Abstract
Background:
An FQHC in Denver, Colorado developed and implemented an interprofessional care model to collaboratively manage type 2 diabetes mellitus (T2DM). Utilizing the 340B program, the team protocolized ADA Guidelines to promote the early adoption of first-line medications, glucagon-like peptide1 receptor agonists (GLP1 RAs) and sodium-glucose cotransporter 2 inhibitors (SGLT2is) to improve patient outcomes.
Objectives:
To assess the impact of interprofessional collaborative management versus standard care on early initiation of a SGLT2i and/or GLP1 RA as first-line therapies to enhance (1) deprescribing of insulin, (2) reducing overbasalization of insulin through insulin de-escalation, and (3) effectively lowering A1C levels in adult primary care patients with T2DM.
Methods:
This was a retrospective chart review of adult patients with T2DM who were initiated on a GLP1 RA and/or a SGLT2i. To determine the effects of initiating GLP1 RA and/or SGLT2i therapy on insulin usage and glycemic control, the total daily dose (TDD) of insulin before initiation was compared with the most recent TDD post-initiation to evaluate deprescribing. To determine the impact on overbasalization, pre-initiation and post-initiation insulin doses were evaluated. The effectiveness of GLP1 RA and/or SGLT2i in lowering A1C levels was determined by comparing the A1C prior to initiation with the A1C postinitiation. To evaluate the influence of interprofessional collaborative care on insulin deprescribing, overbasalization, and diabetes control, relevant measures were compared between patients receiving collaborative care versus standard care.
Results:
Of the 60 total patients treated with insulin, 46.6% were deprescribed insulin, with a majority in the interprofessional collaborative group (93.1%) compared to standard care (6.9%). A total of 78.3% of patients benefited from a reduction in A1C following the initiation of a GLP1 RA and/or SGLT2i. The greatest A1C reduction was −2.9% in the group receiving metformin in addition to a GLP1 RA and a SGLT2i. Patients who received interprofessional collaborative care had an average A1c reduction of −2.9% compared to—1.1% with standard care.
Conclusion:
Most patients initially overbasalized on insulin experienced a reduction in overbasalization after initiating GLP1 RA and/or SGLT2i. There was a notable A1C reduction, de-escalation, and deprescribing of insulin in patients receiving interprofessional collaborative care.
Keywords
Background
The prevalence of type 2 diabetes (T2DM) is a significant concern in the United States (US), with more than 37 million people having T2DM, which accounts for approximately 90% to 95% of all cases of diabetes. Diabetes is the eighth leading cause of death in the US and the number of adults diagnosed with diabetes has more than doubled in the last 20 years. 1 Underserved populations, including low-income, minority, and rural communities, face a higher burden of T2DM due to socioeconomic disparities, limited access to healthcare, and lifestyle factors. The prevalence and incidence of T2DM vary based on demographic and socioeconomic factors. Federally Qualified Health Centers (FQHCs) play a crucial role in addressing healthcare disparities among underserved populations by offering accessible and affordable medical services, making them pivotal in providing preventive healthcare to these communities. 2
The current American Diabetes Association (ADA) guidelines continue to recommend metformin as a first-line agent, but more recently, added the use of glucagon-like peptide1 receptor agonists (GLP1 RAs) and sodium-glucose cotransporter 2 inhibitors (SGLT2is) as first-line treatments. Since January 2022, the ADA guideline recommends the use of GLP1 RAs and SGLT2is as first-line agents as they lower A1C, promote weight loss, and provide cardiovascular and renal protection.3,4 The guidelines additionally focus on reducing overbasalization of insulin, which is defined as a dose of basal insulin over 0.5 units/kg/day. The guidelines currently recommend optimizing first-line treatments prior to initiating insulin since these treatments provide additional benefits beyond blood glucose. Additionally, these agents are recommended to minimize overbasalization in patients already on insulin therapy. 3 It is important to note the role of insulin therapy in those diagnosed with diabetes in the US; according to the CDC, 10.8% of all US adults diagnosed with diabetes start using insulin within a year of their diagnosis. 1 However, there is a lack of data exploring the discontinuation of insulin after initiating a GLP1 RA and/or a SGLT2i. Furthermore, the impact of GLP1 RA and/or SGLT2i therapy on deprescribing or reducing insulin requirements for T2DM is limited. While studies indicate the potential for decreased insulin use with these medications, there is a gap in exploring complete discontinuation.5,6
