Abstract
Keywords
Introduction
Fecal occult blood tests (FOBTs) are used to detect small amounts of blood in the stool, which can indicate the presence of advanced adenomatous polyps (AAP) or colorectal cancer (CRC).1,2 Two types of FOBTs are available—guaiac (gFOBT) and fecal immunochemical tests (FITs). There is general agreement that FITs have better test characteristics and are easier for patients to use compared with gFOBTs.1,3-7 However, the many brands of FIT on the market and limited information on test characteristics for the outcomes of AAP or CRC make it difficult to decide which FIT to choose.
The gFOBT was developed in the 1860s for CRC screening.8,9 FITs were developed in the 1970s and use antibodies directed against the globin component of hemoglobin to detect blood in the stool.9-11 The US Preventive Services Task Force, the American College of Gastroenterology, and the American Cancer Society promote the use of FIT over gFOBT in CRC screening.12-15 FITs are preferred by patients over colonoscopy3,4 and there are no dietary or medicine restrictions, unlike with the gFOBT.1,5,6 Modeling studies indicate there is no difference in life-years gained when comparing a CRC screening strategy of annual FIT testing with a colonoscopy every 10 years. 16 FITs use 2 immunoassay techniques: an automated laboratory instrument-based immunoturbidometric test and a lateral flow immunochromatographic analysis for point-of-care testing. 1
There are numerous FIT products available. The purposes of this research were to summarize: (1) the FITs currently available in the United States based on the Food and Drug Administration (FDA) and product websites, (2) the 2014 pathology proficiency testing (PT) program results regarding FITs, and (3) the literature related to the test characteristics of FITs available in the United States to detect AAP and/or CRC.
Background
FDA Website and FOBT Approval via the 510(k) Process
The US Department of Health and Human Services FDA website has a section for medical devices, the Clinical Laboratory Improvement Amendments (CLIA) database, that lists the commercially marketed test systems catalogued by the FDA since 2000, as well as those catalogued by the Centers for Disease Control and Prevention prior to that time (www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfclia/search.cfm). This database lists FOBTs that have been cleared by the FDA and associated documentation.
FDA FOBT Test Complexity
CLIA legislation passed by Congress in 1988 was established to ensure accuracy, reliability, and timeliness of patient test results regardless of where the test was performed. 17 FOBTs, both waived and moderate complexity, are classified as class II devices. Waived tests are “simple laboratory examinations and procedures that have an insignificant risk of an erroneous result.” 17 To perform waived tests, the healthcare provider needs to have a CLIA Certificate of Waiver. Moderate complexity tests have to be analyzed in a certified pathology laboratory. Class indicates the intended use of the device and the risk the device poses to a patient with class I being the least risk and class III the greatest risk. For class II tests, a 510(k) application to the FDA is required for clearance and then marketing.
All certified pathology laboratories are required to participate in a PT program for all analytes they test for. The Centers for Medicare & Medicaid Services (CMS), through CLIA, regulates approximately 252 000 laboratory entities where FOBTs are analyzed. 18
FDA Document Number
The FDA requires a 510(k) application to be submitted prior to marketing and selling of FOBTs. The 510(k) must demonstrate the FOBT is substantially equivalent to a device already legally marketed. An application submitter must compare their FOBT to one or more similarly marketed devices to support their substantial equivalency claims. The FDA then determines if the data provided supports the claim of substantial equivalence and categorizes the FOBT as a new device, substantially equivalent for marketing in the United States, or as a device modification. Listed on the 510(k) are the predicate device names and numbers, which indicate the device is safe and effective/substantially equivalent to its predicate. Contrary to what one might see on company websites, the FDA does not “approve” any FOBTs, but provides clearance after review of a 510(k) application.
Methods
The University of Iowa Institutional Review Board reviewed this project and determined it was not human subjects research. Data were collected and summarized from the FDA CLIA database, all CMS-approved PT programs in the United States for the calendar year 2014, and a detailed, literature review on FIT characteristics published in the English language where colonoscopy was used as the gold standard.
