Abstract
The optimal patient panel size (PPS) in primary care and the factors determining it remain unclear. We conducted a meta-narrative review of the literature to evaluate factors influencing PPS and assess its association with patient outcomes. A comprehensive search of electronic databases was performed from inception through December 2023, focusing on original studies reporting factors used to determine PPS and related outcomes (eg, clinical outcomes, process measures, and resource utilization). A total of 48 studies were included, identifying 7 key factors influencing PPS. Smaller panels were associated with improved patient satisfaction, continuity of care, and health promotion, while clinical outcomes, utilization, and costs showed minimal impact by PPS. Panel size was primarily associated with patient age, sex, comorbidities, and practice type and structure. Community-based centers typically managed smaller panels, often staffed by female clinicians and serving socioeconomically disadvantaged populations with greater health needs than hospital-based practices. Female clinicians were also independently associated with managing smaller panels, higher quality care indicators, fewer emergency department visits, and improved patient satisfaction. Determining the ideal PPS is a multifaceted process influenced by practice setting, patient demographics, and clinician characteristics. While practice-related factors showed limited association with PPS, patient-reported outcomes were more closely linked to it. Primary care practices should tailor panel sizes to their patient populations, emphasizing a patient-centered approach and ensuring adequate infrastructure support to optimize care delivery.
Introduction
Patient panel size (PPS) refers to the number of patients under the care of a single clinician. A primary care clinician’s panel serves as a formalized link, a long-term bond of care between a clinician and their patients. Determining appropriate PPS is challenging for primary care practices due to ongoing ramifications of excessive PPS. 1 Challenges in determining appropriate PPS are compounded by a combination of factors, including clinician shortages, increasing demand for primary care services from an aging population, rising numbers of individuals seeking care, and variability in practice infrastructure and patient needs.1,2 Ultimately, the quality of health care delivery is threatened by a delay in patient access to care, which is often due to clinicians handling larger patient panels than they can manage.3,4 Larger PPS may also contribute to physician burnout, which further augments the problem.5,6 Understanding what constitutes an ideal PPS for individual practices may help improve patient satisfaction, continuity of care, clinical outcomes, quality of care, and access, while creating a fair and equitable distribution of workload for clinicians.
Consideration has been given to various strategies to alleviate the current primary care clinician shortage. One proposal has been to train more physicians, but Bodenheimer et al 7 suggest this may not be the only solution. Empowering other health care personnel such as nurses, pharmacists, and medical assistants to be more involved in patient care could reduce the burden on physicians and allow them more time to care directly for patients. Indeed, up to a quarter of a physician’s time may be saved by delegating some aspects of patient care to licensed and non-licensed personnel.7 -9 The idea of transitioning to a team-based care model, in which subpanels of patients are assigned to other clinical and nonclinical staff, would allow for the opportunity to increase physician PPS, while using other resources to achieve maximum efficiency and patient and care teams satisfaction.10,11 Furthermore, exercising PPS control may help primary care practices provide preventative, acute, and chronic care to more patients, while improving patient and physician satisfaction.12,13
“What is the optimal PPS a primary care clinician should care for?” is a complex question thought to be influenced by various factors, including patient demographics, physician characteristics, practice infrastructure, regulatory organizations, payers requirements.4,7,14 Attempts to answer this question have resulted in a body of research around methods and formulas to determine PPS (eg, visits per patient per year, provider capacity per year), but these methods face discrepancies that include miscounting, duplicating, and overestimating.15,16 This leads to the question, “What factors determine the ideal primary care PPS?” To address these questions, we performed a systematic review with the aim of synthesizing up-to-date evidence about PPS in primary care practices to determine whether the PPS is associated with differences in patient-centered and health care delivery–related outcomes. We further aimed to report which predictive factors should be considered when determining an appropriate PPS.
Methods
Literature Search
A medical librarian conducted a comprehensive literature search with input from senior study investigators (A.M.A.D., M.H.M.) with experience in systematic reviews. The search was performed in 6 electronic databases: MEDLINE, EMBASE, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, CINAHL, and Scopus from database inception through December 2023. We included original prospective and retrospective studies that reported various primary care PPSs and the estimates used to create them. Studies using predictive models to ascertain PPS were also included, as were studies reporting relevant outcomes and their relationship to PPS (Table 1). To ensure a comprehensive search, we included international studies and expanded the search to include additional search terms (eg, caseloads, managed panel). Study team members also searched references for additional studies and updated the original literature search.
PICO Criteria and Outline of Variables of Interest.
