Abstract
Objective:
Multicancer Detection (MCD) tests, such as the GRAIL Galleri, offer a novel approach to cancer screening by detecting cancer-specific methylation patterns in cell-free DNA through a single blood sample. This study evaluated an 18-month implementation of MCD testing in a tertiary ambulatory internal medicine clinic.
Patients and Methods:
Between June 2022 and November 2023, 2244 asymptomatic (without symptoms attributed to cancer) patients underwent MCD testing. The study focused on operational workflows, patient and physician education, and diagnostic follow-up of positive results. Standardized materials, including electronic health record (EHR) workflows, FAQs, and diagnostic pathways, were developed to facilitate implementation. Challenges included managing false positives, patient anxiety, costs, and ethical considerations.
Results:
Of the 2244 patients tested, 17 (0.76%) had positive results, and 15 underwent further diagnostic evaluation. Cancer was confirmed in 11 (73.3%) patients, including cases of breast, colon, esophageal, lymphoma, ovarian, and pancreatic cancers. Four patients had no identifiable malignancy despite comprehensive work-up.
Conclusions:
MCD testing is feasible in routine clinical workflows, with 73% of positive cases yielding cancer diagnoses. While promising, further research is required to assess long-term outcomes, cost-effectiveness, and optimal implementation strategies of cancer interception in broader healthcare settings.
Keywords
Introduction
In the past 10 years, over half of the most common cancer types in both adult men and women have shown decreased mortality rates in the United States. 1 In both men and women, melanoma and lung cancer continue to have the sharpest declines in mortality. 1 Unfortunately, mortality rates associated with cancer of the bones and joints, brain, liver, uterus, and pancreas continue to climb. 1 Despite the recent evolution of novel therapies, cancer is still a leading cause of global mortality. Early diagnosis of cancer makes it possible for patients to have less invasive treatment options, with relatively lower morbidity associated with the treatments. Earlier detection of late-stage cancer has been associated with a reduction of cancer-related deaths by 15% within 5 years, highlighting the significant need for better cancer testing. 2 In addition to risk reduction and prevention, there is a need for better options for early interception of cancers. The concept of cancer interception is the proactive approach to identifying and intervening early in cancer development and progression to invasive disease. 3 Currently, population-wide screening is only recommended for a few cancers, such as breast, colorectal, cervical, and (in high-risk individuals) lung. Cancers that have demonstrated increased mortality rates, such as pancreas and ovarian, have had no reliable biomarkers for interception or population-based screening tests in asymptomatic patients.4,5
Blood is inherently well suited as a potential biomarker source for cancer screening as it may contain circulating tumor cells, microRNA, and/or cell-free DNA (cfDNA) “shed” from a hitherto occult cancer. 6 This potential has led to the development of novel blood-based, high-performance genomic technologies allowing for early detection of signals from multiple cancers, giving rise to a new paradigm of screening tests called multi-cancer detection (MCD) tests. 6 MCD blood tests rely on analyses from cancer-specific DNA methylation patterns and have the potential to detect numerous cancer types in a single blood sample. A model of cancer interception using MCD testing published by the AACR concluded that late-stage (III+IV) incidence could be reduced by 78%. 7 Furthermore, cancer-related deaths in adults aged 50 to 79 years could be reduced by MCD testing. 7 The projections of MCD testing on Late-Stage incidence using Multi-State Disease were further validated in other studies, which further supported that MCD screening could lead to reductions in late-stage cancer incidence.8,9 MCD evaluation for 50 distinct cancer types became commercially available after a US Food and Drug Administration Investigational Device Exemption was obtained to validate the test.
The MCD test represents a potential groundbreaking advancement in cancer interception diagnostics, detecting multiple cancer types from a single blood sample draw. This test could, therefore, augment current preventive cancer screening and prevention strategies. While the technology holds promise for earlier detection and improved patient outcomes, a significant gap exists in the literature regarding its implementation within health systems. Current studies primarily focus on the accuracy, sensitivity, and specificity of MCD tests.8,9 There is limited research on integrating MCD into routine clinical workflows, addressing potential ethical considerations, and assessing its cost-effectiveness on a large scale. Also, challenges such as clinician training, patient education, and the impact on healthcare resource utilization remain underexplored, highlighting the need for comprehensive studies on the practical and systemic aspects of MCD deployment in healthcare settings. The present report describes the implementation of the MCD test in an ambulatory general internal medicine (GIM) clinic, including the clinical details of patients with positive MCD tests, the diagnostic methodology used to follow up on MCD-positive patients, both patient and clinician education to facilitate informed decision-making and electronic health record (EHR) integration processes.
