Abstract
Low back pain is the most common musculoskeletal complaint accounting for over 30 million visits to primary care physicians annually. Serious pathology is found in less than 1% of these visits. Therefore it is often a challenge to distinguish worrisome findings requiring further workup and treatment from common complaints of pain. Gout is an inflammatory arthritis that most commonly affects the appendicular skeleton. It is characterized by the saturation of uric acid and deposition of monosodium urate crystals in joints and tissues. Spinal involvement is rare and is not typically considered on the differential diagnosis for a patient presenting with acute low back pain. We present such a case of a 35-year-old male who presented with intractable back pain, highlighting the need to recognize signs and symptoms that raise suspicion for spinal gout.
Introduction
Acute low back pain (LBP) is defined as pain lasting less than 12 weeks that “originates below the costal margin and above the inferior gluteal folds, with or without referred or radicular leg pain.” 1 The majority of adults in North America will experience acute LBP at some point in their lives.1,2 In the United States, it is the fifth most common reason for a physician visit, accounting for 30 million visits annually.3,4 Most acute LBP is self-limited, non-specific, and very rarely a harbinger of a serious condition.5 -8 A complaint of acute LBP is challenging for primary care clinicians to quickly and efficiently determine whether or not additional workup for a possible serious condition is required. In this case report, we present an unusual but serious condition causing back pain that can be easily treated once the correct diagnosis is established.
Case Presentation
A 35-year-old male presented to the emergency department with intractable back pain. The previous night while working as a hotel manager, he had back spasms in his lumbar region. The pain radiated down his right leg, was exacerbated by any movement, and rendered him unable to stand. He denied preceding trauma, bowel or bladder dysfunction, saddle anesthesia, intravenous drug use, malignancy, recent weight loss, or night sweats.
His history was significant for gout, confirmed by uric acid crystals in intraarticular fluid from left knee aspiration years prior. His flares were historically treated with NSAIDs, colchicine, and prednisone. He had never been on urate-lowering therapy. The last gout flare of his left knee was 3 months prior, treated with oral prednisone and indomethacin and resolved entirely. Two weeks ago, he started having “gout” pain in his right knee and ankle for which he started taking ibuprofen.
Initial vital signs in the emergency department were temperature 37.4°C, pulse 110 beats/min, respirations 18 breaths/min, BP 153/75 mmHg, SpO2 98% on room air. Two hours later, he became febrile to 38.3°C and remained tachycardic. He had normal cardiovascular and pulmonary examination. No rash or erythema was noted. Neurologic exam revealed normal muscle strength in the lower extremities, with normal reflexes, and no sensation abnormalities. Back spasms limited his ability to participate in the musculoskeletal exam, but a left knee effusion was noted. There was no midline tenderness to palpation of his spine.
Laboratory studies revealed WBC of 12,400 cells/cm3, creatinine of 1.21 mg/dL, c-reactive protein (CRP) of 51.0 g/L, and erythrocyte sedimentation rate (ESR) of 29 mm/h. After receiving morphine 12 mg, ketorolac 15 mg, ketamine 50 mg, and diazepam 5 mg, he still had significant pain and was unable to stand. Despite no neurologic deficits, a magnetic resonance imaging (MRI) was obtained due to his fever, labs, and lack of improvement with analgesia. MRI showed mild enhancement and peri-facet edema associated with the right L2 to L3 and bilateral L3 to L4 facet joints. No discitis, osteomyelitis, abscess, or fluid collection was observed. Radiologist commented that these findings “could be degenerative in nature representing active facet arthritis, however septic facet arthritis could also be considered given the clinical presentation.” Blood cultures were obtained, and he was subsequently admitted to the family medicine inpatient service for further management.
He remained unable to participate in his musculoskeletal exam secondary to pain, with any movement, including changing position in bed, excruciatingly painful. He remained afebrile while off antibiotics and antipyretics. Acute spinal gout was suspected rather than septic facet arthritis because he lacked any risk factors for septic arthritis such as known immunodeficiency, IV drug use, skin infection, or diabetes. His uric acid level was 10 mg/dL. A dual-energy CT of the lumbar spine (Figures 1 and 2) revealed erosive bony changes of the right L1 to L2, left L2 to L3, and right L5 to S1 facet joints. Differential attenuation of the soft tissue highlighted in green was seen particularly at the right L1 to L2 and left L2 to L3 facet joints, characteristic of uric acid crystal deposition.

