA (Analysis of variance) was applied to evaluate the difference between groups (age, sex, and education level). The PCPC scores were reported with mean ± SD and ceiling/floor effects. The item-dimension correlation analysis was conducted to confirm the contribution of each item to the concept.
Confirmatory Factor Analysis
We conducted a confirmatory factor analysis (CFA) to examine the factor structure and dimensionality of the Cantonese version of PCPC. To address the missing data, we used full information maximum likelihood (FIML) approach, which utilizes all available data points to produce unbiased model parameter estimates. 47 The key aspects of the model, including model fit indices, factor loadings, and the correlation between factors, were calculated to assess the construct validity of the Can-PCPC instrument. 48 We used the acceptable cut-off criteria from Hu and Bentler 49 :
standardized root mean square residual (SRMR ≤ 0.08)
root mean square error of approximation (RMSEA ≤ 0.06),
comparative fit index (CFI ≥ 0.90), Tucker-Lewis index (TLI ≥ 0.90)
Item factor loadings ≥0.50 were generally considered acceptable. 50 The discriminant validity of the Can-PCPC was assessed using the Heterotrait-Monotrait (HTMT) ratio/. 51 A threshold of 0.85 was used for the HTMT ratio to indicate acceptable discriminant validity, an HTMT value exceeding this cut-off suggests that the constructs are not sufficiently distinct.
Reliability
We calculated McDonald’s omega to estimate the internal consistency reliability of Can-PCPC, which is a generalized form of coefficient alpha, and is considered more appropriate than alpha for multidimensional scales. 52 Given the 8-dimensional structure of the instrument, we computed both the omega total (ωt) and omega hierarchical (ωh) for the overall scale and the omega total (ωt) for each factor, as these are the primary forms of coefficient omega. 53 An acceptable value for these reliability indices is typically considered to be no less than .7. 54 To examine test-retest reliability, we used the 2-week follow-up PCPC scores and calculated intraclass correlation coefficients (ICC). ICC values falling in the range of .60 to .80 are considered to indicate good reliability. 55
Criterion-Related Validity and Convergent Validity
To assess criterion-related validity, we calculated Pearson correlation coefficients between the PCPC scores and (1) the number of chronic conditions and (2) the number of medical specialties the subjects receive care from. This analysis was grounded in the hypothesis that patient-centeredness diminishes with greater clinical complexity.
56
We further evaluated convergent validity by estimating the correlation between PCPC scores and PEI scores. Correlation coefficients for each PCPC dimension (across the 3 indicators) are denoted as
The analysis was performed using R 4.2.2. A
Results
Characteristics of Participants
Table 1 displays the characteristics of 500 participants. The majority of participants were female (77.2%), while 22.8% were male. The average age of all participants was 73.0 years (SD = 5.6), ranging from 58 to 93 years. Using the Hong Kong Census and Statistics Department age classification,
59
the largest groups were 70 to 74 years (33.8%), 65 to 69 years (27%), and 75 to 79 years (22%), showing the age distribution align with the original PCPC validation sample. The mean Can-PCPC score of all participants was 3.35 ± 0.63 (range = 1-5). Females scored significantly higher than males (
Demographic Characteristics and Mean Can-PCPC Score for Participants.