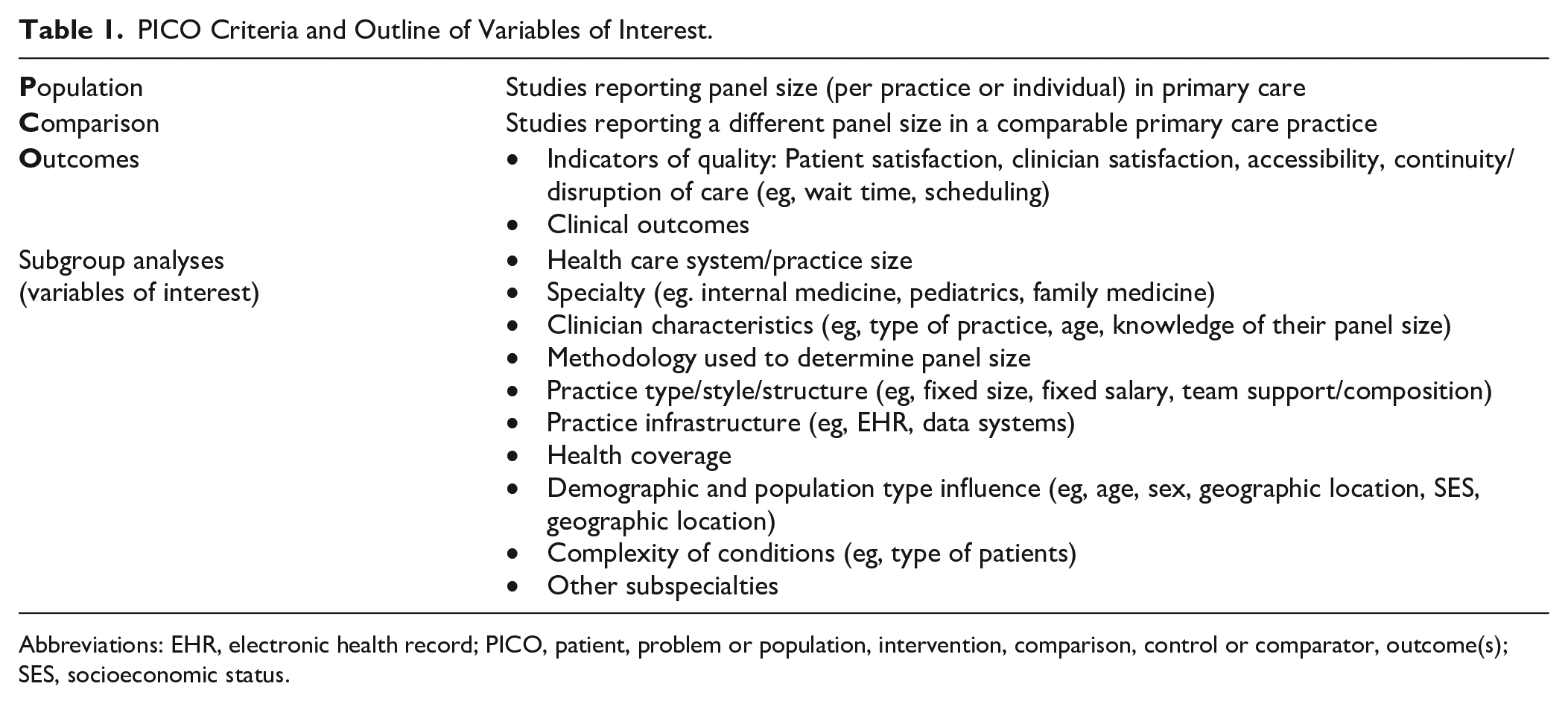
Abbreviations: EHR, electronic health record; PICO, patient, problem or population, intervention, comparison, control or comparator, outcome(s); SES, socioeconomic status.
Study Eligibility
The search criteria included studies that explored the impact of any factors on PPS. We also included articles that associated patient care outcomes with PPS. After excluding duplicates, we identified original studies eligible for further review by screening abstracts and titles. If a study was deemed relevant, the manuscript was obtained and reviewed in full-text version for further assessment by the reviewers. All steps were done in duplicate, independently and blindly by 2 reviewers.
Data Extraction
We extracted data in duplicate and blindly on various study characteristics, including author, year of publication, aim, study design, setting, practice and PPS characteristics, clinical outcomes, and estimated effects of PPS. We also extracted factors associated with predicting PPS, including clinician and patient characteristics and comorbidities.
