Abstract
Introduction:
Many patients with sore throat receive antibiotics from their general practitioners (GPs); however, only a minority are affected by bacterial infection, often streptococcal A (Strep A). Rapid diagnostic test can improve diagnosis, guide treatment decisions and direct patients when required for appropriate self-care. This study investigates the potential benefit of Strep A rapid tests in pharmacies for sore throat patients, with the aim of maximising healthcare resources.
Methods:
Participants over 16 years old were screened using FeverPAIN scores, and those with high scores were tested for Strep A. To estimate avoided GP visits, participants completed a questionnaire on treatment choice (Accident & Emergency (A&E) GP visit, pharmacist, self-care) if the diagnostic service was unavailable.
Results and Conclusion:
Majority (86%) tested negative. The questionnaire revealed that 67% could avoid GP visits. When extrapolated to the UK, this could result in an estimated annual saving of £263 million for the National Health Service (NHS) Despite not being recommended for routine use by NICE, the study suggests that such rapid diagnostic testing for Strep A, combined with FeverPAIN screening, could prevent antimicrobial misuse, alleviate NHS pressure and empower pharmacists. This is particularly relevant considering the recent implementation of the pharmacy-first service.
Keywords
Introduction
Sore throat is a common condition in primary care. Most sore throats are self-limiting, and symptomatic treatment can help alleviate discomfort. 1
Approximately 95% of sore throats are viral, yet antibiotics are prescribed in about 60% of General Practitioner (GP) consultations for sore throat.2 -4 Minor health conditions like sore throat account for 20% to 40% of GP workload. 1 Furthermore, the COVID-19 pandemic, characterised by symptoms including sore throat, led to increased antibiotic prescribing.5,6 Antibiotic resistance is a growing global concern, predicted to cause high fatality rates and further strain the already under-resourced National Health Service (NHS).7,8 To combat this, the NHS and Public Health England advocate antibiotic stewardship to reduce inappropriate antibiotic use.9 -11
As antibiotics are ineffective against viruses, their unnecessary use in viral infections drives antibiotic resistance, 3 leading to the emergence of superbugs such as methicillin-resistant Staphylococcus aureus (MRSA). 12 Streptococcus A is the primary cause of bacterial infections in patients with a sore throat, with the remaining 20% attributed to group C and G Streptococcus. 13
NICE guidelines recognise Streptococcus A as a prevalent bacterial pathogenic cause of sore throats and recommend immediate or delayed use of antibiotics in people with a FeverPAIN score of 4 or 5 or a Centor Score of 3 or 4. 14 Both FeverPAIN and the Centor score are clinical prediction rules (CPRs) used to inform antibiotic prescribing strategies.
FeverPAIN assigns a score (maximum 5) based on specific criteria (Table 1); a high score indicates greater severity and likelihood of bacterial infection. Centor scoring is similar but with a maximum of 4 (Table 2).15,16
FeverPAIN Scoring Criteria.

Centor Scoring Criteria.

However, a recent randomised controlled trial suggested that FeverPAIN and Centor scores poorly discriminate between the presence and absence of Streptococci, concluding they have limited roles in detecting streptococcal throat infections and may encourage overprescription in primary care. 17
Rapid diagnostic testing for Strep A can facilitate decrease in antibiotic usage and prevent the healthcare expenses associated with denying antibiotics to patients who require them the most. 18 A recent Cochrane review concluded that rapid diagnostic tests for sore throat could reduce antibiotic prescriptions by 25%. 19
NICE guidance suggests that rapid testing for group A streptococcal infections in primary care is neither cost-effective nor significantly reduces antimicrobial prescribing or improves patient outcomes.13,14 However, this analysis did not consider the use of rapid tests to manage Strep A outbreaks or their impact in pharmacy settings, where evidence suggests they might be more beneficial. 13 Between 2014 and 2015, a feasibility study showed that point-of-care testing (POCT) can be delivered in community pharmacies and potentially reduce inappropriate antibiotic use. 20
Offering symptomatic treatments with over the counter (OTC) medicines has done little to deter healthcare professionals from prescribing antibiotics, and patients continue to seek them. This may be due to a lack of patient awareness; GPs may feel pressured to prescribe antibiotics, as patients often perceive them as more effective.4,21
In this challenging environment, community pharmacists are emerging as key players in promoting appropriate antibiotic use and delivering symptomatic care to patients.
