Abstract
Background:
Chronic kidney disease (CKD) management was largely centered around renin-angiotensin-aldosterone system inhibitors (RAASi) optimization, until recent emergence of novel therapeutics. However, slow adoption of guideline-directed therapy leaves patients vulnerable to disease progression. In 2022, a data-driven informatics approach was introduced to track real-time adherence to best practices.
Methods:
This multi-center, ambidirectional cohort study analyzed data from a shared electronic health record system in a public healthcare cluster in Singapore, comprising 7 primary care institutions and 3 tertiary care hospitals. Patients aged ≥21 with CKD, managed between 1st March 2022 and 31st March 2024, were included. Prescription trends for RAASi, sodium-glucose co-transporter-2 inhibitors (SGLT2i), non-steroidal mineraloreceptor antagonists, and statins were examined, alongside albuminuria monitoring and comprehensive care uptake.
Results:
Among 34 217 patients, mean age was 72 ± 12 years; 57% received RAASi, 21% SGLT2i, and 66% statins. Among those meeting therapeutic indications, RAASi uptake remained stable at 74%, with 40% receiving ceiling doses. SGLT2i uptake doubled but remained below 40%, with lower adoption in non-diabetic and non-obese patients. Only 21% of albuminuric CKD G1-3b patients received optimal combination therapy with RAASi, SGLT2i, and statins despite only 4% hyperkalemia prevalence and 2% with systolic BP <110 mmHg. Among albuminuric CKD G3 patients with 5-year end-stage kidney disease risk ≥15%, 28% received optimal therapy. One-third lacked albuminuria monitoring and were less likely to receive comprehensive therapy.
Conclusions:
Gaps persist in CKD care, particularly among non-diabetic, non-obese patients, and those without albuminuria monitoring. Health informatics-driven interventions can facilitate real-time process evaluation and adherence to best practices amid evolving treatment landscapes.
Keywords
Introduction
For decades, renin-angiotensin-aldosterone system inhibitors (RAASi) and cardiovascular risk reduction have been the foundation of chronic kidney disease (CKD) management; however, RAASi offers only a 20% to 28% relative risk reduction in long-term kidney failure, and the global prevalence of end-stage kidney disease (ESKD) continues to rise.1 -3 CKD is also a major independent risk factor for cardiovascular disease, with mortality increasing exponentially as kidney function declines. 4 Recent advances, including sodium-glucose co-transporter-2 inhibitors (SGLT2i), non-steroidal mineralocorticoid receptor antagonists (nsMRA), glucagon-like peptide-1 receptor agonists (GLP-1RA), and potassium binders, have expanded therapeutic options. Yet, real-world uptake remains suboptimal, leaving patients vulnerable to irreversible CKD progression and cardiovascular complications.5 -7 Barriers include systemic inefficiencies, drug affordability, provider-inertia, and limited awareness among non-nephrologists. 8
Traditionally, nephrologists were only involved in CKD care at later stages. However, rising CKD prevalence and increased emphasis on early intervention and cardiovascular risk reduction have necessitated re-examination of this model.9,10 In 2014, our hospital launched the Nephrology Evaluation, Management, and Optimization (NEMO) program, protocolizing RAASi initiation in primary care via coordinator-assisted primary care clinics with the oversight of a nephrologist. This evolved into the nationwide Holistic Approach in Lowering and Tracking Chronic Kidney Disease (HALT-CKD) program, involving all public institutions, emphasizing early detection and cardiovascular risk optimization.11,12 However, HALT-CKD’s reliance on manual reminders and coordinators posed scalability challenges amidst growing therapeutic complexity.
In 2022, we introduced a health informatics dashboard enabling real-time visualization of prescribing metrics across primary and tertiary care. Real-time data assessing CKD best-practice implementation was shared on a quarterly basis with primary care physicians, guiding adaptive interventions. This study examines prescribing patterns for RAASi, SGLT2i, MRA, and statins, including RAASi dose intensity. We explored gaps in CKD care and identified cohorts missing evidence-based therapeutics to inform population health strategies. We hypothesize that the use of a real-time health informatics platform across a multi-center public healthcare system is associated with improved adoption of guideline-directed medical therapy in CKD patients.