Early implementation of guideline-based treatments in the primary care setting could result in de-escalation and/or deprescribing insulin, reduction in overbasalization, and improved A1C reductions. Delays in adoption of current guidelines in the primary care setting are multifactorial. Primary care offices must proactively create mechanisms for dissemination of new guidelines. These practices are not standardized and vary greatly from clinic to clinic. Additionally, many of the recommended medications in current guidelines are newer medications to the market and are often cost-prohibitive to patients. Through the 340B program, FQHCs can improve patient access by allowing them to offer low-cost medications. Many FQHCs additionally offer interprofessional collaborative care to enhance patient care and outcomes. One of the benefits of collaborative interprofessional care is to help promote early adoption of guidelines to enhance patient outcomes. Furthermore, collaborative interprofessional practice, involving healthcare providers from various disciplines, has been shown to increase confidence and efficacy when prescribing newer medications, such as GLP1 RAs and/or SGLT2is, enhancing patient outcomes and quality of care. 7
An FQHC in Denver, Colorado developed and implemented an interprofessional collaborative care model, which consisted of primary care providers, a clinical pharmacist, and clinical pharmacy students on rotation, with assistance by a registered nurse and behavioral health specialist when appropriate, to collaboratively manage chronic diseases. Utilizing the 340B program, there was an effort to promote the early adoption of first-line medications as recommended by clinical guidelines to improve patient outcomes. Although many of the patients were collaboratively managed by the interprofessional team, some patients maintained standard management by the primary care provider (PCP) only. The objective of this quality improvement study is to compare the impact of implementing the current diabetes guidelines in the context of collaborative interprofessional management of patients with T2DM, as opposed to standard management. The specific aims of the study are to assess the impact of initiating a GLP1 RA and/or SGLT2i as first-line therapies, to (1) result in deprescribing of insulin, (2) reducing overbasalization of insulin through insulin de-escalation, and (3) effectively lowering A1C levels in adult primary care patients with T2DM.
Methods
This was a retrospective chart review of adult patients with T2DM who received primary care services at a FQHC in Denver, Colorado. This quality improvement initiative was exempt per the Colorado Multiple Institutional Review Board. Patient data was extracted from the electronic health record, Greenway. Patients were included if they were aged 18 to 79 years with a diagnosis of T2DM between January 2021 and May 2023. Patients were excluded if a GLP1 RA and/or SGLT2i was newly initiated and had not yet been titrated, if they had a diagnosis of T1DM, if they had not been seen in the past 12 months by a PCP, transferred care to another clinic, or were deceased during the study period. Data collection was performed by the clinical pharmacy team, which included patient demographic information (age, sex, ethnicity, weight, and BMI), renal function, diabetes treatment regimen during the study period, A1C, total daily dose (TDD) of insulin prior to initiation of GLP1 RA and/or SGLT2i (if applicable), A1C and TDD of insulin following initiation and/or titration to the highest tolerated dose of a GLP1 RA and/or SGLT2i, and enrollment status into the interprofessional collaborative care program. Patients were disenrolled from the program upon reaching their goal A1C or if they did not actively engage in the program after 3 patient visit attempts.