Risk of Bias Assessment
We used the Newcastle-Ottawa Scale (NOS) to appraise the risk of bias (methodologic quality) in the included observational longitudinal studies and a modified 3-item NOS for cross-sectional and case series studies.
Data Synthesis
Across the included studies, we observed considerable heterogeneity in study methodology, outcome reporting, and methods for determining PPS and predicting factors. Therefore, we chose to use a metanarrative approach rather than pursue conventional systematic review and meta-analysis. The study team members (A.M.A.D., W.H.F., H.M., P.B.) synthesized the narrative evidence according to the RAMESES guidelines, 17 a proposed guiding analytic framework, by mapping out benchmarks or predictors in studies that reported various PPSs and the factors that influenced these PPSs in their respective primary care practices as shown in Figure 1.
Proposed analytic framework to explore patient panel sizes in primary care.
Results
The initial search yielded 1329 studies. After abstract screening, full-text reviewing, and additional reference searches, a total of 48 studies were identified meeting the inclusion criteria (Figure 2). The studies were conducted in or presented data from various countries, including the US (30), UK (11), Canada (7), and Spain (1). Thirty-one studies examined PPS and care delivery–related outcomes (Table 2)12,18 -47 and 20 investigated predictive factors of PPS (Table 3).10,26,27,41,42,48-62 Three studies26,27,41 analyzed both (included in Tables 2 and 3). Across all data, 32 panels were analyzed with a median size of 1824 patients (IQR: 1493-2260), while panel sizes ranged from 265 to 13 147 patients. In US-based studies, which included 14 panels, the median panel size was 2263 patients (IQR: 1888-2504), with a similar range of 393 to 13 147. Reporting on solo and group practices, including their sizes, was limited and inconsistent across studies. Family medicine practices were the primary focus in both datasets (~65%), while community internal medicine practices had moderate representation in US studies. Pediatric-focused practices were least reported.

Flowchart of study selection.
Summary of Included Studies on Panel Size and Care Delivery–Related Outcomes.
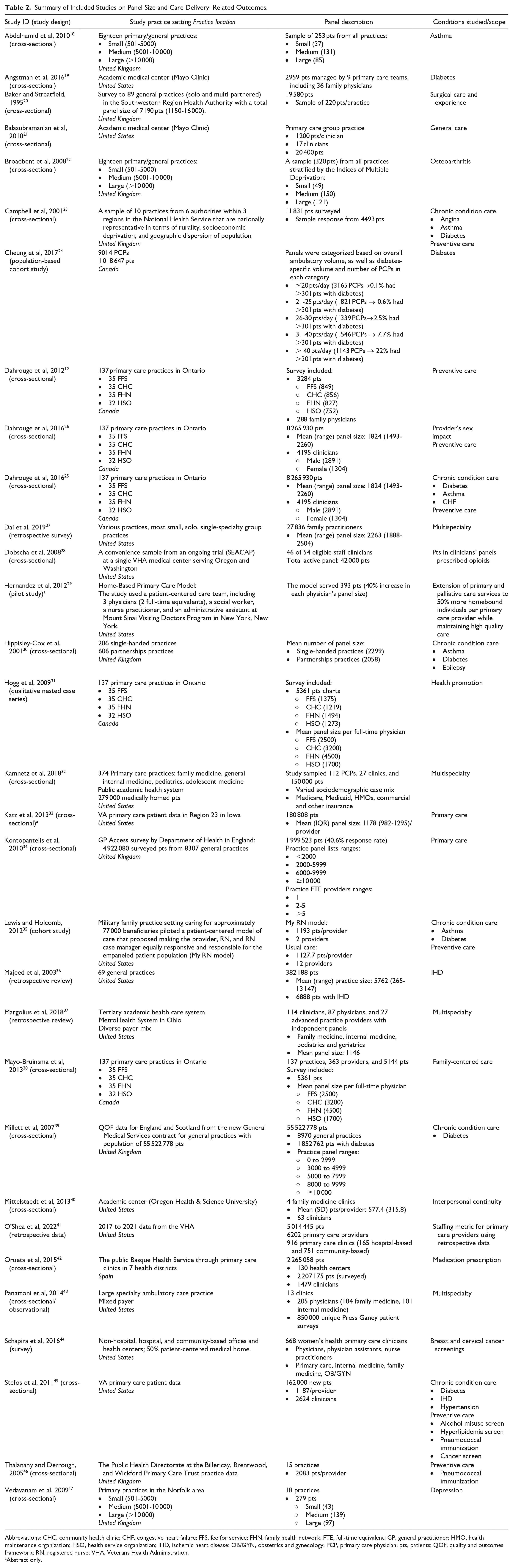
Abbreviations: CHC, community health clinic; CHF, congestive heart failure; FFS, fee for service; FHN, family health network; FTE, full-time equivalent; GP, general practitioner; HMO, health maintenance organization; HSO, health service organization; IHD, ischemic heart disease; OB/GYN, obstetrics and gynecology; PCP, primary care physician; pts, patients; QOF, quality and outcomes framework; RN, registered nurse; VHA, Veterans Health Administration.