To explore the role of community pharmacists in the diagnosis and management of sore throat with the use of POCT service, a study was carried out to quantify the potential benefit in terms of healthcare resource usage and appropriate diagnosis of Strep A infection.
Materials and Methods
Service Overview and Participant Screening
A cross-sectional study was conducted from September 2018 to February 2019 in 29 pharmacies across England, which were supplied with free OSOM® Strep A test kit. Participants aged 16 or older who visited the pharmacist for a sore throat infection and were willing to participate were screened.
Training and Awareness
Pharmacy staff were trained to use the FeverPAIN scoring system for initial assessments. Participants presenting with ‘red flag symptoms’ (e.g. trismus, difficulty breathing, neck swelling, torticollis), atypical symptoms or severe presentations (unilateral or chronic symptoms, signs of sepsis, or recent antibiotics) were immediately referred to their GPs. Across the 29 pharmacies, pharmacists completed a training package on implementing the Strep A sore throat service and managing sore throats. Training modules covered diagnostic testing, data collection, symptomatic relief and the importance of antimicrobial stewardship. Service awareness was increased through in-pharmacy materials, online advertising and direct communication.
Data Collection and Analysis
Data from consultations, including patients’ names, gender, date of birth and address, were collected. By signing the consent form (Appendix 1), patients confirmed they were aged 16 or above and agreed to participate. Data were collected using a standard pharmacy form. Anonymised copies were sent by pharmacies and collated for electronic analysis using Microsoft Excel. In cases of missing data, listwise deletion would be applied and only completed cases will be included.
Screening and Testing Process
Screening involved observing for signs of viral sore throat infection which include cold-like symptoms such as cough, runny nose and hoarseness.22,23 Participants with signs of viral infection were assessed using the FeverPAIN score. Those with higher scores (4-5) were tested for Strep A infection, a colour immunochromatographic assay providing qualitative detection of Group A Streptococcus antigen within 5 min. Participants requiring medical attention were referred to their GP. For this study, all participants who should have been referred to their GP for antibiotics were identified as potential GP referrals (Figure 1).

Flow chat indicating the protocol of the swab test procedure in community pharmacies.
Individuals without signs of viral symptoms and those with red flag symptoms were excluded from the study and not assessed using the FeverPAIN score. Participants with a low FeverPAIN score were recommended over-the-counter symptomatic treatment according to the protocol.
Although FeverPAIN scoring was the primary tool, pharmacists were also permitted to use their clinical judgment if physical examination suggested a bacterial infection, thus ensuring alignment with real-world practice.
Survey
Patients answered questions about their sore throat condition, symptoms, treatment actions taken and satisfaction with the service. They also completed a questionnaire on their likely treatment choice (A&E, GP visit, pharmacist, self-care) if the diagnostic service had not been available (Appendix 2). This was used to estimate potential GP appointments that could be avoided in sore throat management.
Estimating Cost and Resource Utilisation
The economic impact of this service was estimated by assessing the proportion of participants with a viral infection who might have unnecessarily consulted their GP for antibiotics instead of seeking symptomatic treatment from a community pharmacy. Details of the unit costs are listed in Table 3.
Unit Cost of Resource Utilisation.