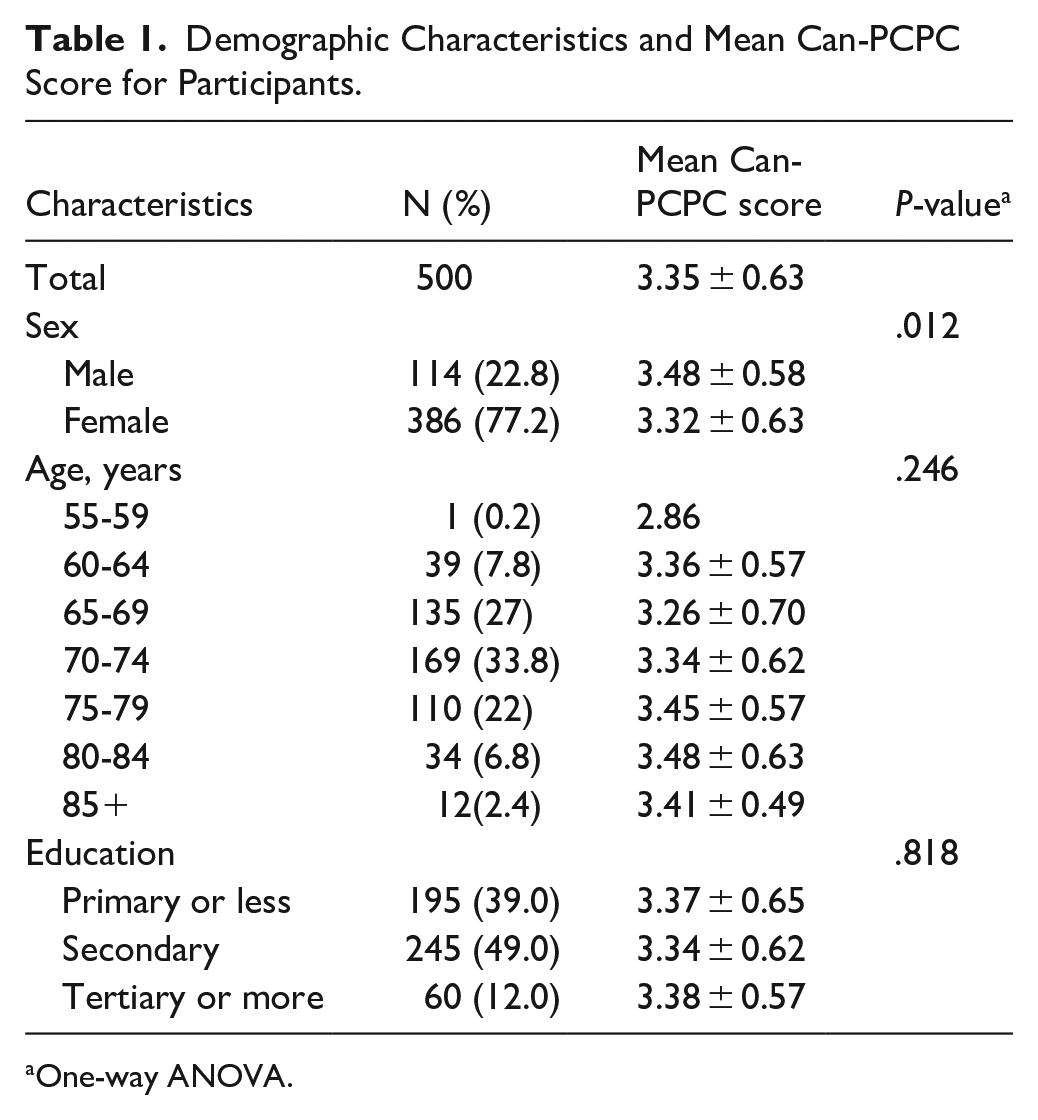
One-way ANOVA.
Item Statistics
Table 2 shows that item 23 (“I was made aware of the possibilities for more intensive emotional support.”) was rated as the most difficult item with a mean score and standard deviation of 2.38 ± 1.41, whereas item 18 (“With a referral, all my information was passed on correctly.”) was rated as the easiest item with a mean score and standard deviation of 4.08 ± 0.98. Five items across 3 domains (item 16, 21, 22, 23, and 31) showed floor effects, ranged from 21.6% to 43.7%. Ten items across 6 domains, including patients’ preference, physical comfort, coordination of care, continuity and transition, access to care, and family and friends (item 1, 6, 7, 10, 14, 15, 17, 18, 25, and 33) showed ceiling effects, ranged from 21.3% to 39.1%. The item-dimension correlation coefficients were all greater than or equal to .65, except for .61 for item 3 (“I was involved in decisions about my treatment.”) and .63 for item 12 (“In the treatment room(s) and at the counter there was sufficient privacy.”).
