Abstract
Introduction:
Treatment of cutaneous warts with intralesional vitamin D3 has demonstrated promising results in over 35 studies with methodological limitations.
Methods:
We conducted a randomized, double-blind, placebo-controlled trial in our primary care procedure clinic of intralesional vitamin D3 12 000 IU vs placebo in cutaneous warts every 4 weeks up to 3 times. Warts were assessed 4, 8, 12, and 24 weeks after the initial injection. The primary outcome was complete wart resolution. Reduction in wart size was a secondary outcome.
Results:
Between October 2020 and April 2023, we enrolled 77 participants with 1 or more cutaneous warts (41 and 36 in vitamin D3 and placebo groups, respectively) with untreated (32%) and recalcitrant (69%) warts. At 24 weeks, the index wart had resolved in 12 (30%) of vitamin D3 and 10 (31%) placebo participants. Treatment with vitamin D3 had no effect on wart surface area in an adjusted model nor wart resolution in analysis adjusted for baseline wart size, multiple warts, and the vehicle (OR 0.31 [95% CI] 0.01-10.3; P = 0.51).
Conclusions:
These results demonstrate a lack of benefit of intralesional vitamin D3 in cutaneous warts.
(ClinicalTrials.gov number NCT04278573)
Keywords
Introduction
Cutaneous warts are caused by human papillomaviruses (HPVs) which infect keratinocytes in the epidermis and include common, plantar, and flat warts. Spontaneous remission occurs in most cases of cutaneous warts in children within 2 years but may take several years in adults, particularly among those without intact cellular immunity. A Cochrane review of 21 randomized clinical trials of destructive treatments for warts reported a wart regression rate for placebo of 27% over a mean of 15 weeks. 1
Destructive therapies, including topical salicylic acid and cryotherapy with liquid nitrogen, are often used for the initial treatment of warts,2,3 but up to one third of cases become recalcitrant to destructive therapies.2,4 Recalcitrant warts are often treated with intralesional injections of immune stimulating antigens, like Candida or MMR, topical antimetabolites like 5-fluorouracil, or an intralesional cytotoxic antibiotic like bleomycin.2,5
Vitamin D has immunomodulatory properties that may augment the immune response to cutaneous HPV.6,7 Keratinocytes produce vitamin D and can enzymatically metabolize vitamin D by 25-hydroxylation (CYP27A1) and 1α-hydroxylation (CYP27B1) to the active metabolite, 1,25-dihydroxyvitamin D (1,25(OH)2D). The 1,25(OH)2D metabolite binds to nuclear vitamin D receptors to regulate gene expression, which can have intracrine effects in the keratinocyte or paracrine effects on immune cellular responses in the skin. 8 Activation of toll-like receptors by vitamin D in macrophages induce an antimicrobial peptide, which may augment immune recognition of HPV of other viruses. 9 Topical application of an analog of 1,25(OH)2D, calcipotriol, suppresses proliferation and stimulates terminal differentiation of epidermal keratinocytes and has been used successfully for skin conditions like psoriasis 10 and ichthyosis. 11
Intralesional injection of warts with vitamin D3 has demonstrated promising results in over 35 case series and comparative trials in Turkey, India, Egypt, and Pakistan including 3 in 2025. In 8 randomized placebo-controlled unmasked trials, 6 in Egypt12-17 and 2 in India,18,19 investigators reported wart clearance rates of up to 90% in study subjects within 6 months, though 1 trial showed no benefit. 19 Despite the methodological limitations in these trials, intralesional vitamin D3 has been recommended in recent meta-analyses and review articles5,20-24 including 1 from the US in 2025, 25 though some are not convinced.26,27
Our objective was to determine if intralesional vitamin D3 is effective in the treatment of cutaneous warts.
Methods
A randomized, double-blind, placebo-controlled trial was conducted from October 2020 to April 2023 in our primary care procedure clinic following the recommendations of the British Association of Dermatologists for studies of wart therapies 2 to evaluate the effect of intralesional injections of vitamin D3 into cutaneous warts. Participants were randomly assigned in a 1:1 ratio to receive either vitamin D3 or placebo vehicle every 4 weeks up to 3 times and followed them for 6 months. The protocol was approved by the Institutional Review Board of the Mayo Clinic, approved by the U.S. Food and Drug Administration (FDA) for an investigational new drug (IND) application, and registered on ClinicalTrials.gov (NCT04278573). Declaration of Helsinki and Good Clinical Practice guidelines were followed.
