Abstract
Introduction
Chronic diseases are an epidemic affecting the United States population, contributing to 7 of the 10 top causes of death. In 2012, approximately half of US adults were reported to have one or more chronic disease(s). Hypertension and diabetes, 2 of the most common chronic diseases, affect 70 million and 29 million Americans, respectively. 1 Socioeconomic status is inversely associated with the prevalence of diabetes and hypertension 2 with links to disparate disease control in medically underserved areas.3-5
Federally Qualified Health Centers (FQHCs), funded by The US Department of Health and Human Services’ Health Resources and Services Administration (HRSA), 6 provide health care services to the medically underserved population. Diabetes control of A1c <9% and blood pressure control of <140/90 mm Hg have been included in the Core Clinical Measures put forth by HRSA. 7 Consequently, FQHCs also focus on population management of diabetes and hypertension and achievement of these goals. 8
Appropriate management of these conditions, including proper use of medications, can lead to better disease control, decrease disease-related complications, and improve overall health. Pharmacists have been shown to positively impact chronic disease outcomes through medication therapy management (MTM), which involves a multifaceted approach of reviewing medications, identifying and remedying medication-related problems, providing disease state management and self-management education, addressing medication adherence issues, and considering preventative health strategies to optimize medication-related health.9-13 An MTM service includes a comprehensive medication review to ensure that the patient’s medication-related needs have been met and all of his/her medications are appropriate, effective, safe, and convenient. At the end of the visit, a care plan is developed and shared with the patient and the primary care provider to resolve and prevent any drug therapy problems by eliminating unnecessary medications, initiating appropriate medications, adjusting dosage regimens, addressing adverse reactions, and increasing the patient’s willingness and ability to adhere to the medication regimen.14,15 Unfortunately, pharmacists are often a missing or underutilized member of the healthcare team.
This project aims to evaluate the impact of pharmacist-provided MTM in FQHCs on chronic disease intermediate outcomes. The primary objective is to report the number of patients seen be a pharmacist who achieve (1) A1c in control, defined as <9% and (2) blood pressure in control, defined as <140/90 mm Hg. The secondary objectives are to (3) describe number and type of medication-related problems (MRPs) identified and resolved by pharmacists providing MTM in FQHCs and (4) identify potential (pADEs) and actual adverse drug events (ADEs), and refer patients to diabetes self-management education classes, as needed.
Methods
This was a multisite, prospective, descriptive pilot study approved by The Ohio State University’s and Ohio Department of Health’s Institutional Review Boards. Three FQHCs involved in the study were selected based on established MTM services in the areas of diabetes and hypertension, provision of care via distinct staffing models, and geographic diversity across the state. A pharmacist representative from each site attended a formal orientation held in January 2014 during which each site presented on their model of care (Table 1). An oversight committee was established for the project, which included representatives from the FQHC project sites, the state department of health, nonprofit professional organizations supporting community health centers and pharmacists, as well as all seven colleges of pharmacy in the state of Ohio. This committee, named the MTM Consortium, met quarterly to provide guidance on project progress, troubleshoot site-related issues that arose, and discuss ways to impact MTM practice in the state with this project through connecting to stakeholders such as the payors, the state’s Health Information Exchange, and other public health entities and professionals.
Sites Demographics and Practice Models in Ohio Chronic Disease MTM Project in FQHCs by Month, March 2014 to December 2015.

Abbreviations: MTM, medication therapy management; FQHC, Federally Qualified Health Center; COPD, chronic obstructive pulmonary disease; EHR, electronic health record; FTE, full-time equivalent; IPPE, introductory pharmacy practice experiences; APPE, advanced pharmacy practice experiences.
Active sites participated in the study; health center networks may have had pharmacists providing MTM for chronic disease patients at other sites in their network that were not formal study participants.
Medicaid expansion went into effect in Ohio in January 1, 2014.
For PrimaryOne Health and AxessPointe, the insurance breakdown is reported for their network of community health centers. For Health Partners of Western Ohio, the insurance breakdown is reported for the active site.