Methods
Study Population
The study was conducted as a pragmatic trial in an executive health clinical practice where patients presented with MCD tests done elsewhere and sought information on their test results. In order to be proactive in providing education to patients while also learning from the experience, the study team provided patients with information on the role of MCD testing as an option to augment but not replace standard established screening tests. After extensive analyses of the performance of the commercially available MCD test and institutional experience with the Pathfinder study using the Grail Galleri test, a clinical decision was made to offer the MCD testing to our patients. 10 Access to the Grail MCD testing began on 01 June 2022. The study was approved by the Institutional Review Board (IRB) to retrospectively abstract data from the EHR for patients who had provided prior research consent. This IRB review and approval was determined to be exempt under section 45 CFR 46.104d, category 4. To be eligible for this study, all patients had to have been empaneled within the GIM executive health practice and would have had prior research authorization within their EHR file. In addition, they would have completed MCD testing between 01 June 2022 and 30 November 2023. If a patient had more than 1 test collected and resulted, only the first test (the “index” test) was included, and subsequent tests were excluded from the final analysis. Descriptive statistics were reported as median (interquartile range) or number of observations (percent of total). Statistical analyses were performed using R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria).
While considering MCD as a clinical option, we identified several operational and clinical practice processes that needed to be implemented before the test was introduced on 01 June 2022 (see supplemental Figure 1). The clinical practice leadership worked with institutional stakeholders in preparation for MCD implementation, and these included multidisciplinary teams: Cancer Center, Department of Medicine, Patient Education, Radiology, and multiple specialties (ie, gastroenterology, hematology, and endocrinology) where potential positive cases would be evaluated. In addition, input was also obtained from institutional leaders, who had been site investigators for the first research study (
Physician Education
It was recognized that successful implementation of the test offering in the clinical practice would require physician understanding and support, especially given the uncertainties and legitimate concerns related to using a test that was not yet FDA-approved. Hence, physician education was deemed a top priority, and specific MCD-related educational materials were prepared for physicians working in the practice who would be responsible for ordering the test and evaluating positive results. In addition, in collaboration with institutional

Snapshot of frequently asked question document created by our general internal medicine executive health team as well as pathfinder 1 investigators from our institution.
EHR Integration
Following the decision by the clinical practice to implement MCD, the clinical informatics team comprehensively evaluated the ability to integrate with the institutional EHR (Epic Systems Corporation; Verona, Wisconsin). The EHR orders for the MCD test were created after the decision was made to offer the test with 2 options (Figure 2): a mail-out (mail order) and on-site (lab collection). The idea behind providing the mail order option was to have test results before the patient came for their yearly in-person general medical exam visit. This would allow for a diagnostic workup at the institution if the MCD were positive, optimizing the patient experience. The order clearly outlined that MCD was a screening, not a diagnostic test (Figure 2). In addition, education was given regarding cohorts for whom testing was not recommended (ie, pregnancy or those undergoing active cancer treatment within the past 3 years). In addition, the difference between the 2 order options is clearly outlined (Figure 2). The MCD test was processed at the Grail company (Menlo Park, CA) testing laboratory. The results were placed in the company’s Grail Provider portal, which interfaces with the institutional EHR.
