Abstract
Introduction
Under the Affordable Care Act (ACA), Medicare beneficiaries receive free coverage for Annual Wellness Visits (AWVs), which is a yearly office visit where the beneficiary can discuss the plan of preventive care for the coming year. During a visit, patients are evaluated with a Health Risk Assessment questionnaire, and may receive health advice, routine measurements, screening schedules, advance care planning, and other tasks related to prevention.
Prior research has found that the use of AWVs rose from about 7% (3.2 million visits) in 2011 to 20% (10.4 million visits) in 2016. 1 The utilization rates in 2016 vary from about 7% in Hawaii to 33% in Rhode Island. In 2014, The AWV utilization rates in non-Hispanic whites were 45% and 88% percent higher, respectively, than African Americans and Hispanics, and the rates in metropolitan residents were about 60% higher than rural residents. Medicare has reported that it spent about $1.2 billion on AWVs in 2016. 2
Few studies have examined the effectiveness of AWVs. Some researchers have argued that there is little benefit from annual examinations such as a physical examination, 3 but which differ from an AWV in that AWVs are not limited to physical health issues, and are instead guided by US Preventive Services Task Force (USPSTF) recommended preventive services. Others who have focused specifically on AWVs have found support for personalized prevention visits for all beneficiaries.4-6 This study examines whether AWVs result in more recommended preventive care, on average and in racial/ethnic minorities and rural residents in Medicare fee-for-service (FFS) beneficiaries after Centers for Medicare and Medicaid Services adoption of ACA policies in 2011.
Study Data and Methods
We examined physician and outpatient claims of 2011-2014 for a sample drawn from approximately 3.18 million beneficiaries enrolled in Medicare 5% FFS, based on health care visit histories. To parse the effect of the AWV on follow-up screening from competing health care visit patterns, we were concerned that the follow-up rates may have been influenced by other previous visits or screening episodes (“S/P”) and which may have occurred sufficiently close to and prior to the wellness visit. Furthermore, the likelihood of a wellness visit may be influenced by the presence of these other visits or episodes, resulting in confounding bias. Thus to reduce bias, we conducted a stratified analysis by implementing a lookback period on patients with wellness visits (set at 90 days) to scan for the presence of S/P. Beneficiaries were classified into 3 groups: (1) those with initial/subsequent (G0438, G0439) wellness visit without indication of S/P during lookback period of 90 days, (2) those with wellness visits after S/P occurring during 90 days prior, and (3) control group with random reference date without S/P during 90 days prior or wellness visit at any time (Figure 1). All beneficiaries were required to have at least 1 year FFS enrollment prior and post reference date with no indication of additional wellness visit during 365-day follow-up. For each candidate control, the reference date was selected at random among the available candidate months in order to diminish temporal and seasonal biases.

Five percent Centers for Medicare and Medicaid Services (CMS) sample selection.
Common screening event codes covered by Medicare during 2012-2013, including DTaP (diphtheria, tetanus, and acellular pertussis)/DTP/Td/influenza/pneumonia vaccinations, screening for cardiovascular disease, colorectal cancer, female breast cancer, prostate cancer, diabetes, and bone mass were then scanned in the Medicare physician services and outpatient files during 1 year after but not including reference date, with the objective of assessing the effect of wellness visit group on overall and individual screening rates. The HCPCS (Healthcare Common Procedure Coding System) codes used to identify screening and preventive services are shown in the appendix; primary sources used to select the codes are described in the notes.
