Abstract
Introduction:
Hepatitis C virus (HCV) treatment with pan-genotypic direct acting antivirals is highly effective, given an evidence-based simplified treatment algorithm. Yet access to treatment is limited among vulnerable populations.
Objective:
We assessed the effectiveness of site-specific HCV treatment workflows on HCV care for vulnerable populations in Austin, Texas.
Methods:
Patients diagnosed with chronic hepatitis C enrolled in care at a study site were eligible for this prospective, single-arm clinical trial. We assessed the proportion of participants that: (1) were prescribed treatment, (2) initiated treatment, (3) completed treatment, (4) were assessed for cure, and (5) achieved cure. We also evaluated implementation using the reach, effectiveness, adoption, implementation, and maintenance (RE-AIM) framework.
Results:
Of 62 participants, 89% had ever experienced homelessness and 94% had ever used drugs. An estimated 66% (95% CrI, 42%-84%) were prescribed treatment and 49% (95% CrI, 26%-70%) initiated treatment. An estimated 38% (95% CrI, 20%-58%) completed treatment, 14% (95% CrI, 4%-44%) were assessed for cure, and 10% (95% CrI, 2%-35%) achieved cure.
Conclusions:
We identified gaps along the HCV care cascade between: (1) enrolled to prescribed treatment and (2) completed treatment to assessed for cure. Site-specific HCV treatment workflows were insufficient to engage participants in care and avoid treatment delays. Novel approaches are needed and these may include patient outreach, patient navigation, test-and-treat protocols, and removing financial or payor barriers to medication access.
Trial Registration:
Registered on ClinicalTrials.gov on July, 14, 2022. Identifier: NCT05460130. https://clinicaltrials.gov/ct2/show/NCT05460130.
Keywords
Introduction
Hepatitis C virus (HCV) is a leading cause of liver disease with more than 2.4 million people living with HCV in the United States (US).1,2 Direct acting antivirals with a >95% cure rate and an evidence-based simplified treatment algorithm make HCV elimination possible.3,4 However, vulnerable populations, such as people experiencing homelessness (PEH) and people who inject drugs (PWID), have limited access to HCV treatment.4,5 The prevalence of HCV is as high as 53% among PEH and greater than 40% among PWID.6 -9 In the US, most new infections are primarily associated with injection drug use. 10 Achieving HCV elimination goals, such as the US 5-year national plan to eliminate HCV and the World Health Organization’s 2030 goal for hepatitis elimination, requires targeting vulnerable populations.3,5
Patients with HCV have been successfully treated in primary care settings.11,12 When PEH and PWID are retained through treatment, HCV cure rates are similar to the general population.13,14 However, in PEH and PWID there are significant barriers to HCV care limiting treatment completion. These barriers include missed appointments, incomplete laboratory assessments, lack of medication access, and struggles with medication adherence. 15
In this study, site-specific HCV treatment workflows addressing barriers specific to PEH and PWID were implemented across 7 clinic sites in Austin, Texas. The participant proportions completing each step of the HCV care cascade, including cure were evaluated. 15 Unlike previous studies which have evaluated cure among participants who initiated treatment, we included all patients eligible for HCV treatment. We also evaluated the implementation of these treatment workflows using the reach, effectiveness, adoption, implementation, and maintenance (RE-AIM) implementation science framework.