FDA CLIA Database
The CLIA database was searched and 122 FOBTs were listed as of July 22, 2016. For each FOBT, a search of the Internet using the name of the product and manufacturer, guaiac, fecal immunochemical, and fecal occult blood test was conducted to determine whether the product was a gFOBT or a FIT. If the product could not be found on the Internet, a search of PubMed was conducted.
Proficiency Testing Programs
Pathology laboratories participate in PT programs to ensure that procedures for analyte testing give accurate results. Currently, there are 7 PT programs in the United States that distribute samples for FOBTs. 19 PT program 2014 data were obtained from the programs’ Internet posting or customer service. The programs included (1) American Academy of Family Physicians Proficiency Testing (AAFP-PT), (2) American Association of Bioanalysts (AAB) Proficiency Testing Service, (3) American College of Physicians (ACP) Medical Laboratory Evaluation (MLE) Program, (4) American Proficiency Institute (API), (5) the College of American Pathologists (CAP)–Surveys (large laboratories), (6) the CAP External Comparative Evaluation for Laboratories EXCEL (small laboratories), and (7) Wisconsin State Laboratory of Hygiene (WSLH). Results that included specific FOBT test system/manufacturer names were obtained from 6 of the 7 PT programs. The AAFP-PT testing service was only able to provide the data in aggregate for manufacturers. Thus, their results were not included in this analysis.
Data from PT programs did not indicate the superiority or inferiority of instruments, reagents, or other materials. 20 PT is required on all analytes conducted in pathology laboratories that are not waived under CLIA. However, if waived tests are run in a CLIA-certified lab, then it is to the advantage of the laboratories to also test those analytes that are waived. 21 If waived FITs are performed in a physician’s office, a CLIA Certificate of Waiver must be obtained and PT is not required. 17
gFOBTs were eliminated from the PT sensitivity/specificity analysis because FITs are now preferred for CRC screening.12,13,15 The PT review included 15 distinct FITs and a total of 12 730 samples.
Literature Review
As of March 11, 2016, there were 447 publications listed in PubMed using the search term “fecal immunochemical test.” There were 72 articles for review after excluding those not written in English, non-research articles and research that did not assess FIT accuracy for AAP and/or CRC in human subjects. After reviewing these abstracts, 26 were found to have information on the accuracy of FITs, with all subjects undergoing a colonoscopy or flexible sigmoidoscopy after FOBT collection.22-47 Two authors (JMD, BTL) reviewed these papers independently to find the results of FIT test accuracy for AAP and/or CRC. After reviewing the articles, those that presented results of FITs no longer on the market, such as FlexSure OBT and HemeSelect, were excluded,23,24,40,41 not used in the United States,30,34,35 not named in the paper, 36 and those where colonoscopy was not used on all subjects.24,36,40,41 For papers presenting sequential results of incremental study populations,26,31,32,37,39,42,45,46 we chose the paper with the largest subject number26,39,46 since that provided the most reliable estimates of test accuracy for AAP and/or CRC. Thus, 13 papers were found in the English language on test characteristics of FITs, using colonoscopy as the gold standard.22,25-29,33,38,39,43,44,46,47
Data Analysis
The PT programs’ results for each FIT brand evaluated in 2014 were reviewed in detail and summarized. At each testing interval, 2 or 3 blinded synthetic fecal samples were sent to participating pathology laboratories and analyzed for the presence of spiked hemoglobin with FITs used in their laboratory. Results were reported back as either positive or negative for hemoglobin. PT programs reported whether the results were correct or incorrect for each FIT and the overall percentages correct/incorrect were tallied across FIT devices. The test system/manufacturer sensitivity and specificity for synthetic fecal samples and the overall sensitivity and specificity for each FIT device across testing programs, along with 95% Agresti-Coull binomial proportion confidence intervals were calculated, 48 using SAS version 9.4 (SAS, Inc, Cary, NC).