Methodology
Study Design and Population
We conducted a multi-center, ambidirectional cohort study using shared electronic health record data from a public healthcare cluster in Western Singapore, comprising 7 primary care institutions and 3 tertiary care hospitals with ~2500 acute beds. All patients ≥21 years with CKD who had any outpatient clinic visits within the healthcare cluster between 1st March 2022 and 31st March 2024 were included. ESKD patients were excluded.
Definitions
CKD was defined using ICD-10 diagnosis codes for “Chronic Kidney Disease,” “Chronic Kidney Failure,” “Chronic Renal Disease,” “Chronic Renal Failure,” “Renal Impairment,” or any 2 consecutive estimated glomerular filtration rate (eGFR) at least 90 days apart of <60 mL/min/1.73 m2 (using the 2009 CKD-Epidemiology Collaboration equation). 13 ESKD was defined by diagnosis codes of “End-stage kidney disease,” “End-stage kidney failure,” “End-stage renal disease,” or “End-stage renal failure.”
Data Source
De-identified demographic, laboratory, and prescription data were extracted from our healthcare cluster’s Next Generation Electronic Medical Record using EPIC Hyperspace May 2022/2023. Data from all clinic encounters were updated quarterly from March 2022. Censoring occurred at dialysis, kidney transplantation, death, or study end (31st March 2024).
Clinical Characteristics
Comorbidities were identified from ICD-10 diagnosis codes. Diabetes was determined by diabetes registry linkage and glycated hemoglobin readings above 6.5%. Obesity was defined as a body mass index (BMI) of ≥27.5 kg/m2, based on the World Health Organization Asian BMI cut-offs. 14 eGFR under 15 mL/min/1.73 m2 was imputed as 14 mL/min/1.73 m2 for analysis. 5-year kidney failure risk equation-Southeast Asia (KFRE-SEA) scores, previously validated in our local population, were calculated. 15
Prescription Data
Electronic prescription records were reviewed every 6 months. The 1st period of 1st March 2022 to 30th September 2022 covered 7 months, followed by 6-monthly intervals till 31st March 2024. Patients were classified as RAASi, SGLT2i, or MRA users if they were prescribed any of these medications during each interval. Individual medications were identified based on medication grouper IDs (Supplemental data S1). RAASi doses were expressed as percentage of ceiling therapeutic dose (Supplemental data S2).
Statistical Analysis
Statistical analyses were performed in IBM SPSS Statistics version 29 (Armonk, NY, USA).
We described the cohort’s clinical profile. We next evaluated the prescribing rates of RAASi, SGLT2i, MRA, and statins over time in patient cohorts who met the respective indications for the former drugs. We assessed uptake of comprehensive care at latest clinic visit by categorizing patients based on albuminuria monitoring. Lastly, we evaluated the combination use of RAASi, SGLT2i, and statins among albuminuric CKD patients at their latest clinic visit. Continuous data was reported as mean (±standard deviation) or median (interquartile range). Categorical variables were compared using the Chi-square test. Statistical significance was taken at the 5% level with 2-tailed tests. Binary multivariate logistic regression was performed to evaluate associations between comorbidities and medication use.
Results
We studied 34 217 patients (Table 1). Mean age was 72 ± 12 years, 57% were male. Mean eGFR was 50±18 mL/min/1.73 m2; 57% had diabetes. At latest visit, 57% received RAASi, 21% SGLT2i, 0.4% non-steroidal MRA (nsMRA), and 66% statins. Two-thirds underwent albuminuria or proteinuria monitoring during follow-up.
Patient Characteristics.

Note: All variables are reported as mean ± SD, frequency, and percentage.
eGFR calculated using the CKD-EPI formula.