Methods
Implementation of Site-Specific HCV Treatment Workflows
We conducted a prospective single-arm clinical trial of site-specific HCV treatment workflows at Healthcare for the Homeless (HCH) sites serving PEH and PWID within a Federally Qualified Health Center (FQHC) in Austin, Texas. 16 Research and clinical staff co-designed and implemented site-specific workflows at clinics in August 2022. The clinical workflows were developed based on the simplified treatment algorithm and were designed to address barriers identified by both patients and clinic staff. The workflows included tasks to address differing staff ratios, roles, clinic space, and characteristics of the vulnerable patient population seen at each clinic site. 15 Additional details about this approach, the study methods, and determination of sample size, have been published.15,17 This study was approved by the University of Texas at Austin’s Institutional Review Board (IRB) and CommUnityCare’s Research and Quality Improvement Committee. 17 This research was reported using applicable CONSORT Guidance (Supplemental File 1). 18
Setting
This study was implemented in 7 primary care clinics serving vulnerable populations. The study clinics included a clinic housed inside a local homeless shelter (Austin Resource Center for the Homeless (ARCH)); 2 traditional brick-and-mortar clinics, 1 focused on medically complex PEH (Care Connections [CareCo]) and another on treatment of opioid use disorder (Medication Assisted Treatment [MAT] Clinic); as well as walk-up mobile and street medicine clinics (at 3 Mobile Sites and the Street Team).15,17
Participants
Patients aged 18 years and older, diagnosed with chronic hepatitis C (HCV antibody positive with a detectable viral load), and enrolled in care at 1 of the 7 study clinics were eligible for study participation. Patients were excluded from study participation if they met any of the following criteria: human immunodeficiency virus (HIV) or hepatitis B virus (HBV) co-infection; decompensated cirrhosis; HCV treatment experienced; status post liver transplantation or active on liver transplant waitlist; currently pregnant; could not provide informed consent; or provider or patient desired GI/hepatology specialty referral for treatment. Study participants gave written, voluntary, informed consent at enrollment. 17 Enrollment eligibility was directed by the simplified HCV treatment protocol requirements at the time of the study.
Recruitment and Enrollment
Enrollment occurred from September 29, 2022 through March 29, 2023. Patients with a positive HCV antibody test and detectable viral load between September 1, 2021 and March 29, 2023 were identified through electronic medical record (EMR) review. Potentially eligible participants were recruited and enrolled during scheduled appointments, when present at Mobile sites, or seen for care by the Street Team. The research team implemented assertive outreach strategies for this vulnerable population, including field visits with clinic staff and maintaining a presence in clinics to catch potentially eligible patients. An offer of treatment or treatment initiation was not required for enrollment. Study enrollment was not required to receive treatment.
Data Collection
Data were collected by review of EMRs and through interviews from September 2022 to December 2023.
Demographic and clinical data were collected from the EMR including: date of birth, sex, gender, race, ethnicity, preferred language, medical comorbidities, HCV Ribonucleic Acid (RNA) viral load, HIV, HBV, sustained virologic response 12 weeks after treatment completion (SVR12), and date of treatment prescription, medication pick-up, and treatment completion.
In participant interviews conducted at study enrollment, participants self-reported socioeconomic status, health care coverage/payor, education, social support, employment, housing, substance use, alcohol use, incarceration, tattoos, and psychiatric diagnoses. 17 Among participants who initiated HCV treatment, interviews were conducted at treatment completion to capture medication adherence and treatment side effects.
Key Variables
Housing status was determined by asking participants where they usually slept in the last 7 days. The Department of Housing and Urban Development (HUD) definition of homelessness and published literature informed our 5 mutually exclusive housing categories: unsheltered homeless, sheltered homeless, unstably housed (transitional housing, group home, and couch surfing), housed and formerly homeless, and never homeless.19,20 Opioid use disorder (OUD) treatment was determined by self-report of being on buprenorphine or methadone for OUD. Substance use and injection drug use (IDU) were categorized as current use (if used within the 30 days prior to enrollment) or having ever used.
A prescription for treatment in the EMR was used to indicate that treatment was offered. Treatment initiation was defined as having a first bottle of medication dispensed. However, dispensing records were not systematically updated in the EMR, and if missing at the time of review, record of medication refills or treatment completion was used to infer treatment initiation. Laboratory assessment result was used as a proxy for completed treatment, with time restrictions in place where the HCV RNA test result had to be at least 8 or 12 weeks after starting treatment, depending on the medication. In the absence of test results, other markers for treatment completion were used including self-report, date of last bottle pickup +28 days, or clinical documentation of treatment completion in a note in the EMR. If treatment initiation or completion could not be determined from the EMR, data were collected directly from the participant or care team. The study duration was designed to allow sufficient time for treatment completion. 17
Clinical and Implementation Outcomes
Primary clinical outcomes were the proportion of all study participants completing steps along the HCV care cascade, including the proportion who (1) were prescribed treatment, (2) initiated treatment, (3) completed treatment, (4) were assessed for cure (HCV RNA SVR12 laboratory test), and (5) achieved cure, defined by SVR12. We also quantified the average number of days from treatment offered to initiated. The RE-AIM framework was used to evaluate implementation outcomes of the site-specific workflows. 21
Analysis
Using the “intention-to-treat” principle, all enrolled participants were included in the analysis, whether or not they initiated treatment. Descriptive analyses were conducted using the dyplr and tidyverse packages in R software (v4.2.3). 22 We applied a Bayesian probit model to estimate the marginal prevalence for each of the binary outcomes in the care cascade using Stata v18 and its suite of Bayesian functions. 23 The model itself was fit with random intercepts only (by clinic, to account for correlation among participants from the same clinic) and used inverse-gamma priors with prior shape and scale both set to 0.01. An analytic formula was applied to compute the marginal prevalence from the within-cluster intercept. We report the posterior median as the marginal prevalence point estimate and the associated equal-tailed 95% credible interval (CrI) as its uncertainty range. GEE and GLMM approaches were initially considered but proved difficult to implement. Given the small sample size (which became progressively smaller further down the care cascade) and the relatively large number of clusters, estimation of the marginal prevalences with maximum-likelihood methods raised a number of computational issues that were avoided by use of the Bayesian probit model.