Abstract only.
Summary of Studies on Predictive Factors of Panel Sizes.

Abbreviations: CHC, community health clinic; FFS, fee for service; FHN, family health network; FTE, full-time equivalent; HSO, health service organization; MEPS, Medical Expenditure Panel Survey; NAMCS, National Ambulatory Medical Care Survey Series; POINT, Permanente Online Interactive Network Tool; pts, patients; UCSF, University of California San Francisco.
Reported outcomes varied between studies and included clinical outcomes, processes, patient-reported outcomes, practice-centered outcomes, and system-related outcomes. Studies that explored the relationship between PPSs and health care delivery–related outcomes (ie, patient- and practice-centered outcomes) are summarized in Table 4.12,18 -20,22 -28,30 -47,52,55,57,58,60,62 Findings from all studies that highlighted the relationship between predictive factors and their respective PPSs are summarized in Table 5.10,13,15,21,24,27,32,37,41,43,48 -51,53,54,57,59 -64 Figure 1 provides a mapped-out analytic framework to follow these reported findings.
Summary of Study Outcomes and Effect Size.

Abbreviations: ACEI, angiotensin converting enzyme inhibitor), ARB, angiotensin receptor blockade; BMI, body mass index; DM, diabetes mellitus; ED, emergency department; FCC, family-centered care; FTE, full-time equivalent; IHD, ischemic heart disease; LDL, low-density lipoprotein; NP, nurse practitioner; OR, odds ratio; PA, physician assistant; PCMH, patient-centered medical home; PCP, primary care physician; pts, patients; RR, relative risk; SE, standard error.
Summary of Predictive Care Models.
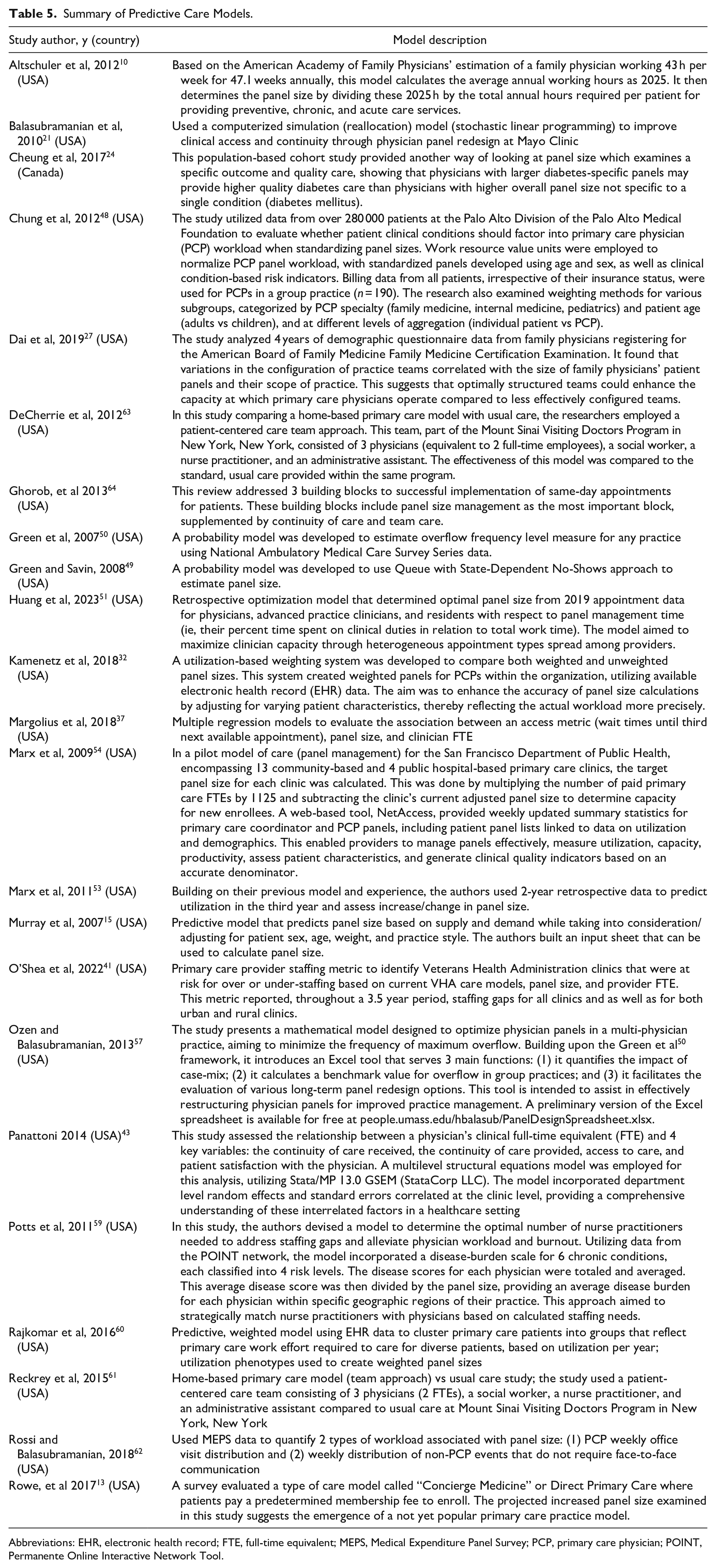
Abbreviations: EHR, electronic health record; FTE, full-time equivalent; MEPS, Medical Expenditure Panel Survey; PCP, primary care physician; POINT, Permanente Online Interactive Network Tool.
PPS and Care Delivery–Related Outcomes
Of the 31 studies examining PPS and care delivery–related outcomes, 23 were cross sectional or survey studies, 3 were retrospective chart review, 2 were cohort studies, 1 had a mixed methods design, and 1 was a quasiexperimental pilot study. Table 2 incudes the baseline characteristics, clinical outcomes, and reported effect sizes of these studies. We identified reported trends between PPS and (1) patient-centered outcomes (clinical and wellness outcomes); (2) patient-reported outcomes (patient satisfaction); (3) quality of care measures; and (4) utilization. A detailed narrative of these findings is included in Supplemental Appendix.
Patient-centered outcomes (clinical outcomes)