Uptake of Comprehensive CKD Management
We report the results of the key CKD medication uptake over the 2-year study period. Among albuminuric patients, RAASi uptake remained stable at 74% over 2 years; 69% to 71% received ≥50% of ceiling doses, 40% achieved maximum dose (Figure 1).

Percentage of albuminuric patients on ACE-i/ARB.
SGLT2i uptake nearly doubled among patients with eGFR ≥ 20 ml/min/1.73 m2 but remained below 40% across diabetes and albuminuria subgroups (Figure 2). SGLT2i uptake was particularly low (<15%) among non-diabetic and non-albuminuric CKD patients. Figure 2 illustrates the timeline of SGLT2i uptake in relation to local regulatory actions and concerted efforts by physicians to promote its adoption.

Percentage of patients on SGLT2i (eGFR ≥ 20 ml/min/1.73 m2).
The use of nsMRAs among diabetic patients remained <1% despite an increase in late 2023 (Figure 3).
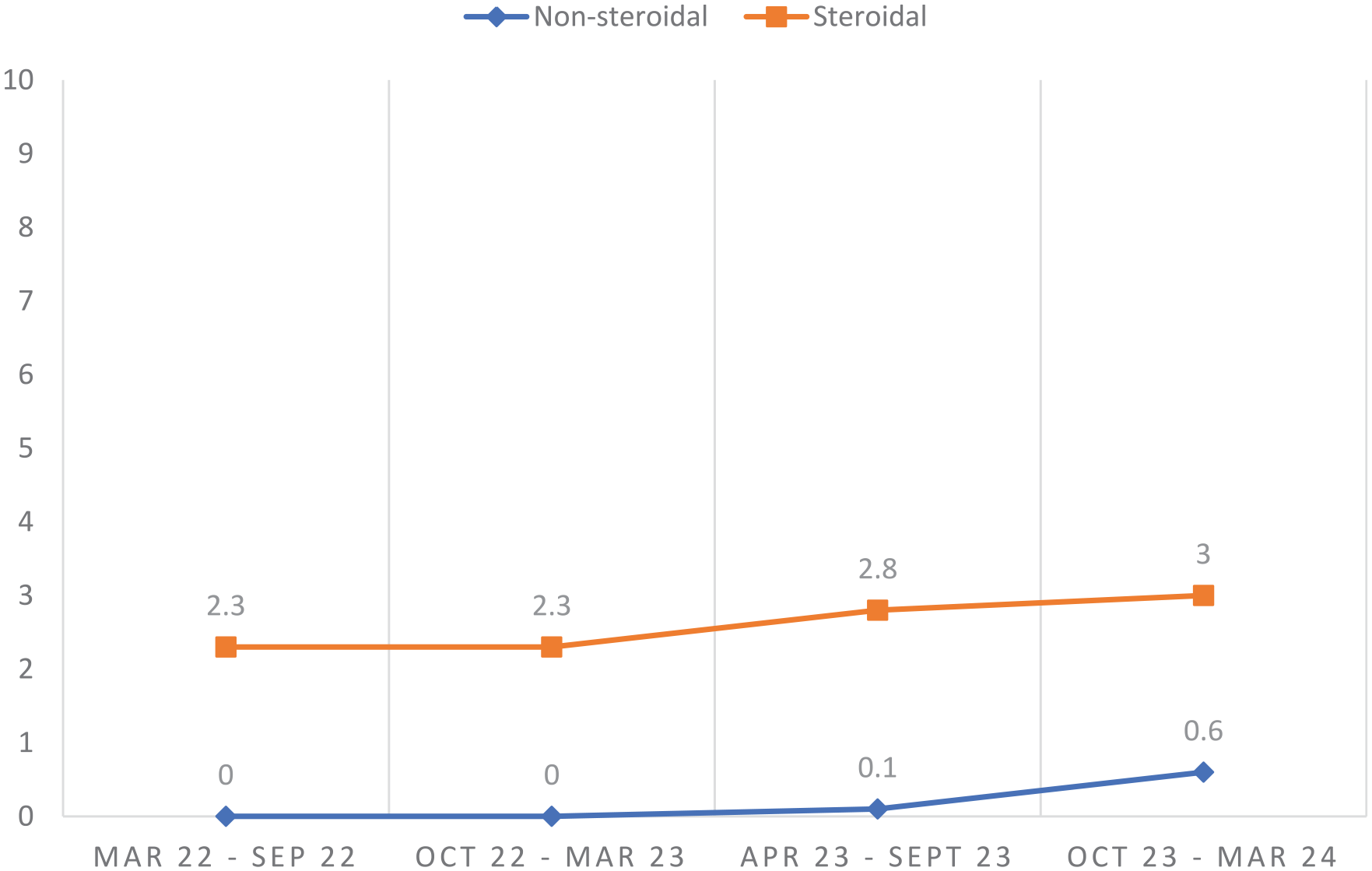
Percentage of diabetic patients on mineralo-receptor antagonists.
