Abstract
Purpose:
A case of a patient receiving warfarin for pulmonary embolism (PE) concomitantly with rifampin for treatment of active pulmonary tuberculosis (PTB) is presented. A successful clinical intervention whereby the patient achieved therapeutic anticoagulation after switching to an alternative rifamycin antibacterial, rifabutin, is described.
Summary:
The drug-drug interaction between warfarin and rifampin is well known and documented. However, to our knowledge, no case reports of the interaction between warfarin and rifabutin have been published, and literature describing this interaction is lacking. We describe the case of a 27-year-old African American female referred to a pharmacist-managed anticoagulation clinic for treatment of PE with warfarin. The patient was also being treated for active tuberculosis with rifampin, isoniazid, pyrazinamide, and ethambutol. Warfarin was initiated and over the course of 1 month was continuously increased to a total weekly dose (TWD) of 140 mg without ever achieving the target international normalized ratio (INR) of 2 to 3. In an attempt to reach the target INR, rifampin was switched to rifabutin to minimize the drug-drug interaction with warfarin. Six days after this switch, the target INR was achieved with a lower warfarin TWD of 115 mg. Rifabutin interacts with warfarin to a lesser degree than rifampin and may be considered as an alternative in patients taking warfarin who require treatment with a rifamycin.
Conclusion:
For patients in whom therapeutic anticoagulation with warfarin has been difficult, the use of rifabutin may be considered in place of rifampin when the concomitant use of a rifamycin is required.
Highlights
Limited data available supporting the use of Rifabutin instead of Rifampin to limit the drug drug-interaction with warfarin.
To our knowledge, this is the first published case report of a successful rifabutin use as an alternative to rifampin in a patient taking warfarin.
Our clinical observation suggests that patients in whom therapeutic anticoagulation with warfarin has been difficult, the use of rifabutin may be considered in place of rifampin when the concomitant use of a rifamycin is required.
Introduction
Despite the increased utilization of direct oral anticoagulants, vitamin K antagonists (VKA) remain a therapeutic option for the treatment of thromboembolic disease. Current treatment guidelines recommend VKA as an option for the management of pulmonary embolism (PE) with a target international normalized ratio (INR) of 2 to 3. INR is a blood test used to determine the anticoagulation effects of warfarin. 1 Numerous drug interactions impact both the safety and efficacy of warfarin use. Warfarin is metabolized by many cytochrome P-450 (CYP) enzymes, including CYP2C9, CYP2C19, CYP2D6, CYP2C8, CYP2C18, CYP1A2, and CYP3A4. 2 Inducers and inhibitors of these enzymatic pathways affect the safety and efficacy of warfarin. For example, CYP2C9 is involved in metabolism of the more potent S-enantiomer, and CYP1A2 and CYP3A4 are involved in the metabolism of the R-enantiomer. 2
In reports of the interaction between warfarin and rifampin, high total weekly doses (TWD) of warfarin have been described in order to maintain therapeutic antithrombotic activity, with notable risks of adverse events following discontinuation of rifampin therapy.3 -7 Rifamycin antibacterial agents are known to induce several isoenzymes, including CYP3A4, CYP1A2, CYP2C19, and CYP2C9, all of which may impact warfarin metabolism.8 -10 Rifampin is a highly active, well-utilized and studied therapy in many regimens recommended for active pulmonary tuberculosis (PTB), 11 and is known to have more potent CYP isoenzyme induction than alternative agents in the class, including rifabutin.12,13 The interaction between rifamycins and warfarin is significant and often requires a 2- to 3-fold increase in warfarin dose. 11 Rifampin established adverse effects include hepatotoxicity, renal failure, hemolysis, thrombocytopenia, and hypersensitivity reactions. 14 On the other hand, warfarin is known to be associated with bleeding, skin necrosis, and nephropathy. 15
There is ample literature to support the drug-drug interaction between warfarin and rifampin, but there is a lack of data regarding the extent of interaction between warfarin and rifabutin. The vast majority of literature on rifabutin drug interactions discusses use with antiretrovirals or azole antifungals, but there is little to no mention of warfarin.