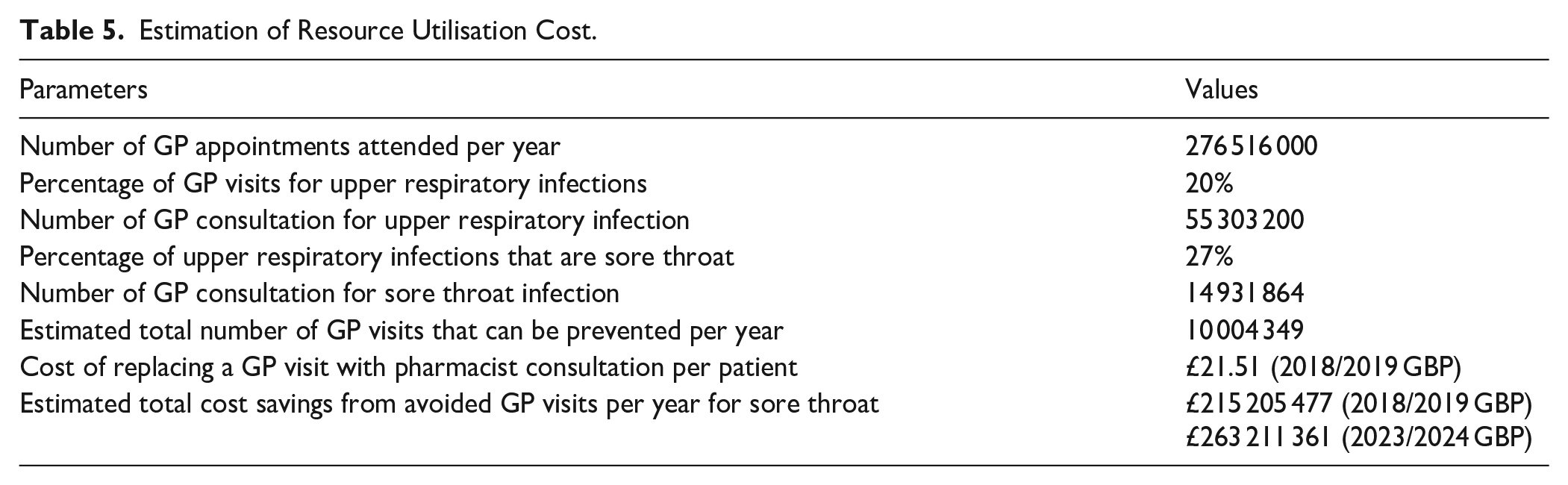
To estimate the national impact, we extrapolated findings using National Health Service (NHS) Digital data on annual General Practitioner (GP) consultations for sore throat. The percentage of participants who avoided GP visits due to the pharmacy service was applied to national GP attendance figures. Data from the NHS Digital were used to determine the total number of GP appointments attended per year between February 2018 and January 2019 for upper respiratory infection (URI). The number for sore throat consultations were there after extrapolated from this data.25,26 Cost savings were then estimated using the difference in consultation costs between pharmacists and GPs. Inflation adjustments were performed using the CCEMG – EPPI Centre Cost Converter to express values in 2023/2024 GBP. 24
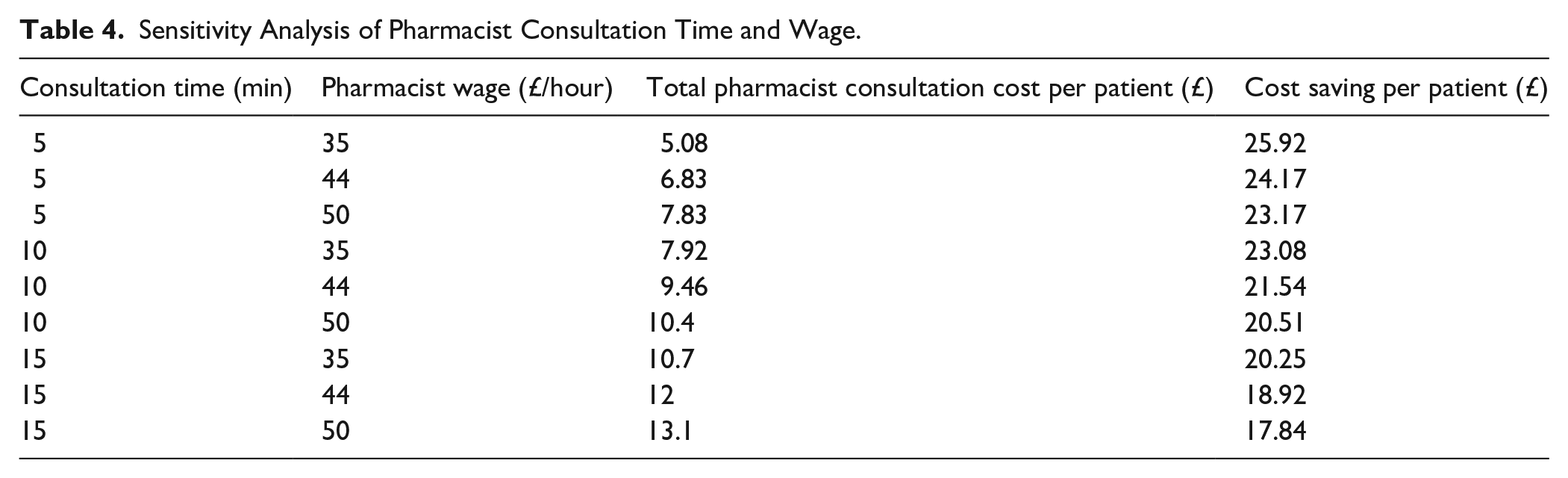
A sensitivity analysis was conducted to evaluate the impact of variations in pharmacist consultation time and hourly wage on cost savings.
Results
Recorded data was available for 59 participants at study completion based on individuals willing and available to take part in the study. A larger proportion of patients were female (63%) compared to male (37%). Most participants were between 26 and 64 years old (61%), 22% were aged 16 to 25 and 17% were over 65.
Out of the 59 participants, 36 were swab tested (in and out of protocol) while 23 participants did not undergo swab testing as these patients had either sign of viral infection, low FeverPAIN score or red flag symptoms (Figure 2). Twenty-two participants had low FeverPAIN score (0-3), while 21 had a high score (4-5). Despite a high FeverPAIN score in all participants, 86% (18/21) tested negative for Strep A, and only 14% (3/21) were positive. The protocol was not adhered to in 8 participants where they were swab tested based on the pharmacist’s clinical judgement.

Swab Test Service participant schematic. Participant journey through Swab Test Service: (a) within protocol and (b) all participants.
According to the pre-set protocol, only patients showing high FeverPAIN score should be swab tested. However, during the pharmacy consultation, some patients showed signs of bacterial infection signs but a low FeverPAIN score, pharmacists applied their clinical judgement by swab testing these participants for confirmation.
Of the total participants, 12 tested positive for Strep A and were potential GP referrals, leaving 47 participants. At the start of the study, when the multi-options questionnaire was administered, 34 participants opted for GP visit if the service was unavailable, 19 would consult the pharmacy and 9 opted self-care.
At the end of the service, 32 participants followed the correct full protocol outlined (Figure 2a). Majority of these were diagnosed with non-bacterial infections, with 11 negative tests for Strep A, 18 not tested due to low FeverPAIN score and 3 participants referred to a GP based on positive test results. Participants with low FeverPAIN score were recommended symptomatic treatment.
Of the 34 participants who initially selected the option to visit a GP, only 11 required GP appointments. Implementing the swab test protocol potentially saved 23 GP visits. Those testing negative for bacterial infection via the swab test were advised to seek over-the-counter treatment and instructed to seek GP consultation if symptoms worsened or showed no improvement after 7 days, in line with the protocol.