Evaluation of Implementation
A convenience sample of clinic staff from each clinic site were interviewed to measure implementation outcomes. 17 Implementation outcomes were qualitatively and quantitatively measured and evaluated in aggregate, using data extracted from the EMR and collected during interviews with clinic staff. 21
Adoption reflected the proportion of providers who were trained. 17 Fidelity captured appropriateness, timing, and frequency of ordering laboratory assessments. Guided by the RE-AIM framework, the research team conducted 7 in-person, audio-recorded, qualitative interviews with providers (medical doctors and nurse practitioners) across all clinic sites to assess implementation, maintenance, and acceptability of the HCV treatment workflows. 21 Our qualitative data analysis was informed by the Framework Method from Gale et al. 24 Interviews were audio recorded, transcribed, and analyzed using thematic descriptive and interpretive coding techniques by two coders. A qualitative matrix was created to identify and categorize major themes, identify characteristics of the data, and identify relationships between themes. 24 Due to the small sample size, implementation outcomes were analyzed in the aggregate, rather than by clinic site.
Results
Study Participants
We approached 73 (43%) of the 171 patients eligible for our study. We were unable to approach 57% of eligible patients because we could not locate them or they did not come to a clinic site during the enrollment period. We enrolled 68 (93%) of those approached (Figure 1), 6 of whom were later withdrawn due to no longer meeting the study criteria (4 no longer eligible for the simplified treatment algorithm, 1 not treatment naive, and 1 with an undetectable viral load).

Study flow diagram.
Participants were majority white (79%), non-Hispanic (77%), male (74%), and an average age of 52 years (Table 1). Most participants were patients of the brick-and-mortar clinics – CareCo (30%) and MAT (29%) – compared to the shelter, mobile, or street sites. Most had experienced homelessness at some time in life (89%), with 39% currently experiencing sheltered or unsheltered homelessness. The Travis County Medical Access Program (MAP), which, in the absence of state-expanded Medicaid, provides health care coverage for those living at or below 200% of the federal poverty level, provided medical coverage to 52% of participants. 25 The majority of participants graduated high school (31%) or its equivalent through GED (34%). Of the 53% employed, over half generated income in the informal sector of the economy through day labor (23%) or panhandling/informal work (38%).
Study Sample Population Characteristics.

Clinic distribution was categorized by the clinic where the participant was enrolled, since a patient may be seen at more than 1 clinic.
Five Housing Status categories were collapsed into two: (1) Unhoused Homeless (unsheltered homeless, sheltered homeless) and (2) Housed (unstably housed, housed formerly homeless, housed never homeless).
Health Care Coverage/Payor is not mutually exclusive, 6 SPs had multiple types of coverage/payor.
Listed Substances included marijuana or K2, cocaine or crack, prescription stimulants, methamphetamine, heroin, PCP or ketamine, and prescription opioids.
A positive AUDIT-C Score was ≥ 3 for females and ≥ 4 for males.
Mental Health Conditions included diagnoses of depression, anxiety, bipolar disorder, post traumatic stress disorder (PTSD), schizophrenia or schizoaffective disorder, and other mental health conditions.