To determine if initiation of a GLP1 RA and/or SGLT2i allowed for deprescribing of insulin, the TDD of insulin before initiation was compared with the TDD post-initiation. To determine if the initiation of a GLP1 RA and/or SGLT2i allowed for de-escalating the dose of insulin to reduce overbasalization, which is defined by the ADA as a basal insulin dose exceeding 0.5 units/kg/day, the units/kg/day of insulin prior to initiation were contrasted with the units/kg/day post-initiation. To determine if the initiation of a GLP1 RA and/or SGLT2i effectively lowered A1C levels, the A1C before initiation was compared to the A1C post-initiation. To determine the impact of interprofessional collaborative care in adult primary care patients with T2DM, the rates of deprescribing and de-escalation of insulin to reduce overbasalization and reductions in A1c were compared to rates seen in patients managed with standard care. Descriptive analysis was reported for all variables evaluated except for pre-post de-escalation of insulin, which was analyzed using the Fisher’s exact test with a priori significance level of 0.05.
Results
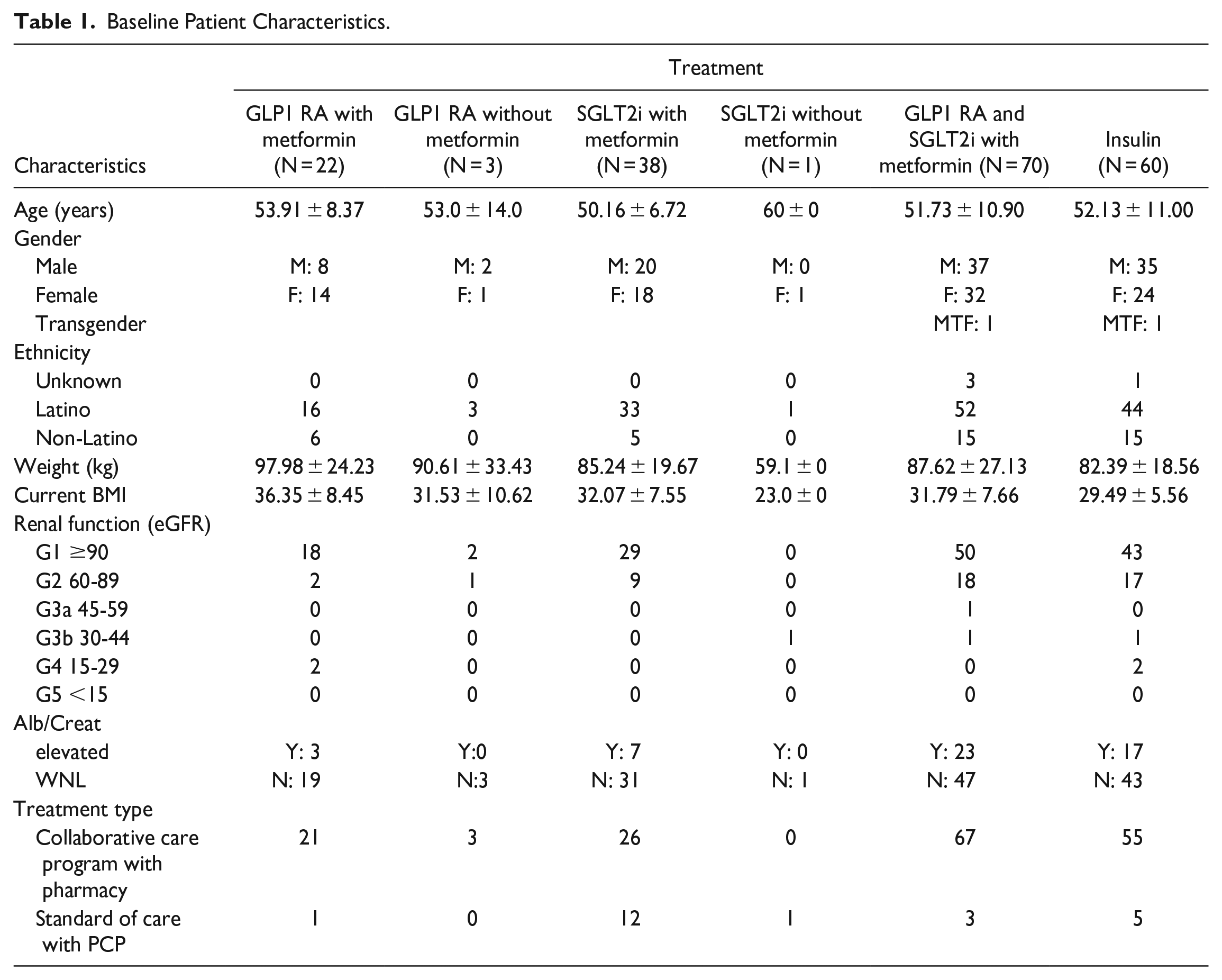
Two hundred and sixty-four patients were included in the study. Seventy patients were excluded from the study analysis (35 had not been initiated on, or were newly initiated on a GLP1 RA and/or SGLT2i which had not yet been titrated to an optimized dose, 2 had a diagnosis of T1DM, 28 were not seen in the past year, 3 transferred medical care to another clinic and 2 were deceased), with a total of 194 patients included in the analysis. The average age of patients was 53 years with 52.28% being male, 46.7% being female, and 0.5% being transgender. The average BMI was 30.7 kg/m2 and most patients were Latino (76.14%). Twenty-five percent had a chronic kidney disease (N = 50). The majority of patients (87.8%) received interprofessional collaborative care. Most patients received a combination of Metformin with a GLP1 RA and SGLT2i (N = 70) followed by metformin and a SGLT2i (N = 38) and then metformin and a GLP1 RA (N = 22) with very few patients (N = 4) receiving a GLP1 RA or SGLTi without metformin (see Table 1).
Baseline Patient Characteristics.
Deprescribing Insulin