Results
FDA CLIA Database
The initial FIT listed in the CLIA database was the SmithKline FlexSure OBT, which is no longer on the market. Historically, CMS reports that early FITs were considered substantially equivalent to the first gFOBT. 49
Based on the search for FOBTs in the CLIA database, 122 FOBT analytes with 60 distinct 510(k) numbers were retrieved; 65 were FITs, 56 were gFOBT, and one was an orthotolidine reagent method (see Figure 1). Of the 65 FITs listed, 60 are CLIA-waived, and 5 are moderate complexity, non-CLIA-waived (See Table 1). There are 25 distinct 510(k) numbers for the 60 CLIA-waived FITs and 3 distinct 510(K) numbers for the automated FITs. Only 2 automated FITs are sold in the US Test systems with the same 510(k) numbers have the same manufacturer (see Table 1). For example, Immunostics, Inc, is listed as the test system/manufacturer for these 4 FITs, hema-screen SPECIFIC Immunochemical Fecal Occult Blood Test, Consult Diagnostics iFOBT, Henry Schein OneStep+ iFOBT, and Select Diagnostics Immunochemical Fecal Occult Blood Test. These 4 FITs have identical 510(k) numbers but are sold under different labels. Three of these 4 FITs are included in the PT program results.
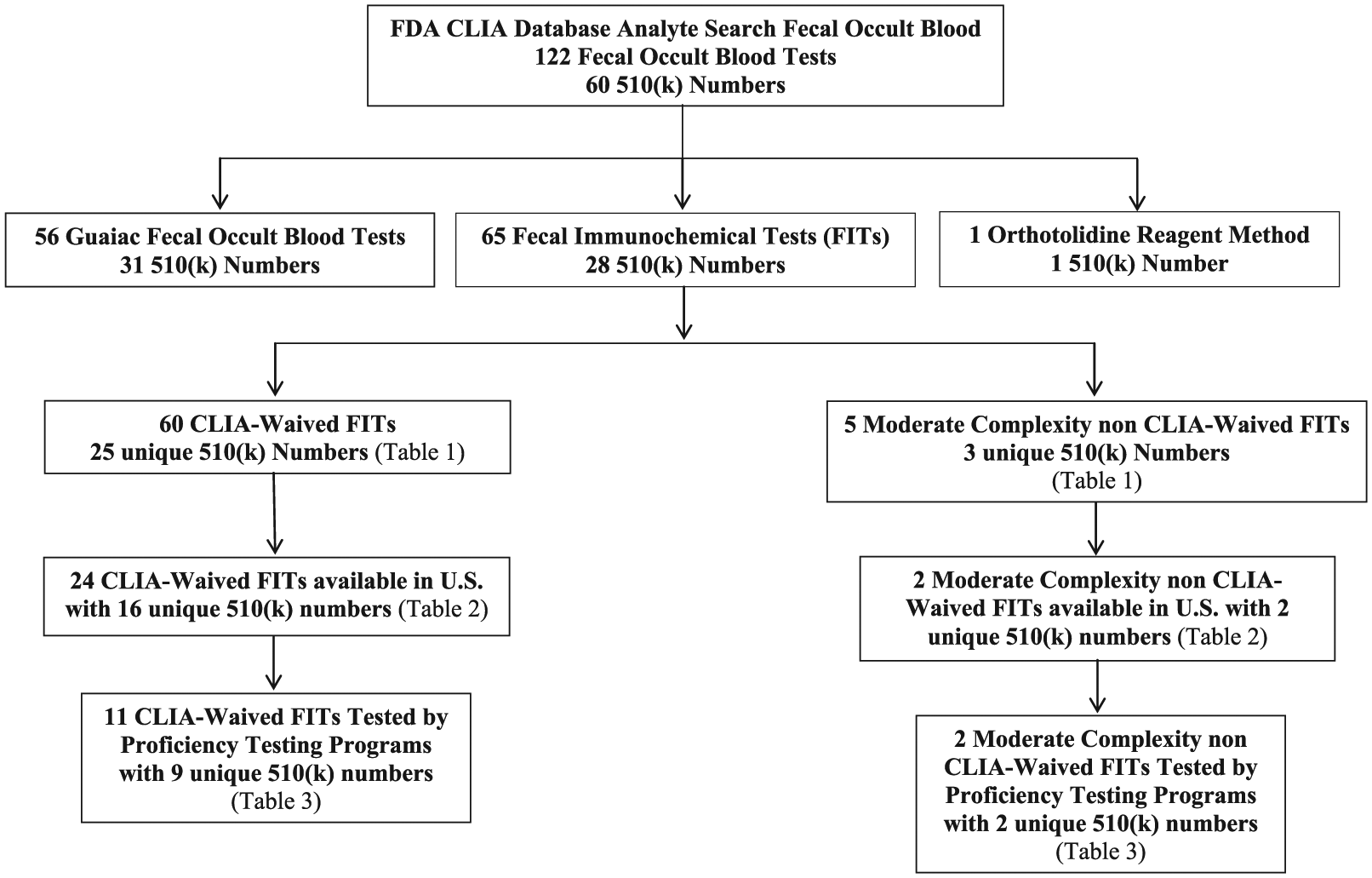
FDA CLIA database fecal occult blood search focusing on distinct FITs available in the United States and number tested by proficiency testing programs. FDA, Food and Drug Administration; CLIA, Clinical Laboratory Improvement Amendments; FIT, fecal immunochemical test.
FDA 510(k) Document Number and Test System/Manufacturer a for CLIA-Waived and Moderate Complexity FITs. b
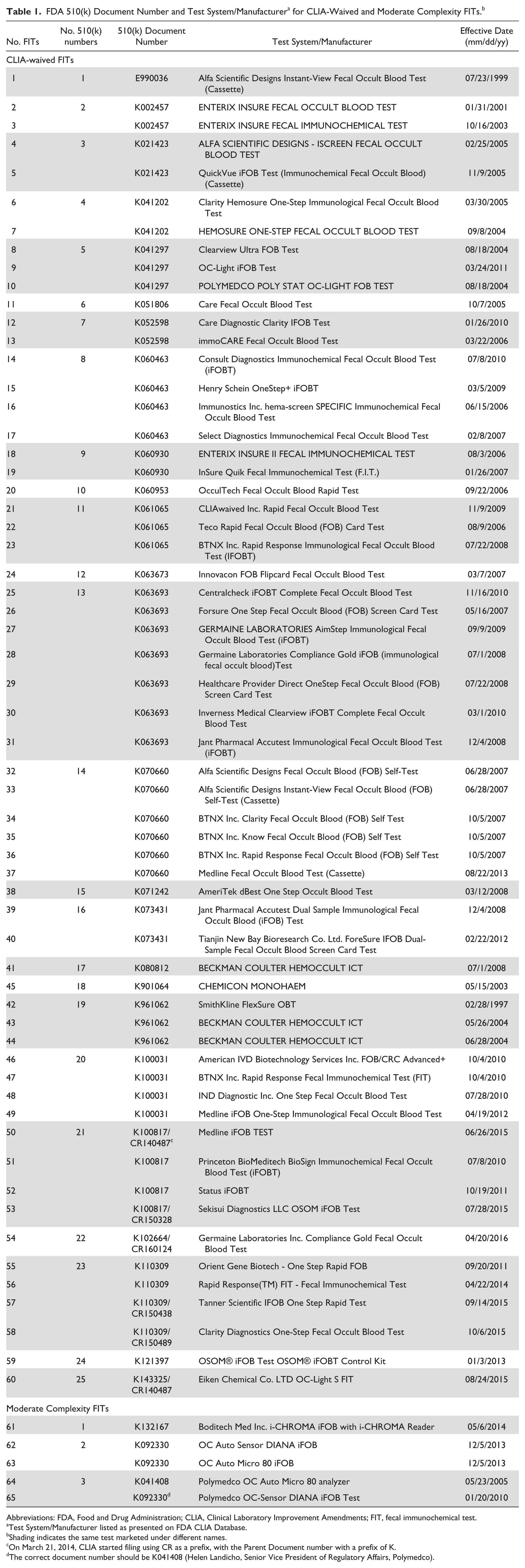
Abbreviations: FDA, Food and Drug Administration; CLIA, Clinical Laboratory Improvement Amendments; FIT, fecal immunochemical test.