Because of the observational study design, rates may differ due to the effect of other confounders and not exclusively due to the group membership, resulting in biased comparisons. In order to correct for this type of bias, we weighed the sample using inverse probability weights for multiple groups (IPTW). 7 IPTW is comparable to propensity scoring but, by using suitable IPTW weights, the weighted sample among the 3 groups should have similar distributions of confounders, enabling a fair comparison between groups provided there is no hidden imbalance. A multinomial logistic regression was fit to the data in order to calculate the group membership probabilities needed for the weights. The covariates list used in the model included all possible 2-way interactions between the covariates in Figure 2, excluding the comorbidities which were entered as main effects only. The quality of the IPTW weights was strengthened by the use of rescaling factor in order to reduce the presence of excessively large weights and their influence. Furthermore, the covariates balance among the groups was evaluated by assessing standardized differences scores. Confounders considered as predictors of membership and outcome consisted of beneficiary race (white, black, other), gender, age category at reference, Charlson comorbidity score (NCI Kablunde 2000 version 8 ), semester of reference date (2012-2013), National County Economic Distress from Appalachian Regional Commission 9 (2013 version), and NCHS rural urban 10 status from the Centers for Disease Control and Prevention. In addition, the broader set of individual comorbidity groups (n = 18) of the Charlson score were entered into the model as separate indicators in order to achieve balance by individual morbidities. Finally, accessibility to primary care at patient’s county centroid was also included as a potential confounder. This index was based on a 2-step floating catchment area method, 11 where the number of 2013 primary care providers at each county was extracted from the Area Health Resource File. 12

Unweighted covariate distributions.
As a secondary analysis, cumulative screening incidences for overall and individual screening rates were then compared also across groups using Kaplan-Meier failure curves. In addition, we performed subgroup analysis by economic distress, rurality, race, age, and Charlson index category in order to assess possible effect heterogeneity within disparity strata and discover potential differences which may contribute to literature relating wellness visits to disparities
Since covariate distributions by strata were not likely to be similar, we further assessed balance for each subgroup, adjusting for covariates with largest imbalances if needed. To assess differences between initial and subsequent wellness, a separate IPTW analysis was conducted comparing patients who were assigned to their initial wellness visit (G0438) vs patients who were assigned to a subsequent wellness visit (G0439). Next, in order to examine the effect of provider wellness visit volume on screening, we linked wellness visit providers after 2012 to wellness visit volumes from their Medicare Provider Utilization and Payment Data (MPUPD) and then conducted an IPTW analysis comparing beneficiary screening rates across provider volume quartiles. Finally, we conducted separate analysis with varying look-back period in order to assess the sensitivity of the results as a function of look-back period.
Limitations that the IPTW method may fail to address include the potential of hidden latent confounders which may result in a spurious association. These would include potential drivers of AVW use and preventive screening use which have not been controlled for. Sample limitations may affect the generalizability of the results. The beneficiaries in this sample are older Medicare beneficiaries who follow the distributions in Figure 2, which may not be representative of the national population or a cohort of interest. Furthermore, the design of the study required fee for service Medicare restriction, which excludes effect of AWV under other types of plans and scenarios.
Results
A total of 52 300 beneficiaries were included in the wellness visit only group (W), 20 850 were included in the wellness visit with prior S/P group (P + W), and 586 000 were included in the control group (Figure 1). The majority of patients resided in middle ‘transitional” economic status counties (60%), lived in a metropolitan area (66%), were white (83%), slightly more female (53%), had a mean age of 70 years (SD = 13.1) with 19% age group ≤64 years, had no Charlson index defined comorbidities (68%), and had a most frequent comorbidity of diabetes (17%). Percentages varied between groups (Figure 2), with noticeable differences being control age category ≤64 years (22% vs 11% other groups combined), and P + W group having more comorbidities (57% with 0 comorbidities vs 72%) and having more diabetes (24% vs 15%). After weighting the sample to match the control distribution, the maximum standardized difference balance diagnostic dropped from 0.35 to 0.02, resulting in relatively close rates between groups (Figure 3).
