Abstract
Introduction:
Direct to consumer stem cell and regenerative interventions (SCRIs) for various medical conditions have increased in popularity due to unmet medical needs and the promise of SCRIs to meet those needs. These interventions may have varying levels of safety and efficacy data and many lack sufficient scientific data to be marketed. The direct to consumer SCRI industry has received significant attention due to potential physical, economic, and emotional harms to patients. Patients may seek the counsel of their primary care providers when considering stem cell therapy for their condition.
Methods:
Here we describe strategies primary care providers can utilize when counseling patients.
Results:
Although we recommend constructing these discussions around individual patients’ needs, one can utilize a general approach consisting of 4 parts. First, providers should recognize what information the patient is seeking and what is their understanding of stem cell and regenerative medicine. Next, providers should convey evidence-based information at the level of patients understanding so that they are aware of the risks, benefits, and descriptions of possible procedures. Throughout the conversations, attempts should be made to guide patients to a trusted resource that can provide additional information. Finally, providers should make an effort to address misinformation in a way that is nonjudgmental and patient-centered to make the patient feel safe and comfortable.
Conclusion:
Effectively communicating risk information by primary care providers to patients is important given the harms reported from direct-to-consumer SCRIs. Correcting misinformation remains a priority when discussing SCRI’s. Providers should strive to offer patients with additional resources such as the opportunity for consultation with a specialist or a consultation service dedicated to informing patients about regenerative medicine.
Introduction
Direct to consumer stem cell and regenerative interventions (SCRIs) for various medical conditions have increased in popularity due to unmet medical needs and the promise of SCRIs to meet those needs. Though predominantly still in a translational research stage, early success stories have catapulted regenerative medicine into a fast growing medical specialty, with potential to advance care for chronic conditions.1-8 As with many novel technologies, great promise may bring even more hype. One platform driving that hype is the misinformation about stem cell therapies that have recently permeated traditional areas of medicine such that patients are actively seeking them and speaking with their physicians.9,10
Social science studies suggest that the general public considers stem cells as having the power to heal a variety of medical conditions, resulting in growing demand and subsequent emergence of a direct-to-consumer market where characteristically unspecialized and even predatory clinics offer unproven regenerative labeled therapies such as stem cells and more recently exosomes.11-14 The term “unproven” is used to mean that there is varying levels of scientific evidence to demonstrate safety and efficacy of a SCRI but that many still lack sufficient scientific data to be marketed.
Stem Cell Seeking Patients
We have previously published data about individuals seeking stem cells and regenerative interventions (SCRIs), who are generally older with various chronic medical conditions having previously explored conventional treatment options or parents of ill children seeking potential treatments. 9 While orthopedic conditions are a common area of interest for individuals seeking SCRI’s, other medical conditions in the specialities of cardiology, neurology, and pulmonary have also been shown to be areas of interest. 9
Patients considering SCRIs are often confronted with large amounts of misinformation, much of which is unbalanced, inaccurate, or misleading.10,15-20 It remains unclear how patients and families differentiate between misinformation and evidence-based literature.9,21 While potential patients may undertake research about SCRIs, including online searches, visiting clinics, or by attending stem cell seminars, 9 some may remain uncertain or apprehensive regarding such treatment options. Several factors can influence a patient’s decision to seek experimental SCRIs. These may include personal beliefs, risk perception, illness severity, thoughts of vulnerability or fear, mistrust toward conventional health care and research, and interpersonal interactions with people including experts.9,22-24
When considering a regenerative therapy or clinical trial, it is imperative that patients and caregivers are provided with an evidence-based discussion about the procedure including risks, benefits and any associated costs.23,24 This should largely be done by disease specialists with expertise in the conditions that patients are seeking assistance with. Nevertheless, it can be a challenge for patients, their families, and their primary medical providers to know where to find such disease specific expertise. Deciding how best to manage patients’ informational needs, especially given the rapid changes in the science of regenerative therapies, remains a significant challenge.