Dual-energy analysis suggests that the involvement particularly at right L1 to L2 (highlighted in green) may be related to uric acid calculi deposition.
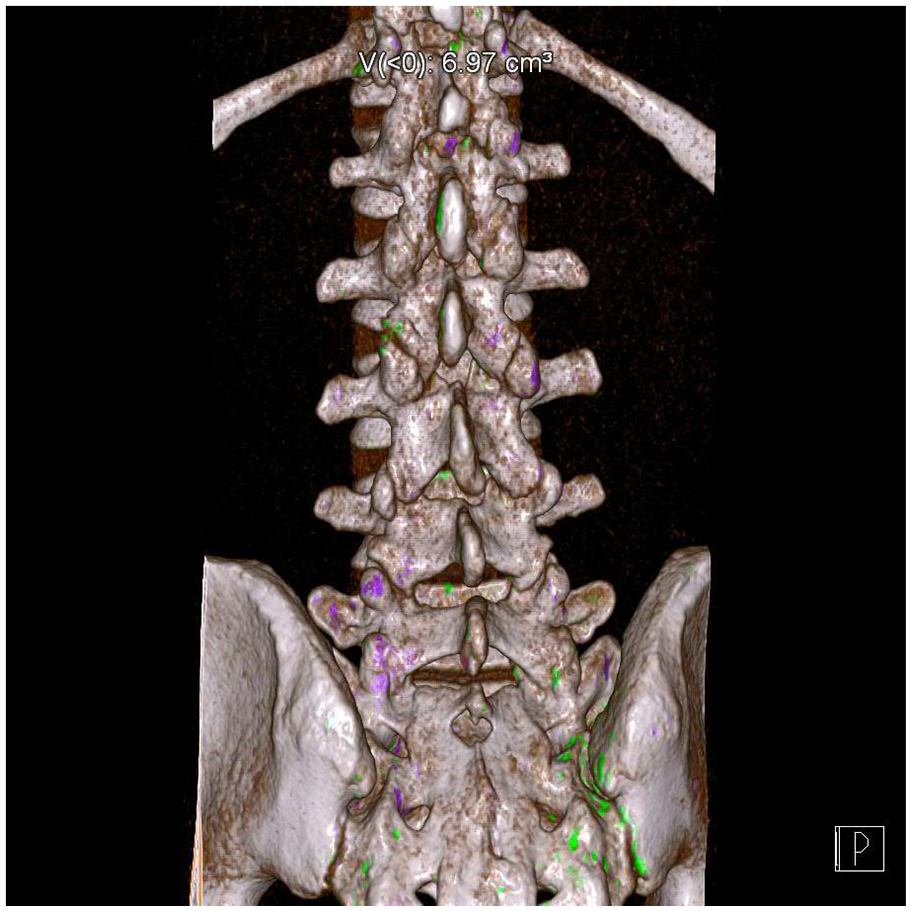
Differential attenuation of the soft tissue highlighted in green is seen particularly at the right L1 to L2, left L2 to L3 facet joints, and right L5 to S1 facet joints, characteristic of uric acid crystal involvement.
He was treated with IV methylprednisolone 125 mg. The following day, his pain decreased by 80% and he could ambulate unassisted and participate in physical therapy. IV methylprednisolone was continued 1 more day and was transitioned to oral prednisone taper as outpatient (40 mg/days × 3 days, then decreasing by 5 mg/days every 3 days). Rheumatology was consulted who assisted with developing his treatment plan which, in addition to steroids, consisted of initiation of urate lowering therapy (allopurinol 300 mg daily, titrated outpatient) with a target uric acid of less than 5 mg/dL because of erosive changes and colchicine 0.6 mg twice daily prophylaxis.
At outpatient follow-up 1 week following hospital dismissal, prednisone taper was continued as he continued to improve. Two weeks later he had recurrent flare of his gout symptoms in his back and left knee. This was treated with IV methylprednisolone, increased dose of oral prednisone and re-taper, intra-articular aspiration and injection of prednisone in the left knee, and continuation of his allopurinol and colchicine. Uric acid dropped to 6.2 mg/dL and rheumatology felt this was an expected flare in the setting of fluctuating uric acid levels during initial urate lowering therapy.
Discussion
Low back pain (LBP) is a leading presenting complaint in many ambulatory care visits. Most cases of LBP resolve spontaneously, however there are rare cases of serious pathology that can cause significant morbidity and mortality. The challenge for primary care physicians is to “find the needle in the haystack” accurately and cost effectively.