The Item Statistics and the Factor Loadings of the Cantonese Version of PCPC.

Model Fit and Discriminant Validity
The confirmatory factor analysis (CFA) tested an 8-dimensional model using the full information maximum likelihood method. Table 3 shows the model fit indices were evaluated to assess its structure. The standardized root mean square residual (SRMR = 0.073) fell within the acceptable range (SRMR ≤ 0.08). However, other indices, including the root mean square error of approximation (RMSEA = 0.064, 90% CI = [0.061, 0.068]), comparative fit index (CFI = 0.871), and Tucker-Lewis Index (TLI = 0.856), failed to meet recommended thresholds (RMSEA ≤ 0.06, CFI ≥ 0.90, and TLI ≥ 0.90), indicating that the model required improvement.
Model Fit of the Cantonese Version of PCPC (CFA Results). a

Abbreviations: AIC, Akaike information criterion; BIC, Bayesian information criterion; CFI, comparative fit index; RMSEA, root mean square error of approximation; SRMR, standardized root mean square residual; TLI, Tucker-Lewis index
Model 1: including all 35 items
Model 2: including 30 items (removed items with a factor loading less than 0.5, ie, item 3, 8, 9, 24, and 25)
The confirmatory factor analysis was conducted using full information maximum likelihood (FIML) to address the missing variables.
All standardized factor loadings were statistically significant, confirming the items’ contributions to their respective constructs. Although most factor loadings exceeded the recommended threshold of 0.5, 5 items – item 3 (0.476), item 8 (0.452), item 9 (0.353), item 24 (0.489), and item 25 (0.496) – fell below this cutoff. After removing these items, the revised 8-dimensional model with 30 items demonstrated improved fit indices, with CFI = 0.925, TLI = 0.913, RMSEA = 0.055 (90% CI = [0.050, 0.059]), and SRMR = 0.049. All factor loadings in the revised model exceeded 0.5, providing strong evidence for the validity of the latent constructs. Thus, the CFA results support the hypothesized structure and dimensionality of the model.
While the revised 30-item model demonstrated a superior fit, the items with low factor loadings primarily focus on patient concerns and shared decision-making, which are essential components of patient-centered care. Based on expert feedback, the theoretical significance of the 5 low-loading items was emphasized. Although the model indices are less satisfactory, the RMSEA value <0.08 indicates a reasonable error of approximation, and the CFI and TLI values are close to the threshold, suggesting a moderately acceptable fit, particularly in the context of culturally adapted instruments. 60 Sensitivity analyses confirmed that the 2 models are generally consistent. Therefore, consistent with cases in previous cross-cultural research,60-62 the original 35-item scale was retained to ensure the instrument’s theoretical completeness and cross-cultural consistency, therefore prioritize conceptual integrity over statistical optimization, aligning with expert recommendations that advocate preserving complete item sets when reduction risks compromising the instrument’s construct representation. Subsequent analyses were conducted using the 35-item model.