AxessPointe developed CDTM for warfarin and insulin in September 2015. Health Partners of Western Ohio has no formal CDTM, but use integrated care team visits during which a pharmacist is part of the care team at the visit.
Collaborative visit involves patient cared for by pharmacist, physician provider, and other care team members (ie, social worker, dietician).
Residents are Postfraduate Year 1 (PGY1) or Year 2 (PGY2) Pharmacy Practice Residents who are licensed pharmacists seeking post-PharmD clinical training.
Pharmacy student involvement includes as pharmacist extenders in delivery of MTM as well as data collection and reporting. These may include first through third-year students, who complete rotations called Introductory Pharmacy Practice Experiences (IPPEs) as well as students in their final experiential year completing what are called Advanced Pharmacy Practice Experiences (APPEs).
Patient recruitment began March 1, 2014. Patients were recruited at each FQHC from reports created with each site’s electronic health record (EHR). Patients were included if they were aged 18 to 75 years, had a diagnosis of hypertension and/or diabetes with diagnosis occurring at least one year prior, were seen for a medical visit(s) at least once in the last year, and had a most recent A1c >9% or a most recent systolic blood pressure ≥140 mm Hg or diastolic blood pressure ≥90 mm Hg. They were excluded if pregnant, diagnosed with end stage renal disease, or had received a pharmacist visit at the site within 1 year prior to enrollment.
Once enrolled, patients received MTM from a pharmacist at each FQHC. While each site and pharmacist provides care with site-specific protocols and workflow models, pharmacist interactions aligned with core elements of MTM.14,15 MTM services involve a review of all of the patient’s medications with them to ensure they understand why they are taking each medication, how to use them properly, as well as side effects to watch out for and other relevant education points specific to each medication. Pharmacists review the patient’s chart to analyze past medical history, including progress notes and laboratory values, to assess for safety and efficacy of the patient’s medication regimens. Pharmacists providing MTM collaborate with the patient and the physician or prescribing provider to identify medication changes that could benefit the patient, educate the patient on lifestyle or behavior changes, and set goals with the patient related to their chronic diseases. These services are provided through a variety of interactions, including face-to-face, on-site visits, electronic communications via email and/or texting, as well as telephone visits and follow-ups according to each site’s standards of usual care.
Data were reported monthly in aggregate regarding primary and secondary outcomes including the number of patients seen per month who were uncontrolled or controlled for A1c, defined as <9% and blood pressure, defined as <140/90 mm Hg. The A1c goal was set at <9% to align with value-based quality metrics from HRSA, which guide evaluation of FQHCs as well as the National Quality Forum (NQF) of the National Committee for Quality Assurance, specifically NQF measures 18 and 59, which are performance measures set forth by the Centers for Disease Control and Prevention.8,16 Additionally, sites reported number of patients with A1c between 8% and 9%, between 7% and 8%, and less than 7% to evaluate more rigorous achievement of guideline-driven disease management goals. 17 The sites tracked and reported number of MRPs, ADEs, and pADEs identified as well as referral to outside disease self-management education programs. Based on clinic structure and evolving documentation trends, not all sites reported on MRPs and ADEs/pADEs every month. Thus, the data presented are underrepresenting actual interventions at the clinic.
Data were aggregated across all 3 pilot sites using Microsoft Excel. Each outcome measure was examined for trends over time for the project as a whole and monitored monthly to ensure data quality. All statistical analyses were completed using the software SAS and Excel.