Chronic Conditions included diagnoses of diabetes, heart disease, hypertension, chronic kidney disease, and chronic lung disease.
Most participants had at least 1 tattoo (76%), with nearly half from unlicensed sources such as prison (30%) or a friend/self-administered (19%). A vast majority had a history of incarceration after the age of 18 years (92%). Most participants were currently using alcohol (64%) and/or other substances (69%), while almost all participants had ever used substances (92%).
The majority of participants had at least 1 mental health condition (81%), with 74% having more than 1. Mental health conditions included depression (71%), anxiety (69%), post-traumatic stress disorder (PTSD; 44%), and bipolar disorder (40%). Fifty-eight percent had a dual diagnosis of a mental health condition and current substance use. The majority of participants had at least 1 chronic physical health condition (60%), hypertension being the most prevalent (48%).
HCV Care Cascade and Implementation of Site-Specific HCV Treatment Workflows
At the end of the 16-month data collection period, participants were at various steps along the HCV care cascade (Figure 2). All study participants were diagnosed with HCV. An estimated two-thirds (66%, 95% CrI, 42%-84%) of participants were offered treatment (Reach). Forty-nine percent initiated treatment (95% CrI, 26%-70%), with an average of 57 days between being offered and initiating treatment. An estimated 38% (95% CrI, 20%-58%) of all participants completed treatment, 14% were assessed for SVR12 (95% CrI, 4%-44%) and 10% achieved cure (95% CrI, 2%-35%; ; Effectiveness). When including only participants who initiated treatment in the denominator, 26% completed treatment and 20% were cured.11,12
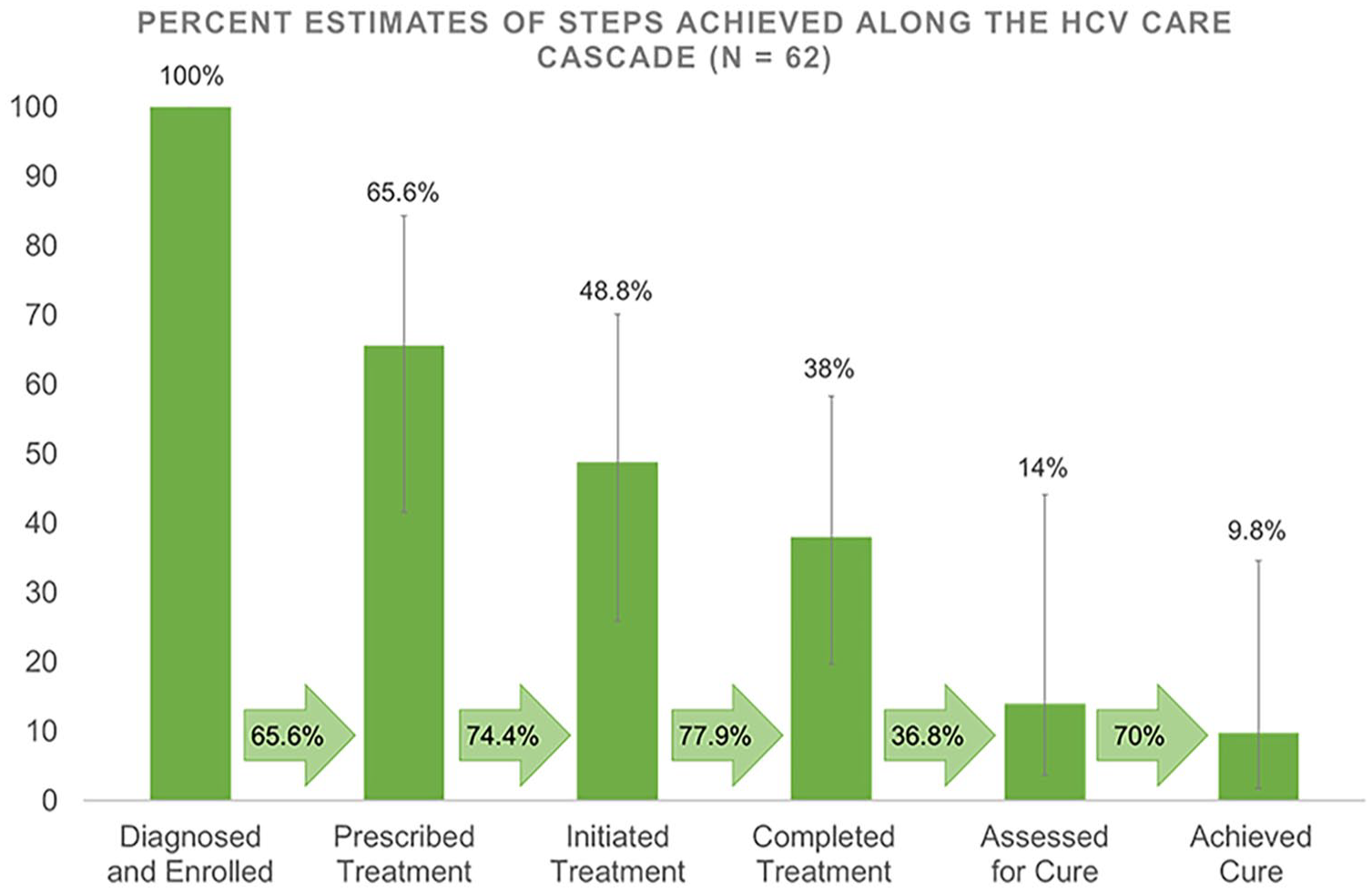
Cluster-adjusted estimates and 95% CrI of steps achieved along the HCV care cascade.
Among those interviewed at treatment completion (N = 20), 45% reported side effects during treatment and 65% reported missing medication doses for reasons including forgetting to take the medication and running out of medication.
Nine providers were trained on the simplified treatment algorithm and to implement the site-specific HCV treatment workflows across 7 clinical sites (Adoption). Providers reported seeing more patients with HCV and feeling more empowered to treat HCV rather than referring patients to GI/hepatology specialists. Additionally, workflows clarified the division of labor across teams and departments, making the Patient Assistance Program (PAP) easier and more efficient for patient medication access.