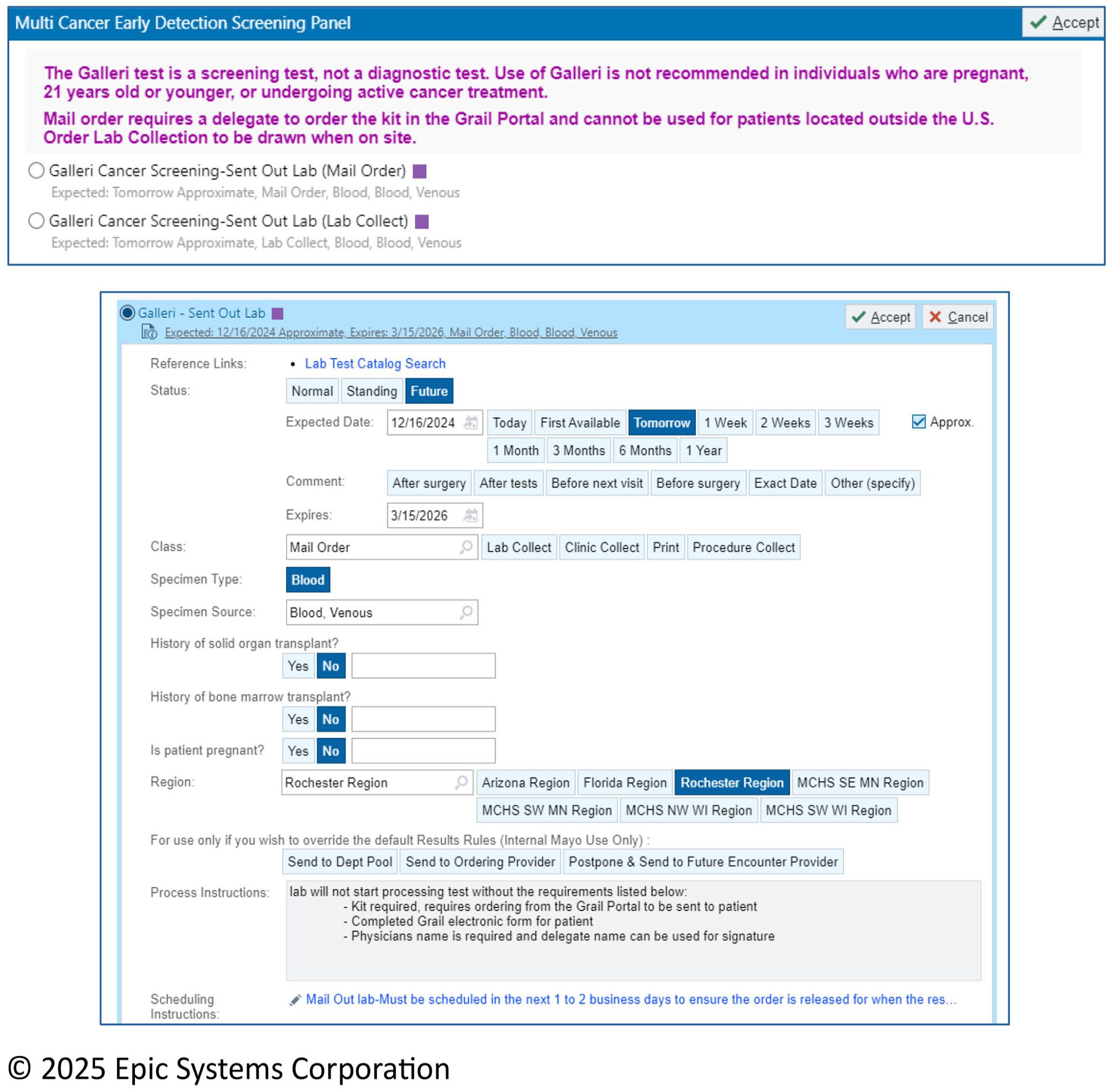
Snapshot of MCD order set.
In addition to displaying the results in the Results Section of the institutional EHR, an automatic In-basket result is sent to the ordering physician. In addition, physicians received an email that the results were available, but this was viewed as redundant and quickly discontinued. Initially, the MCD did not have a flag if the test was abnormal, but this was changed after implementation (Figure 3). Furthermore, the company was advised to deliver all positive results directly to the clinical practice leadership in addition to the ordering physician so they would serve as backup to prevent delayed action in the diagnostic workup of the positive signal. Smart phrases were created to standardize patient messaging when testing was positive. When a test resulted positive, the ordering physician immediately called the patient and informed them of the finding and its implications and recommended plans for further evaluation.

Snapshot of Results as they appear in the EPIC HER.