Results
At the conclusion of phase 1, December 31, 2015, 706 total patients had been enrolled in the project, 422 with uncontrolled diabetes, 434 with uncontrolled hypertension, and 150 with both uncontrolled diabetes and hypertension. Of the 422 enrolled patients with uncontrolled diabetes, 52.84% (n = 223) had an A1c that was brought into control, defined as <9%. Seventy-two patients (17.06%) achieved an A1c between 8% and 9%, 19.19% (n = 81) of patients achieved an A1c <8% and ≥7%, and another 16.59% (n = 70) of patients achieved an A1c <7%. (Figure 1) The number of patients with blood pressure at goal, defined as <140/90 mmHg has improved from 0% in control at baseline to 65.21% (n = 283) in control at the preliminary data collection point of December 2015. (Figure 2)
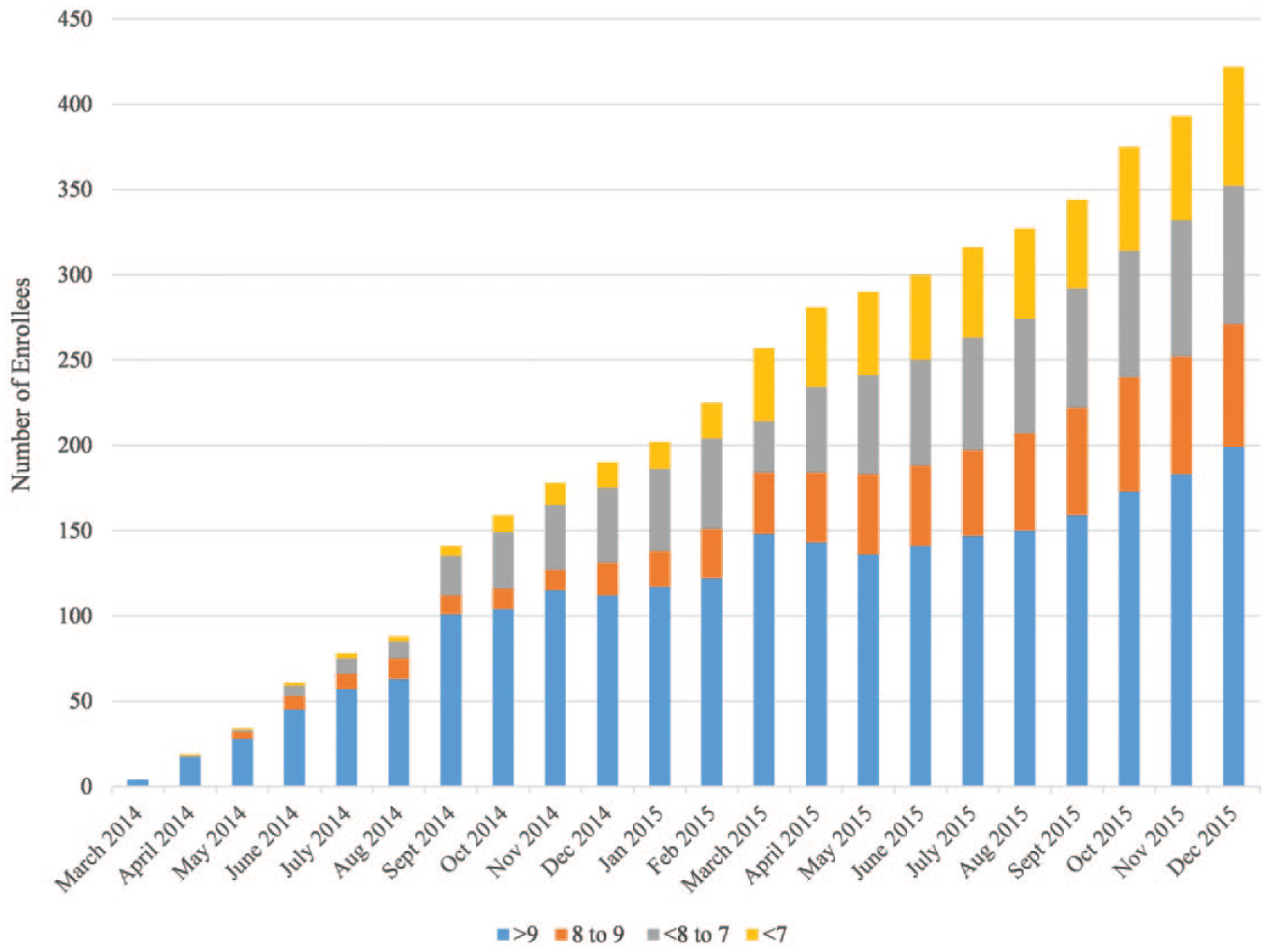
A1c measurement of medication therapy management (MTM) project enrollees by month, Ohio, March 2014 to December 2015. Since the beginning of the project, 223 diabetic patients who were enrolled in MTM services decreased their A1c to a level considered in control (<9%). Among those, 70 lowered their A1c to a level considered tight control (<7%).