At the latest clinic visit, among patients aged <80 years who met indications for statin use (eGFR < 60 ml/min/1.73 m2, diabetes, ischemic heart disease, stroke, or peripheral vascular disease), 72% (n = 14 707) were on statins. Of the 69% of patients with lipid profile measured, 71% achieved low-density lipoprotein <2.6 mmol/L.
Albuminuria and Proteinuria Monitoring and RAASi, SGLT2i, and Statins Use
One-third of CKD patients lacked albuminuria or proteinuria monitoring. As shown in Table 2, patients monitored for albuminuria or proteinuria were more likely to be on RAASi, SGLT2i, and statins across all CKD stages at latest clinic visit.
Albuminuria/Proteinuria Monitoring and Use of CKD Therapeutics.

Optimal Combination of RAASi, SGLT2i, and Statins in Early, Albuminuric CKD Patients
We arbitrarily defined optimal CKD therapeutics as a combination of RAASi ≥50% ceiling doses, SGLT2i, and statins, in line with recommendations from the Kidney Disease: Improving Global Outcomes (KDIGO) 2024 clinical practice guidelines for the evaluation and management of chronic kidney disease. 16 Of patients <80 years with CKD G1-3b (eGFR ≥ 30 mL/min/1.73 m2) and albuminuria (≥ 3 mg/mmol), only 21% received this combination (Supplemental Table S3). Non-diabetic and non-obese patients were less likely to receive optimal therapy. Hyperkalemia (4%) and low systolic blood pressure <110 mmHg (2%) were uncommon. In CKD G3a-G3b patients with ≥15% 5-year kidney failure risk (KFRE-SEA), only 28% received optimal therapeutics, 65% received 1 to 2 agents, 6% received none. Patients of Malay ethnicity had the highest proportion on optimal therapeutics, followed by Indian and Chinese patients (P < .001). Additional subgroup analysis revealed that both Malay and Indian patients had higher rates of diabetes compared to Chinese patients (81% and 84% vs 65%, P < .001), while obesity was most prevalent among Malay patients (61% vs 47% in Indian and 33% in Chinese, P < .001). Patients with tertiary care contact were more likely to be on optimal therapy compared to those managed exclusively in primary care (26% vs 19%; P < .001).
Binomial logistic regression was performed to analyze further factors associated with optimal combination CKD therapy, compared to 2 or fewer therapeutics of the latter. Presence of diabetes, ischemic heart disease, obesity, mean eGFR, and mean uACR were selected for inclusion based on clinical and statistical significance in the findings illustrated in Supplemental Table S3. Non-diabetic and non-obese patients were more likely to be lacking in optimal treatment, as compared to diabetic and obese patients (OR 0.31, 95% CI 0.25-0.38, and OR 0.69, 95% CI 0.59-0.81) (Supplemental Table S4).
Discussion
Despite robust evidence supporting RAASi, SGLT2i, and statins in CKD management, this study reveals substantial underutilization in real-world practice. RAASi and SGLT2i have been demonstrated to reduce major cardiovascular events and ESKD risk; statins similar reduce atherosclerotic events in CKD patients.16 -20 However, only a fifth of patients in our cohort with albuminuric CKD received optimized triple therapy. RAASi use remained stable at 74%, with only 40% reaching ceiling doses, despite low rates of hyperkalemia (4%) and low systolic blood pressures of <110 mmHg (2%).