Test System/Manufacturer listed as presented on FDA CLIA Database.
Shading indicates the same test marketed under different names.
On March 21, 2014, CLIA started filing using CR as a prefix, with the Parent Document number with a prefix of K.
The correct document number should be K041408 (Helen Landicho, Senior Vice President of Regulatory Affairs, Polymedco).
Of the 60 CLIA-waived FITs, 24 FITs with 16 unique 510(k) numbers are available for purchase in the United States (see Table 2). The 5 moderate complexity FITs have 3 unique 510(k) numbers, but only 2 are available in the United States (OC-Auto Micro 80 and OC-Sensor Diana). These 2 use identical liquid-vial collection containers, reagent, calibrator, and controls, but the analyzers have different throughput rates.
CLIA-Waived and Moderate Complexity FITs Available for Purchase in United States as of July 22, 2016 With FDA Applicant, Respective 510(k) Document Number, Effective Date, and Accuracy in Human Studies. a

Abbreviations: FDA, Food and Drug Administration; CLIA, Clinical Laboratory Improvement Amendments; FIT, fecal immunochemical test; PT, proficiency testing; PPV, positive predictive value; NA, not available.
Shading indicates the same test marketed under different names.
Assessed by Proficiency Testing Programs.
Dry slide collection method.
Proficiency Testing Programs provide only the single sample result.
Taken from manufacturers’ product brochure.
Proficiency Testing Programs’ Accuracy of FITs
There were 57 433 synthetic stool samples used for analysis of gFOBTs and FITs for the 7 PT programs in 2014. However, no information is available on the types of FOBTs analyzed for the 2552 AAFP-PT samples, leaving 54 881 samples available from 6 PT programs for analysis. Of these, 12 730 (23%) were FITs and 42 151 (77%) were gFOBTs. Of the 12,730 FIT samples, the following FITs were most frequently evaluated: (1) Hemosure One-Step Immunological Fecal Occult Blood Test (30%), (2) Beckman Coulter Hemoccult ICT (27%), (3) OC-Light iFOB (12%), (4) OC-Auto Micro 80 (12%), and (5) QuickVue IFOB (9%). These 5 tests accounted for 90.7% of the FITs evaluated. PT programs evaluated 13 FITs, 11 (9 distinct 510(k) numbers) of the 24 CLIA-waived FITs and 2 automated FITs (see Table 3).
Proficiency Testing Programs’ Fecal Immunochemical Test (FIT) Totals and Accuracy for Spiked Samples by Test System/Manufacturer for 2014.