We recruited local adults with cutaneous lesions consistent with a plantar, common, or flat wart through advertisements and flyers. Interested participants were screened for eligibility and submitted wart photos for virtual review by one of the investigators.
Subjects were excluded from the study if they had used destructive treatments in the prior month, immunoadjuvant therapy for warts in the prior 4 months (e.g., Candida), high-dose oral vitamin D supplementation exceeding 4000 IU daily in the prior 3 months, pregnancy, lactation, facial or genital warts, or were considered immunosuppressed by disease or medication. Written informed consent was obtained from enrolled participants who were scheduled for an initial clinical visit up to 1 week later by one of the physician investigators. Study drug was ordered from the research pharmacy.
Vitamin D3 for injection 40 000 IU/ml was prepared in a vehicle of 10:1 propylene glycol and dehydrated alcohol 28 by our Production Lab Pharmacy, a licensed compounding facility. This non-commercially available formulation has a long history of use in the U.S. in intramuscular and subcutaneous administration for hypovitaminosis D. An initial IND application for intralesional injection into cutaneous warts was approved by the FDA on July 18, 2020 (IND 146953) for use of this formulation in warts. Potency was confirmed and impurities excluded by an independent laboratory (ARL Bio Pharma, Oklahoma City, OK) and all sets of compounded vials were cultured to ensure safety.
Participants were randomized to receive either placebo or vitamin D3. The randomization code in blocks of 4 was generated by the research pharmacy and concealed from participants and study personnel until completion of statistical analysis. The experimental arm received 12 000 IU (0.3 ml) of vitamin D3; the control arm received an equal 0.3 ml volume of vehicle alone, both provided in a 1.5 ml clear vial as a clear solution. Study investigators were alerted to any potential adverse drug reaction. A research supervisor had authorization to break the randomization code for an individual subject for any serious adverse events.
At the first visit, the diagnosis of wart was confirmed on dermoscopy based on disruption of the dermatoglyphics (skin-lines) by typical hyperkeratosis with visible capillaries in the base. 2 Warts were photographed and 2 diameter measurements at right angles recorded on a spreadsheet for calculation of surface area based on the area of an ellipse. Photodermoscopy images of the warts were stored in the medical record.
Study enrollment commenced in October 2020. In the case of multiple warts, the investigator and participants made a shared decision about which wart to inject (usually the largest, most symptomatic). Only 1 wart was injected but up to 7 other warts were measured for change throughout the study.
For each injection, the skin was prepared with alcohol, and ethyl chloride spray was used to provide topical anesthesia for injection. A volume of 0.3 ml study drug was injected with a 1 ml Luer-lock syringe and ¾” 30g needle directed from the edge of the index wart under the central base into the wart. Any leakage was estimated and recorded. The subjective pain score rating from 0 to 10 during, immediately following, and 5 min after the injection was recorded. One week after the injection, participants were surveyed for post-injection symptoms of pain, redness, itching, malaise, myalgias, and fever. Subjects were evaluated by a physician investigator at 4, 8, and 12 weeks (±7 days) after the initial injection. Evaluation included dermoscopy and measurements. Those with a persistent index wart underwent up to 2 additional injections at 4 and 8 weeks.
The study was paused in December 2020 after the first 17 participants received their initial wart injection (designated “phase 1”) as 3 participants complained of persisting burning pain and had hemorrhagic vesicles that we suspected were due to intradermal injection of dehydrated alcohol in the vehicle. A new formulation with sesame oil vehicle was approved as a modified IND by the FDA on June 29, 2021, and used for the intralesional injections of the subsequent 60 participants (designated “phase 2”) beginning in September 2021 with the additional exclusion criteria of sesame allergy.
The primary outcome variable was prespecified as complete regression of the injected wart assessed by visual exam and photodermoscopy at each visit. The prespecified secondary outcome variable was reduction of the surface area of the index wart and other non-injected warts. Dermoscopic wart measurements were repeated by another investigator of the dermoscopic images. Any dimensional difference greater than 1 mm was adjudicated by re-examination of the images. The dimensions of both investigators were independently used to calculate surface area and the mean of the 2 used for size change analysis. Wart resolution was diagnosed by loss of hyperkeratosis and re-establishment of normal dermatoglyphics without capillaries.