To our knowledge, only one case report was identified describing similar induction properties with rifampicin and rifabutin to warfarin therapy in a Japanese patient. 16
Herein, we report a case of a patient receiving warfarin therapy for PE concomitantly with rifampin for treatment of active PTB. We describe a successful clinical intervention whereby the patient achieved therapeutic antithrombotic therapy after switching to an alternative rifamycin antibacterial, rifabutin.
Case Report
A 27-year-old African American female was referred to a pharmacist-managed anticoagulation clinic for treatment of a PE. Her medical history is significant for recent cesarean section complicated by a cesarean wound infection requiring debridement, active PTB, tobacco use, and alcohol abuse. No other disease states were identified. Her PTB medication regimen consisted of rifampin 600 mg twice weekly, isoniazid 900 mg twice weekly, pyrazinamide 3000 mg twice weekly, ethambutol 3200 mg twice weekly. The patient’s PE was initially treated with enoxaparin 70 mg subcutaneously every 12 h, and warfarin 5 mg daily. Warfarin was selected for this patient due to denied insurance coverage for a direct oral anticoagulant agent. Tobacco use was reported to be 4 to 5 cigarettes daily and alcohol intake was reported at 16 to 24 beers per week. During each encounter, the patient denied any changes to tobacco or alcohol use. She also declined tobacco cessation counseling or reduction in alcohol intake.
For treatment of active PTB, the patient was initially started on a daily 4-drug regimen by the local department of health services at the same time treatment with warfarin began. The tuberculosis regimen was subsequently changed to a directly observed twice-weekly regimen after 3 weeks of treatment. During this time, she was receiving rifampin and warfarin. From day 1 of warfarin therapy until day 31, INR ranged from 1.1 to 1.3, which was below the target INR of 2 to 3, despite providing booster doses as well as gradually increasing the TWD to up to 140 mg based on our institutional warfarin dosing adjustment protocol (Figure 1). The patient was being bridged with enoxaparin injections during this time but was beginning to complain of severe injection site pain in conjunction with her recent cesarean section.
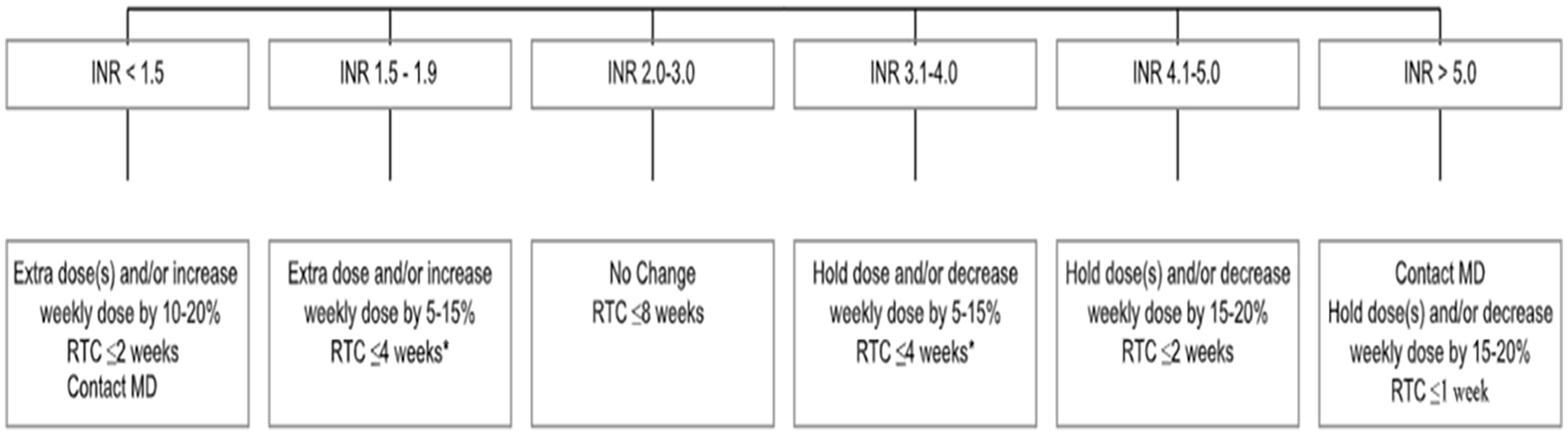
Dosage adjustment protocols for low intensity INR (Goal 2-3).