I never felt like I had to do a lot of leg work, someone was already on the cases as I ordered the [HCV] medication, already making sure that the PAP [Patient Assistance Program] application was being completed appropriately. There was someone following up on the PAP application. It was great. It was very easy to do. I didn’t have to do anything. [Most] of the time when I order PAP medications for people, I have to actively follow up on these things. But, this was the easiest PAP application I’ve ever had to do. (Provider at 1 clinic site)
However, fidelity to site-specific treatment workflows was low. Additional diagnostics were often ordered for participants, when not required by the simplified treatment algorithm, including HCV genotype (73%) and abdominal ultrasounds for non-cirrhotic participants (FIB-4 score < 3.25; 20%). Only 22% of participants with a lab-ordered genotype had payors known to require genotyping for drug coverage (ie, certain Medicaid or Medicare plans). In addition to payor requirements, providers ordered unrecommended tests based on prior experiences of finding hepatological cancers in some patients and to err on the side of caution (Implementation).
I order [ultrasound] for all of [my patients]. Because I’ve had several patients get liver cancer. And maybe I’m a little shy about that. So, for me, I err on the side of caution and everybody gets an [ultrasound] order. (Provider working at 1 clinic site)
I did order a couple patients ultrasounds. And to be fair, there was one patient that we found, HCC (Hepatocellular Carcinoma) in. So, after that I was like, everyone needs an ultrasound. . .The other thing is for patients with Medicare or Medicaid, those got tricky because sometimes their insurance would [say] we need this and they have to order it. And so, for some patients with Medicare or Medicaid, I would just order the extra tests that I knew insurance might need, just so that we can move forward with treatment. (Provider working across multiple clinic sites)
During RE-AIM qualitative interviews with clinic staff, suggestions were made to increase fidelity to the workflows, including (1) periodic re-education on the simplified treatment algorithm and reminders to staff to implement the simplified workflows, and (2) an added step to check-in with patients on medication adherence and interactions during treatment. It was also recommended to create a centralized registry of patients with HCV appointments to actively target outreach and engage patients not recently seen in clinic. Providers requested increased access to gastroenterology consultation services for complex cases. All providers recommended training other providers on the simplified treatment algorithm and many believed the site-specific workflows increased engagement in care and improved patient outcomes (Maintenance).
