Abstract
Objectives:
To describe member adherence to a mail-based, health insurer-sponsored gap closure program for colorectal cancer (CRC) screening using multi-target stool DNA (mt-sDNA; Cologuard®) tests.
Methods:
Combined patient data from Exact Sciences Laboratories LLC and data from mass-mailed mt-sDNA orders placed by a large Medicare Advantage Insurance Plan were analyzed (03/01/2023-06/30/2023). Adherence and time to test return were the primary and secondary outcomes, respectively. Their association with patient characteristics was evaluated using multivariable regression.
Results:
Among the 3201 member-patients included (86.6% aged 65-75 years; 58.7% female), adherence rate was 49.4%, and mean time to test return was 25.5 days. After multivariable adjustment, the odds of test return were significantly higher among 65- to 75-year-olds (odds ratio [OR] = 1.59 relative to 45- to 64-year-olds), those living in small towns (OR = 1.43 relative to metropolitan-located individuals), and with digital outreach via both SMS and email (OR = 4.31 relative to no digital outreach). Time to test return was shorter in 65- to 75-year-olds than in 45- to 64-year-olds and was not associated with other patient characteristics.
Conclusions:
Mass-mailed mt-sDNA tests for CRC screening were associated with an overall adherence rate of about 50% in this Medicare Advantage population, with higher likelihood of test return among patients receiving digital outreach.
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer-related deaths in the United States (US) and is estimated to account for 8.7% of all cancer-related deaths in the US in 2024.1,2 Early detection of CRC through screening can help improve outcomes. 3 Previously, the US Preventive Services Task Force (USPSTF) recommended all adults aged 50 to 75 years be screened for CRC; in 2021, this age range was revised to 45 to 75 years. 4
Guideline-recommended CRC screening modalities include endoscopic methods, such as colonoscopy or sigmoidoscopy, and less invasive, stool-based tests that can be performed at home, such as high-sensitivity guaiac fecal occult blood testing (gFOBT), fecal immunochemical test (FIT), and multi-target stool DNA (mt-sDNA) test (marketed as the Cologuard® test; Exact Sciences, Madison, WI).5,6 Access to screening is crucial to the early detection of CRC, which may help reduce subsequent health and financial burden on patients, 7 and adherence to screening recommendations is also critical to ensuring screening effectiveness. Per estimates based on the US standard population in 2020 (ie, prior to the revised recommendations by the USPSTF), 72% of adults aged 50 to 75 years were up to date with CRC screening by any modality. 8 More recently, according to the National Health Interview Survey in 2021, the average up-to-date CRC screening prevalence in the US for those ≥45 years of age was estimated at 59%. 9 Meanwhile, a 2022 US population-based analysis estimated that 44% to 46% of screening-eligible, average-risk individuals aged 45 to 75 years did not have up-to-date CRC screening. 10 Thus, strategies aiming to increase the initiation of CRC screening and the completion of initial and subsequent tests are needed.
Testing for CRC is generally ordered in point-of-care (PoC) settings, where health care providers discuss the importance of screening with their patients and prescribe a CRC test. This ordering approach for mt-sDNA screening has been shown to result in an adherence (ie, completion of an ordered test) of 66.8% in a US nationally insured population 11 and 71.1% in a Medicare population. 12 However, the requirement of patient–provider interactions at the PoC to initiate CRC screening could be a barrier to testing, which may result in a substantial portion of the screening-eligible population remaining unscreened and missing the opportunity to be diagnosed with CRC early when treatment is more effective.
Drawing on lessons from other health-related programs such as the US immunization schedule for children 13 and COVID-19 surveillance testing, 14 an approach to expand program outreach and completion rate is through an opt-out framed recruitment strategy. In the context of CRC screening, this would entail the implementation of mass-mailed tests via a gap closure program, in which a test kit is shipped directly to each patient eligible for CRC screening after an insurer or a health system receives bulk prescriptions from a provider. Such a program has the potential to increase the absolute numbers of patients being screened for CRC by reaching individuals who may otherwise not initiate screening themselves.
A pilot study among Medicare Advantage enrollees receiving mailed FIT outreach has reported an adherence rate of 29%. 15 To date, the potential impact of mass mailing on mt-sDNA screening has not been rigorously evaluated or reported. Given the convenience and non-invasive nature, mass-mailed mt-sDNA tests can be conducive to the initiation and completion of testing in a broad population, which may in turn improve overall CRC screening rate. Hence, this study aimed to assess adherence to a single mt-sDNA test in a population of patients insured with Medicare Advantage plans who received mass-mailed tests via a gap closure program and to compare adherence rates across selected patient characteristics.