Advances in Regenerative Medicine
Some notable successes in the field of regenerative medicine include autologous epidermal grafts for epidermolysis bullosa, skin grafts for acute burn injuries and CAR-T immunotherapies for various hematologic malignancies.25-30 Point of care, autologous cell-based therapies, termed orthobiologics, have shown some benefit for orthopedic conditions, such as bone marrow aspirate concentrate for avascular necrosis of the femoral head during hip decompression surgery and platelet rich plasma for management of knee pain secondary to osteoarthritis.31,32 Additionally, platelet rich plasma is commonly utilized by cosmetic and dermatologic practices for treatment of androgenic alopecia and some skin conditions.33,34 Despite these milestones and ongoing research efforts, there remains a direct-to-consumer market for unproven stem cell interventions, most notably in the United States.12,15,35
Direct-to-Consumer Market
The direct-to-consumer SCRI industry has received significant attention due to potential physical, economic, and emotional harms to patients. Many of these experimental interventions are offered outside of controlled clinical studies and are sold directly to patients. 36 These interventions may lack scientific evidence, product characterization as well as safety and efficacy data. It is estimated that there are over 2700 clinics in the United States offering SCRI. 12 While over 60% comprise musculoskeletal indications, all medical specialties are represented to some degree.37,38 The uncertainty of the indications, nature of products being used, safety and qualification of providers performing some interventions creates a need for patients seeking regenerative solutions to search for help from trusted resources. The resulting environment has led to significant complications due to illicit use of stem cell therapies by providers operating outside their area of expertise.14,39-44
Counseling Patients
Many scientific and medical institutions have gone to great lengths to inform patients about possible harms in undertaking unproven stem cell and regenerative interventions, including the International Society of Stem Cell Research, the Food and Drug Administration (FDA) and Stem Cells Australia amongst others.14,45-51 Despite these efforts, it remains unclear whether such messaging effectively influences patients and if patients regard these sources reputable and believable. 22
We have launched a consult service and have engaged with over 6500 patients seeking regenerative care. This service provides aims to provide clinical navigation for patients interested in regenerative care and direct patients to scientifically based options of care. If no such options are available, patients are provided with education on the state of stem cell research for their condition. 52 In our previous assessment of patient satisfaction, the majority of patients found the consult service to be helpful and informative, many expressing gratitude for the information shared. 9
From our experience, we can translate lessons learned to other medical specialties and providers. Primary care physicians are a highly trusted source of medical information to their patients53-55 and can likely provide patients with similar counseling. Studies show patients will trust their primary care physician and feel empowered when provided counsel from their primary care provider.56,57 Though not always experienced in SCRIs, primary care providers will commonly have subspecialty relationships connecting them to the emerging regenerative field.24,58
Recognizing the Difficulty of Consultations Around SCRI
We’ve previously published challenges that physicians have described when discussing unproven stem cell interventions with patients. 21 These included tensions between supporting a patient’s decision to undergo such treatment and being worried about harms that could befall their patients, addressing family pressure and stem cell hype and lack of patient understanding about SCRIs, responding to high expectations or demands from patients, and patients viewing conventional medicine with distrust. In a time when physicians may be increasingly asked to respond to health misinformation and counsel on interventions lacking scientific data, these strategies may be of interest to other physicians responding to patient questions about unproven SCRIs.

In order to help patients navigate this confusing space of misinformation, providers can communicate evidence-based information to patients about the unproven stem cell and regenerative industry while also recognizing that each patient’s has a unique set of circumstances. Theories of evidenced based medical counseling acknowledge patient exposure to misinformation, while providing appropriate sources of information and encouraging shared decision making between patients and providers. 64 Although we recommend constructing these discussions around individual patients’ needs, the authors recommend utilizing a general approach consisting of 4 parts.
First, providers should recognize what information the patient is seeking and assess their understanding of stem cell and regenerative medicine. Next, providers should convey evidence-based information so that they are aware of the risks, benefits, and descriptions of possible procedures. This should be communicated at a level appropriate to the patient and teach-back techniques can be used to establish what the patient has learned from the discussion. Throughout the conversations, attempts should be made to guide patients to a trusted resource that can provide additional information. Finally, providers should try to address misinformation in a way that is nonjudgmental and patient-centered to make the patient feel safe and comfortable.59,63 These 4 parts can best be summarized as the “Four E’s,” see figure below.