Our case of a young man presenting with intractable LBP in the setting of an extensive history of appendicular gout is an example of this conundrum. Axial gout is an infrequent presentation of gout and can lead to diagnostic confusion during evaluation of LBP which encompasses a myriad of other possible underlying diagnoses. Our patient presented with fever, tachycardia, leukocytosis, and elevated inflammatory markers in setting of poorly controlled pain with inability to ambulate despite intravenous analgesics, warranting inpatient hospitalization. With attention to his clinical history, physical examination, and further testing including uric acid level and dual-energy CT (DECT) of his lumbar spine, we were able to identify axial gout as etiology for his presentation.
Current practice guidelines recommend a focused history and physical examination to judiciously stratify severe cases of LBP and determine need for further imaging and workup. 7 Presence of red flag symptoms including fever, vertebral tenderness, limited range of motion of the spine or significant motor weakness, and/or progressive sensory deficit are indicative of more serious causes of back pain and warrant prompt workup rather than watchful waiting. Additionally, the severity of back pain may be out of proportion to physical findings, signaling the need for further workup to astute clinicians. Prompt work-up in setting of clinical findings can include plain radiography, MRI, and blood tests including complete blood count, ESR, and CRP.7,8 MRI remains the preferred mode of imaging over computed tomography (CT) after initial plain radiography, as it provides better visualization of soft tissue. 9 However distinguishing amongst the various considerations of infectious, intrinsic, systemic, or referred etiology, a comprehensive evaluation including understanding the context of the patient’s history is imperative to guide proper diagnosis and management.
Gout is an inflammatory arthritis characterized by the deposition of monosodium urate crystals in joints and tissues. Incidence is up to sixfold higher in men compared to women and increases with age. 10 Risk factors include a number of comorbid conditions such as metabolic syndrome, cardiovascular and renal disease. Gout typically involves the appendicular skeleton, classically the first metatarsalphalangeal (MTP) joint, but it can involve small or larger joints in the extremities. It is unusual to involve the axial skeleton. However, in patients with poorly controlled gout, prevalence of axial involvement has been reported as high as 35%.11,12 Spinal gout can present similarly to back pain of various etiologies and can include symptoms of radiculopathy and claudication. Fever, leukocytosis, and elevated ESR are common, often mimicking a spinal infection. Cases of axial gout are more commonly found in the lumbar spine than the cervical and thoracic spine. 13
Arthocentesis is the diagnostic standard and most accurate method of confirming diagnosis of gout, however it is not applicable in many cases and can impose increased risk of infection. Plain film radiographs, CT, and MRI have limited use in the identification of axial gout, as findings can be non-specific, which can lead to misdiagnosis. However, dual-energy CT imaging is known to have high sensitivity (up to 100%) and specificity (up to 89%) to detect and quantify crystal deposition that can be used in both diagnosis and follow-up of gout.14,15 In our case, facet arthrocentesis was deemed unnecessary in setting of a positive dual-energy CT and known history of appendicular gout.
Treatment of acute gout flares typically involves oral glucocorticoids, nonsteroidal anti-inflammatory drugs (NSAIDs), colchicine, and urate lowering therapy to prevent recurrence. Intravenous glucocorticoids, given in our case, is helpful in cases of polyarticular involvement. Despite the gout flare our patient experienced 2 weeks after initiation of urate-lowering therapy (ULT), recent studies do not show worse outcomes with initiation of urate-lowering therapy (ULT) during acute gout flares.16 -18 The American College of Rheumatology now recommends starting ULT in patients with tophi, radiographic evidence of joint destruction attributable to gout, CKD stage 3 or greater, serum uric acid >9 mg/dL, nephrolithiasis, or 2 or more attacks within a year. 19 ULT should be titrated to a goal serum uric acid <6 mg/dL and patients should be covered by anti-inflammatory prophylaxis with NSAIDs, steroids, or colchicine for 3 to 6 months.
Conclusion
Axial involvement is a rare presentation of gout and is often missed as symptoms mimic alternate etiologies. Suspicion for spinal gout should remain high in the setting of intractable low back pain, especially in individuals with elevated risk factors or a history of appendicular gout. This case highlights the need for familiarity of the prevalence of axial gout and the sensitive and specific imaging tools available that enable accurate diagnosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