SGLT2i uptake doubled over 2 years but remained under 40%, despite robust evidence from CREDENCE in 2019, followed by DAPA-CKD and EMPA-KIDNEY in short succession.18,19,21 Regulatory approvals followed, with the U.S. Food and Drug Administration endorsing canagliflozin, dapagliflozin, and empagliflozin for CKD patients between 2020 and 2023.19,21,22 Locally, the Health Sciences Authority similarly acted promptly, approving canagliflozin in December 2020, dapagliflozin in October 2021, and empagliflozin in December 2023 to reduce CKD progression and end-stage kidney disease.
Despite expedited approval, the use of SGLT2i remained below 20% from March to September 2022, reflecting global barriers such as cost and clinical inertia. 23 To mitigate this, local nephrologists and policymakers advocated for subsidies and engaged in multidisciplinary education (Figure 2). Dapagliflozin was added to the Medical Assistance Fund in August 2022, allowing for patients to receive up to 50% subsidies for the drug, depending on their household income, improving access for financially needy patients. The local diabetes and CKD guidelines were updated in May 2023 and Oct 2023, recommending SGLT2i as foundational therapy.24,25 Empagliflozin’s inclusion in the Standard Drug List in December 2023 further improved affordability by allowing all residents and permanent residents, regardless of household income, access at subsidized rates. 26 Real-time visualization of SGLT2i uptake allowed nephrologists to continually assess the effectiveness of policy and educational strategies and refine interventions in response. The implementation of evidence-based therapies for chronic diseases often lags behind clinical evidence, influenced by factors such as the strength of guideline recommendations. RAASi adoption followed this pattern, with pivotal trials first published in early 2000s and incorporation into the first KDIGO clinical practice guideline for CKD only in 2012.1,22,27 Even years later, uptake remained suboptimal. RAASi use in patients with CKD in the United States was 40% by 2014 and had plateaued after the 2000s, with only 17% of patients with CKD and Type 2 diabetes initiated on RAASi within a year of diagnosis. 28 The historical experience and current developments highlight the need for responsive healthcare policy to match emerging evidence.
A granular analysis of treatment patterns also revealed suboptimal therapeutic optimization among patients <80 years with early-stage (G1-3b) albuminuric CKD, when preventive efforts are crucial. Only 21% received triple therapy of optimized RAASi, SGLT2i, and statins. Furthermore, 50% of CKD G3 patients without this combination were at high risk of kidney failure (KFRE-SEA risk of ≥15% over 5 years), underscoring the mismatch between risk and management. Non-diabetic and non-obese patients were less likely to receive optimized therapy, reflecting a prevailing concept that combination therapy may primarily be for diabetic kidney disease and metabolic health. However, emerging evidence highlights their comparable risk for adverse outcomes, including heart failure and end-stage kidney disease, necessitating a broader therapeutic focus beyond the traditional high-risk diabetic and obese population.29,30
Higher rates of combination CKD therapy among Malay patients may partly reflect higher clinical risk profile with greater burden of diabetes and obesity, which was demonstrated in previous local studies.11,31 Further population-based studies will be needed to further explore ethnic disparities and the role of social determinants of health in chronic disease burden, management and treatment equity. Patients co-managed in tertiary care had higher use of combination CKD therapies compared to those managed in primary care alone. This may reflect greater clinical complexity, increased specialist involvement, or stronger adherence to treatment guidelines. Notably, tertiary care in this study included all specialty services, not just nephrology, highlighting the potential of multidisciplinary specialist care to support comprehensive risk factor management. These findings underscore the need for integrated care models to optimize CKD management across healthcare settings.