Diagnostic Evaluation
One of the key gaps we identified early in the implementation process was the absence of a standardized approach to evaluating positive MCD tests. A paucity of published studies supports the best approach for any positive MCD signal. A clinical practice leadership task force was convened to review the
Diagnostic Algorithm for Positive MCD Signal based on Multidisciplinary Expert Opinion.


Snapshot of MCD expanded MCD positive.
Patient Education
A critical component of operationalizing MCD implementation in clinical practice is ensuring that patients are well-informed and express an understanding of the test’s benefits, risks, and limitations. Patient education materials were developed, and patients had to attest that they had reviewed and understood them (Table 2). This attestation was offered as a pragmatic guardrail, an alternative to the IRB-informed consent research pathway, and a practical approach to patient education when choosing to proceed with MCD testing before their face-to-face physician appointment. Attestation approaches have been utilized in other pragmatic trials, including cologuard.11 -13 The education materials included a patient education video (Figure 5) and a written pamphlet. Both were sent through the appointment request form (which the patient fills out before face-to-face appointments) to allow the attestation to be delivered at home when utilizing the home lab option. The education materials were created by the clinical practice team and approved with the assistance of the institutional Patient Education Department.
Key Attestation Points of MCD Patient Education Video.


Snapshot of MCD Patient Materials and Attestation.
Results
The demographics of the study population are highlighted in Table 3. The study cohort involved 2244 patients who were seen over 18 months between 01 June 2022 and 30 November 2023. Of the study cohort, 917 (40.9%) patients were female and 1327 (59.1%) were male. The majority of the population studied were non-Hispanic or Latino (n = 2129; 94.8%), 51 (2.3%) were Hispanic/Latino, and 64 (2.9%) were of unknown ethnicity. The race breakdown of our study population was that 2102 (93.7%) patients were white, 21 (0.9%) patients were black, 72 (3.2%) patients were Asian, and the remaining 49 (2.2%) patients identified as other. Additional factors were evaluated, including the educational background of our study cohort. Of note, 1727 (77.0%) had an associate’s degree or higher. Regarding smoking history, 1643 (73.2%) patients were never smokers, and 574 (25.6%) patients were either past