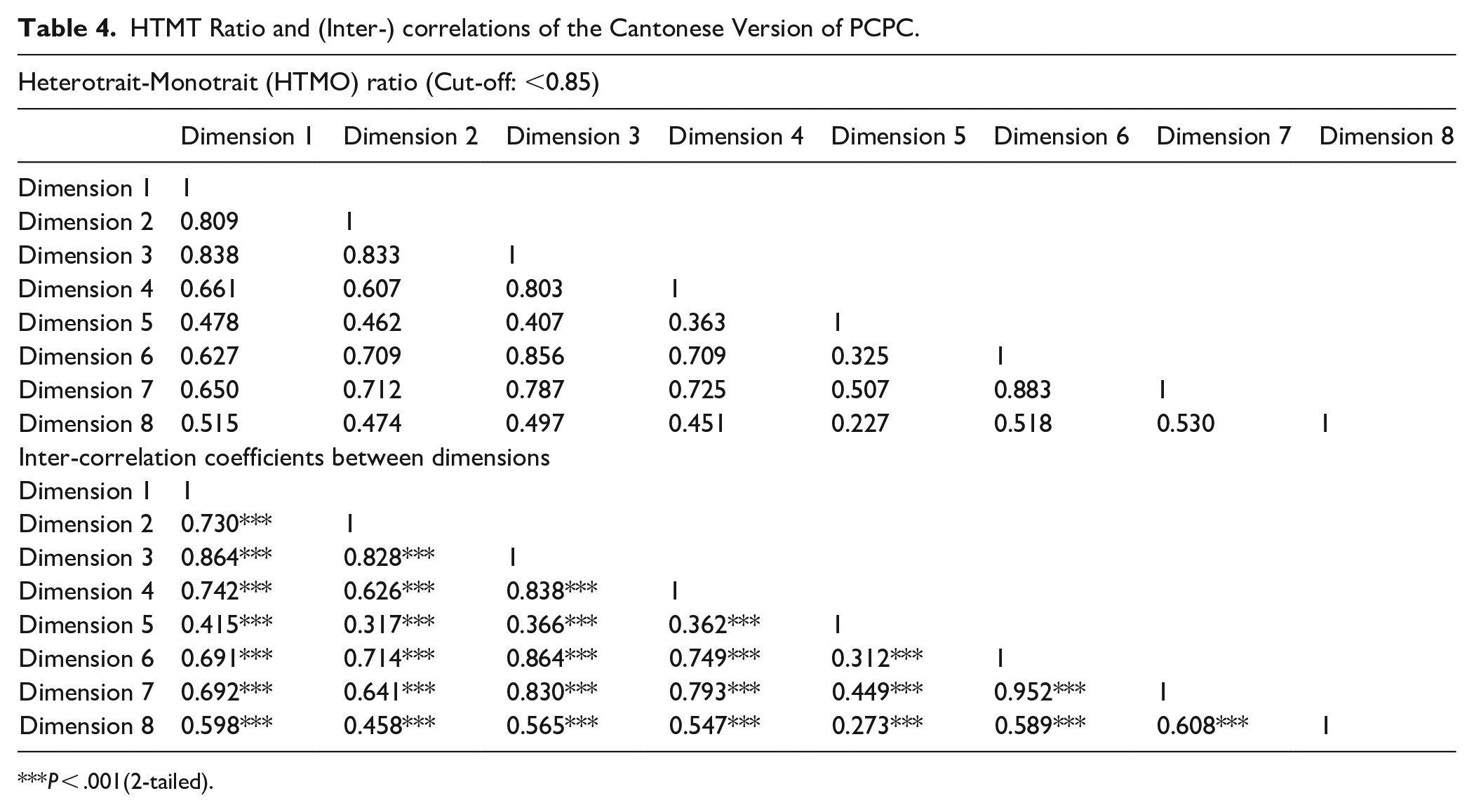
The HTMT results, which are shown in Table 4, indicated that most constructs demonstrated acceptable discriminant validity, with values below that recommended cut-off of 0.85. The HTMT value between Dimension 6 and Dimension 7 (0.883) exceeded this threshold, suggesting that these 2 constructs are not sufficiently distinct. Therefore, the model demonstrates acceptable discriminant validity for most dimensions.
HTMT Ratio and (Inter-) correlations of the Cantonese Version of PCPC.
Reliability
The reliability of the scale is presented in Table 5. The total Omega (ωt) for the multidimensional scale was .91, indicating excellent internal consistency. This suggests that the dimensions in the scale are highly interrelated and measure the same underlying construct reliability. Additionally, the total Omega (ωt) and hierarchy Omega (ωh), which represent the overall reliability for the total 35 items and the proportion of variance attributable to a general factor, was .96 and .78, respectively. The reliability of each dimension was also examined, the total Omega values for each dimension ranging from .79 to .95, indicating all dimensions demonstrated good reliability.