Discussion
In this single-arm trial of implementing site-specific HCV treatment workflows, we used a population health approach and applied the “intention-to-treat” principle, defining the denominator as all enrolled participants, including participants who did not initiate treatment or were lost-to-follow-up. Approximately half of all study participants initiated treatment (49%), and only 10% achieved cure. We learned that simplified, site-specific HCV treatment workflows designed to optimize in-clinic factors are incomplete solutions to improving HCV treatment in PEH and PWID. These workflows must be supplemented with interventions to address factors outside the clinic to improve cure rates in this vulnerable population. These may include targeted outreach and navigation support, and the elimination of financial and regulatory barriers to accessing curative medicines. Despite the small proportion cured, we anticipate lasting change beyond our study because of the provider training and the human-centered design approach to develop the simplified HCV treatment workflows. More providers and clinics are now treating HCV than before our study.
On average, across Texas there is a 31% gap between diagnosis and cure in the general population, but here, in this vulnerable population, the gap was as large as 90%. 10 We identified 2 main gaps along the HCV care cascade: (1) diagnosed and enrolled to initiated treatment, and (2) completed treatment to achieved cure. Potential reasons for delays in treatment initiation may include payor requirements of diagnostic tests not in the simplified treatment algorithms (eg, genotype and ultrasound) and patients not returning to clinic after screening or diagnosis. Approximately two-thirds of participants had a genotype ordered and less than half of patients diagnosed initiated treatment. As HCV guidelines have evolved, policy changes are needed to expand medication access without unnecessary diagnostic tests. We have already seen examples of policy changes, including Texas Medicaid revoking the requirement to demonstrate sobriety from substance or alcohol use. Additionally, after our enrollment period, the HCV simplified treatment algorithms were updated to include guidelines to treat patients co-infected with HIV/HCV (excluded from our study per guidelines at the time). 26
The majority of patients who complete treatment are clinically cured, therefore focusing on the first care gap and those diagnosed who are not getting treatment should be the priority when considering next steps. This requires considering and addressing barriers between diagnosis and treatment such as the transient nature of the population, mobile phone access, competing needs, and the impact of mental health disorders on time management. 15 Furthermore, reduction of financial barriers to medication access is critical, such as ensuring Medicaid programs cover evidence-based HCV treatment without unnecessary prior authorization requirements or other delays. In addition to policy changes, increasing acceptability and implementation of test-and-treat approaches where patients initiate treatment the day of diagnosis may address the gap between diagnosis and treatment initiation and reduce the time to treatment initiation.
Limitations
Use of a single-arm design allowed for only a descriptive analysis of HCV treatment in this population. Comparative studies are needed to better understand the intervention’s impact on HCV treatment.
Self-reported data collected from patient interviews and clinical care team reports may have been limited or incorrect due to poor recall or in case of patients, distrust when sharing sensitive information. 13 However, warm handoffs from clinic staff to the research team helped build trust between the research team and participants, potentially mitigating this issue. Manual chart review may be subject to human error therefore, we double checked missing fields and triangulated data with other sources where applicable (eg, treatment initiation and completion). Additionally, due to missing data and varying lengths of time to achieve these steps, proportions along the HCV care cascade in Figure 2 are conservative estimates. Patients may achieve steps along the care cascade in the future.
Our final study sample size did not meet our pre-specified target. At the local level, a city ordinance criminalizing camping or soliciting money in public areas went into effect in Austin in May 2021, coinciding with the study timeline. This may have compromised linkage to care, recruitment, enrollment, and follow-up among PEH. 27 Multiple outreach and navigation strategies were implemented to improve recruitment while aiming to minimize impact on clinic implementation of the site-specific workflows. Clinic staff helped to identify eligible patients approaching the clinic as walk-ins. The sexual health navigator, whose role includes helping patients navigate the HCV care cascade, called eligible patients to engage them in care. Ultimately, this yielded few additional appointments scheduled, since many patients have no or inconsistent phone access. Research staff traveled with clinic staff at the mobile and street clinics where some patients lived or camped, to help find them. Additionally, the generalizability of our findings may be limited to patients who engage in care. Though our sample size was small, we were able to derive proportions completing steps along the HCV care cascade and gain relevant insight into steps where future interventions will be most useful.
