Abstract
Background:
Syncope is a common presenting complaint in the outpatient setting and requires a thoughtful and meticulous approach to history-taking skills. This is crucial in discerning cardiac versus non-cardiac etiologies, underscoring the pivotal role of healthcare professionals in the diagnostic process.
Case Report:
This is a case of a 76-year-old male who presented to the primary care clinic following an episode of syncope with abrupt loss of consciousness. Given the presentation, there was a high suspicion of cardiac etiology, and further testing was pursued. An electrocardiogram (ECG) demonstrated normal sinus rhythm with a pre-existing first-degree atrioventricular (AV) block. The Holter monitor was without sustained arrhythmias. A dobutamine stress echocardiogram was negative for myocardial ischemia, though a resting transthoracic echocardiogram demonstrated findings consistent with cardiac amyloidosis. Further evaluation with cardiology confirmed the diagnosis of transthyretin (ATTR) cardiac amyloidosis. He was initiated on appropriate treatment and underwent a dual chamber implantable cardioverter defibrillator (ICD) given his presentation. The patient ultimately succumbed to his disease 5 years after his diagnosis.
Conclusion:
If the cardiac etiology of syncope is suspected, further cardiac structural evaluation is necessary. Cardiac amyloidosis, although rare, is a cause of cardiogenic syncope that should not be overlooked. It should be considered in the differential diagnosis in the appropriate clinical setting, highlighting the need for a broad and comprehensive approach to diagnosis.
Introduction
The heart is one of the most commonly affected organs in transthyretin (ATTR) amyloidosis, with cardiac involvement leading to serious complications such as arrhythmias and valve disease, which can be fatal. Patients may be misdiagnosed for years before coming to the correct diagnosis, which emphasizes the necessity of maintaining a high index of suspicion for cardiac amyloidosis. Early diagnosis and prompt recognition are crucial, as they can significantly improve patient outcomes and slow disease progression. The workup for syncope requires a thorough evaluation to identify potential underlying causes, with a focus on cardiac etiologies when patients present with concerning symptoms. This highlights the importance of vigilance among healthcare professionals, ensuring they remain alert to the possibility of cardiac amyloidosis when evaluating patients with syncope.
Case Report
We present a case of a 76-year-old male presenting to the primary care office following an episode of syncope. Earlier that morning, the patient was sitting at their kitchen table reading a newspaper when he experienced light-headedness followed by a sudden loss of consciousness and a fall to the ground. He woke up lying on the floor underneath the table without recollection of how he got there. His wife witnessed the event, and she believed the patient was most likely unconscious for 10 to 15 s. The patient regained consciousness without confusion, slurred speech, or loss of bladder or bowel continence. His blood pressure and blood sugar were checked at home and were normal.
Subsequent to the syncopal episode, he endorsed intermittent sensations of light-headedness during the day but no further syncope. He hit his head during the syncopal event, but outside of a contusion on his left forehead, he had no headache, mood alternations, change in cognition, speech deficit, or gait changes. He denied chest pain, numbness, tingling, and shortness of breath at rest but did endorse a history of exertional shortness of breath when walking more than 100 yards before the syncopal event.
The patient’s past medical history was significant for hypertension, hyperlipidemia, impaired fasting glucose, obesity (BMI = 29 kg/m2), GERD, nonmelanoma skin cancer, history of colon polyps with a family history of colon cancer, and prior bilateral carpal tunnel correction surgery. He was a non-smoker with no known history of coronary artery disease. Medications were significant for allopurinol, aspirin, zolpidem, atorvastatin, losartan, diltiazem, pantoprazole, and terazosin.
On examination in the office, the patient had stable vital signs, including normal orthostatic blood pressures. The Dix-Hallpike maneuver was negative. Cardiac auscultation failed to reveal murmurs, rubs, or gallops. Jugular venous distention was not appreciated. His lungs were clear to auscultation bilaterally. The remainder of the physical exam was unremarkable.
There was a concern for cardiogenic syncope, given the abrupt loss of consciousness and the patient’s cardiac risk factors. Further testing included an electrocardiogram (ECG), which showed normal sinus rhythm with sinus arrhythmia and unchanged first-degree atrioventricular (AV) block. A 24-h extended telemetry monitor demonstrated some atrial and ventricular ectopy but was without sustained arrhythmia. Initial laboratory work included a complete blood count (CBC), creatinine, potassium, sodium, glucose, and TSH, which were within normal range. Troponin T was normal at <0.01 ng/mL. D-dimer was elevated at 515 ng/mL (normal <250 ng/mL). NT-Pro B-Type Natriuretic Peptide (NT-proBNP) was elevated at 302 pg/mL (normal <116 pg/mL). Chest computed tomography (CT) with IV contrast was performed due to elevated D-dimer and was negative for pulmonary embolism.