McDonald’s Omega and Test-retest Reliability for the Cantonese Version of PCPC.

Based on the 8 dimensions (multidimensional composite score). The McDonald’s Omega Total value for the overall 35 items was 0.96. The Omega Hierarchical value for the 35 items was 0.78.
For test-retest reliability, among the 35 items, 21 fell within the range of moderate reliability (ICC = .50-.75) range, while the remaining 14 items scored below .50. 63 Among these, 7 items fell between .40 and .50, while the item showing the worst test-retest reliability is item 30 (“The information I received was well explained.”), with an ICC of .22.
Criterion-Related Validity and Convergent Validity
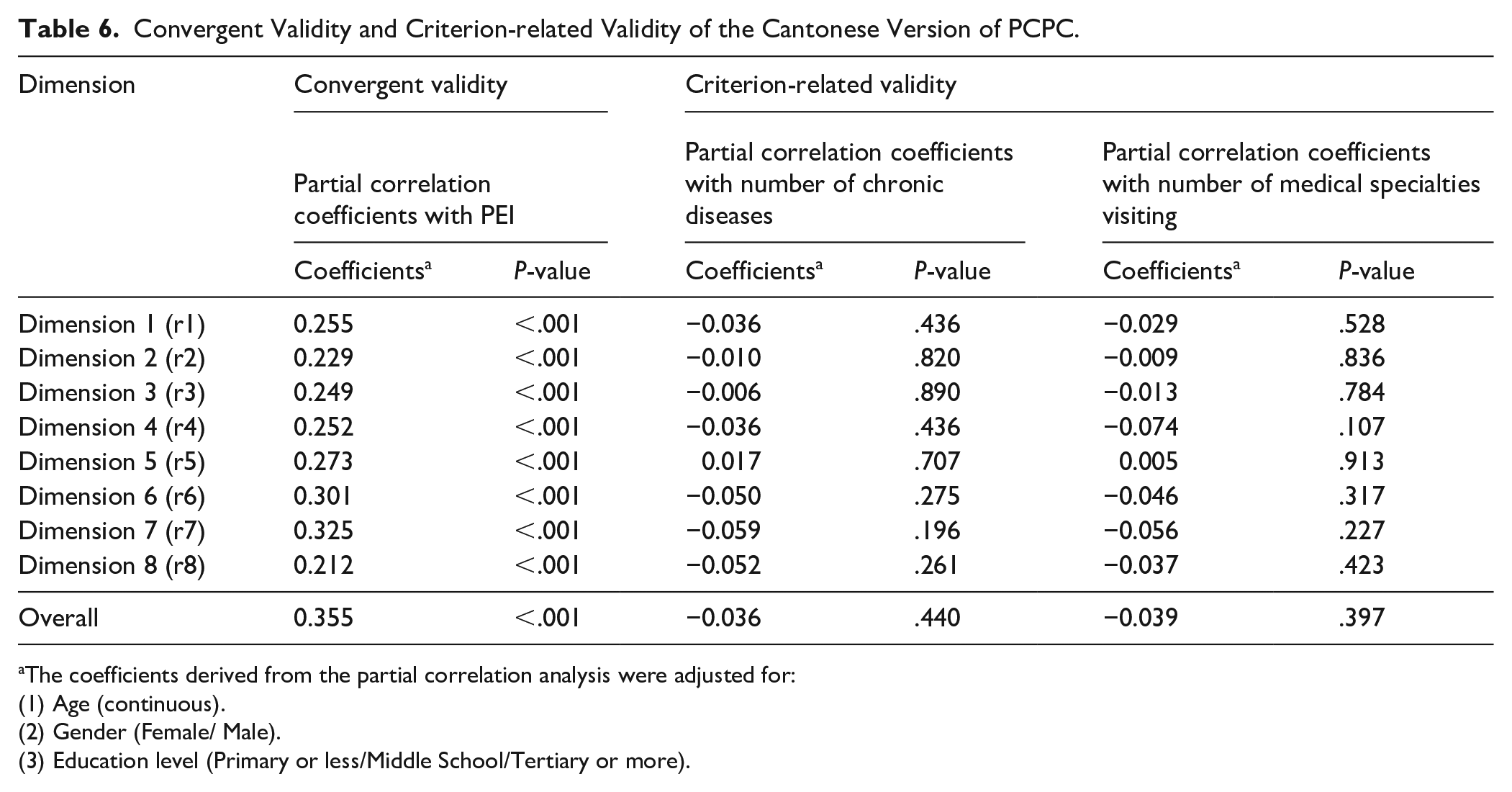
Number of Chronic Conditions and Number of Medical Specialties the Subjects Receive Care From
The scale’s criterion-related and convergent validity results are summarized in Table 6. After adjusting for age, gender and education level, the partial correlations between each PCPC dimension and the number of chronic conditions were as follows:
Convergent Validity and Criterion-related Validity of the Cantonese Version of PCPC.
The coefficients derived from the partial correlation analysis were adjusted for:
(1) Age (continuous).
(2) Gender (Female/ Male).
(3) Education level (Primary or less/Middle School/Tertiary or more).
Patient Enablement Instrument Score
The Can-PCPC scores demonstrated a statistically significant positive correlation with PEI scores (
Discussion
In this study, we evaluated the psychometric properties of the Cantonese version of the PCPC among Hong Kong residents with multimorbid chronic conditions, following standard translation and cultural adaptation procedures. CFA was conducted based on the original 8-dimensional model, the model fit and discriminant validity were assessed. Additionally, reliability was assessed using McDonald’s Omega, the criterion-related validity and convergent validity was examined using Pearson partial correlation coefficients.