Methods
Data Source
Laboratory data from the manufacturer of the mt-sDNA test, Exact Sciences Laboratories LLC (ESL; Madison, WI) and data from mass-mailed orders placed by a large Medicare Advantage Insurance Plan (MAIP) between March 1, 2023, and June 30, 2023, were used. All data were de-identified and compliant with the Health Insurance Portability and Accountability Act (HIPAA), and this study was considered exempt research.
Study Design and Sample Selection
This study included patients aged 45 to 85 years who were MAIP enrollees and received a mass-mailed mt-sDNA test kit between March 1, 2023, and June 30, 2023. Mass-mailed mt-sDNA test kits referred to kits that ESL shipped directly to patients with verified eligibility, after MAIP received bulk prescriptions from a provider via a gap closure program. Patients were excluded from the analyses if it had been <270 days between the date of shipment and the date when the data were pulled for analyses and were not insured under a Medicare Advantage plan.
As standard for the mt-sDNA test, the accompanying patient navigation includes 24h a day, 7days per week, and 365 days per year of support for both patients and providers (with translation services for over 240 languages). Specialized support staff are available to provide personalized outreach based on patient preference. The standard outreach includes a welcome call, along with postal mail, email, and/or text reminders.
Measures
Study measures included patient demographic characteristics such as age category, sex, race, ethnicity, geography, median household income by patient ZIP code, preferred language, and type of outreach (ie, no digital, partial digital with SMS or email only, or full digital with both SMS and email).
Outcomes
The primary outcome of interest was mt-sDNA adherence rate, which was defined as the percentage of eligible patients who completed and shipped the test kit, with the return test kit shipment received by ESL and a valid test result (positive or negative) obtained within 270 days of the initial shipment date. The secondary outcome was time to test return, which was defined as the number of days from the date of test kit shipment to the patient (ie, the start date) to the date of receipt of a test kit by ESL (ie, the end date).
Statistical Analyses
This observational study utilized a convenience sample and did not have a pre-specified effect size for the primary outcome. Descriptive statistics were used to summarize demographic characteristics, adherence rates, and time to test return of the study population. Categorical variables were summarized using counts and percentages and continuous variables were summarized using means.
Adherence rates and mean number of days to test return were compared across categories of patient characteristics (ie, age category, sex, race, ethnicity, geography, median household income by patient ZIP code, preferred language, and type of outreach; results for race, ethnicity, and preferred language are not reported due to substantial missingness). Statistical comparisons were conducted using Chi-square test or Fisher-Exact test (when observations were fewer than 5 in at least 1 cell of the comparison table) for adherence rates, and independent t-test (2 groups) or analysis of variance (≥3 groups) for mean number of days to test return.
The relationship between selected patient characteristics and adherence rate (binary outcome) was assessed using a logistic regression model and reported using adjusted odds ratios (OR) and 95% confidence intervals (CI). The association of selected patient characteristics with time to test return (continuous outcome) was assessed using a linear regression model with log transformations and reported using exp(estimate) and its 95% CI. For both regression models, covariates were included if they had significant p values (ie, P < .05) in the descriptive, univariable analysis. The covariates included in the final models were age category (reference [ref]: 45-64 years), geography (ref: metropolitan), and type of outreach (ref: no digital).
All statistical analyses were conducted using R version 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Sample Characteristics
A total of 3228 mass-mailed mt-sDNA tests were shipped to individual patients between March 1, 2023, and June 30, 2023. After excluding 27 patients who did not meet the study criteria (delayed outreach n = 14, unreachable phone type n = 13), the final sample comprised 3201 patients.
The demographic characteristics of the study sample are summarized in Table 1. Most patients (86.6%) were aged 65 to 75 years and 58.7% were female; a substantial proportion of patients (~90%) had missing data on race, ethnicity, and preferred language. A majority of patients lived in metropolitan areas (71.2%) and in a ZIP code with median household income ranging from $25 000 to <$75 000 (83.6%). The most common outreach methods were partial digital via SMS (61.8%) and non-digital (33.6%).
Demographic Characteristics of the Study Sample.

Race, ethnicity, and preferred language are not presented due to substantial missingness (~90%).