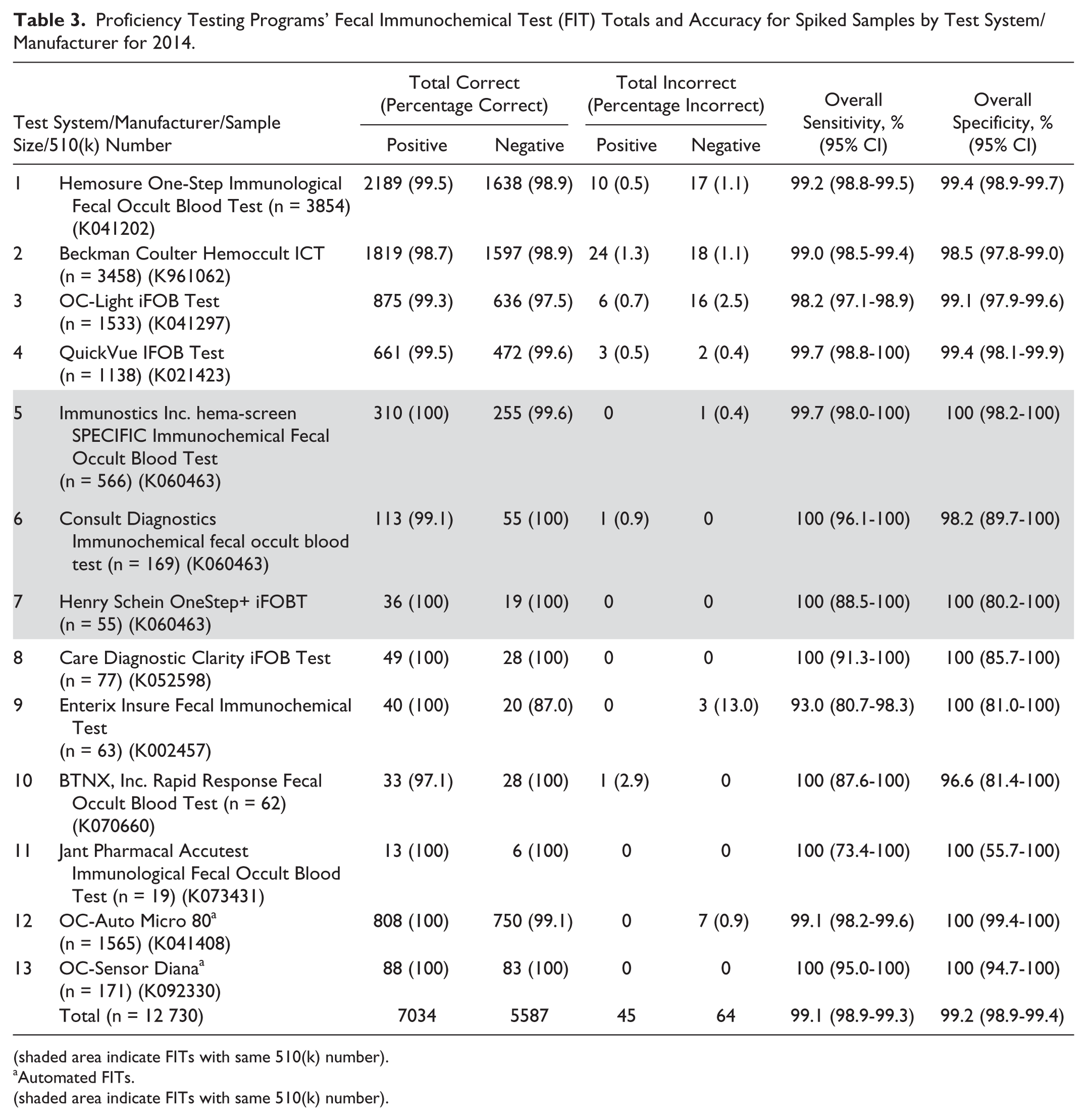
(shaded area indicate FITs with same 510(k) number).
Automated FITs.
(shaded area indicate FITs with same 510(k) number).
For the waived FITs, sensitivity ranged from 93.0% for Enterix Insure Fecal Immunochemical Test to 100% for 5 test system/manufacturers and specificity ranged from 96.6% for BTNX, Inc Rapid Response Fecal Occult Blood Test to 100% for 5 test system/manufacturers (see Table 3). The automated FIT, OC-Sensor Diana, had a sensitivity of 100% and specificity of 100%, with similar results for the OC-Auto Micro 80.