A transthoracic echocardiogram was obtained to evaluate for structural heart disease. This demonstrated findings consistent with cardiac amyloidosis, including normal left ventricular chamber size with mildly increased concentric wall thickness and normal systolic function with left ventricular ejection fraction (LVEF) of 55% without regional wall motion abnormalities. There was biatrial chamber enlargement and increased left ventricle filling pressures but no significant valve disease or pericardial effusion. The global left ventricular longitudinal strain was abnormal at –13% (normal is more negative than –18%) with an apical sparing pattern, which can be seen in patients with infiltrative cardiomyopathy such as cardiac amyloidosis (Figure 1). A dobutamine stress echocardiogram was also completed, which was negative for ischemia, with an increase in LVEF from 55% to 70%.
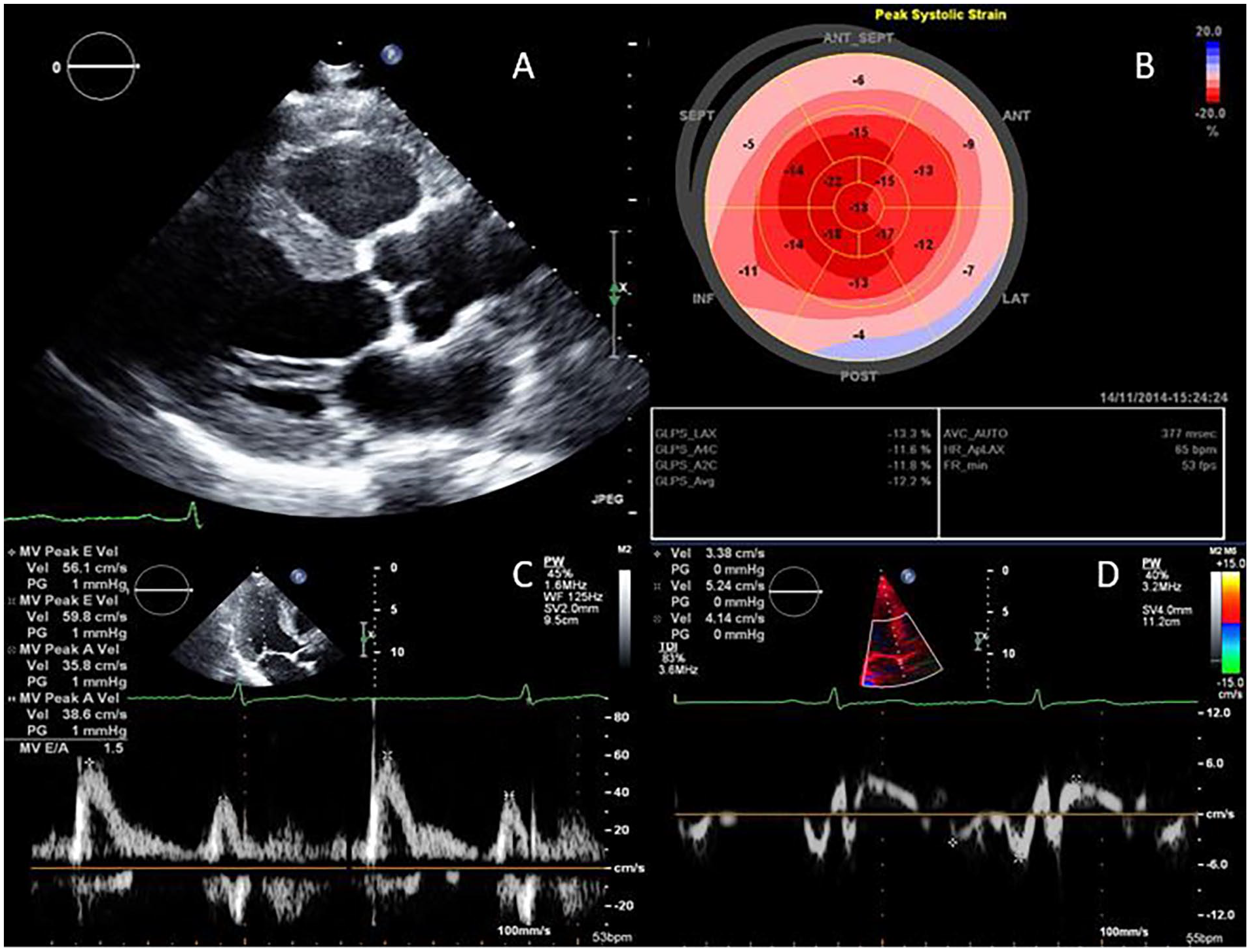
Images from transthoracic echocardiogram. (A) is a still image from the parasternal long axis view demonstrating normal left ventricular chamber size with mildly increased concentric wall thickness, the septal wall measured 12 mm and the posterior wall measured 14 mm. (B) shows abnormal left ventricular global longitudinal strain (−13%) in an apical sparing pattern, a pattern which can be seen with infiltrative cardiomyopathy such as cardiac amyloidosis. (C) and (D) are the mitral inflow and medial mitral annular tissue Doppler, respectively. The medial e’ was very abnormal at 0.03 cm/s, with E/e’ of 20 consistent with increased left ventricular filling pressures.