We estimated that a sample size of 30 subjects in each group (60 total) would have 95% confidence and 80% power to show a difference between an approximate 60% wart resolution in the vitamin D3 group based on the results of literature referenced above12-19 and 25% wart resolution in the control group based on the natural history of warts.1,2 Data were entered with Research Electronic Data Capture (REDCap) and exported for statistical analysis with BlueSky Statistics Version 10.3.1, R version 4.13. The lme4 package was used to fit models in R. A multivariable generalized mixed effects logistic regression model was used to compare the primary outcome of complete wart regression between the 2 groups over 24 weeks, adjusting for baseline wart size, multiple warts, and the study phase (vehicle). A linear mixed effects regression model accounting for repeated measures over time was used to assess the effect of vitamin D3 on the surface area of warts, adjusting for initial surface area. A term for phase 1 or phase 2 of the study included in the regression models assessed the effect of vehicle on outcomes. P values were obtained from t-tests using Satterthwaite’s approximations for degrees of freedom in the linear mixed model and from Wald z-tests for the generalized mixed effects logistic regression model. Differences in the proportion of adverse events between groups were compared with Fisher’s exact test, and differences in pain scores were compared with the t-test assuming unequal variances. P values <.05 were considered significant.
Results
Of 428 subjects screened for eligibility, 77 were enrolled (41 and 36 in vitamin D3 and placebo groups, respectively) (Figure 1). The median (range) age of 77 enrolled subjects was 34 (19-67) years, and 58 (75%) were female (Table 1).
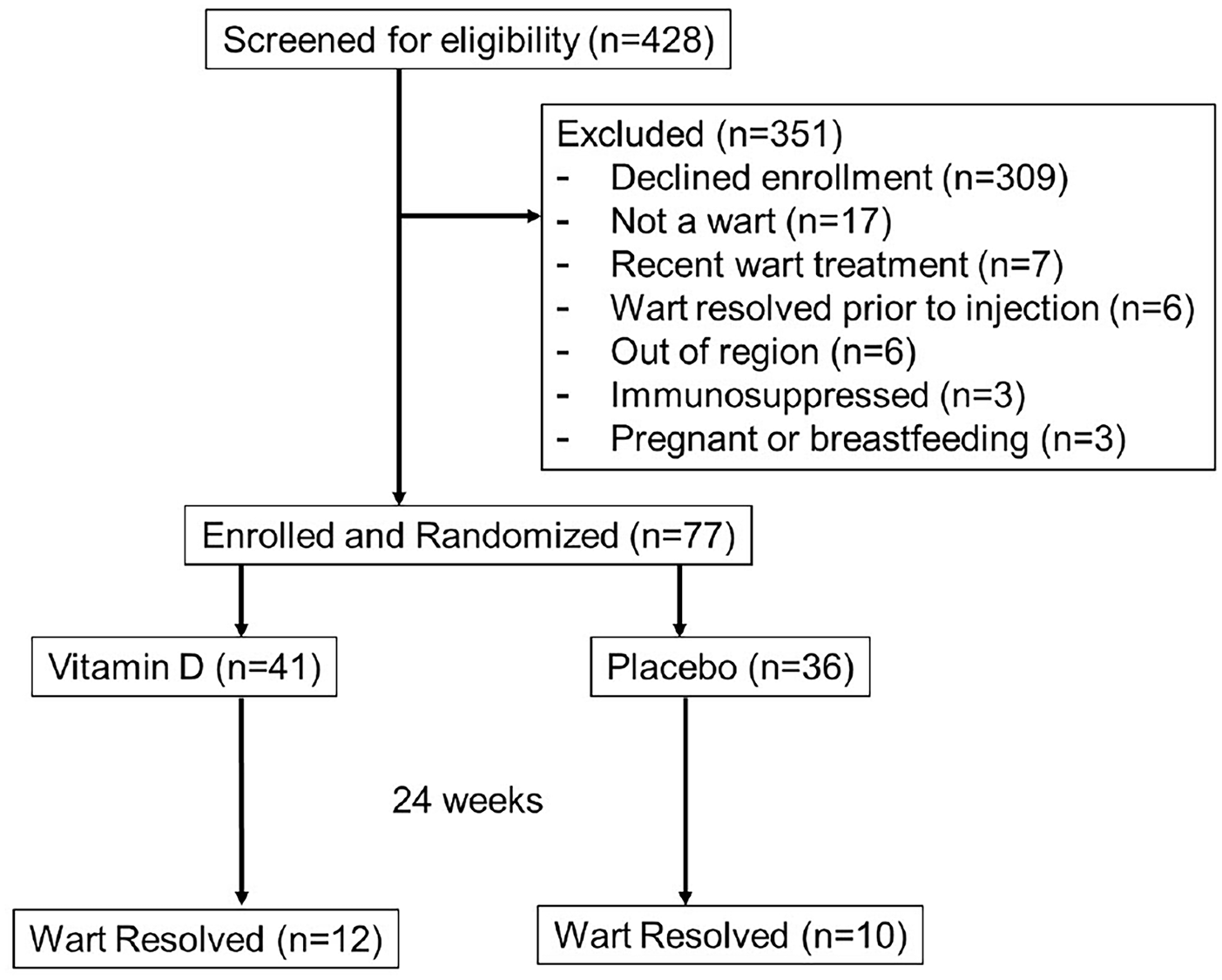
CONSORT flow diagram.
Subject Characteristics.