or current smokers. The alcohol use history of our study cohort revealed that 398 (17.7%) patients reported not drinking alcohol (never/no/not currently), while 1678 (74.8%) patients reported presently using alcohol, and an additional 168 (7.5%) patients had unknown alcohol use history. Notably, in our study cohort, 471 (21.0%) patients had a prior cancer diagnosis before undergoing MCD testing.
Participant Demographics and Lifestyle.
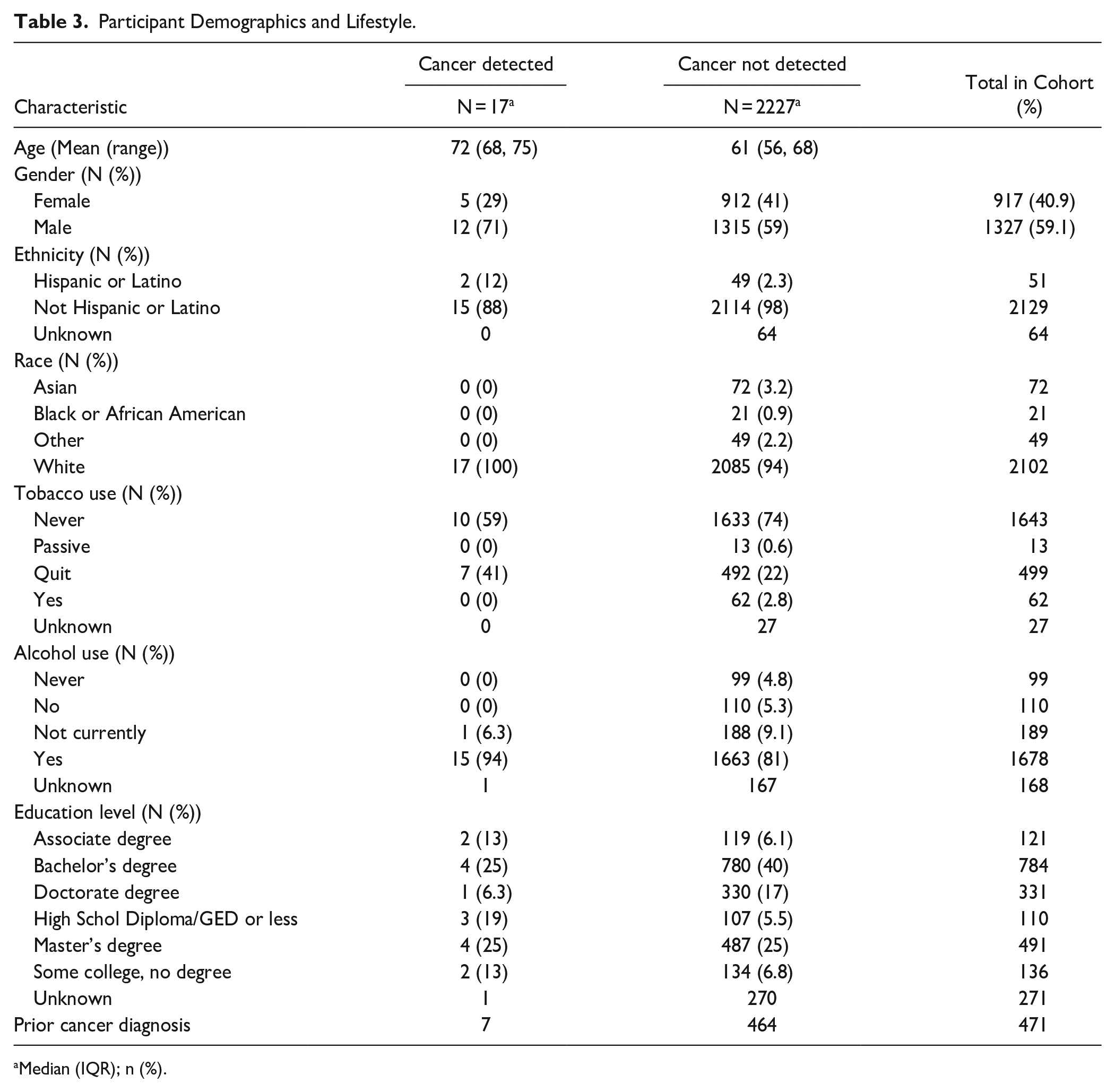
Median (IQR); n (%).
Of the 2244 patients evaluated in this study, 17 (0.76%) had a positive MCD test result. Further subgroup analysis revealed that 12 (71%) patients were male, and the remaining 5 (29%) were female. Further retrospective chart review was performed on the 17 patients with MCD-positive testing. Table 2 highlights these patients’ pertinent clinical history, MCD signal, diagnostic workup findings, and clinical outcomes (as of 11 September 2024). When reviewing the patients with positive MCD signal testing, 2 of the 17 (11.8%) patients declined further workup following initial MCD positive testing and were excluded from further statistical analysis.
Of the 15 remaining MCD-positive patients who went on to receive complete diagnostic workups, 11 (73.3%) patients were found to have cancer, representing true positives. These cancers included breast (n = 1), colon (n = 2), esophageal (n = 1), lymphoma (n = 2), oropharyngeal, for example, glossotonsillar sulcus cancer (n = 1), ovarian (n = 1), pancreatic (n = 1), tonsillar (N = 1). Diagnostic evaluation for 4 patients with MCD-positive signals did not reveal a source of the signal. These patients received repeat MCD testing in 6 months, with the plan of repeating diagnostic workup if the testing remained positive. Of the 17 patients with positive MCD testing, 13 (76.5%) had more than 1 positive cancer signal localization (eg, head and neck, lung, lymphoid). In total, the 17 patients had 30 positive cancer signal localizations.
Important takeaways from the retrospective analysis included the fact that squamous cell skin cancer of the lower extremity was not detected by MCD testing in patient number 13. Skin cancer focused on melanoma detection is a known limitation of the MCD test. 14 In addition, the outcome for patient number 16 (Table 4) illustrates the importance of identifying appropriate candidates for MCD testing. It became clear that this patient’s level of dementia prevented her from fully understanding the implications of MCD testing, noting that she and her caregiver opted to defer diagnostic evaluation.
Case Series.