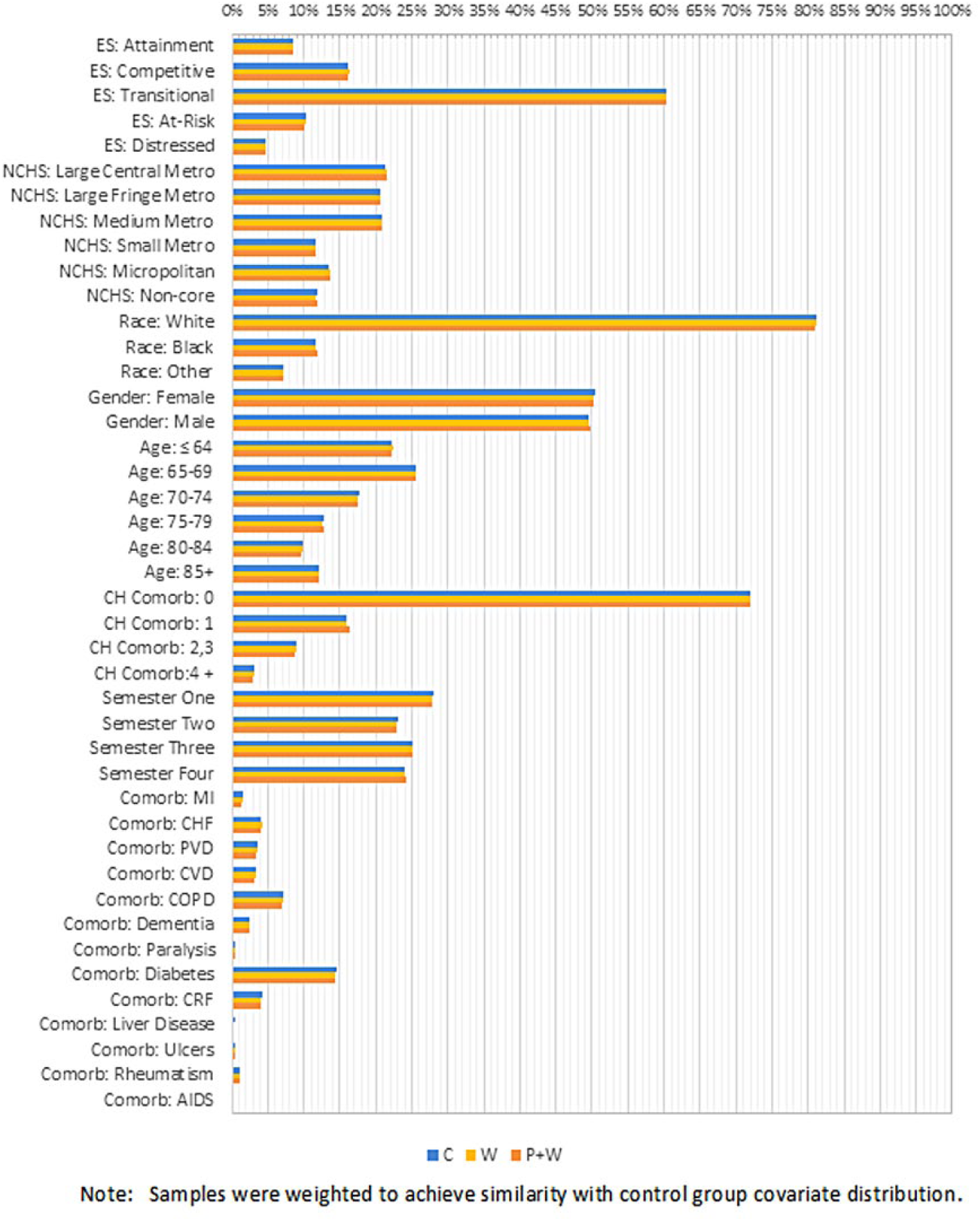
Weighted covariate distributions.
Screening rates after follow-up for the control group were significantly lower compared with the other 2 groups (Figure 4), with a mean of 1.19 different type of screening events (out of 7) compared to 1.68 in the wellness group and 1.99 in the P+W group. The confidence intervals, illustrated as error bars in Figure 4, suggest the comparisons are highly significant (ie, P < .01), which is to be expected due to the large sample sizes. Additionally, control rates were significantly lower for all screening event types (Figure 4). Overall, 63% of the control group had any one of the 7 screening events compared with 82% and 89% for the other 2 groups.