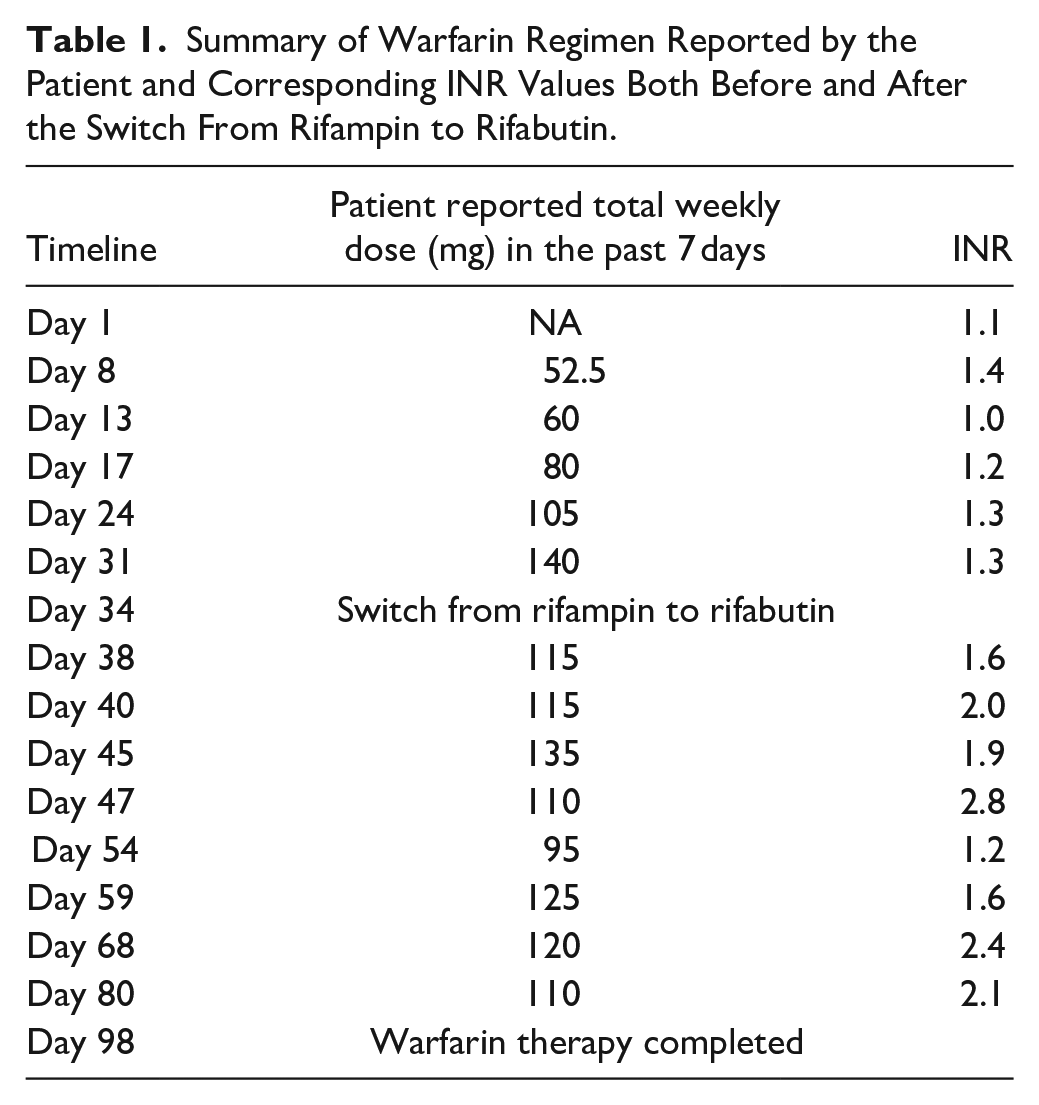
After 1 month of INRs below the target range at a warfarin TWD of 140 mg, rifampin was switched to rifabutin 300 mg twice weekly with the intention of minimizing the interaction with warfarin. Table 1 represents a summary of warfarin regimen reported by the patient and corresponding INR values both before and after the switch from rifampin to rifabutin. Four days after making the switch, the patient’s INR increased to 1.6 while receiving only 115 mg due to missed doses by the patient. In addition, the patient was instructed to resume previous dosing regimen of 140 mg TWD. However, she continued receiving 115 mg resulting in a therapeutic INR of 2.0. Five days later, the patient’s INR was 1.9 on a TWD of 135 mg eliminating the need for extra dosing since INR was close to goal range. She was instructed to continue this dosing and come back in 2 days to re-check INR. Her INR increased to 2.8 while only receiving 110 mg. She was instructed to continue with this dose as her INR remained therapeutic. On day 54, patient’s INR dropped to 1.2 due to only receiving 95 mg during the previous week. Patient was instructed to take an extra dose of 30 mg once. On day 59, the patient dose was adjusted to be 120 mg as a new TWD. A TWD between 120 to 110 mg seemed to be appropriate to maintain INR at goal range from day 68 until day 98 which marked the end of the warfarin therapy. The patient completed 3 months of treatment with warfarin for PE.
Summary of Warfarin Regimen Reported by the Patient and Corresponding INR Values Both Before and After the Switch From Rifampin to Rifabutin.
During this time, the patient was continually assessed and reported no lifestyle modifications in regard to her diet, tobacco use, or alcohol consumption. In addition, no other medication changes were made. These factors were addressed during each patient encounter throughout the course of treatment. Medication adherence was reviewed at each visit by patient report as well as review of prescription refill history. During the first month of therapy, the patient was compliant with the instruction provided, however, adherence issues were encountered during the second month of therapy. TWD was reported based on the amount received by the patient and not the amount prescribed by the provider.