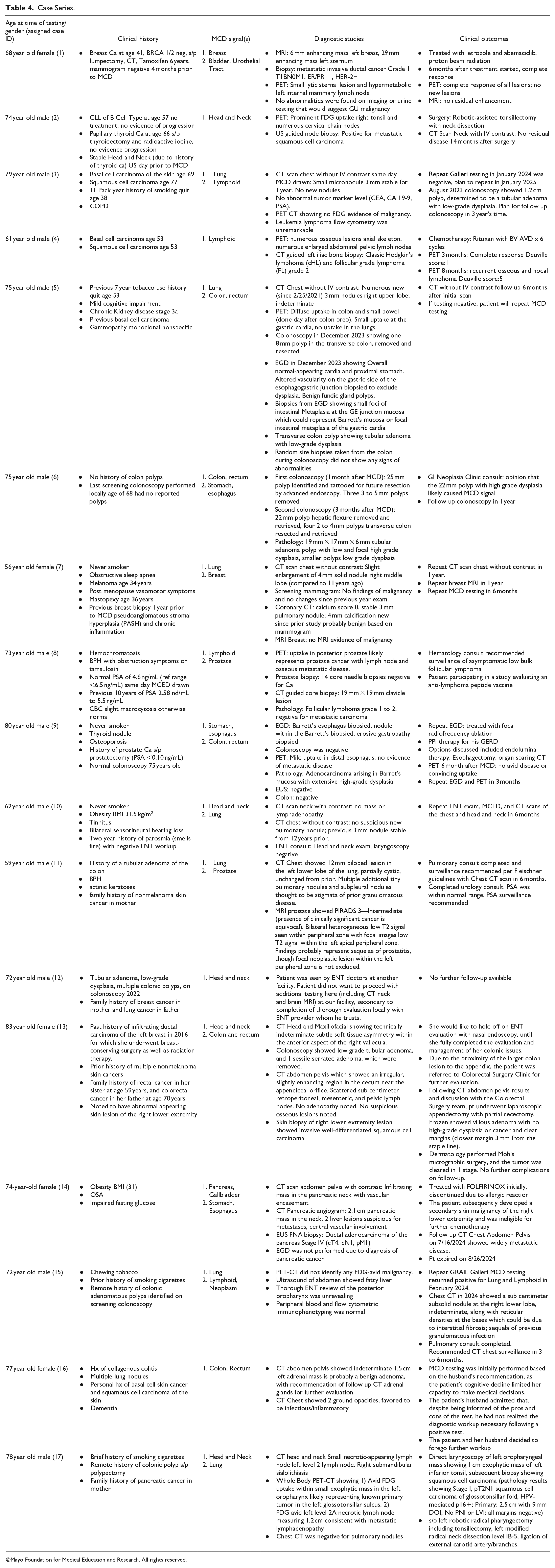
©Mayo Foundation for Medical Education and Research. All rights reserved.
Discussion
The clinical need to perform a pragmatic trial was highlighted when patients seen in the GIM/Executive Health Program clinical practice presented with MCD tests done elsewhere. In the absence of guidelines to facilitate shared decision-making, we implemented a process to enable the education of patients and clinicians regarding the benefits, risks, and limitations of MCD testing and to study the experience to further our understanding and create best practices. The study demonstrated that less than 1% of patients in this population undergoing the test have a positive MCD test. Among those patients who tested positive, 73% were true positives with breast, esophageal, lymphoma, oropharyngeal, ovarian, and pancreas cancers being diagnosed. The remaining 27% of patients with a positive test and negative diagnostic work-up are being followed with a defined plan of repeat testing in 6 months and continued follow-up.