There was a total of 60 patients on insulin (N = 55 in interprofessional collaborative care group and N = 5 in standard care group) with a total of 48.3% (N = 29) deprescribed insulin. Of those deprescribed insulin, 93.1% were in the interprofessional collaborative care group (N = 27) and 6.9% were in the standard care (N = 2). Among the various regimens, the largest number of patients who had insulin deprescribed (55.2%), occurred in the groups receiving interprofessional collaborative care on the combination of GLP-1 RA and SGLT2i with metformin followed by 27.6% in the GLP-1 RA and metformin. Comparatively, under standard care, 1 patient was deprescribed insulin in the SGLT2i with metformin group and 1 patient in the GLP-1 RA and SGLT2i with metformin group (see Table 2).
Patients Deprescribed Insulin Following the Initiation of a GLP1 RA and/or SGLT2i Who Received Standard Care vs Interprofessional Collaborative Care.

De-Escalation of Insulin to Reduce Overbasalization
There were 60 patients receiving insulin with 61.1% (N = 37) initially overbasalized which decreased to 17.5% (N = 11) following the initiation of a GLP1 RA and/or SGLT2i. Of the 8 patients initially treated with insulin plus metformin, 50% (N = 4) were overbasalized. The addition of a GLP1 RA to this group resulted in all patients (N = 8) having de-escalations in dose and eventual discontinuation of insulin therapy. In the patients only on insulin (N = 1), this patient was overbasalized prior to the initiation of a GLP1 RA, which was also de-escalated and then discontinued. There was a total of 11 patients who received metformin, insulin and a SGLT2i during the study period. Of these patients, 57% (N = 7) were overbasalized prior to the initiation of a SGLT2i with 1 patient who was not on insulin, but then required insulin. Following the initiation of a SGLT2i, 36% of patients were overbasalized and 27% were de-escalated (P = .0769), and eventually insulin was discontinued in 100% of these patients. There were 25 of 40 patients on insulin and metformin who were overbasalized (62.5%) prior to the initiation of a GLP1 RA and SGLT2i, which then decrease to only 17.5% (N = 7), which was statistically significant (P < 0.0001). A total of 17 patients from this treatment group (42.5%) eventually had their insulin therapy discontinued (Tables 3 and 4).
Change in Insulin Dose Pre and Post Initiation of Intervention to Manage Overbasalization Through De-escalation.