It is important to note that Hong Kong has one of the world’s fastest-aging populations, with 30% of residents projected to be over 65 by 2039, 64 chronic diseases are highly prevalent, approximately 24.1% of Hong Kong’s population required long-term medical treatment for chronic diseases, 65 yet primary care remains fragmented, as over 70% of chronic disease patients rely on hospital-based care. 66 This reliance often leads to challenges in accessibility and dissatisfaction due to insufficient attention to patients’ needs by healthcare providers. Furthermore, mental health needs are frequently overlooked in Hong Kong’s current primary care system. 67 No valid psychometric instrument currently exists to assess patients’ mental health needs in primary care settings. Although some “Not Applicable” responses occurred, the Can-PCPC includes a dimension with 4 items measuring emotional support provided by primary care clinics, addressing this gap in assessment. Given these challenges, a patient-centered primary care model is essential for effectively managing chronic conditions.
The adapted 8-dimensional framework demonstrated suboptimal fit with our dataset. While the SRMR values indicated adequate alignment, RMSEA, CFI, and TLI indices fell marginally below established thresholds, suggesting discrepancies between our data and the Dutch patient responses used in prior validation. Although removing 5 items from the Can-PCPC improved model fit slightly, the 35-item model retained indices near the margin of acceptable approximation. To preserve the instrument’s theoretical integrity and ensure it captures the nuanced dimensions it was designed to assess, we retained the full 35-item scale for subsequent analyses. Following the precedents in previous studies, this decision prioritizes cross-cultural comparability and instrument integrity despite improvements in model fit from removing low-loading items.60-62 Retaining the fill scale acknowledges cultural adaption needs and maintaining the original construct’s comprehensiveness.