Screening events during 1-year follow-up.
Subgroup analysis (Figure 5) showed the same difference and ordering of effects persisted in each subgroup examined, with some slight variations. A test of parallel trends across subgroups using statistical interaction tests rejected the hypothesis of parallel trends for all groups; this would be expected given the large sample sizes where even trivial differences may be significant. Some of the differences, however, are potentially interesting; for example, when comparing by Area Economic Distress, the control group (C) decreased from 1.27 to 1.09 with increasing levels of distress, whereas the wellness group (W) increased from 1.66 to 1.70, and the P + W group decreased from 2.04 to 1.96. This would suggest the wellness visits are serving to maintain screening rates in distressed areas compared to gradual decrease in controls.

Screening events by subgroups.
The cumulative incidence curves (Figure 6) show the rates by time of follow-up, with rates at the end of one year matching up to the rates in Figure 2. Wellness and preventive screening were consistently higher than the control group in all the charts. A visual classification of trends suggests 3 groups: those with ramp-up period after wellness visits (overall, colorectal cancer, bone mass, breast cancer), those without the ramp (vaccinations, diabetes), and an atypical pattern (CDS - cardiovascular disease screening, prostate). Although there are time periods in which the P + W groups experienced lower rates than the W group, ultimately, the P+W displays higher cumulative rates.
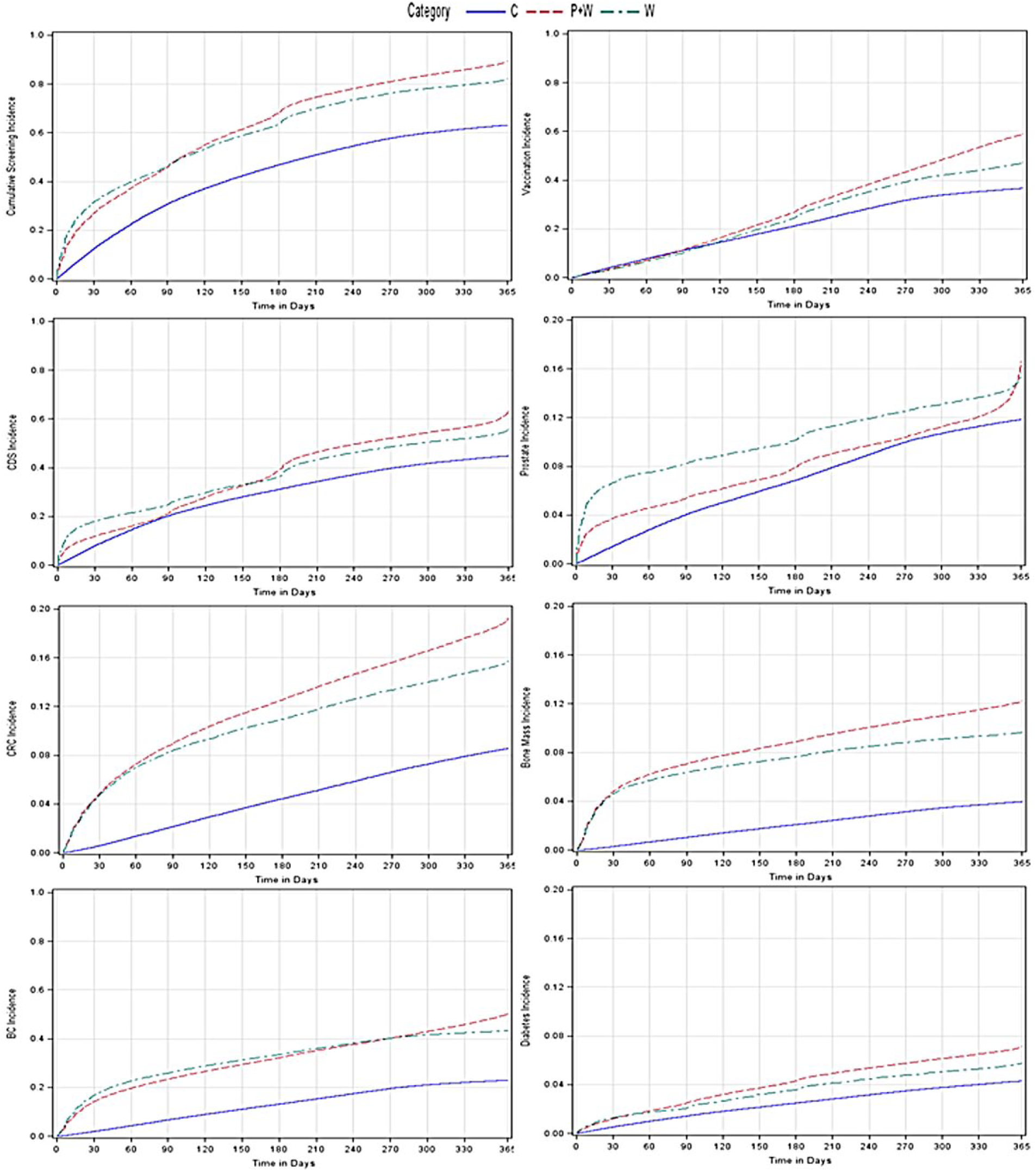
Cumulative incidence curves.
The comparison between Initial Wellness (G0438) versus Subsequent Wellness groups (G0439), conducted only in the wellness subgroups, standardizing to the covariate distribution of the initial G0438 wellness group, and adding the subgroup type in the predictive model to get IPTW weights, resulted in statistically indistinguishable rates (2.10 for both groups, P = .8698). The comparison of provider volume quartiles in subgroup with available volume information suggests an increase from 2.06 in the first quartile to 2.09 in the last quartile, with a difference of 0.03. This suggests increased volume associates with increased chance of screening, but relative increase is small.