Descriptive statistics were computed for FeverPAIN scores, and Strep A positivity rates. The mean FeverPAIN score was 3.33 (Standard Deviation [SD]: 1.41), (95% CI: 2.73-3.93). Among participants tested for Strep A, the positivity rate was 28.57% (95% CI: 8.0%-49.1%).
While missing data was minimal, listwise deletion was applied to ensures that only complete cases were included in the analysis, reducing bias introduced by partial responses.
Estimated Resource Use and Potential Cost Savings
The cost of the OSOM® Strep A Test was £2.16 per application (including VAT). Average hourly wage for an advanced pharmacist practitioner was £44. We estimated that a consultation and application of the test would take approximately 10 min per participant, and therefore, a 10-min consultation was estimated to cost £7.30. Hence, the total cost, inclusive of the testing, was £9.46 per participant. The average cost of a GP consultation (average of 9.22 min) was estimated to be £31. 26 The resulting cost savings for every GP visit prevented per participant was £21.54 (GP costs minus pharmacist costs per participant, £31.00-£9.46).
Results of the sensitivity analysis indicate that as consultation time increases or pharmacist wage rises, the total cost of pharmacist consultation increases, reducing the potential cost savings per avoided GP visit. However, under all tested conditions, pharmacist-led consultations remain a cost-saving alternative compared to GP consultations. The full results are presented in Table 4.
Sensitivity Analysis of Pharmacist Consultation Time and Wage.
The total number of GP appointments attended per year from February 2018 to January 2019 was 276 516 000.
The results from a UK study indicated consultations for sore throat accounted for 27% of all consultations for respiratory tract infections. 2 The extrapolated estimate for the total number of annual preventable GP consultations was estimated as 276 516 000 per year. Twenty percent of all GP consultations are for URIs, and 27% of these URIs consultations are specifically for sore throat. Applying these proportions, the total annual GP visits for sore throat was estimated at 14 931 864. Extrapolating this data to study findings on percentage of GP consultations for sore throat symptoms that can be avoided through pharmacist-led interventions; the projected number of preventable GP consultations per year was 10 002 348.
As 36 participants were tested based on their FeverPAIN score; 34 participants noted that they would have visited their GP. Of these 34 participants, 11 tested positive, implying that 67% (23/34) of GP consultations were avoided. By applying these figures to the number of GP appointments attended between February 2018 and January 2019 in England, it was estimated that approximately 10 million GP visits can be prevented per year. Table 5 shows the monetary value of the preventable GP visits.
Estimation of Resource Utilisation Cost.
Discussion
This study assessed the effectiveness of point-of-care testing (POCT) for identifying Strep A infections and evaluated current screening methods to promote awareness of antibiotic resistance and self-care treatments. A significant portion of participants with high FeverPAIN scores tested negative for Strep A, challenging existing primary care guidelines that rely heavily on FeverPAIN assessment. 14
Our analysis showed that a high FeverPAIN score does not definitively indicate bacterial infection, as a considerable proportion of participants with high scores were Strep A-negative.
These results indicate that while FeverPAIN scoring provides a structured approach, it does not reliably predict bacterial infection. This finding aligns with previous studies questioning the predictive accuracy of clinical prediction rules (CPRs) such as FeverPAIN and Centor in detecting streptococcal infections in sore throat. This also suggests that relying solely on FeverPAIN may result in unnecessary antibiotic prescribing. We therefore recommend using rapid diagnostic tests as an adjunct to clinical assessment. However, an important strength of this study is that pharmacists exercised clinical judgment alongside FeverPAIN scoring. For example, some participants with low FeverPAIN scores but clinical signs suggestive of bacterial infection were swab-tested, ensuring more accurate identification of Strep A cases. 17
The FeverPAIN questionnaire was chosen over CENTOR scoring based on NICE recommendations, suggesting it has a greater probability of identifying bacterial infections. 27
It has been reported that some GPs have positive view of rapid testing for strep but worried about increase in GP attendance for sore throat. 28 The results of this study suggest that not only could unnecessary GP visits be avoided but also that more patients could be directed to the most appropriate form of treatment (symptomatic relief).