The reliability of the overall scale and its dimensions was assessed using McDonald’s Omega. The total Omega values for each dimension ranged from ω = .79 (Dimension 5, Access to Care) to ω = .95 (Dimension 1, Patients’ Preference), all of which exceeded the threshold of .7, indicating strong reliability. These results align closely with Cramm and Nieboer’s prior validation study, reporting Cronbach’s alpha equals to .89 for the full scale, and .72 to .92 across dimensions. 30 While both studies demonstrate robust reliability across dimensions, the current analysis, employing McDonald’s Omega, offers a more nuanced understanding of the scale’ hierarchical structure. Specifically, it distinguished between the general factor and the specific dimensions by reporting multidimensional construct reliability values, as well as the total Omega and hierarchical Omega for all items. This approach provides deeper insights into the relative contributions of the general factor and the individual dimensions, enhancing the interpretability of the scale’s reliability. Convergent validity was further supported by significant positive correlations between PCPC scores and PEI scores, confirming that PCPC measures similar patient-centered care concepts as other validated instruments across linguistic and cultural contexts – as demonstrated by the Korean PCPC adaption and the patient-centeredness measurement tool for inpatients in Korea. 48
The unsatisfactory CFA results may reflect systemic and cultural differences in chronic disease management between Hong Kong and the Netherlands. First, despite both regions being high-income with advanced medical systems, their healthcare funding models diverge: the Netherlands employs social health insurance, whereas Hong Kong relies on a mixed public-private system. 68 In Hong Kong, patients dependent on General Outpatient Clinic (GOPCs) for chronic disease management often face limited accessibility and less patient-centered care compared to the Netherlands, 69 where the original PCPC study was conducted. Second, cultural norms in Hong Kong’s healthcare system may influence patient expectations. Historically, patients have preferred doctor-directed care over patient-centered appropraches,70,71 potentially diminishing the perceived relevance of certain PCPC items. Third, Hong Kong’s cultural habits and patient preferences drive widespread traditional Chinese medicine (TCM) use, particularly among older adults, 72 may affect care patterns. With approximately 3.17% of outpatient consultations involving exclusive TCM services, and many patients integrating both medical systems, older adults raise in traditional Chinese contexts frequently choose TCM practitioners (eg, herbalists) due to habits, perceived treatment efficacy, and personal recommendations.73-75 This suggests some items from PCPC may lack relevance for TCM-oriented patients who occasionally use Western primary care. Consequently, poor adherence to conventional treatments is common, with 44.8% of chronic disease patients classified as having “poor adherence.” 76 Patients may adjust dosages, discontinue consultations, or halt treatments prematurely upon symptom improvement, complicating the alignment of responses with the PCPC framework. These systemic, cultural, and behavioral factors may contribute to the suboptimal factor loadings and model fit indices.
Some limitations were identified during the study. First, some items in the instrument received a high number of “non-applicable” responses, indicating that they were not relevant to all patients. For example, the items related to Dimension 4 (Continuity and Transition) do not suit for patients who have no experience being transferred to another health care professionals. While the original author argued that if these items are applicable to specific patients, they are crucial for the outcomes. 30 Second, this study included multimorbid patients in New Territories East Cluster (NTEC), although NTEC’s demographics align well with Hong Kong overall pattern, 77 representativeness is not a significant issue, while individuals in other clusters should also be considered. Third, this study exclusively involved patients from public-sector care settings in Hong Kong. While chronic disease management occurs predominantly in the public sector and creating heavy pressure on the healthcare system, 78 the omission of individuals using private services (including family physicians) limits private-sector representation and may affect generalizability of the study population. Moreover, a significant gender imbalance exists in the sample, with 77.2% of the total sample being females (n = 386) and only 22.8% being males (n = 114). This selection bias limits the generalizability of findings, particularly for male-specific conditions like benign prostatic hyperplasia and the broader male population, necessitating further research. Another limitation is the absence of a direct assessment of acceptability due to time and resource constraints. Although operational acceptability was partially evaluated through expert review and missing data analysis (5 items > 5% “NA” responses, ranging from 5.6% to 9.0%), we did not conduct a direct assessment of acceptability, including aspects such as comprehensibility, administrator burden, and patient burden, through the participant feedback mechanisms (eg, cognitive interviews).79,80 Furthermore, the developed Cantonese version of the PCPC instrument requires additional validation studies to: (1) incorporate broader cultural nuances across diverse populations, and (2) evaluate critical psychometric properties, such as responsiveness, 81 cross-cultural validity, and measurement invariance The current absence of these validations limits the comprehensiveness and the applicability in other diverse settings – particularly given varying patients demographics within Hong Kong’s public and private healthcare context. Additionally, to strengthen the generalizability, addressing region and gender imbalances in sampling strategies and assessing variations in clinical service providers’ usage patterns during information collection were recommended in future studies.