(a) Preventive services: Smaller practices were often associated with a higher likelihood of being up-to-date with preventitive services,12,25 though this was not consistently reported.30,44,60 (b) Diagnostic tests: Generally, practice size was not associated with the number of diagnostic tests ordered per patient.18,22,47 (c) Health promotion: Better health promotion was associated with smaller PPSs in 1 study.
31
(d) Clinical services: Health education was not associated with PPS.18,22,47 (e) Diabetes care: While several studies found no association between PPS and diabetic care,22,25,36 others reported favorable23,39 or unfavorable19,61 outcomes for diabetic care with larger panels (≥2500). (f) Medication prescription rate: The consensus among the studies was that PPS did not impact medication prescription rates for various conditions, including asthma, and depression.18,22,25,35,36,47 Two studies found that larger PPS was strongly and negatively correlated with opioid prescriptions.28,42
Patient-reported outcomes
(a) Patient satisfaction and experience: Larger PPSs were either negatively correlated with20,28,34,45 or had no significant effect on patient satisfaction.19,23,35 A 1000 PPS increment was reportedly associated with the following reduction in experience and satisfaction: 2.4% reduction relating to getting through on the phone; 1.0% reduction relating to scheduling an urgent appointment; 1.9% reduction relating to scheduling an advanced appointment; and 1.2% reduction relating to scheduling an appointment with a particular doctor
34
Reduced full-time equivalent (FTE) work hours were also associated with higher patient satisfaction.
43
Mohr et al
65
surveyed 222 primary care clinics in the Veterans Health Administration (VHA) system and noted that when workload increased, patient quality of life ratings were negatively impacted. However, higher levels of relational climate (ie, clinicians communicating effectively and providing mutual support to accomplish patient care goals) were associated with better care.
65
(b) Family and Personal History Reporting/Communication: Larger panels in a practice were associated with lower odds of patients reporting family-centered care (ie, lower focus on individualized, family-centered care aspects due to higher workload or insufficient time per patient)
38
; for every 1000 additional patients in a practice, the odds of patient-reported family-centered care dropped by 8%. Family-centered care was defined as consideration of hereditary conditions in the patient’s family, household income, and living situations, as well as awareness of the signs of child abuse.