Our cost analysis revealed that swab testing for Strep A in pharmacies can significantly reduce healthcare costs and decrease unnecessary antibiotic prescriptions. Extrapolated nationally for NHS England, this could save approximately £215 million (2018/2019 GBP) and with currency adjustment to account for inflation, it amounted to £263 million (2023/2024 GBP). Findings from the sensitivity analysis also supports the economic advantage of pharmacy-based Strep A testing, demonstrating that even under conservative assumptions, the approach remains a viable strategy for reducing NHS costs.
Rapid diagnostic tests for sore throat management are approved and reimbursed in pharmacies in countries like France and Portugal but not in the UK. 29 NICE’s economic analysis deemed rapid tests in GP settings not cost-effective. 13 While our findings suggest significant cost savings when POCT is introduced in pharmacies solely from direct cost of GP and pharmacist consultation time, this indicates that a full economic analysis in pharmacy setting could be favourable.
A recent publication by NHS England acknowledged increased use of rapid testing for Strep A directly by consumer, and therefore recommends consideration of CPRs alongside the rapid testing, however routine use of it not recommended citing cost as one of the reasons. 30
An economic evaluation in 5 Canadian provinces showed cost savings of 1.3 to 2.6 million Canadian dollars per year when treating sore throat using rapid tests in pharmacies compared with GP settings. 31
At the time of our study, a similar one was conducted by NHS Wales using CENTOR or FeverPAIN scores between November 2018 and February 2020. 32 Analysis of the first 5 months, involving over 1700 participants, revealed fewer than 30% of swab-tested individuals were Strep A-positive. Approximately 92% of respondents said they would have contacted their GPs if the service hadn’t been available. 33 This service received high patient satisfaction, and pharmacists felt empowered to provide clinical services and educate patients. 34 Health Technology Wales concluded a 100% probability of POCT for Strep A being cost-effective in pharmacies at a threshold of £20 000 per QALY, though further research is needed. 17
A recent systematic review suggests potential benefits of using CPRs as a triage step before rapid testing for Strep A. 35
These findings support the integration of rapid diagnostic testing as an adjunct to FeverPAIN scoring, allowing for more precise differentiation between bacterial and viral infections. Such an approach can enhance antimicrobial stewardship and reduce inappropriate antibiotic use. This would also empower pharmacists to distinguish accurately bolstering their confidence in antimicrobial prescribing. By conducting swab tests, pharmacists can make informed decisions, ensuring patients receive the most effective and efficient treatment.
Following the recent implementation of the NHS ‘Pharmacy First Scheme’, this study warrants further large-scale research to validate findings and inform policy decisions.
The introduction of POCT was notably beneficial during the COVID-19 pandemic. This approach can also be applied to managing sore throat, especially considering the 2022 Strep A outbreak. Therefore, this service will enhance pharmacists’ role as antimicrobial stewards, mitigating antibiotic resistance and improving patient outcomes.
Limitations
While this study provides valuable insights into the feasibility of pharmacy-based Strep A testing, several limitations should be considered. A key limitation of our study was its small sample size, possibly due to the campaign being limited to a single pharmacy chain. This limits the generalisability of findings. Consequently, while we have presented national-level extrapolations of potential cost savings, these figures should be interpreted as illustrative estimates only. Larger-scale, multi-centre trials would strengthen the reliability and external validity of our findings and are recommended to confirm these cost-saving projections.
Although pharmacists followed a protocol based on FeverPAIN scoring, some exercised clinical discretion in testing participants who did not meet strict FeverPAIN thresholds. While this reflects real-world clinical practice, it introduces variability in adherence to the protocol and may have influenced diagnostic accuracy.