The validation of the PCPC is a very timely action that corresponds to the continual expansion of the role of primary care in the healthcare system of Hong Kong. This urgency stems from the region’s hybrid public-private healthcare system,82,83 and its rapidly aging population, which has resulted in increasingly prevalent multimorbidity. Consequently, the continued reliance on tertiary-care-oriented system proving to be no longer affordable in the short future. 84 People with multimorbidity represent the group in the community that require the higher volume of healthcare utilization. 85 Developing the Can-PCPC can be highly useful for assessing the perceived patient-centered care by the multimorbid individuals who received services in the primary care settings. The assessed Can-PCPC results can help to provide feedback and directions for recognizing multimorbid patients’ complex needs, 86 enabling primary care providers to improve care coordination and develop tailored care plans that accommodating local primary care workflows and resource constraints. Thus, reducing unnecessary clinical consultations and emergency visits. Moreover, enhancing services for multimorbid populations in primary care settings will strengthen the role of primary care providers in Hong Kong healthcare context while diminishing reliance on tertiary care. This shift could partially alleviate significant societal burdens. Although further validation is required for practical implementation, the PCPC remains an effective tool to measure the degree to which this population is being managed appropriately and effectively.
Conclusions
The Cantonese version of the Patient-Centered Primary Care (PCPC) instrument was validated for assessing patient-centered care in Hong Kong’s multimorbid population. It demonstrated strong reliability and convergent validity with patient enablement instrument, though confirmatory factor analysis yielded marginally acceptable model fit. As the first culturally adapted tool measuring patient-centeredness for Cantonese-speaking multimorbid patients, it addresses the research gap in the local primary care settings. However, the suboptimal CFA results and lack of criterion-related validity suggest the need for further studies to expand its application to broader populations, refine its structure and assess its predictive value for real-world health outcomes. This tool provides the theoretical foundation for future patient-centered primary care research in Hong Kong and similar cultural regions, guiding improvements in primary healthcare systems.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251368995 – Supplemental material for Psychometric Validation of the Cantonese Version of the Patient-Centered Primary Care Instrument for Multimorbid Patients in Hong Kong
Supplemental material, sj-docx-1-jpc-10.1177_21501319251368995 for Psychometric Validation of the Cantonese Version of the Patient-Centered Primary Care Instrument for Multimorbid Patients in Hong Kong by Jinglin Gan, Peipei Hu, Tak-Ka Tang, Cheuk Ling Lee, Chen-Wei Liu, Eliza Lai-Yi Wong, Francisco Tsz Tsun Lai and Benjamin Hon-kei Yip in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We sincerely appreciate all the participants for actively participating in this project.
Ethical Considerations
This study is in compliance with the Declaration of Helsinki. Ethics approval was obtained from the Chinese University of Hong Kong Survey and Behavioral Research Ethics Committee (Ref. No. SBRE(R)-21-009)) and the Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee (Ref. No. 2021.284).
Consent to Participate
All participants provided written (electronic) informed consent prior to study participating in the study, in accordance with the approved ethics protocol.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Food and Health Bureau under Health and Medical Research Fund (HMRF 18190361).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are not available due to ethical concerns and privacy. Requests for access to these data should be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