Therefore, switching the patient from rifampin to rifabutin is the most likely explanation that a target INR was achieved with an 18% decreased TWD of warfarin. Figure 2 represents a summary of warfarin regimen and INR values.

Summary of warfarin regimen and INR values. Rifampin was switched to rifabutin on Day 34.
Discussion
Warfarin is a substrate for many cytochrome P450 isoenzymes, 2 which is the primary reason for the large number of drug-drug interactions with warfarin. Of the rifamycins, rifampin has the most profound effect on warfarin metabolism, followed by rifapentine, and lastly rifabutin. 17 Rifampin is an inducer of many isoenzymes, and specifically a strong inducer of CYP1A2, CYP2C19, CYP2C9, and CYP3A4, which directly affect warfarin metabolism. 8 It is also a minor substrate of CYP1A2 and CYP2C19, and a major substrate of CYP2C9 and CYP3A4. 8 Studies have reported a 50% to 80% decrease in warfarin serum concentrations when used with rifampin. 18 In a warfarin-stable patient, the maximum effect of the interaction occurs within 5 to 10 days,5,17 several weeks are required for the interaction to resolve upon discontinuation of rifampin.3,5,17,18 As a result, the dose of warfarin is typically doubled empirically when rifampin is added to a patient’s regimen.5,11,18,19 In the case reported here, the patient was receiving doses of warfarin up to 20 mg daily while also taking rifampin, and only achieved a maximum INR of 1.3. These results are consistent with numerous published case reports of patients requiring up to 30 mg/day of warfarin when also taking rifampin.3 -7
In contrast, rifabutin is a strong inducer of only one isoenzyme that affects warfarin metabolism, CYP3A4. 9 It is also a substrate of this same isoenzyme, so chronic administration results in auto-induction of rifabutin,12,16 but to a lesser extent than with rifampin. 12 One study reported that rifabutin resulted in 29% drug clearance compared with 90% drug clearance with rifampin. 12 Another pharmacokinetic study reported that the maximally achievable induction of CYP3A4 with rifabutin is 2-fold lower than with rifampin. 13 Rifabutin is more extensively distributed to the tissues than rifampin due to its higher lipid solubility, resulting in a higher volume of distribution. 12 It also has a long elimination half-life of 45 h. 9 These factors are thought to contribute to the delayed and less severe interaction seen with rifabutin and warfarin.