Adherence Rate and Time to Test Return
Overall, the adherence rate was 49.4%, with the mean time to test return being 25.5 days (Table 2). The adherence rate was higher among those aged 65 to 75 years (50.8%) relative to those aged 45 to 64 years (39.9%) and 76 to 85 years (41.7%; P < .001). Email only and SMS only outreach had higher adherence (57.9% and 51.1% respectively) compared with no digital outreach (43.4%), with the highest adherence rate being among patients receiving outreach via both email and SMS (75.9%; P < .001). Adherence was slightly higher for small town- (55.3%) and micropolitan-located (52.6%) individuals relative to those living in rural (46.9%) and metropolitan (48.2%) areas (P = .044). There were no statistically significant differences in time to test return across any of the patient characteristics (Table 2).
Adherence Rate and Mean Time to Test Return Overall and by Patient Characteristics.
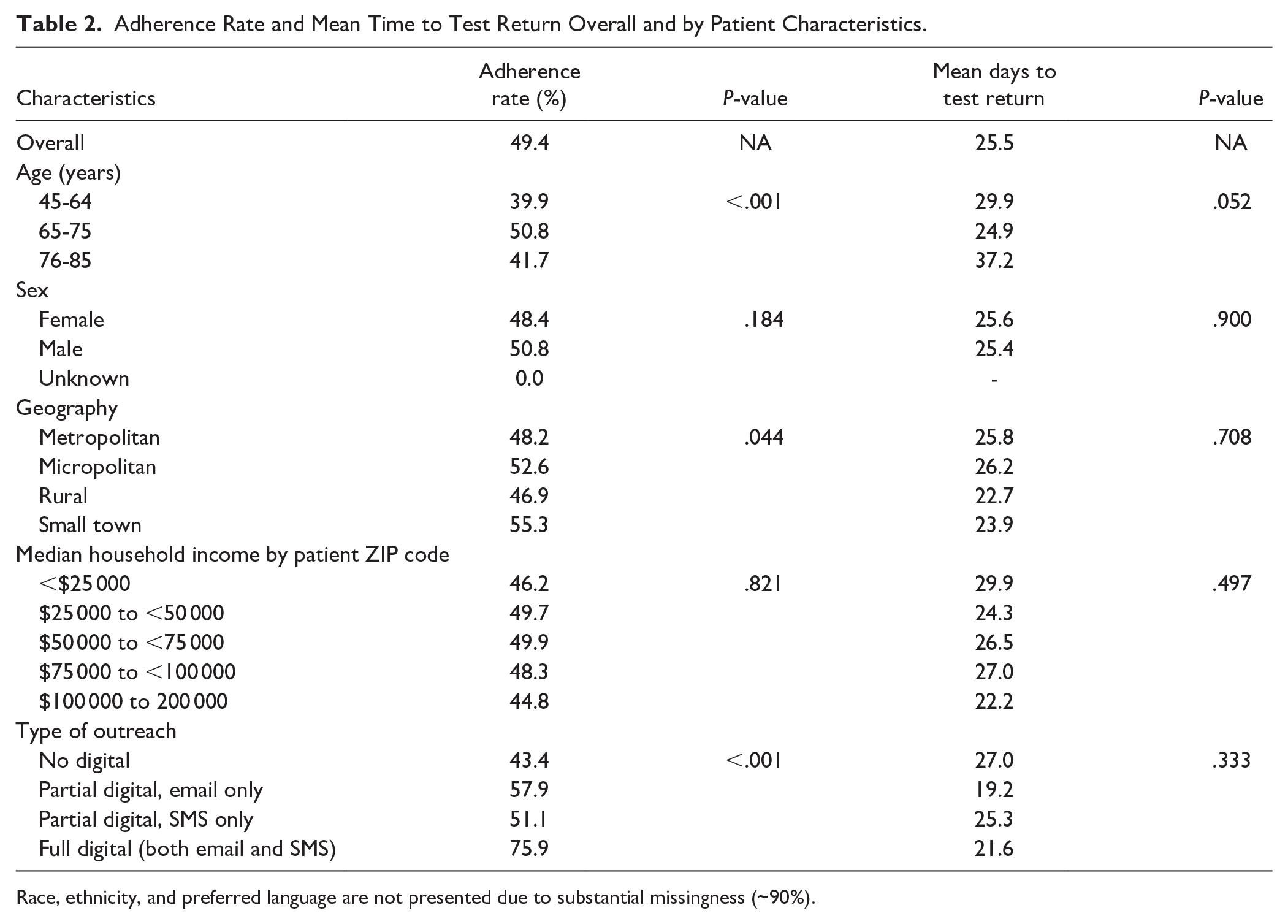
Race, ethnicity, and preferred language are not presented due to substantial missingness (~90%).