Most warts were on the plantar foot (64%) or a finger (23%), and 32 (42%) had more than 1 wart. The median surface area of the primary wart was 15.9 (1.4-144.5) mm2. The median duration of the primary wart was 5.0 (0.42-25) years, and 48 (69%) were recalcitrant to previous treatment of the index wart, predominantly with salicylic acid or cryotherapy.
The first 17 participants in phase 1 who received the initial injection with the propylene glycol and dehydrated alcohol vehicle only received the first injection but were followed for 24 weeks. The 60 phase 2 participants received vitamin D3 or placebo in sesame oil vehicle. Of the 77 participants enrolled, 72 completed 24 weeks of follow-up. All subjects who dropped out were in phase 1. Three participants in the placebo group dropped out: 1 moved out of state, 1 no longer wanted to participate and didn’t provide a reason, and 1 dropped out to pursue treatment of the wart. One participant in the verum group dropped out as they did not show for the final visit without reason. One participant was never injected as it was determined their wart had spontaneously resolved prior to the first visit. Two subjects who had a single injection in phase 1 re-enrolled in phase 2. Two subjects who had a single injection in phase 1 re-enrolled in phase 2. Due to the 10-month time interval achieving FDA approval between phases, these subjects restarted the protocol (Figure 1).
At 24 weeks, the index wart had resolved in 12 (30%) and 10 (31%) in the vitamin D3 and placebo groups, respectively. There were no differences between the groups in the proportion of participants achieving resolution of the index wart at any time point (Figure 2). Vitamin D3 had no effect on wart resolution in a multivariable generalized mixed effects logistic regression model, adjusting for baseline wart size, multiple warts, and the vehicle (OR 0.31 [95% CI] 0.01-10.3; P = 0.51) (Table 2).