Quality of care measures
(a) Continuity of care: Continuity was found to be associated with PPS in some studies, with continuity of care improving with smaller PPSs,12,18,44,62 and to have no effect in others.23,33,40 Dahrouge et al
26
found that continuity of care was optimized with PPSs of 1800 to 2700 patients per clinician. Lewis and Holcomb
35
found that implementing a coordinated care model that included the same physician and nurse per patient significantly improved continuity of care and the clinician’s ability to see their own patients, even with larger PPSs. Another VHA study of 180 808 patients with the aim of implementing a comparable model found no effect on continuity of care when PPSs were increased.
33
Increment of clinician FTEs was associated with better continuity of care.
43
(b) Access to care: Access was measured by various methods. Generally, the association between access to care and PPS was positive with smaller PPSs23,34 and negative with larger PPSs, resulting in delays.19,20,38 One study found that implementing a patient-centered model of continuous care increased access in larger panels.
35
Lower clinician FTEs was associated with less access,
37
and increased FTEs were directly associated with better access to care.
43
The creation of a weighting system accounting for complexity resulted in changes to practice closure, increased total patients, and improved access. After this weighting was implemented, patients’ perceived access improved in family medicine and general internal medicine clinics.
32
Egede et al
66
conducted a study within a primary care clinic at an academic medical center in the Midwest over the course of 24 months, reporting improved access to care (as well as provider productivity and patient satisfaction) of approximately 22% by integrating various practice changes, including: consistent visit lengths; addition of early morning and evening hours; handoff of completion of pain management agreements to registered nurses; creation of a team-based care model including 2 advanced practice providers to each team to increase patient volume; standardization of visit length; and incorporation of biweekly meetings with scheduling staff and administration to improve scheduling. Additionally, O’Shea et al
41
introduced a “gap staffing” metric to assess staffing adequacy in VHA clinics, finding significant staffing gaps, especially in rural areas. They found that lower clinician FTEs were associated with larger panel sizes, which in turn led to less access to care, highlighting the importance of balanced staffing for optimal patient access.
41
Utilization and cost
(a) Adherence to treatment: Smaller PPS was significantly associated with improved adherence to certain preventive care (eg, metabolic panels, mammography and colorectal cancer screening),
35
though the significance was not consistent with other quality indicators.
18
(b) Cost: There was a paucity of data about patient empanelment in relation to cost.
Predictive Factors of PPS
There were 20 studies included in this category. Table 3 incudes the baseline characteristics of the included studies, clinical outcomes, and any reported effect sizes. Factors contributing to the calculation of appropriate PPSs included (1) patient, (2) clinician, and (3) practice-related characteristics, as well as (4) utilization and cost. Despite their crucial relevance to PPS, there remains insufficient reporting regarding practice structure resources (eg, team support and composition), practice infrastructure resources (eg, electronic health record, data systems, information technology support), health type coverage, and other integrated subspecialty services within primary care. A detailed narrative of these findings is included in Supplemental Appendix.
Patient-related characteristics
(a) Age and sex: Chung et al 48 summarized the evidence using data from over 280 000 patients and employing work relative value units to standardize primary care physician panel workload. Standardized panels were created using age and sex and clinical condition–based (comorbidities) risk indicators. According to the study, for pediatric panels, age and sex–based adjustment is probably sufficient, but for adult panels, further adjustment reflecting patient clinical conditions is warranted. The authors concluded that PPS standardization should account for unusual but predictable coding patterns. 48
(b) Comorbidities: In addition to Chung et al, 48 Ozen and Balasubramanian 57 found that when patients have more comorbidities, physicians should consider decreasing PPS.
Clinician-related characteristics
Dahrouge et al 26 found that female clinicians had significantly lower PPSs and their patients were more likely to have received recommended cancer screening and diabetes management, fewer emergency department visits and hospitalizations, and higher referral rates.