Another pressing concern is that one-third of patients lacked proteinuria or albuminuria surveillance, limiting risk assessment beyond eGFR. This undermines timely intervention, with data showing 63% higher ESKD risk with just a 1-year diagnostic delay and corresponding lifestyle and pharmacological intervention. 32 A risk-based approach to CKD therapies, targeting highest risk patients for accelerated and intensified treatment, offers a more effective strategy for optimizing outcome. 33
Therapeutic optimization in CKD involves complex interdependencies among the physician, the patient, and systemic factors. 5 Medication discontinuation during acute kidney injury episodes and failure to resume therapy thereafter further compound these challenges. 34 Additionally, multimorbidity, polypharmacy, and pill burden impede adherence, necessitating innovative strategies such as combination pills to improve adherence and lower cost. 28 Emerging therapies, including nsMRAs and GLP-1RAs, represent the next frontier in CKD therapeutics. Incorporation of these additional treatments into routine CKD care in the current landscape will require systemic efforts to address existing barriers and facilitate widespread adoption.
Our findings highlight actionable targets for adaptive interventions. First, real-time prescribing data revealed low SGLT2i uptake despite updated guidelines and regulatory approvals, prompting multidisciplinary educational efforts and subsidy expansion. Second, identification of high-risk patients not receiving optimized therapy supports the development of risk-based dashboards to prompt proactive treatment reviews. Third, the association between lack of albuminuria monitoring and suboptimal therapy underscores the need to integrate testing within the primary care workflows. Finally, disparities in treatment among non-diabetic and non-obese patients suggest the need to reframe CKD as a cardiometabolic condition beyond the traditional risk factors. These insights support targeted, data-driven clinical decision support tools to improve therapeutic uptake and equity across the CKD care continuum.
Strengths and Limitations
Existing literature on the real-world uptake of RAASi, SGLT2i, and statins across primary and tertiary care in the modern era of expanding CKD therapeutics is scarce. This study addresses this gap. The strength of this study is the large CKD population of over 30 000 patients and the ability to access CKD management within the entire western medical cluster of Singapore, spanning primary and tertiary care. Health informatics as a resource provides us quarterly updates on the adoption of guideline-directed medical therapy and currency to the data we present. Limitations include inability to capture individualized RAASi dose tolerability nuances and lack of qualitative data on patient or provider perspectives, which could be explored with future language models. Additionally, our analysis focused on ESKD risk and not the broader cardiovascular risk profile of our CKD patients. Despite these limitations, our findings can be a foundation for future initiatives to enhance CKD management and therapeutic intervention.
Conclusions
This study reveals significant gaps in the real-world implementation of guideline-directed therapy for CKD despite strong clinical evidence and regulatory efforts. Therapeutic optimization remains suboptimal, particularly in non-diabetic and non-obese patients, and even in patients at high risk of kidney failure. Underdiagnosis of albuminuric CKD further limits risk-tiered interventions. Multifaceted strategies, including enhanced monitoring, education, behavioral science to improve adherence strategies, and policy-driven cost reductions are essential. Our study highlights the feasibility of leveraging health informatics to generate real-time disease management insights, facilitating quality improvement initiatives and integration of comprehensive CKD care including emerging therapies.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251365314 – Supplemental material for Multi-Center Health Informatics to Examine the Implementation of Guideline-Directed Medical Therapy in Chronic Kidney Disease
Supplemental material, sj-docx-1-jpc-10.1177_21501319251365314 for Multi-Center Health Informatics to Examine the Implementation of Guideline-Directed Medical Therapy in Chronic Kidney Disease by Yan Ting Chua, Horng-Ruey Chua, Priyanka Khatri, Shilpa Rastogi, Sky Wei Chee Koh, Valerie Ma and Clara Lee Ying Ngoh in Journal of Primary Care & Community Health
Footnotes
Ethical Consideration
The study was approved by our cluster’s Domain Specific Review Board (approval number 2023/00413), which waived the need for informed consent due to the use of anonymized data and data privacy maintained by our informatics office as the trusted third party.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AstraZeneca Singapore funded the electronic data retrieval for the study but had no role in study conduct, analysis or reporting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All authors had access to the data and played a role in the writing of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