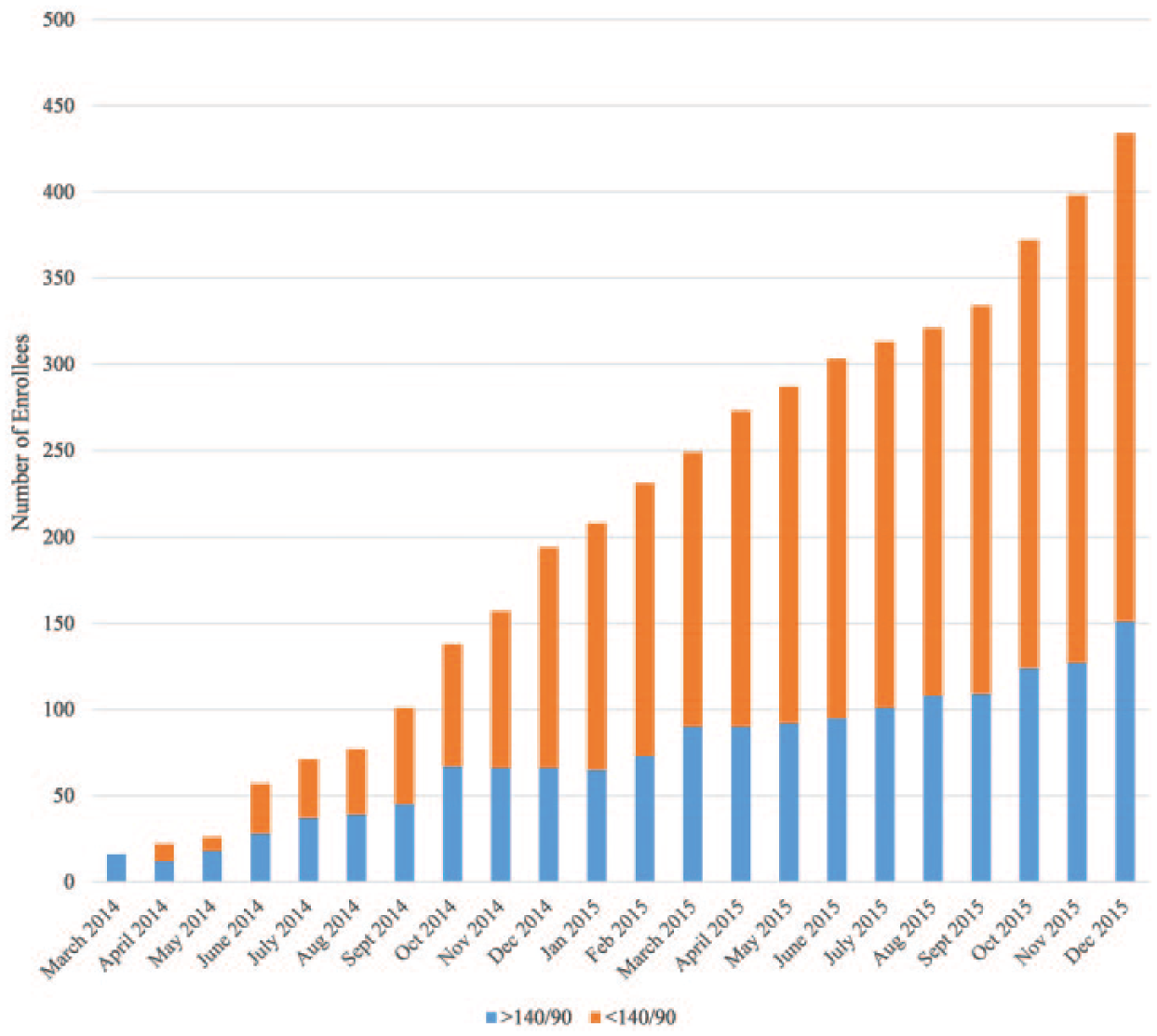
Blood pressure measurement of medication therapy management (MTM) project enrollees by month, Ohio, March 2014 to December 2015. Since the beginning of the project, 283 hypertensive patients who were enrolled in MTM services decreased their blood pressure to a level considered in control (<140/90 mm Hg).