While the role of MCD testing in cancer interception is still evolving, research is steadily increasing worldwide. The
The growing repository of studies supporting MCD testing in cancer assessment is not limited to the US. In England, the National Health Service (NHS) has partnered with GRAIL to perform a pragmatic, prospective RCT called NHS-Galleri. This study aims to determine whether MCD testing can reduce late-stage cancer incidence. Over 140 000 people from the general population have been enrolled thus far. The study will evaluate the clinical significance of population screening with MCD testing. 15
In a multi-center, prospective, observational study at NHS hospital sites in England and Wales, participants with non-specific symptoms or symptoms potentially due to gynecologic, lung, or upper/lower gastrointestinal cancers underwent MCD testing. The MCD testing isolated cDNA, and tests were performed blinded to clinical outcome. MCD predictions were compared with diagnoses obtained by standard care to establish the primary outcomes of overall positive and negative predictive value, sensitivity, and specificity. Of the 6238 recruited participants, the MCD test detected a cancer signal in 323 (5.2%) cases. Cancer was ultimately diagnosed in 244 of these MCD-positive cases, yielding a positive predictive value of 75.5%, negative predictive value of 77.6%, sensitivity of 66.3%, and specificity of 98.4%. 16 This study represents one of the first large-scale prospective evaluations of MCD diagnostic testing in a symptomatic population, suggesting a potential role in clinical decision-making for this population. Our cohort was asymptomatic, perhaps explaining the significant difference in true positive cases (0.76% versus 5.2%).
The assessment of real-world performance characteristics is essential in validating the utility of any new health screening technology, which we have attempted to do herein. It is equally important to assess the perceptions of patients and the medical community with its implementation. In particular, input from primary care physicians in ongoing clinical trials will be critical as they will play an essential role in ordering and performing follow-up on MCD testing. In preparation for the implementation of MCD testing in clinical practice settings outside of tertiary care centers, we developed pre-visit education for patients via video and printed materials, clarifying the uncertainties associated with the test, the potential false positive test and its implications, the need for additional detailed testing for positive tests, cost and lack of insurance coverage at this time. Patients then discuss with the physician whether they wish to do the test. In addition, ongoing education of the clinical team was also set up to ensure that standard practices were followed to facilitate and ensure learning from these processes. The results of the clinical team feedback will be reported in a future report.
One of the challenges to the pragmatic implementation of MCD and other novel tests is the buy-in needed from primary care physicians (PCP). The Vanguard study was a pilot that utilized focus group evaluations to report on the perceptions of PCP and laypersons of different clinical trial designs and their willingness to participate. 17 It was found that both primary care physicians and laypersons expressed concerns and reluctance about a study design in which MCD test results would not be returned to the Vanguard control group (intended effect). 17 Still, they also noted that they were open to participation in MCD clinical trials if there was transparency regarding whether MCD testing would be run on provided biospecimens and if results would be eventually returned so that they could deliver those results to their patients. The findings of this study provided insights to guide clinical trial designs and plan prospective evaluation of MCD testing.
Outside of involvement in clinical trials concerning MCD, there have been notable concerns that PCPs have a voice regarding the use and implementation of MCD in the primary care setting. It is essential to note that the literature on MCD utility in the primary care setting is still very early, with more studies needing to be performed to assess precisely how these assays will impact primary care practices. Review articles aimed at evaluating the thoughts of PCPs regarding MCD testing have identified concerns regarding how to counsel patients on the nature of MCD testing effectively, what the overall financial impact of MCD and potential subsequent testing could be on the patient, as well as how best to manage patient fears regarding a positive MCD result. 18 The main takeaway from these studies is that integrating MCD panels into primary care practice requires careful planning and consideration for managing increased clinical load, interpreting results, downstream testing, and managing costs. 19
In this study, we evaluated the GRAIL Galleri MCD test for implementation in clinical practice. However, GRAIL Galleri is one of many MCD tests currently available. Several other tests are being studied in multiple populations. The OverC is an MCD test used in the THUNDER (THe UNintrusive Detection of EaRly-stage cancers, NCT04820868) study that aimed to evaluate the performance of enhanced linear-splinter amplification sequencing, a previously described cell-free DNA (cfDNA) methylation-based technology, in the early detection and localization of 6 types of cancers in the colorectum, esophagus, liver, lung, ovary, and pancreas. This trial showed that the OverC test had high sensitivity, specificity, and accuracy of predicted origin in detecting these 6 types of cancers. 20 The CancerSEEK test used next-generation sequencing of mutations and protein markers to detect esophageal, stomach, liver, pancreatic, colorectal, ovarian, lung, and breast cancers early.21 -23 The EpiPanGI Dx was focused on the early detection of gastrointestinal cancers through a cell-free DNA methylation pattern study. However, These tests are not FDA-approved but available for commercial use. 24
While this report highlights the findings of implementing MCD testing in clinical practice, the pragmatic approach has limitations. The homogeneity of the population studied in this report makes it difficult to extrapolate the findings to larger populations. Understanding the real-world clinical utility of MCD testing requires extensive population-based studies with longer-term follow-ups, noting that several trials are ongoing. However, this report provides insight into being agile and responding to clinical decision-making when any technology or testing is rapidly becoming available commercially while large-scale studies to look for the impact of a test are still evolving. A detailed assessment of the effects of the MCD test on patients and clinicians is also needed, especially regarding false positives. Future studies on the cost-effectiveness and generalizability of the test as a screening tool and the risk of overdiagnosis with early detection will also be needed as research on MCD progresses. In addition, the testing frequency needs to be explored as the current study looked at single testing time points, and more frequent testing could improve cancer interception.