Results of the regression models evaluating the association of selected patient characteristics with adherence rate and time to test return are shown in Table 3. Patients in the 65 to 75 years age group were more likely to return the test compared with those in the 45 to 64 years age group (OR = 1.59, 95% CI = 1.27-1.98, P < .001), and those living in a small town were more likely to return the test compared with those living in a metropolitan location (OR = 1.43, 95% CI = 1.13-1.81, P = .003). The likelihood of returning the test was generally higher among patients with any digital outreach compared with no digital outreach. Specifically, patients receiving partial digital SMS only were 40% more likely to return the test (OR = 1.40, 95% CI = 1.20-1.63, P < .001), and those receiving full digital with both email and SMS had more than a 4-fold increase in their likelihood of test return (OR = 4.31, 95% CI = 2.72-6.81, P < .001). Meanwhile, patients 65 to 75 years of age had time to return that were about 12% lower (ie, shorter) than those 45 to 64 years of age (95% CI = 1%-23% lower return times, P = .034); no other patient characteristics were associated with significant differences in time to test return.
Association of Patient Characteristics With Adherence Rate and Time to Test Return.

Abbreviations: CI, confidence interval; ref, reference.
Discussion
The overall adherence rate of close to 50% found in this study among patients insured under Medicare Advantage plans who were shipped a mass-mailed mt-sDNA test via a gap closure program appears higher than that reported for FIT, another home-based CRC testing modality, in a similar patient population. In a study among Medicare Advantage enrollees receiving a mailed FIT kit, 29% of screening-eligible patients completed and returned the kit within 12 months. 15 Compared with prior studies on adherence to mt-sDNA tests ordered in PoC settings, the adherence to mass-mailed mt-sDNA tests found in this study was lower than those reported for a US nationally insured population (67%) 11 and in a Medicare population (71%). 12 Notably, the current study population comprised patients covered under Medicare Advantage, who were found to have a slightly higher CRC screening rate of 57% compared with 50% in those with traditional Medicare in a large-scale US study. 16 Efforts to increasing these CRC testing rates among the Medicare population is particularly important given this population is within the age range recommended for CRC screening.4,17
Adherence rates varied by certain patient characteristics. This study found that patients in the 65 to 75 years age group were more likely to return the mt-sDNA test kit than those in other age groups. This may be partially explained by a potential lack of awareness among younger individuals about the USPSTF-recommended starting age for CRC screening, which was changed from 50 to 45 years in 2021, as well as the recommendation for selective screening among those aged 76 to 85 years. 4 This study also found that adherence rates were higher with digital outreach modalities, with an adherence rate of over 75% among those receiving both email and SMS, who were also found to be 4 times more likely to return the test compared with no digital outreach, suggesting that patient navigation support and outreach via digital channels may help improve CRC screening rates. In this regard, each ordered Cologuard test kit includes access to a patient navigation program, where patients may receive emails and/or SMS reminders in addition to live support and automated phone calls 18 ; these additional digital outreach channels may have helped increase screening adherence to mt-sDNA testing. Moreover, despite not having any pretest counseling, the mean time to completion of the test in this cohort (25.5 days) was remarkable. Collectively, these results provide crucial insight into the patient characteristics associated with adherence to CRC screening, which may inform strategies for improving screening rates and early detection of CRC.
The current study was relatively unique considering the mass mailing aspect. Unlike tests requested at the PoC, which rely on patient and provider encounters and discussions on the need for CRC screening/adherence before a test is ordered, mass-mailed test orders via a health insurer-sponsored gap closure program are shipped to eligible patients without having been requested at the PoC. Therefore, the return of a mass-mailed test kit reflects a patient’s initiative to complete CRC screening. Mass-mailing screening strategies may improve CRC detection and reduce disease burden by potentially reaching patients who are not prescribed CRC screening in PoC settings, including the large proportion of asymptomatic patients who may otherwise not seek medical services until their cancer has progressed to a stage that is more difficult to treat. One population-based study has found that over 55% of screen-detected CRC are asymptomatic, 19 and early detection has been shown to improve outcomes in individuals with average risk of CRC who may not exhibit any symptoms, 20 highlighting the potential benefits of expanding screening access to a larger population. By eliminating the need for PoC encounters to initiate CRC screening, mass mailing could increase the absolute numbers of individuals who complete CRC screening and provide an opportunity to help close the CRC screening gap between the current state and the 80% goal set by CRC societies in the US. 21
Furthermore, mass mailing may be a potential cost-effective investment for insurance payers. Mass mailing may help reduce additional health care costs associated with appointments and procedures (eg, with colonoscopy screening). 22 It may also help reduce disease management costs by allowing the detection of cancer early when treatment has a higher likelihood of success. 23 Additionally, the convenience of home-based mt-sDNA testing may help further improve CRC screening rates and early detection of CRC and pre-cancerous lesions. 24 Most importantly, through increased screening, a mass-mailing strategy has the potential to improve the quality of patient lives and reduce the societal and individual burden associated with the management of CRC identified at later stages. 25 Further research on mass-mailing screening strategies for CRC using mt-sDNA should aim to identify interventions that increase adherence rates, especially in groups with low return rates; understand how adherence rates for mass-mailed mt-sDNA tests vary in other populations (eg, patients with commercial insurance); and assess rescreening rates among those with a valid test return given the suggested 3-year interval of mt-sDNA testing.