Literature Review
As described in the Methods section, 13 studies presented the accuracy of FITs for AAP and/or CRC, using colonoscopy as the gold standard. Information on FIT accuracy in human studies is available for only 4 CLIA-waived FITs on the US market (see Table 2).26,27,33,44,46 These studies were heterogeneous, conducted on screening or mixed screening, and high-risk populations in Canada, 44 China, 46 Germany, 26 Taiwan, 27 and the United States. 33 The outcomes differed across studies and were typically either AAP or CRC, or the 2 combined. Table 2 shows which products were tested, the size of the study population, the test detection limits, and the sensitivities, specificities, and positive predictive value for the outcome. In general, the sensitivity for any specific outcome varied within product across studies and by outcome across products. The sensitivity for CRC or AAP reported by Levy, et al 33 was 4.0% for the Polymedco OC-Light, whereas the much larger study by Chiu et al 27 reported 28% sensitivity for AAP and 78.6% for CRC. 27 The Levy, et al study had the FIT mailed in for testing, and the Chiu et al study had the specimen collected the day before the colonoscopy, stored in the refrigerator, and brought into the clinic the day of the colonoscopy which may have affected the results. The sensitivity reported for advanced neoplasms or screening relevant neoplasia for the QuickVue IFOB was 59.6%, 26 for OC-Light FOB was 28%, 27 and for Beckmann Coulter ICT was 23.2%. 44 Corresponding specificities were 70.2%, 93.5%, and 95.8%. There were 2 studies that reported sensitivities separately for AAP and colorectal cancer.27,46 The Wong et al study using Hemosure One-Step showed sensitivity for CRC of 54.5% and AAP of 33.1%. 46
For the 2 automated FITs, there were 8 studies assessing FIT accuracy (see Table 2).22,25,28,29,38,39,43,47 Sensitivity for AAP across studies was highly variable, from 25.7% 25 to 49.5. 28 Sensitivity for colorectal cancer varied from 65% 39 to 92.3. 38 For studies reporting the sensitivity for CRC separately from AAP, sensitivities for colorectal cancer were higher than for AAP.
Discussion
Based on our review of the FDA database, there are 26 unique FITs currently on the U.S. market, with 24 waived under CLIA and two automated tests. FITs comprise 23% of the market based on proficiency testing results, with the rest being gFOBT. The 2 automated tests are used quantitatively in many countries in order to adjust the numbers of individuals sent for diagnostic colonoscopy. These are not approved by the FDA for quantitative analysis in the United States.
Of the 26 FITs on the market, 13 with 11 distinct 510(k) numbers were tested in PT programs. Most FITs tested extremely well with sensitivities for 11 of 13 FITs at 99% or higher and specificities for 10 of the 13 FITs at 99.4% or higher. However, samples used by PT programs are simulated fecal material spiked with an unknown amount of hemoglobin that is likely not at the lower limits of sensitivity for FIT products being assessed. PT programs will not share how much hemoglobin is in their spiked samples, as that is considered proprietary information. Tests used for CRC screening need to be simple, reliable, and test well under usual conditions. 50 Health care providers and patients deserve to know that the FITs being used provide accurate test results. There is very little published, population-based research on CLIA-waived FITs done at a single point in time, using colonoscopy as the gold standard. Only 5 studies of 4 unique CLIA-waived FITs (out of 16 total) have been conducted.26,27,33,44,46 The 2 automated FITs have been studied in larger populations than the CLIA-waived FITs and generally have favorable sensitivities and specificities for CRC and/or AAP.22,25,28,29,38,39,43,47
Lee et al 51 conducted a systematic review and meta-analysis to assess the diagnostic accuracy of FITs for CRC. 51 Nineteen research studies, 2 from the United States, were included in the study with both qualitative and quantitative FITs. The overall sensitivity for CRC across these 19 studies of 113 360 total patients was 79% and specificity was 94%. Sensitivity and specificity was not studied for AAP. 51 Another review of 21 studies using either CLIA-waived or automated FITs demonstrated that FITs have a sensitivity for CRC ranging from 0.25 to 1.00 and specificity of 0.83 to 0.99. 52 A systematic review and meta-analysis of qualitative FDA-cleared FITs used in US studies would be worthwhile to continue to explore accuracy for CRC and AAP. This cannot be done without more data. Researchers in Germany have evaluated 20 qualitative and 1 quantitative FIT for hemoglobin in spiked samples and determined the detection limits for hemoglobin do not match the manufacturer’s specifications. 53 German researches recommend screening programs avoid using qualitative FITs due to differences in detection limits with large variations in sensitivity and specificity. 54
A good FIT is one that has a high sensitivity and specificity for screening relevant neoplasia. FITs are recommended regularly over time, in order to have the best chance of detecting occult blood. However, in a real-world setting, many issues can affect the sensitivity and specificity of FITs.33,50,55 There is a seasonal variation in FIT results with lower positivity rates in warmer weather, due to degradation of hemoglobin.55,56 Allison et al 57 have also recommended all companies use a standard collection device and probe with a known buffer for comparison across studies. There is no accepted international quality control standard for use with FITs.