MRPs are described in Table 2. Because of limitations in documentation software at individual sites, these secondary outcomes were reported variably. All sites reported MRPs; however, reporting occurred from the start of the study through December 2015 for 2 sites and through May of 2014 for the third site. For ADEs/pADEs, one site reported monthly between April 2014 and May 2014; another site reported March 2015 through December 2015, and the third site did not report on these outcomes. Overall during this time frame of reporting, the 2 sites identified 169 potential adverse drug events and 84 ADEs. No sites reported referring patients to diabetes self-management education programs outside of their clinic sites during this project.
Number of Medication-Related Problems (MRP) Identified and Remedied in the Ohio Chronic Disease MTM Project in FQHCs by Type, March 2014 to December 2015.
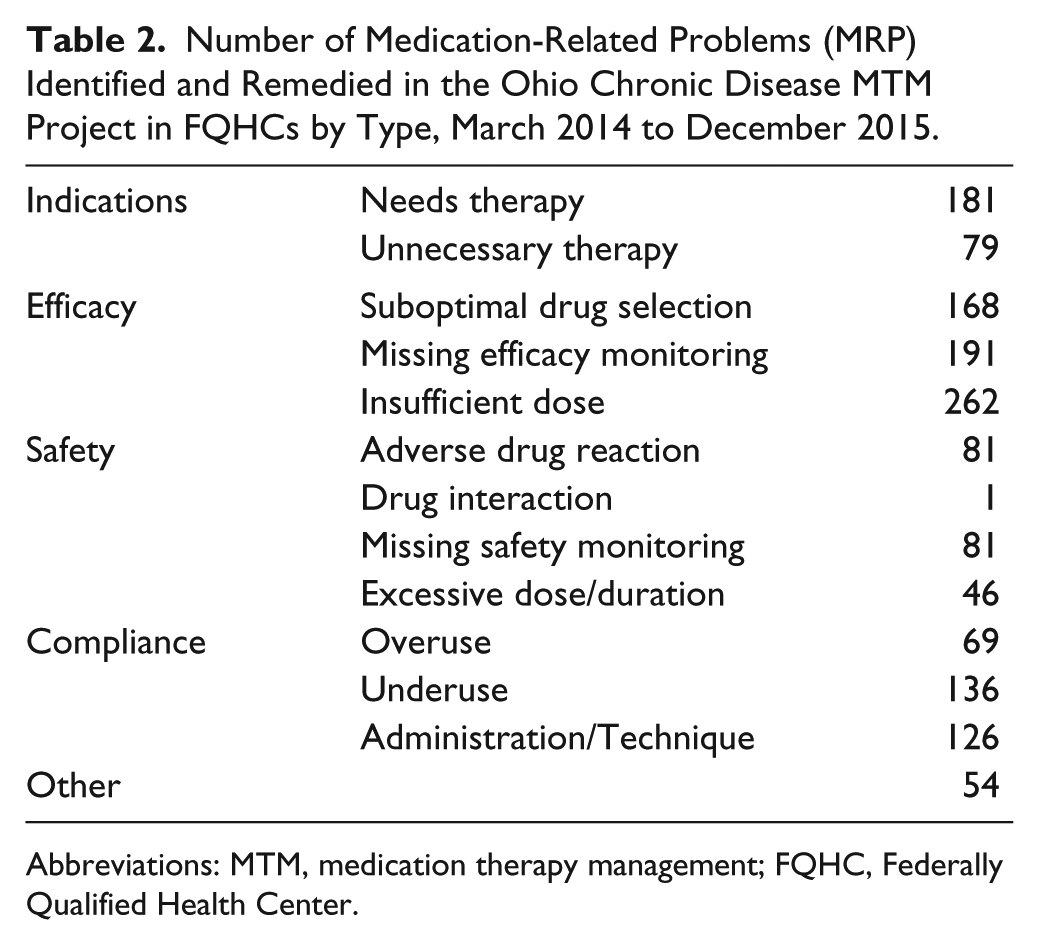
Abbreviations: MTM, medication therapy management; FQHC, Federally Qualified Health Center.