The findings of this study should be considered with some limitations. First, there was high missingness for race, ethnicity, and preferred language information; hence, the impact of these factors on adherence and time to test return could not be assessed. Second, it was assumed that the insurer assessed test eligibility criteria for each individual (ie, aged 45 years or older and at typical average risk of CRC) who received a mass-mailed test kit, but this information could not be ascertained. Third, this study only assessed the rate of return of a single test; future studies should assess continued adherence to screening guidelines (eg, mt-sDNA testing every 3 years). Additionally, we understand that our decision to exclude patients from the analyses if it had been <270 days between the date of shipment and the date when the data were pulled for analyses may introduce bias towards a favorable study result; however, we feel that our current analysis was based on a suitable cut point for the dataset, and we did not analyze those patients who fell within the range of less than 270 days. Fourth, this study focused on mt-sDNA tests that were mass-mailed, and the results may not be generalizable to populations who receive PoC screening; this factor should be considered when comparing the current results to studies that included populations who ordered tests at PoC. Lastly, the current data did not allow for the assessment of adherence to follow-up colonoscopy among those who tested positive with mass-mailed mt-sDNA tests; this question warrants further investigations with mass-mailing strategies.
Conclusions
Mass-mailed mt-sDNA tests shipped directly to patients via a health insurer-sponsored gap closure program yielded an adherence rate of about 50% in a Medicare Advantage population. Digital outreach programs, particularly with both email and SMS, were associated with a higher likelihood of test return. Mass-mailing screening strategies have the potential to improve CRC screening rate by reaching individuals who may otherwise not interact with the health care system. Future studies evaluating follow-up testing and treatments among patients who tested positive with mass-mailed mt-sDNA tests are warranted.
Footnotes
Acknowledgements
Medical writing and editorial assistance was provided by Flora Chik, PhD, MWC, an employee of Analysis Group, Inc. and funded by Exact Sciences Laboratories LLC.
Author Contributions
All authors have made substantial contributions to the conception or design of the study, or the acquisition, analysis, or interpretation of data, drafting the manuscript and revising it critically for important intellectual content, and have provided final approval of this version to be published and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG, TP, QL, AP, WKJ, ABO, and PL are employees of Exact Sciences Corporation and own stock/stock options. DE has a consulting agreement with Exact Sciences to provide support regarding research design and methodology, with proceeds paid to Mayo Clinic. MD is an Associate Professor of Medicine at Duke University and has no financial interests to disclose. JK is an inventor of Mayo Clinic intellectual property under license to Exact Sciences and has received grant support from a sponsored research agreement between Mayo Clinic and Exact Sciences. AMF has been a consultant for AbbVie, Amgen, Centivo, Community Oncology Association, Covered California, EmblemHealth, Exact Sciences, Freedman Health, GRAIL, Harvard University, Health & Wellness Innovations, Health at Scale Technologies, MedZed, Penguin Pay, Risalto, Sempre Health, the State of Minnesota, U.S. Department of Defense, Virginia Center for Health Innovation, Wellth, and Zansors; has received research support from the Agency for Healthcare Research and Quality, Gary and Mary West Health Policy Center, Arnold Ventures, National Pharmaceutical Council, Patient-Centered Outcomes Research Institute, Pharmaceutical Research and Manufacturers of America, the Robert Wood Johnson Foundation, the State of Michigan, and the Centers for Medicare and Medicaid Services.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Exact Sciences Laboratories LLC.
Ethical Considerations
The study was considered exempt research under 45 CFR § 46.104(d)(4) as it involved only the secondary use of data that were de-identified in compliance with the Health Insurance Portability and Accountability Act (HIPAA), specifically, 45 CFR § 164.514.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
The data that support the findings of this study are available from Exact Sciences Laboratories LLC. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors with the permission of Exact Sciences Laboratories LLC.