Conclusions
Implementing site-specific HCV treatment workflows in primary care clinics treating vulnerable populations is feasible and can be optimized, including iterative testing and adaptation in future interventions, which may improve patient outcomes. Future studies need to explicitly test the effectiveness of patient outreach, patient navigation, and removal of financial barriers. Once an intervention is demonstrated to be effective, a cost analysis could help policymakers to better understand the cost implications.
However, successfully treating and curing HCV in these vulnerable populations, requires innovative methods to engage and retain patients in care. 13 Based on our findings, the following themes may be key focus on in future research: (1) expand beyond in-clinic interventions to include targeted outreach and navigation support across the HCV care cascade with community health partnerships; (2) continue to remove financial and policy barriers to medication access; and (3) increase acceptability and implementation of test-and-treat approaches, to reduce delays and barriers between diagnosis and treatment initiation. 5 Addressing homelessness, addiction, and the multiple other social determinants of health at a community level can facilitate efforts to engage these populations in care. 13 Successfully engaging and curing this population will be necessary to reach HCV elimination.
Supplemental Material
sj-doc-1-jpc-10.1177_21501319251330622 – Supplemental material for Implementation of Site-Specific Hepatitis C Virus Treatment Workflows for Vulnerable, High-Risk Populations: A Prospective Single-Arm Trial
Supplemental material, sj-doc-1-jpc-10.1177_21501319251330622 for Implementation of Site-Specific Hepatitis C Virus Treatment Workflows for Vulnerable, High-Risk Populations: A Prospective Single-Arm Trial by Anmol Desai, Kia Reinis, Lauren O’Neal, Patrick Chang, Cristal Brown, Michael Stefanowicz, Audrey Kuang, Deepak Agrawal, Tim Mercer and Darlene Bhavnani in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We would like to thank CommUnityCare for their input as our clinical partner, as well as patients and staff. We would also like to thank Paul J. Rathouz for his advice on our statistical analysis.
Author Contributions
DA, DB, CB, and TM, as co-principal investigators and co-investigators, conceived the Erase Hep C study, that encompasses this single-arm prospective study. AD, KR, LO, TM, and DB contributed to the development of the data collection tools and qualitative interview guides. AD, DB, KR, LO, PC, and TM synthesized and curated the data. AD and KR conducted the interviews with participants and qualitative analysis of the qualitative interviews with clinic staff. AD, DB, and PC conducted the analytical design and formal analysis. DB and TM acquired funding for this study. AD, AK, CB, KR, MS, and TM conducted the investigation process. AD, DB, KR, LO, and TM developed methodology and design of the study. As clinicians familiar with the study sites and clinic staff, TM, CB, MS, and AK provided guidance on the methodology to conduct this study at each clinic site. AD and KR administered the project with supervision and overall guidance from DB and TM, as co-principal investigators of the Erase Hep C study. AD drafted the initial version of the manuscript, with significant feedback from KR, LO, DB, and TM in editing subsequent versions. AD managed the references and finalized the manuscript after edits and review by all co-authors. AD wrote the content for the background. AD wrote the methods section, with PC writing the analysis portion, and critical review from TM and DB. TM, DB, LO, and KR provided critical review of the results. All authors contributed to the conception and design of the study. All authors contribute to reviewing and editing iterative drafts of this manuscript. All authors read and approved the final manuscript.
Ethical Considerations
The Erase Hep C study was approved by the University of Texas at Austin’s Institutional Review Board (IRB) and CommUnityCare’s Research and Quality Improvement Committee.
Consent to Participate
All participants provided written informed consent prior to enrollment in the study.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors report grant funding from Gilead Sciences. The trial was sponsored by The University of Texas at Austin.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclose receipt of the following financial support for this work by a grant from Gilead Sciences, Inc (Award Number: IN-US-987-6016) and core funds of the Dell Medical School at The University of Texas at Austin (to DB and AD). The content is solely the responsibility of the authors. The trial sponsor is The University of Texas at Austin Dell Medical School.
Data Availability
De-identifiable data is available to researchers and other stakeholders working to expand access to HCV treatment upon request. Identifiable data of those who provided written informed consent for future use may be shared with collaborating researchers in future studies. Data collection instruments and statistical code can be made available upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