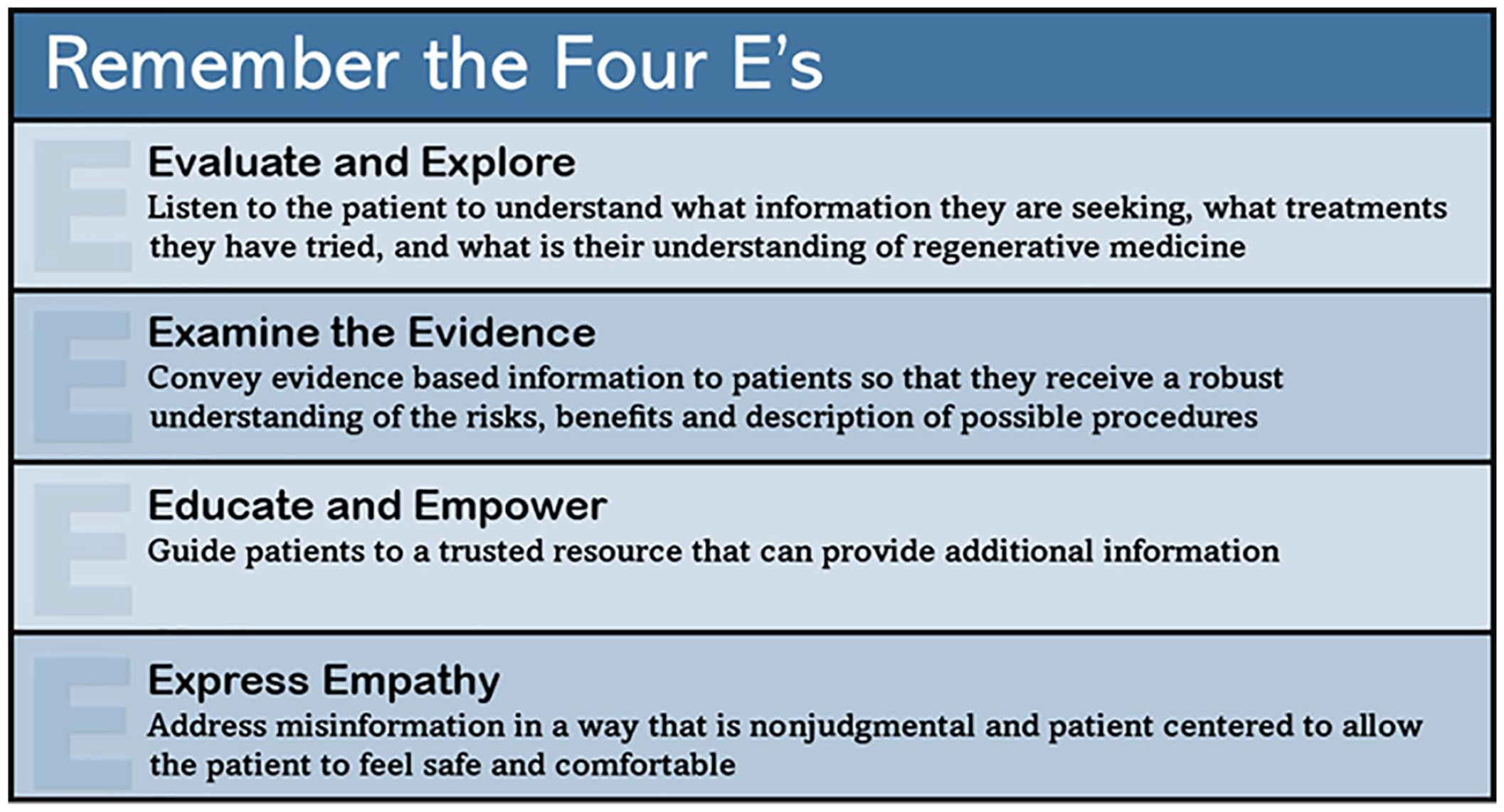
Conclusion
Primary care provider recommendations and strategies for patient engagement surrounding SCRIs can help contextualize the messaging patients receive. Effectively communicating risk information by primary care providers to patients is important given the harms reported from direct to consumer SCRIs.
Correcting misinformation remains a priority when discussing SCRI’s. We acknowledge that more research is needed as to the effectiveness of patient eduction in not only in correcting misinformation and sharing knowledge, but also possibly changing health behavior.
Providers should strive to offer patients with additional resources such as the opportunity for consultation with a specialist or a consultation service dedicated to informing patients about regenerative medicine.
Scientific organizations and academic institutions can help disseminate information to primary care providers to help them better understand the experimental SCI space and identify resources for patients.61,62
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge support for this work from the NIH/National Institute on Aging (R21AG068620) and Mayo Clinic’s Center for Regenerative Medicine.