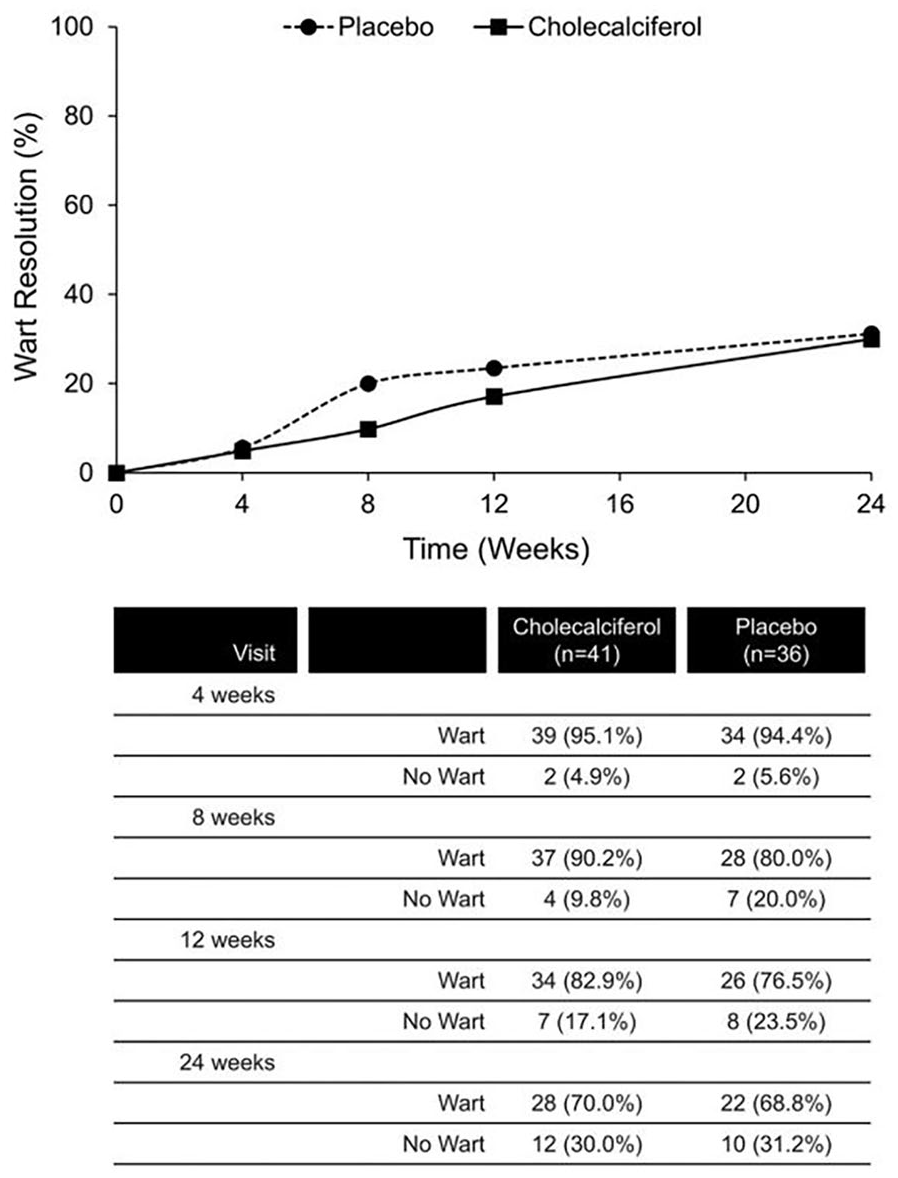
Wart resolution following intralesional injection of cholecalciferol (N = 41) or placebo (N = 36).
Multivariable Repeated Measures Analysis of Wart Resolution and Reduction in Surface Area.
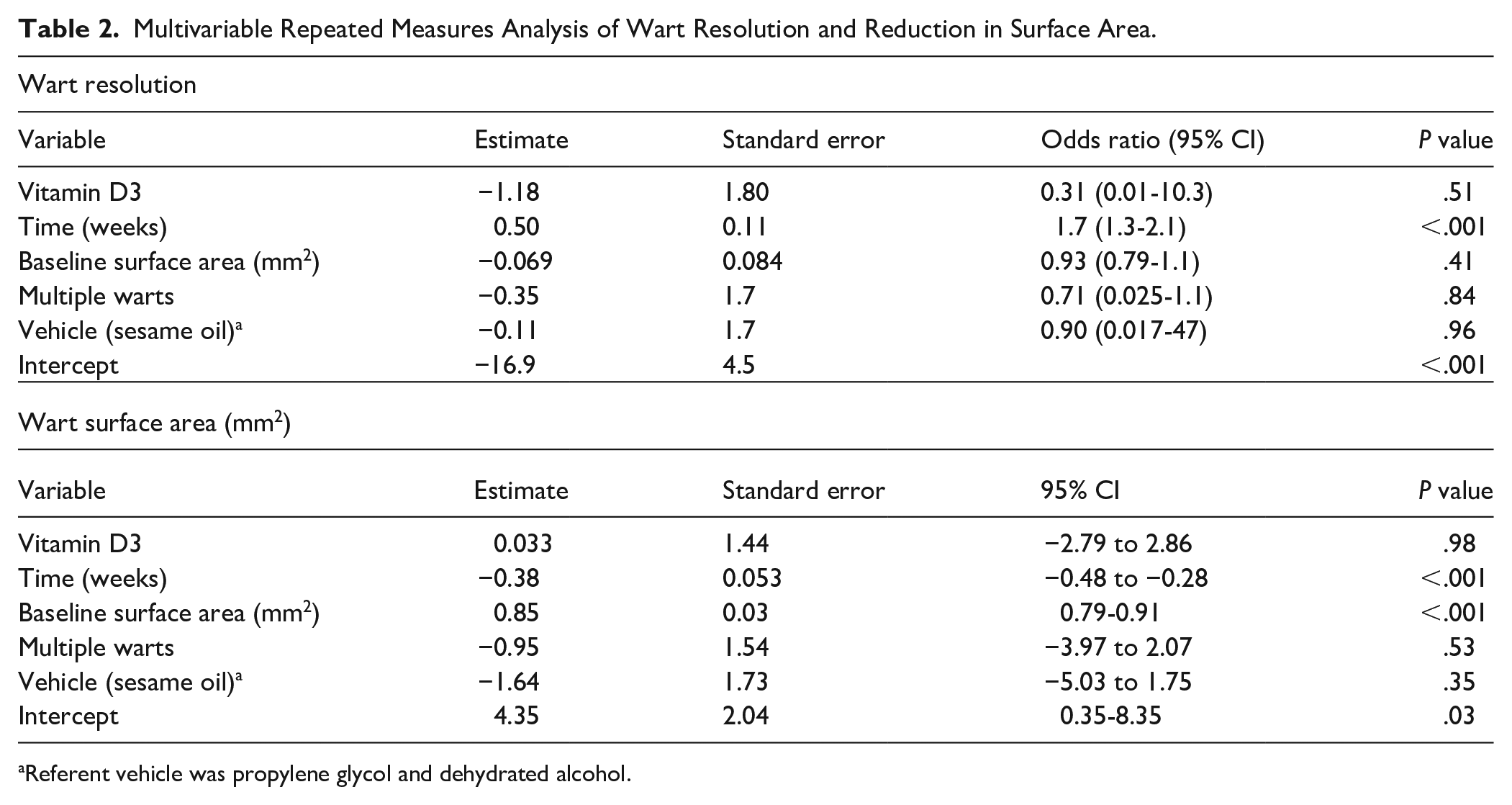
Referent vehicle was propylene glycol and dehydrated alcohol.
The baseline size of the wart and multiple warts were not predictive of wart resolution. Only time was significantly related to the likelihood of wart resolution. In a repeated measures linear regression model adjusted for the same variables, the change in wart surface area in the vitamin D3 group was 0.033 (95% CI −2.79 to 2.86; P = 0.98) mm2 which did not differ from the placebo group. The vitamin D3 group did not demonstrate significantly greater pain, redness, or itching compared with the placebo group. Propylene glycol and alcohol used for the 17 participants in phase 1 of the trial were associated with greater pain after injection than the sesame oil vehicle used for the 60 participants in phase 2 of the trial (P = .01); but the vehicle had no effect on wart resolution (P = 0.96) or surface area reduction (P = 0.35).
Pain was the most common adverse effect of the intralesional injections. Overall, pain occurred in 84.1%, 74.4%, 62.5%, 43.2%, 15.3%, and 7.4% of subjects immediately, at 1 min, 5 min, and during the first, second, and third weeks after injection, respectively. The pain scores on a scale of 0 to 10 were 7 or greater in 14.8%, 2.8%, 1.7%, 2.3%, 1.7%, and 0.6%, respectively. Mean pain scores did not differ between the vitamin D3 and placebo groups at any time point, except during the first week (Table 3).
Adverse Effects of 176 Intralesional Wart Injections.

Based on t-test assuming unequal variances or Fisher’s exact test as appropriate.
However, mean pain scores were significantly greater during the first, second, and third weeks in those in phase 1 than those in phase 2. Itching, redness, and hematoma were uncommon and occurred with similar frequency in the vitamin D3 and placebo groups, but redness was more frequent with the propylene glycol and alcohol vehicle than with the sesame oil vehicle. The mean estimated volume of fluid that escaped during injections was 0.067 ± 0.073 mL in the vitamin D3 group and 0.064 ± 0.075 mL in the placebo group (P = 0.81).