To analyze the effect of varying lookback period, we extended from 90 days to 180 days and then 365 days. The sample sizes and covariate distributions of the groups change since increasing look-back restricts the pool of available observations to those of beneficiaries in the control (C) and wellness (W) group who did not experience any S/P during greater time periods. Without providing additional details, significant overall effects were still detected for the 180 days period (0.78 in controls vs 1.40 in W vs 1.89 in P + W) and the 365 days period (0.42 in controls vs 1.08 in W vs 1.80 in P + W). We attribute the decreasing rates to conducting the analysis on samples that are less likely to experience preventive visit and screening episodes.
Discussion
Based on these results, screening rates are projected to significantly increase compared with control group for patients who have not received prior preventive care or screening episodes before AWV, either during the prior year or within a smaller time frame. That is, we find evidence in the study sample that AWV are modestly effective in increasing access to preventive services, including cancer screening among those not previously served. Ganjuli et al 1 have noted that early adoption of AWVs has been concentrated in Accountable Care Organizations and certain primary care physicians, suggesting patients who receive AWVs may tend to receive care from organizations and providers vested and motivated in preventive health practices. The fact that these same patients had higher receipt of preventive services within, presumably, the same organization or practice type, could suggest that the association between AWV and screening may be partially affected by increased prevention awareness leading to a policy of reimbursing for AWVs in addition to increased delivery of preventive services. Our findings reflect continuous enrollment in FFS during the study period, and thus do not capture results from managed care plans that may have been more prevention focused, so the association may be understated.
Patient-centric factors also play a role in determining choice of AWVs and screening practices. Findings here are consistent with AWVs being more likely to occur in metropolitan regions and regions with higher socioeconomic indices, along with individual characteristics such as white ethnicity, female gender, and older age groups. Taken together, the association of AWVs on receipt of preventive services observed in these results may be a function of genuine effect of AWV on screening, and potentially, patient factors which we sought to control thru IPTW analysis, as well as to a lesser degree (we hypothesize) unseen practice-driven factors that may affect both AWV and screening due to receiving care from organizations/ providers with a strong preventive care management and culture.