While there is a wealth of information regarding the interaction between rifampin and warfarin, there is a paucity of literature describing the interaction between rifabutin and warfarin. One case report was published in 2011 for a 40-year-old woman who was on warfarin therapy for deep vein thrombosis with an INR maintained between 2 and 3 at a dose of 3 mg daily (TWD of 21 mg). The patient was diagnosed with atypical mycobacteriosis, and was started on rifampicin, clarithromycin and ethambutol. The patient’s INR markedly decreased within 9 days after initiating anti-mycobacterium drugs, suggesting rifampicin-induced warfarin metabolism. The patient was switched to rifabutin since the drug-drug interaction is considered of to be less severe than with rifampicin. In addition, the patient’s warfarin dose was increased to 5.5 mg daily (TWD of 38.5 mg); however, that did not result in a therapeutic INR. The authors concluded that rifabutin exerts an induction effect on warfarin metabolism. This case report finding contradicts our case finding of attaining successful therapeutic anticoagulation with rifabutin. Several reasons might explain the opposing conclusions. First, the increase in the TWD may have been inadequate. This patient might have achieved a therapeutic INR with more aggressive increases in TWD. Another reason might be the time allowed to reach therapeutic INR range. Unfortunately, we were not able to attain all specific information regarding timing and doses as the original manuscript was written in Japanese, so this information was derived from the abstract only. 18
Tuberculosis and PE both require several months of treatment,1,11 so finding alternative regimens that allow both conditions to be appropriately treated is ideal. In the vast majority of case reports published describing warfarin and rifampin, patients rarely achieve an INR within the desired range, despite significant increases in warfarin dosages.3 -7 By using rifabutin as an alternative to rifampin, therapeutic anticoagulation was able to be achieved while also appropriately treating tuberculosis. In our patient case, an INR within the desired range was achieved with a reduction in TWD of warfarin by 18% from 140 to 115 mg when rifampin was switched to rifabutin. We acknowledge that the presented case findings should be interpreted in the context of few limitations. First, the patient exhibited non-compliance issues and providers instructions were not followed appropriately. To account for this limitation, dosing was reported based on what the patient received rather than what was instructed to be taken. This provides better representation of the effects on the INR. In addition, patient also had a recent pregnancy and cesarean section surgery, this may have affected the physiological responses to warfarin. To our knowledge, this is the first published case report of a successful rifabutin use as an alternative to rifampin in a patient taking warfarin.
Conclusion
The interaction between rifampin and warfarin is well known but is under-appreciated in clinical practice due to the relatively infrequent concomitant use of these medications. Rifabutin has less impact on the metabolism of warfarin than the other rifamycins, although there is little evidence in the literature supporting the use of rifabutin over rifampin in patients also taking warfarin. Successful anticoagulation with warfarin was achieved in this patient after switching her tuberculosis treatment from rifampin to rifabutin. Target INR values were attained with lower TWDs of warfarin when used with rifabutin than is typically required with rifampin. In addition, the patient was able to discontinue bridge therapy with enoxaparin injections earlier by achieving a target INR sooner. For patients in whom therapeutic anticoagulation with warfarin has been difficult, the use of rifabutin may be considered in place of rifampin when the concomitant use of a rifamycin is required.
Key Points
A 27-year-old female experienced subtherapeutic INRs for over 1-month despite incremental dosage increases in warfarin while concomitantly taking rifamycin.
Rifampin is an inducer of many isoenzymes (CYP1A2, CYP2C19, CYP2C9, and CYP3A4) which may affect warfarin metabolism, whereas rifabutin is a strong inducer of only one isoenzyme that affects warfarin metabolism, CYP3A4.
For patients in whom therapeutic anticoagulation with warfarin has been difficult, the use of rifabutin may be considered in place of rifampin when the concomitant use of a rifamycin is required.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
An ethical board approval and patient informed consent were not required for our study. We present a case report and describe only clinical observations without involvement of any experimental interventions.