Discussion
This multisite, prospective, descriptive pilot study displayed improvement in diabetes and hypertension clinical markers associated with pharmacist provision of MTM. A1c goal achievement occurred in 52.84% of patients and hypertension control was reported in 65.21%. Pharmacists identified and resolved more than 1400 MRPs and addressed multiple adverse drug event issues.
The impact pharmacists have on improving chronic disease management is well documented with studies showing cost-effectiveness for pharmacists affecting blood pressure and diabetes9,11,12,18-20 and improvement in clinical markers.9-13,21-23 However, sparse data are available specifically for pharmacist-provided MTM with vulnerable populations, such as those cared for in FQHCs. A few studies have been published describing pharmacist involvement in Annual Wellness Visits (AWV), 24 medication reviews,25,26 and telepharmacy, 27 provided in medically underserved urban and rural communities with positive results, including reductions in hospitalizations and emergency room visits as well as reductions in total medications. 25
The data presented in this article are novel due to the impact shown on diverse populations served across the state of Ohio as well as inclusion of a variety of practice models used to address needs of communities and provide opportunities for sustainable MTM services. This project is the first of its kind to partner FQHCs, a state department of health, nonprofit professional organizations supporting community health centers and pharmacists, colleges of pharmacy, and pharmacies to enhance MTM provided to medically underserved patients. Pharmacy and public health have collaborated in recent years; opportunities exist to further enhance these partnerships to improve the health and well-being of the nation through access and optimization of medication therapy by pharmacists.20,28
Limitations exist to the data presented. One key consideration is that the data was collected in aggregate as part of this observational study and no randomization or control group were used in this demonstration project; patients served as their own controls. Thus, other factors may have influenced the reported results and only associations can be implied. Additionally, investigators did not track subjects lost to follow-up. Not all practice sites collected and reported MRP, pADE, and ADE outcomes and the MRP data may have had some subjectivity to interpretation by individual pharmacists based on the nature of interpreting and reporting this type of categorical data. New hypertension guidelines were released 29 after the study started. In response, investigators adjusted the Excel reporting tool to identify patients older than 60 years whose blood pressure was deemed controlled, but greater than 140/90 mm Hg. The models of care varied for each site, which was a strength for the external validity of this project. However, since the protocols for care were developed to be successful within each individual clinic environment, there was variation in the way care was provided among the sites. This poses a limitation in internal validity. A key limitation of the reported data is that only individuals with uncontrolled disease are recruited as part of this project. As recruitment is ongoing and patients achieve control of A1c and blood pressure, new, uncontrolled patients are continuously being enrolled into the study. Thus, the impact of the pharmacist-provided MTM on these chronic disease outcomes may be underestimated in the effect reported in this article.
This pilot study was phase 1 of a 2-phase, 5-year project. In phase 1, best practices of pharmacist-provided MTM with FQHCs were identified and impact on intermediate outcomes was shown. In phase 2, which will take place through June 30, 2018, up to 9 additional pharmacists will integrate MTM with FQHCs across the state of Ohio and report impact on A1c, blood pressure, and MRPs, while continuing to identify novel, sustainable models of pharmacist-provided MTM.
Conclusion
Pharmacist-provided MTM may improve chronic disease intermediate outcomes for medically underserved patients in FQHCs through a variety of practice models. Public health practitioners and pharmacists have excellent opportunities to collaborate to enhance the health of those living with chronic diseases through MTM and should seek sustainable models to include pharmacists in the health care team.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was supported by the Cooperative Agreement Number, DP004817, funded by the Centers for Disease Control and Prevention. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the Centers for Disease Control and Prevention or the Department of Health and Human Services.