For the group of patients who received preventive services during the past year, AWV appears to likewise show a marked increase over control group, and are projected to experience higher screening rates than the AWV group, with disparity increasing as look-back period increases. The impact of AWV, however, may be reduced due to the influence of other preventive care/screening episodes during the past period on future period and presence of latent proactive factors in this group which may be contributing to the increase in rates. We surmise the hypothetical removal of AWV in this group may results in diminished rates but still be well above control rates.
Perhaps most important, if the strong associations observed here are accepted as fully or mostly genuine effects of AWV, AWVs may then be an important policy tool to reduce disparities in preventive services, such as cancer screenings and vaccinations, especially in disadvantaged groups which tend to lack prior preventive services. These groups include rural, low socioeconomic status, males, and African Americans populations. Policies to increase AWV penetration may involve increasing awareness of these visits with community health workers, proposing financial incentives, and increasing the willingness to schedule for these visits. Furthermore, AWVs may increase utilization of screenings and preventive services, not only because they raise awareness and motivate patients but also possibly because they motivate providers and raise their investment into their patients’ care. Thus, AWVs may also strengthen the therapeutic relationship between the patient and the provider, which in itself improves quality and outcomes of patient care.
Given the size of the screening effects found in this study together with the still sizeable underutilization of free AWV documented in other studies, 1 policies of no-cost access to AWVs, and their promotion to patient and practices could result in gains in early disease detection and improved health outcomes in the population through increased delivery of preventive services. At the very least, if increases in screening rates and disease prevention are desirable health care objectives, continuing a healthcare coverage of no-cost AWV begun by Affordable Care Act is highly warranted. In summary, given the benefits of AVW, there is a need for future research to determine effective community or practice-based interventions that increase use of AVWs in disadvantaged populations or patient groups.
Footnotes
Appendix
Table of HCPCS Codes to identify Screening and Preventive Services.

| Other Preventive Visits: • Office E/M: 99201-99205,99211-99215 • Office Consultations: 99241-99245 • Preventive Medicine Services: 99381-99387, 99391-99397,99401-99404,99420-99429,99450 • Special E&M services: 99455-99456,99499 |
| Other Wellness Visits: • G0402-G0405,G0101,Q0091 |
| Vaccinations: • Influenza: 90630,90653-90657,90661,90662,90672-90674,90685090688,Q2035-Q2039,G0008 • Pneumococcal: 90732,90670,90714,90715,90736,90746,G0009 • DTaP/DTP/Td:90698,90700-90703,90714,90715,90718,90719,90720,90721,90722,90723 |
| Screening Events: • Cardiovascular Disease Screening (CDS): 80061,82465,83718,84478,G0446 • Colorectal Cancer Screening (CRC): G0104-G0106,G0120-G0122,G0464,G0328,00810,74263, 74270,74280,81528,82270,82272,82274,44401-44408,45330-45350,45378-45398,45330-45335, 45337-45342, 45345-45347, 45349-45350, 44388-44394, 44397, 45355, 45378, 45393, 45398 • Breast Cancer Screening: 77057,G0202,G0203,77052,77063,77092,76083,76376,76377,76645 • Prostate Cancer Screening: G0102,G0103 • Diabetes Screening: 82947,82950,82951,G0108,G0109 • Bone Mass Screening:76977,77078,77080,77081,G0130 |
Notes:
• The source for “Other Preventive Visits” codes was the AMA CPT 2016 Manual.
• Primary source for vaccinations was from the CDC Immunization Information Systems (IIS): (https://www2a.cdc.gov/vaccines/iis/iisstandards/vaccines.asp)
• Primary source for other screening events was from the Medicare Learning Network Preventive Services Chart. (https://www.cms.gov/Medicare/Prevention/PrevntionGenInfo/medicare-preventive-services/MPS-QuickReferenceChart-1.html)
• CRC screening codes were supplemented with additional codes from KP Hedis Insight (https://provider.ghc.org/open/providerCommunications/hedisInsight/index.jhtml)
• Breast cancer screening codes were supplemented with internal expert review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