As the role of MCD testing evolves, there is excitement about the potential for early cancer interception and early treatment with less toxic treatment options, which could potentially improve overall survival, longevity, and quality of life for cancer patients. 25 However, enthusiasm for MCD testing must be balanced against the test performance limitations and potential sources of harm, including prohibitive costs for the test, the diagnostic workup of positive results and subsequent treatment costs. Identifying specific populations most likely to benefit will be important as research on MCD testing progresses. As evidence becomes available in the future, creating guideline-directed algorithms for evaluating and managing positive MCD testing and educating patients and clinicians before implementation are necessary steps if the test is being considered in any clinical practice.
Conclusions
The 18-month prospective cohort study highlights the feasibility of implementing MCD testing within a tertiary referral center. The findings demonstrate that MCD testing can effectively identify cancers in asymptomatic (without symptoms attributed to cancer) populations, with 73% of positive cases yielding a new cancer diagnosis. These results underscore the potential of MCD testing to complement existing cancer screening protocols, enabling earlier detection and intervention. However, challenges such as false positives, patient anxiety, cost considerations, and the integration of MCD testing into routine clinical practice require further investigation. As the role of MCD testing continues to evolve, its potential to transform cancer interception and improve patient outcomes remains a promising frontier in precision medicine.
Supplemental Material
sj-jpg-1-jpc-10.1177_21501319251329290 – Supplemental material for Implementation of a Multicancer Detection (MCD) Test in a Tertiary Referral Center in Asymptomatic Patients: An 18-Month Prospective Cohort Study
Supplemental material, sj-jpg-1-jpc-10.1177_21501319251329290 for Implementation of a Multicancer Detection (MCD) Test in a Tertiary Referral Center in Asymptomatic Patients: An 18-Month Prospective Cohort Study by Ryan T. Hurt, Aditya K. Ghosh, Brian M. Dougan, Elizabeth A. Gilman, Bradley R. Salonen, Jay Adusumalli, Sara L. Bonnes, Carl A. Andersen, Amirala S. Pasha, Sanjeev Nanda, Erin M. Pagel, Christina D. Verness, Steven D. Crowley, Steven W. Ressler, Jewel J. Samadder, Richard J. Presutti, Aadel A. Chaudhuri, William Sanchez, Ivana T. Croghan, Christopher R. Stephenson and Karthik Ghosh in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
A special thanks to the faculty and staff of the Executive Health Program and the Division of General Internal Medicine for their help in identifying the patient data needed and all the patients who gave research authorization for the use of their clinical data. Without their participation, this study would not have been possible.
Author Contributions
All the authors participated in the study concept and design, data analysis and interpretation, and paper drafting and revision, and they have seen and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Mayo Clinic Division of General Internal Medicine in Rochester, Minnesota.
Ethical Approval and Consent to Participate
In accordance with the Declaration of Helsinki, this study was reviewed and determined to be exempt under section 45 CFR 46.104d, category 4. Mayo Clinic Institutional Review Board (IRB) approved the informed consent waiver.
Consent to Publish
Not applicable.
Ethical Standards
This study was determined to be EXEMPT under 45 CFR 46.104d, category 4 by the Mayo Clinic Institutional Review Board, which had ethical oversight for this study. In addition, the authors assert that all procedures contributing to this work comply with the ethical standards of the Mayo Clinic IRB guidelines on human experimentation in accordance with the Declaration of Helsinki of 1975, as revised in 2008.
ORCID iDs
Availability of Data and Materials
All data supporting the study findings are contained within this manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