Practice-related characteristics
Community-based settings predict smaller PPSs and poorer populations with higher medical comorbidities. 55 One study suggested the difference in PPSs between community and hospital settings may be due to the larger number of FTE staff at hospital-based clinics; PPS per clinical FTE, a more accurate measure of clinical burden, was slightly higher for community settings vs hospital-based clinics. 54
The addition of nurse practitioners and physician assistants within family physician practices was associated with increased PPSs. 27 Beyond the composition of clinical teams, the physical office layout and support staffing in clinics did not predict changes in PPS. 55 Reckrey et al 61 found that with the support of a nurse practitioner, an administrative assistant, and a social worker, physicians were able to increase their PPS. Potts et al 59 found that hiring nurse practitioners to work with physicians who have patient populations with high comorbidities helped maintain manageable PPSs while improving patient and physician satisfaction. Porter et al 58 used a hypothetical panel of 2500 patients in a simulation study to quantify time needed to provide care in a single-physician versus team-based care model. They noted that a primary care physician alone would need 26.7 h a day (14.1 h/day for preventive care, 7.2 h/day for chronic disease care, 2.2 h/day for acute care, and 3.2 h/day for documentation and inbox management). The physician’s time would decrease to 9.3 h a day in a team-based structure (2 h/day for preventive care, 3.6 h/day for chronic disease care, 1.1 h/day for acute care, and 2.6 h/day for documentation and inbox management). They concluded that primary care physicians did not have enough time to provide guideline-recommended care alone or in a team-based care model, even though team-based care reduced time requirements by more than half. 58 Huang et al 51 optimized clinician workloads in primary care by modeling patient panel sizes using data from 82 881 patients and 105 clinicians based on their panel management time, improving workload distribution and reducing burnout. The study highlighted significant differences in annual appointment volumes per clinician with an optimization-based approach. 51 O’Shea et al 41 examined a novel “gap staffing” modeling metric to assess the relationship between FTE-based staffing size and panel size to ensure adequate access to care. This metric showed that lower clinician FTEs were associated with larger panel sizes, resulting in less access to care, particularly in rural clinics; thus, emphasizing the need for balanced staffing to optimize patient care and access. 41
Utilization and cost
(a) Annual visits: Two studies concluded that utilization was directly or indirectly predictive of optimal PPS.60,62 Factors considered included estimating the workload required to deal with non–primary care physician specialty visits (non–face-to-face visits) that primary care physicians coordinate. A third study was able to predict PPS based on visit type (ie, preventative, acute, or chronic). 50
(b) Referrals: Orueta et al, 42 analyzing health system data of panels between 1600 and 1890 patients in Spain, found that PPSs larger than 1700 patients did not significantly affect the number of doctor visits or referrals to specialists. On the other hand, Hugo et al 52 found that practice size, proximity to the clinic, female general practitioners, general practitioners with UK-based degree and qualification, and full contraceptive services were all associated with higher referral rates.
(c) Cost: Angstman et al 19 reported that PPS did not predict changes in costs measured as per member per month charges. Hernandez et al 29 found that a 40% increment in PPS for home-based primary care programs resulted in a 15% cost reduction per patient.
Discussion
Our review identified 48 original studies exploring 7 key factors associated with PPS as dynamic and multifactorial outcome, influenced by practice type, patient population characteristics, and organizational structure. Generally, smaller panel sizes were associated with improved patient-reported outcomes, such as satisfaction, continuity of care, and health promotion, while clinical outcomes, utilization, and costs showed minimal association with panel size. Larger panels were linked to increased clinician burnout, reduced patient access, and lower patient satisfaction. A lack of standardized definitions and reporting on PPS characteristics created significant heterogeneity across studies, limiting the ability to draw consistent conclusions. Community-based centers generally managed smaller panels, often staffed by female clinicians, and served populations with greater socioeconomic disadvantages and comorbidities compared to hospital-based settings. Despite these findings, there was no single clear predictor for optimal PPS.
It is estimated that by 2034, there will be an estimated shortage of up to 48 000 primary care physicians. 67 Despite these predictions, studies have demonstrated the risk of insufficient feasibility or sustainability of primary care, highlighting that it would take a primary care physician between 17 to 21 h a day of clinical work to keep up with their patient panel.14,68 In his essay, Bodenheimer 69 hypothesized that primary care’s root problems are due to 2 primary factors: a low percentage of national health expenditure directed to primary care and unmanageably large PPSs. The author, among others, noted that these factors lead to widespread primary care physician exhaustion, cynicism, and burnout, as well as poor patient access, suboptimal medical care, fewer preventative services, and lower patient satisfaction.25,69 -72
We designed our metanarrative review to determine whether there is an association between PPS and outcome measures, as well as to further determine which factors could be used when calculating ideal PPS. In a systematic review, Paige et al 73 concluded that higher PPS was negatively associated with patient health outcomes, clinical quality, patient experience, and health care professional burnout. Similarly, the included evidence demonstrated significant associations between PPS and outcome measures, as well as great variation in how groups and health systems evaluate PPS and the factors and outcomes associated with modifying PPSs for distinct primary care practices. Furthermore, we agree with a study that identified significant variation in how different organizations define primary care panels, the factors for including or removing patients from a panel, and the impact that these variable definitions have on primary care PPS. 74 According to Mayo-Smith et al, 74 given the considerable variation in defining and measuring primary care panels, caution and judgment should be used when comparing reported PPS findings across studies until a standardized definition is created. Understanding and defining PPS in primary care is an essential building block of population-based care. 7 Effective use of this building block may be a key component to the future of effective primary care delivery.