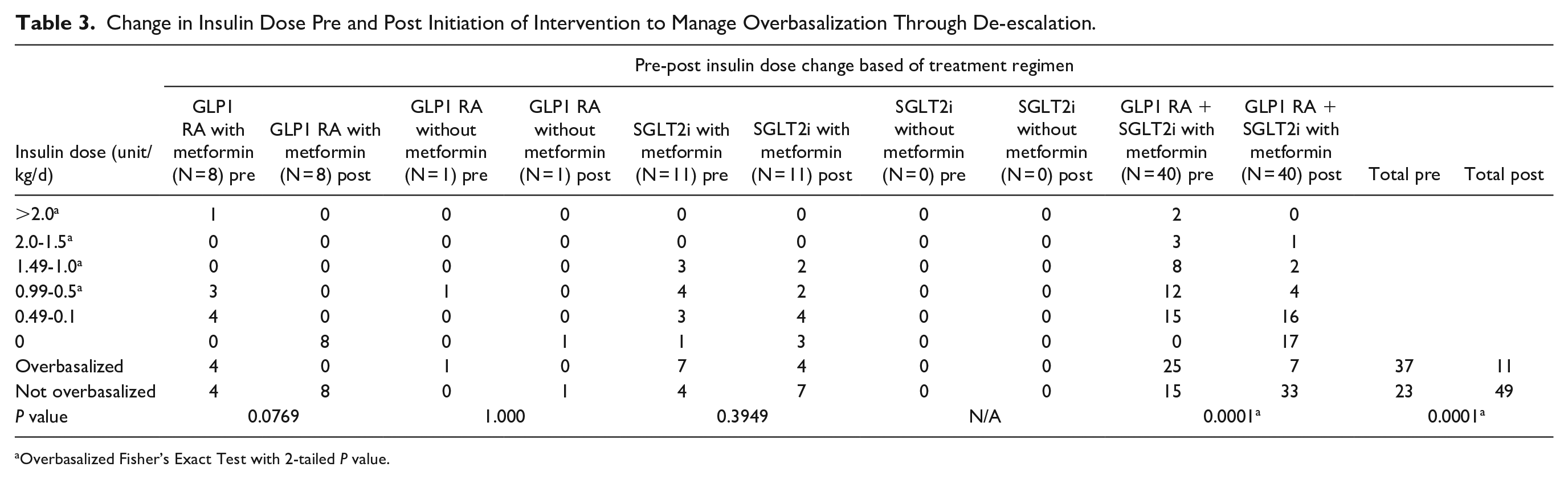
Overbasalized Fisher’s Exact Test with 2-tailed P value.
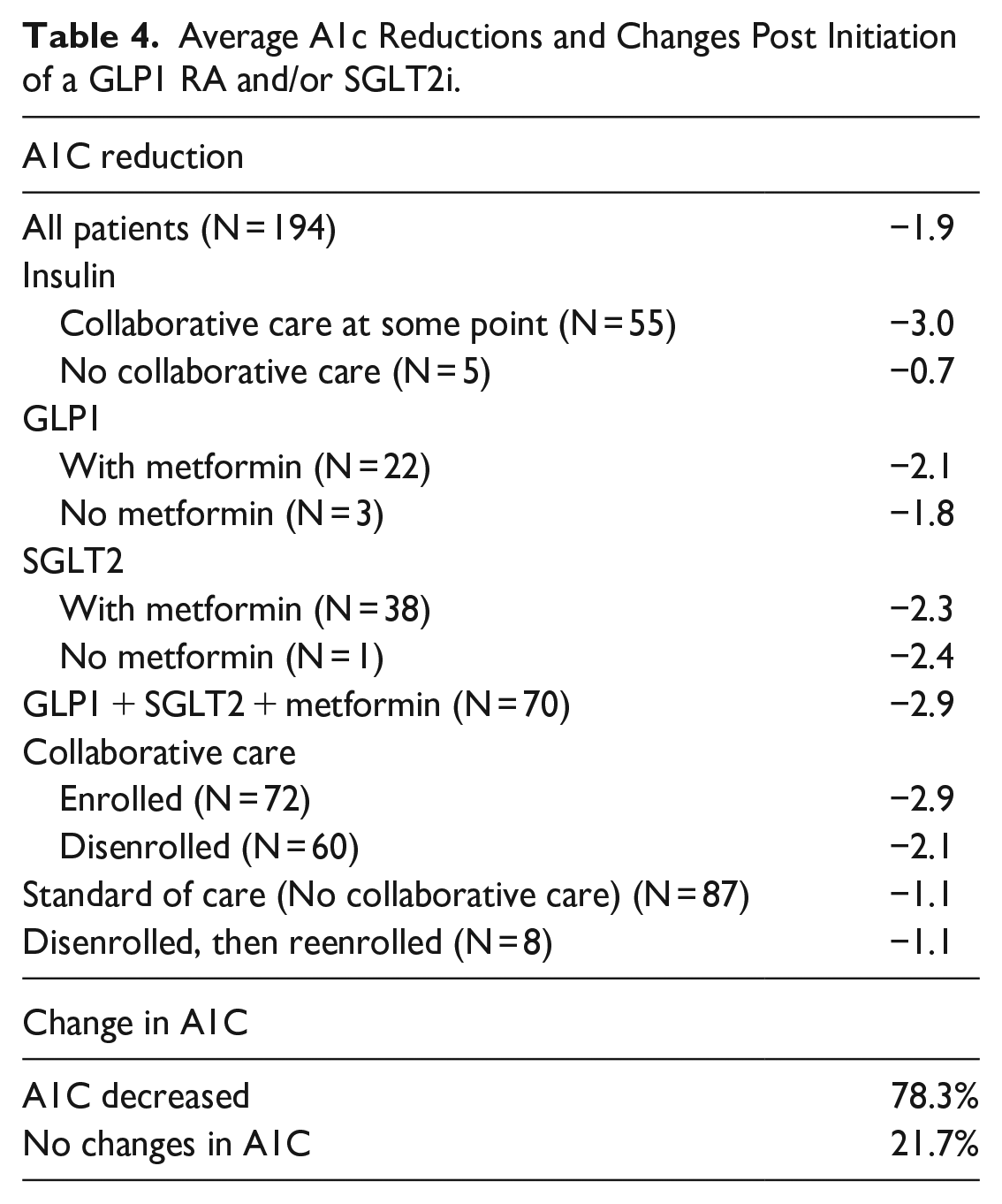
Average A1c Reductions and Changes Post Initiation of a GLP1 RA and/or SGLT2i.
Reduction in A1C
The average additional A1C reduction in patients initiated on a GLP1 RA and/or SGLT2i was −2.1% with 78.3% of patients benefiting from a reduced A1C and no patients having an increased A1C. The greatest additional A1C reduction was −2.9% in the group receiving metformin in addition to a GLP1 RA and a SGLT2i. Patients who received a SGLT2i with or without metformin had a −2.3% and −2.4% reduction, respectively compared to patients who received a GLP1 RA with or without metformin (−2.3% and −1.8%, respectively). Patients who received interprofessional collaborative care had a −2.9% reduction compared to −1.1% with standard care. Data demonstrated that patients disenrolled from interprofessional collaborative care also benefited with a −2.1% A1C reduction.
Discussion
Early initiation of GLP1 RA and SGLT2i medications in patients with T2DM is an effective way to minimize insulin requirements to maintain blood sugar control. Our data demonstrates 61.1% of patients were initially overbasalized which decreased to 17.5% following the initiation of a GLP1 RA and/or a SGLT2i. Furthermore, 48.3% of patients were able to discontinue insulin therapy entirely following the initiation of 1 or both agents. This contrasts with the studies by Van Dril et al 5 and George et al 6 who found that patients’ insulin dosage may be decreased but did not note insulin discontinuation.