Discussion
In this randomized, double-blind, placebo-controlled trial of intralesional vitamin D3 for cutaneous warts, intralesional injection of cutaneous warts with vitamin D3 did not lead to greater wart resolution or reduction in wart surface area than vehicle alone. Spontaneous remission rates were high in this young adult population with 30% of index warts completely regressing by 24 weeks, consistent with the literature. 1
Unlike prior studies of intralesional injection of warts with vitamin D3, our study sought to optimize randomization and masking of the study subjects and investigators to avoid bias. This more rigorous design likely accounts for the negative results of this trial, unlike the results reported in studies without masking, randomization, or controls. Other differences between this study and those of other investigators that could account for different results include the dose and formulation of vitamin D3, the location of warts, duration of warts, size of warts, vehicle, technique and frequency of injection, younger, healthier population, and geographic location.
We acknowledge that this trial has limitations. We used an FDA-approved lower dose of vitamin D3 of 12 000 IU per injection to minimize risk of hypercalcemia and yet provide sufficient vitamin D3 for local immune modulation. Trials in other countries used higher vitamin D3 doses of 30 000 to 1 200 000 IU. In the Middle Eastern and South Asian countries, where previous studies have been done, individuals are at greater risk of vitamin D deficiency due to darker skin pigmentation and sun avoidance. Correction of underlying vitamin D deficiency by the high doses injected in these studies may have contributed to wart regression and confounded results.29,30 Measurement of 25-hydroxyvitamin D levels was not included in our study or other studies with much higher doses of intralesional vitamin D.31,32
In their 2014 guidelines for wart management, the British Association of Dermatologists authors recommended that future studies of cutaneous wart therapies examine single treatment arms rather than combined therapies, accrue sufficient sample size based on formal sample size calculations, ensure true randomization of patients to treatment, report rates for complete cure of warts, ensure masking of outcome to investigators, and follow up both 12 weeks after treatment and 6-12 months post-treatment. 2 This is the first study of vitamin D3 intralesional injection in cutaneous warts to follow these recommendations.
We found no benefit of intralesional injection of vitamin D3 into cutaneous warts.
Supplemental Material
sj-tif-1-jpc-10.1177_21501319251365853 – Supplemental material for Intralesional Injection of Vitamin D3 for Treatment of Cutaneous Warts: A Randomized, Double-Blind, Placebo-Controlled Trial
Supplemental material, sj-tif-1-jpc-10.1177_21501319251365853 for Intralesional Injection of Vitamin D3 for Treatment of Cutaneous Warts: A Randomized, Double-Blind, Placebo-Controlled Trial by Stephen P. Merry, David N. Brennan, Michelle J. Duvall, Joseph B. Stanek, Erin K. O’Dowd and Tom D. Thacher in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We are grateful for statistical consultation from Ross Dierkhising, Jeremy Syrjanen, and Natalie Averkamp. We especially thank all study participants; without their participation, this study would not have been possible.
Authors’ Note
An abstract of this study was presented at the National Association of Primary Care Research Group (NAPCRG) meeting in Quebec City on November 22, 2024. The abstract was published as a supplement to the November 2024 edition of the Annals of Family Medicine.
ORCID iDs
Ethical Considerations
The protocol was developed by the first and last author, approved by the Institutional Review Board of the Mayo Clinic (approval # 19-010219) on January 6, 2020, approved by the U.S. Food and Drug Administration for a new drug application on July 18, 2020 (IND 146953) and a modified IND approved by the FDA on June 29, 2021, and registered on ClinicalTrials.gov (NCT04278573)
Consent to Participate
Written informed consent was obtained from all participants prior to their participation.
Consent for Publication
Not applicable
Author Contributions
Concept and design: Merry and Thacher
Acquisition of data: Merry, Brennan, Duvall, and O’Dowd
Analysis and interpretation of data: Merry, Thacher
Drafting of the manuscript: Merry
Critical revision of manuscript: Brennan, Duvall, Stanek, O’Dowd, and Thacher
Statistical analysis: Thacher
Final Approval: Merry, Brennan, Duvall, Stanek, O’Dowd, and Thacher
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by a professorship endowment from Parker D. Sanders & Isabella G. Sanders for Dr. Tom D. Thacher, Professor of Family Medicine, Mayo Clinic, Rochester.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data will be made available without restriction with publication upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