Practice leaders and policy makers are frequently tasked with the establishment of PPS parameters or targets for their practice. “What are your target PPSs?” and “How did you arrive at that number?” are commonly asked questions at primary care leadership conferences. It should be noted that many factors influence the calculation of ideal PPS, including physician and practitioner preferences, organizational logistics, ancillary support structure, and patient population. 68 Furthermore, according to a systematic review by the VHA, the optimal PPS requires a complex balance of health care system demands (ie, access, quality of care, cost, and patient experience) and the needs of the health care team (ie, preferences, satisfaction, and minimization of burnout).10,72 Shekelle et al 72 noted that although determining optimal PPS is complex, there was a negatively significant relationship of modest size between increasing PPS and various measures of health care quality and experience.
Variability in PPS is influenced not only by practice-specific factors but also by external factors such as regulatory and payer requirements. Organizations such as the National Committee for Quality Assurance (NCQA) and the Joint Commission as well as medical societies and bodies (eg, American Academy of Family Physicians [AAFP]; Society of General Internal Medicine [SGIM]) set standards for care delivery, yet they do not provide concrete guidance on PPS, leading to inconsistencies in its implementation.75,76 Additionally, payer-driven metrics and incentives focusing on cost, quality, and patient experience significantly shape panel size decisions. Addressing these influences is essential for developing a comprehensive and standardized framework for PPS optimization.7,77
There are novel factors that might be considered in future PPS research and frameworks. For example, theories involving patient capacity frameworks especially for chronic care management, such as those proposed in minimally disruptive medicine (MDM), may provide valuable insights. 78 These approaches emphasize aligning care demands with patients’ ability to manage their health, which could inform PPS optimization, particularly for high-need or vulnerable populations.11,79,80
Our review further illustrates the important challenges facing stakeholders and decision makers as they grapple with the complexities of their own practice characteristics and the variability found in the literature. In the current state, it is not feasible to confidently establish optimal PPS for a given practice based on well-established norms or via an extensive review of the literature. Therefore, our metanarrative review demonstrates the need for a standardized and systematic approach to the work of understanding primary care PPS. We propose a framework of 7 categories for conceptualizing primary care panels and practice characteristics and their impact on practice-meaningful outcomes to explore and identify potential commonalities, as included in Figure 1. We have summarized the existing literature into this framework with the hope that it will facilitate future studies in PPS in primary care research.
Strengths and Limitations
Some limitations of our study are inherent to the nature of the study design, observational and nonvalidated predictive model, which may limit generalizability. Additionally, the heterogeneity in reporting outcomes may limit its application. The strengths of this study include its comprehensive nature and selection process following a robust methodologic framework of reporting heterogeneous literature. This novel classification approach could inform many types of future studies and research as noted above. The narrative nature of the summaries may highlight new areas and opportunities for collaboration to direct future practice guidelines and policies.
Conclusions
Our metanarrative review found that the optimal primary care PPS is a multifaceted decision that varies according to practice type and setting. Generally, patient-reported outcomes are more highly associated with PPSs than practice-related factors. For individual primary care practices, patient population characteristics were a major factor for determining appropriate PPS. There was substantial heterogeneity in methodology and approaches to determine PPS and the predictive factors for its determination. Future research should adopt a standardized framework, focusing on the 7 identified factors to better understand and evaluate PPS, ensuring a balance between patient-centered outcomes, system demands, and clinician well-being.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251321294 – Supplemental material for Determining Patient Panel Size in Primary Care: A Meta-Narrative Review
Supplemental material, sj-docx-1-jpc-10.1177_21501319251321294 for Determining Patient Panel Size in Primary Care: A Meta-Narrative Review by Abd Moain Abu Dabrh, Wigdan H. Farah, Heidi M. McLeod, Parisa Biazar, Arya B. Mohabbat, Bala Munipalli, Rachel Garofalo, Robert J. Stroebel, Nilay Shah, Kurt B. Angstman, Richard J. Presutti, Bryan Farford, Jennifer L. Horn, Summer V. Allen, Adam I. Perlman, Ana Lucia Chong Lau, Larry J. Prokop and M. Hassan Murad in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The Scientific Publications staff at Mayo Clinic provided copyediting, proofreading, administrative, and clerical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