The AAFP-PT results were excluded because they provided only aggregated data for all FOBTs, thus, it was not known which FITs were evaluated by that program. However, the AAFP-PT was only 4% of the total proficiency tests evaluated. Proficiency testing results do not provide any information on the accuracy of human samples for AAP or CRC. In addition, laboratories performing point-of-care tests are not required to participate in PT programs. This means that there is little to no information on test accuracy for the majority of CLIA-waived tests being used in primary care offices and Federally Qualified Health Centers in the United States.
Based on the 2014 PT program results, the gFOBT accounts for 77% of the FOBTs, even though the FIT has been determined to be a better test for occult blood in the stool. Proficiency test results are a crude measure for determining the percentage of gFOBTs and FITs used in the United States. Among the FITs, Hemosure One-Step Immunological Fecal Occult Blood Test was evaluated the most with the Beckman Coulter product close behind. The current worldwide use of FITs is primarily automated FITs read with quantitative results.22,25,29,39,51 In the United States, 122 671 physician office laboratories use the CLIA-waived FOBTs 58 and the quantitative FITs used in other countries are either not available or are only able to report qualitative results because they have not been approved by the FDA to report quantitatively.
The 13 FIT brands tested across the 6 PT programs tested extremely well using spiked samples and might be appropriate to use in CRC screening programs. However, there was published information on only 4 of 24 (16 unique 510(k) numbers) CLIA-waived tests in screening populations and/or populations at higher risk. Given the goal of 80% of individuals screened for colorectal cancer by 2018, it is likely that FITs will need to play a greater role in screening, and an estimated 24 million more Americans will need to be screened. 59 Most developed countries have a systematic CRC screening program that uses mailed automated FITs developed in a central or few locations.60,61 A comparative effectiveness study of the test characteristics of commonly used FITs with colonoscopy as the gold standard is essential if we are to know which FITs available in the United States have the best test characteristics. Studies conducted on 4 qualitative, CLIA-waived FITs demonstrate widely varying test accuracy. 33 Do the commonly used FIT brands test positive down to the lower limits of hemoglobin specified in their product inserts and under conditions where stool is present? It is critical to determine the FITs with the best test characteristics in order to implement successful FIT-based screening programs.28,32,62
To answer the question, “which FIT do I use?” a FIT is recommended in the CRC screening guidelines in preference to gFOBT.12,13,15 Reviewing the PT program results, 13 FITs evaluated across programs would be appropriate based on spiked samples. However, narrowing the FITs further, based on the studies that conducted a one-time FIT prior to colonoscopy to assess test accuracy for AAP or CRC, the automated FITs (OC-Auto Micro 80 and OC-Sensor Diana), provided the best sensitivities and specificities across studies. Further studies are warranted prior to recommending FITs that are waived under CLIA for private practice offices.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health, NIH: RC1 CA144907 and the Department of Family Medicine, University of Iowa, Iowa City, Iowa.