The patient was referred to cardiology and underwent further evaluation for cardiac amyloidosis. Serum and urine protein electrophoresis with immunofixation showed no evidence of a monoclonal protein. The abdominal fat aspirate was negative for amyloid. Technetium pyrophosphate (PYP) scan was indeterminate, with minimal myocardial PYP uptake on SPECT/CT images.
Given that the clinical suspicion of cardiac amyloidosis was high, an endomyocardial biopsy was recommended. All endomyocardial fragments stained positive for amyloid deposits. Mass spectrometry confirmed transthyretin (ATTR) amyloid deposition. There were no amino acid sequence abnormalities in the transthyretin protein, most consistent with wild-type or age-related amyloidosis.
Due to the patient’s concerning episode of suspected cardiogenic syncope without other explanation, he underwent successful implantation of a dual-chamber implantable cardioverter defibrillator (ICD). He was also initiated on treatment for ATTR cardiac amyloidosis. The patient ultimately succumbed to his disease 5 years after his diagnosis.
Discussion
Syncope is a common presenting complaint in the outpatient setting, and the clinician must have the skill to determine when a cardiogenic cause should be considered. As outlined by the American College of Cardiology (ACC)/American Heart Association (AHA)/Heart Rhythm Society (HRS) guidelines, the following are outlined risk factors for cardiac causes of syncope: older age (>60 years old), presence of known ischemic heart disease, structural heart disease, previous arrhythmias, sudden loss of consciousness without prodrome, syncope during exertion, syncope during supine position, low number of syncope episodes (1 or 2), abnormal cardiac examination, family history of heritable conditions or premature sudden cardiac death (<50 years of age), or presence of congenital heart disease (Table 1). 1
Risk Factors for Cardiac Causes of Syncope.

Initial evaluation of suspected cardiogenic syncope should include a thorough medical history, physical exam, and electrocardiogram, and then further testing can be considered based on the quality and frequency of symptoms, such as a holter monitor or echocardiogram. 1 Our patient demonstrated several high-risk symptoms that raised suspicion for a cardiogenic syncope, including age >60 years old, male sex, syncope with no prodrome, low number of syncopal episodes, and no prior history of syncope. It is also important to consider the patient’s medications in the setting of syncope. For our patient, medications at the time of the event included diltiazem, terazosin, and losartan. While syncope has been reported rarely with these medications, his blood pressure was normal, with no evidence of orthostatic hypotension.2 -4
Particularly in patients such as our own with no known structural heart disease, a transthoracic echocardiogram can be very beneficial in the evaluation of suspected cardiogenic syncope. This study allows the provider an evaluation of the cardiac structure and function, including an assessment of biventricular size and function, evaluation for wall motion abnormalities which may suggest underlying coronary artery disease, screening for major valvular heart disease, and evaluation for patterns of abnormal wall thickening or thinning which may suggest underlying cardiomyopathy. If any major abnormalities are noted, discussion with and referral to colleagues in cardiology would be recommended. Our patient had typical echocardiographic features of cardiac amyloidosis, including a concentric increase in left ventricular wall thickness, evidence of elevated left ventricular filling pressures, enlarged atria, and abnormal left ventricular strain in a pattern consistent with infiltrative cardiomyopathy, prompting further testing.