Participation was voluntary and restricted to individuals aged 16 and above who sought care at participating pharmacies. This may not reflect the broader population experiencing sore throat symptoms, particularly those who prefer GP consultations or self-care. This could also lead to an overestimation of the impact of pharmacy-based Strep A testing, as individuals who might have sought alternative healthcare options were not considered.
The exclusion of younger patients also affects the applicability of the findings to paediatric populations.
Additionally, patients accessing pharmacies may differ in health-seeking behaviour compared to those consulting GPs, potentially introducing bias in patient characteristics and their likelihood of antibiotic-seeking behaviour. Further research including a more diverse age range and a direct comparison with GP consultations is needed to better understand the full impact of pharmacy-based Strep A testing across different patient populations.
While training was provided to pharmacists, however, their experiences in throat swabbing could impact test results. Technique and personnel have been shown to affect swab performance. 34
Data was collected between September 2018 and February 2019, a period that includes peak respiratory infection seasons. Seasonal fluctuations in sore throat incidence and antibiotic prescribing patterns may impact the generalisability of cost savings estimates when applied to year-round National Health Service (NHS) data.
Conclusions
This study highlights the significant role of community pharmacies in the management of sore throat through the appropriate diagnosis of Strep A infection using rapid diagnostic testing. By effectively identifying Strep A infections, the service reduced unnecessary GP visits and, indirectly antibiotic prescriptions. Potential savings to the NHS was estimated at £263 million per year. These findings challenge current primary care guidelines and underscore the importance of incorporating rapid diagnostic testing in pharmacies considering the recent policy implementation of the NHS’s ‘Pharmacy First Scheme’. This approach aligns with this scheme and addresses the global concern of antibiotic resistance. While further research is needed to assess its impact fully, this study demonstrates the potential of pharmacy-based POCT for Strep A in enhancing patient care and promoting efficient healthcare resource utilisation.
Supplemental Material
sj-pdf-1-jpc-10.1177_21501319251340836 – Supplemental material for Streptococcus A Rapid Diagnostic Testing in England Community Pharmacies: Clinical and Economic Impact of Empowering Pharmacists in Management of Sore Throat
Supplemental material, sj-pdf-1-jpc-10.1177_21501319251340836 for Streptococcus A Rapid Diagnostic Testing in England Community Pharmacies: Clinical and Economic Impact of Empowering Pharmacists in Management of Sore Throat by Kay Edokpayi, Patricia Aluko, Flora Ka Kei Cheng, Darush Attar-Zadeh and Anne Dawson in Journal of Primary Care & Community Health
Supplemental Material
sj-pdf-2-jpc-10.1177_21501319251340836 – Supplemental material for Streptococcus A Rapid Diagnostic Testing in England Community Pharmacies: Clinical and Economic Impact of Empowering Pharmacists in Management of Sore Throat
Supplemental material, sj-pdf-2-jpc-10.1177_21501319251340836 for Streptococcus A Rapid Diagnostic Testing in England Community Pharmacies: Clinical and Economic Impact of Empowering Pharmacists in Management of Sore Throat by Kay Edokpayi, Patricia Aluko, Flora Ka Kei Cheng, Darush Attar-Zadeh and Anne Dawson in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We thank Arwen Caddy (Reckitt, Library & Knowledge Management, Hull, United Kingdom) for review of the manuscript.
Author Contributions
Conceptualisation, K.E. D.A. and A.D. Formal Analysis, P.A and F.C.; Writing – Original Draft Preparation, K.E., P.A. and F.C.; Writing – Review & Editing, K.E., P.A., F.C., D.A., and A.D.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Reckitt Health.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: K. Edokpayi, P. Aluko, F. Cheng and A. Dawson are employees of Reckitt Health. D. Attar-Zedeh has received honoraria from various companies for respiratory related work. Reckitt Health was involved in the design, interpretation and writing of the manuscript. Reckitt had no role in the collection and analyses of the data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