Despite many primary care practice sites not rapidly adopting the newest ADA guidelines, this data supports the importance of early guideline adoption of GLP1 RA and SGLT2i medications as first-line therapies and the significant implications for practice. Shay et al 8 identified how the lack of provider confidence may impact the rapid adoption of newer medications. 8 Fonseca 7 evaluated how patient outcomes are dependent on how providers manage the care and use of the medications for patients at an increased risk for poorly controlled diabetes. Our study highlights how interprofessional collaborative management is an effective mechanism to enhance management of T2DM patients by promoting provider confidence, as evident by the uptake of current guidelines at the practice level. This was achieved through provider education and utilization of a standardized patient care decision-making process through implementation of guideline-based practice protocols. The Collaborative Drug Therapy Management (CDTM) protocols are developed by the clinical pharmacist, which are then reviewed by prescribers for input. This process allows for provider education while enhancing standardized implementation through protocolization. The interprofessional collaboration where providers have input on how the program impacts workflow appears to be an important aspect for CDTM to be effective. Bald et al 9 evaluated the pharmacist-led diabetes CDTM services in 2 primary care settings, but had barriers related to provider uptake and noted providers wanted to explore potential options to better align workflow. 9
The use of the interprofessional collaborative management model has proven to be a significant intervention as evidenced by the average A1C reduction in patients who received interprofessional collaborative care with a −2.9% reduction compared to −1.1% with standard care. Access to affordable medications is a crucial piece of collaborative drug therapy management, particularly since GLP1 RAs and SGLT2is may be cost-prohibitive medications. Through 340B, our clinic site was able to provide access to these medications which not only improved the identified outcomes, but also removed prescriber barriers.
Study Limitations
This research is subject to certain limitations. Its retrospective design may limit the reproducibility of results in broader populations. A small study sample may have limited our ability to demonstrate statistically significant outcomes in all areas related to overbasalization and de-escalation of insulin. However, despite the small sample size, we were able to demonstrate statistically significant outcomes in patients who were initiated on both GLP1 RA and SGLT2i’s, and we were trending toward significance with the GLP1s plus Metformin sample as well as with the total population. Additional studies with a larger sample size would be warranted to elucidate greater clinical impact and broader statistical significance. Given that our study took place in a FQHC in Southwest Denver, where the majority of the population is Hispanic/Latinx, the findings might differ in populations with different cultural backgrounds, such as Asian or Middle Eastern, due to variations in diet and exercise patterns. Additionally, the availability of affordable medications in our clinic might not be representative of other healthcare settings. The academic nature of our clinic, with a substantial presence of students, interns, and residents, facilitates collaborative drug therapy management and the adoption of evidence-based guidelines, which may be challenging for non-academic or private clinics. It’s also worth noting that the study did not assess the impact of patient education on diet and exercise, potentially influencing the observed outcomes.
Implications
Implications for practice arising from this study underscore the importance of adhering to current management guidelines. The study demonstrated enhanced A1C control, reduced overbasalization, and the potential for complete insulin discontinuation by aligning patient regimens with evidence-based guidelines. Collaborative efforts between primary care providers and clinical pharmacists, grounded in a shared, evidence-based protocol for T2DM management, not only optimizes patient care but also establishes mutual accountability among healthcare professionals. In some states, clinical pharmacists are recognized as providers and can readily be integrated into the interprofessional collaborative care team. Clinics that do not have a clinical pharmacist may benefit from exploring ways to expand their interprofessional care team to include this key member. Access to affordable medications emerges as a pivotal factor, fostering provider awareness and boosting confidence in prescribing newly added first-line medications, and promoting standardized practices.
Conclusion
This quality improvement study demonstrated how an FQHC utilizing the 340B program in Denver benefited from an interprofessional collaborative model, which allowed for deprescribing of insulin in 46.6% of patients with a greater impact on deprescribing with the collaborative care model compared to standard care (93.1% vs 6.9%). Additionally, there was a reduction in overbasalization of 43.6% after initiating GLP1 RA and/or SGLT2i. There was a notable A1C reduction of -2.9% in patients receiving collaborative care compared to a −1.1% reduction with standard care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