Cardiac amyloidosis is a complex disease characterized by the deposition of insoluble amyloid proteins in the heart, leading to tissue replacement and subsequent dysfunction.5,6 Roughly 70% of patients with amyloidosis have cardiac involvement.7,8 There are different types, the most common being AL (light chain) and ATTR (transthyretin) amyloidosis. When suspected, initial testing for cardiac amyloidosis should include serum free light chains and serum and urine immunofixation as screening for AL amyloidosis along with a nuclear medicine bone scintigraphy based amyloid imaging study. There should be rapid involvement of hematology-oncology if initial screening labs raise concerns for AL amyloidosis. 9 Clinical features that may suggest AL amyloidosis include a tendency for multi-organ involvement, heart failure, hepatomegaly, nephrotic syndrome, peripheral and autonomic neuropathy, macroglossia, and periorbital purpura. In comparison, in ATTR amyloidosis, common clinical history features include atrial fibrillation, carpal tunnel syndrome (particularly bilateral), biceps tendon rupture, spinal stenosis, and heart failure.
Clinicians should maintain a high index of suspicion for cardiac amyloidosis as it can have similar presenting features as other common cardiac disease processes, including hypertensive heart disease, hypertrophic cardiomyopathy, and ischemic heart disease, leading to treatment delays. 6 In fact, it is reported that approximately 37% of patients did not receive the diagnosis for over 1 year after symptom onset. 10 Additional advanced cardiac imaging is occasionally necessary to help discern between various disease processes. Cardiac magnetic resonance imaging (MRI), with gadolinium enhancement in particular, is a valuable tool for myocardial tissue characterization to aid in the diagnosis and prognostication for patients as the presence and pattern of late gadolinium enhancement vary based on the underlying disease process, is it can help distinguish cardiac amyloidosis versus hypertrophic cardiomyopathy. 11 For the primary care provider, these advanced cardiac imaging modalities will most frequently be guided by consultation with cardiology.
It is hypothesized that an arrhythmia related to previously undiagnosed ATTR amyloidosis caused our patient’s syncopal episode. Cardiac amyloidosis-induced arrhythmias are a well-documented occurrence in the medical literature, ranging from bradyarrhythmias to atrial and ventricular tachyarrhythmias. 12 Atrial fibrillation is the most commonly observed arrhythmia in ATTR-cardiac amyloidosis, with varying reported rates of prevalence between 15% and 70%. Barge-Caballero et al 5 found that one-fourth of patients with transthyretin amyloid cardiomyopathy had at least 1 syncopal episode, and patients in the study with a history of syncope had more advanced disease and a lower survival rate. With rates of atrial fibrillation and other arrhythmias as high as 70%, screening with long-term monitoring should be considered by the primary care clinician in patients with ATTR-cardiac amyloidosis and symptoms of pre-syncope or syncope.13 -17
The data for the use of ICDs in patients with cardiac amyloidosis is limited. Prior studies have shown higher rates of appropriate therapy in patients with AL-type amyloidosis. 18 However, ICD therapy has not been shown to improve mortality in patients with amyloidosis in studies including patients with AL and ATTR cardiac amyloidosis.18,19 Due to unclear benefits, An individualized approach to determining candidacy for ICD placement is required. 20 Our patient was offered an ICD due to his unheralded syncopal event, though he never received any ICD therapies.
Treatment for cardiac amyloidosis is targeted to the underlying protein disorder. Tafamidis is an FDA-approved medication for treating ATTR amyloid cardiomyopathy, and cardiology providers with expertise in cardiac amyloidosis will direct its use. It stabilizes the transthyretin tetramer and has shown efficacy in reducing mortality and cardiovascular-related hospitalizations in those with NYHA class I to III heart failure symptoms and wild-type or variant transthyretin cardiac amyloidosis.21,22 Other investigational agents are also in various stages of clinical development. However, the only therapy shown to improve cardiovascular outcomes in ATTR-CM is tafamidis. 22 If AL amyloidosis is identified, management and treatment will be directed by hematology-oncology specialists.
Conclusion
Maintaining a broad differential for the patient presenting with syncope is important. One of the first important distinctions is whether the presentation has features suggestive of cardiogenic syncope. If so, further cardiac testing to evaluate for structural heart disease and arrhythmias is necessary. Though a less common etiology, cardiac amyloidosis can present with arrhythmia and syncope, and it is worthwhile to consider the diagnosis in the evaluation of cardiogenic syncope.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
