Abstract
Introduction
Chronic obstructive pulmonary disease (COPD) is a leading cause of mortality and morbidity throughout the world causing higher medical costs and social economic burdens.1-3 In 2005, 5% of all deaths worldwide were due to COPD complications. 4 This percentage is predicted to increase by 30% by 2020, and COPD is on target to become the third leading cause of death worldwide. 4
COPD is often under- or misdiagnosed in the primary care setting.5-7 For example, in the United States, for every 1 person diagnosed with COPD, there are another 1.4 people who have not been diagnosed and therefore not captured by health care system. 8 Spirometry, the current gold standard for diagnosing COPD, is often underutilized or used incorrectly in primary care settings5,7 due to lack of expertise and/or difficulty in its use by COPD patients. Therefore, a noninvasive method that is quick and easy to use is needed to diagnose COPD in primary care settings.
Nitric oxide (NO) and NO derivatives are known markers of airway inflammation present in exhaled breath condensate of asthma patients. 9 NO is a noninvasive, time-saving, highly reproducible, and cost-effective method for assessing inflammation.9,10 Measuring NO in exhaled breath condensate (eNO) is a promising noninvasive method that would allow physicians and public health professionals to monitor the progression of COPD in outpatient settings with minimal training. However, studies relating NO derivatives to COPD, and COPD severity remain inconclusive; some have shown an association between eNO and COPD,9,11 whereas others have not. 10 Since, there is no cure of COPD, early diagnosis and treatment remains the only viable option to controlling and decreasing COPD incidence and prevalence rates.
The purpose of this study was to determine if there is an association between eNO, COPD, and COPD severity, using the National Health and Nutrition Examination Survey (NHANES) 2007 to 2010 data.
Methods
The NHANES is an ongoing, stratified, multistage probability program of studies that examines 5000 civilian, noninstitutionalized adults and children every year in the United States. Fifteen counties/locations are visited annually where participants are interviewed to gather demographic, socioeconomic, dietary, and health-related information, physical examinations conducted, and laboratory tests completed.12,13 The total sample size for NHANES 2007 to 2010 was 20 686 (10 149 for 2007-2008 and 10 537 for 2009-2010). The sample was restricted to adults 30 years and older because COPD can be present in younger patients. The final analytic sample consisted of 10 214 individuals.
Dependent Variables: COPD and COPD Severity
The main dependent variable, COPD, was constructed using these 2 questions: “Ever told you had emphysema?” and “Ever told you had chronic bronchitis?” The binary variable was formed by coding individuals with positive responses to either of these questions as 1. Individuals who responded negatively to both questions were coded as 0.
COPD severity was calculated using individual prebronchodilator spirometry data for the COPD population from NHANES 2007 to 2010, converted from milliliters to liters. A ratio was calculated by dividing FEV1 (forced expiratory volume in 1 second) by FVC (forced vital capacity). Observed values less than 70% were divided by predicted values and aligned to the GOLD (Global Initiative for Chronic Obstructive Lung Diseases) standard to determine severity as Stage I, II, III, or IV for analysis. 14 Predicted values were calculated by dividing the observed FEV1 value by the predicted FEV1 value estimated for an individual of similar age, height, gender, and race, using gender- and race-specific reference equations. 15 A correction factor of 0.88 was applied for race/ethnicity category listed as “other.” 15 . Since there were few observations for stages III and IV, data for these 2 categories were combined into 1 category, stage III.
Independent Variable: Exhaled Nitric Oxide
Exhaled NO readings, measured in ppb (parts per billion), were taken during the physical examinations, and the mean of 2 reproducible readings was recorded. For the purpose of this analysis, eNO, a continuous variable, was converted into a categorical variable and grouped into quartiles.
Covariate Variables
Covariates included age, gender, race/ ethnicity, tobacco smoking, body mass index, whether someone smoked in the home, whether the subject ate or drank within the last hour, ate nitrogen rich foods within the last 3 hours, ate nitrogen-rich vegetables within last 3 hours, smoked within the last hour, exercised strenuously in the last hour, used steroids within the last 2 days, had a cough, cold or other respiratory illness in last 7 days, or had ever been told by a health care professional they had asthma.
Statistical Analysis
Data were described using descriptive statistics. Univariable and multivariable binary (COPD) and ordinal (COPD severity) logistic regression analysis were used to calculate unadjusted and adjusted odds ratios, respectively, to test the association of eNO with COPD and COPD severity. Data analysis was performed using Stata statistical software package version 13.0 taking into account survey feature of NHANES. Four-year weights were constructed by taking one-half of 2-year weights of the 2 survey cycles. 16
Results
The average age of study participants was 55.1 years (SD = 15.2 years). Women made up 52% (n = 5230) of the sample, while 48% were male (n = 4984). Non-Hispanic whites made up most of the sample at 71% (n = 4982), followed by non-Hispanic blacks at 11% (n = 1951). Fifty-three percent of the study sample were nonsmoker (n = 5287) followed by 27% ex-smokers (n = 2815) and 20% current smokers (n = 2106). Thirty-five percent of responders were overweight (n = 3446) and 37% were obese (n = 4274) (Table 1).
Descriptive Characteristics of the Study Sample.
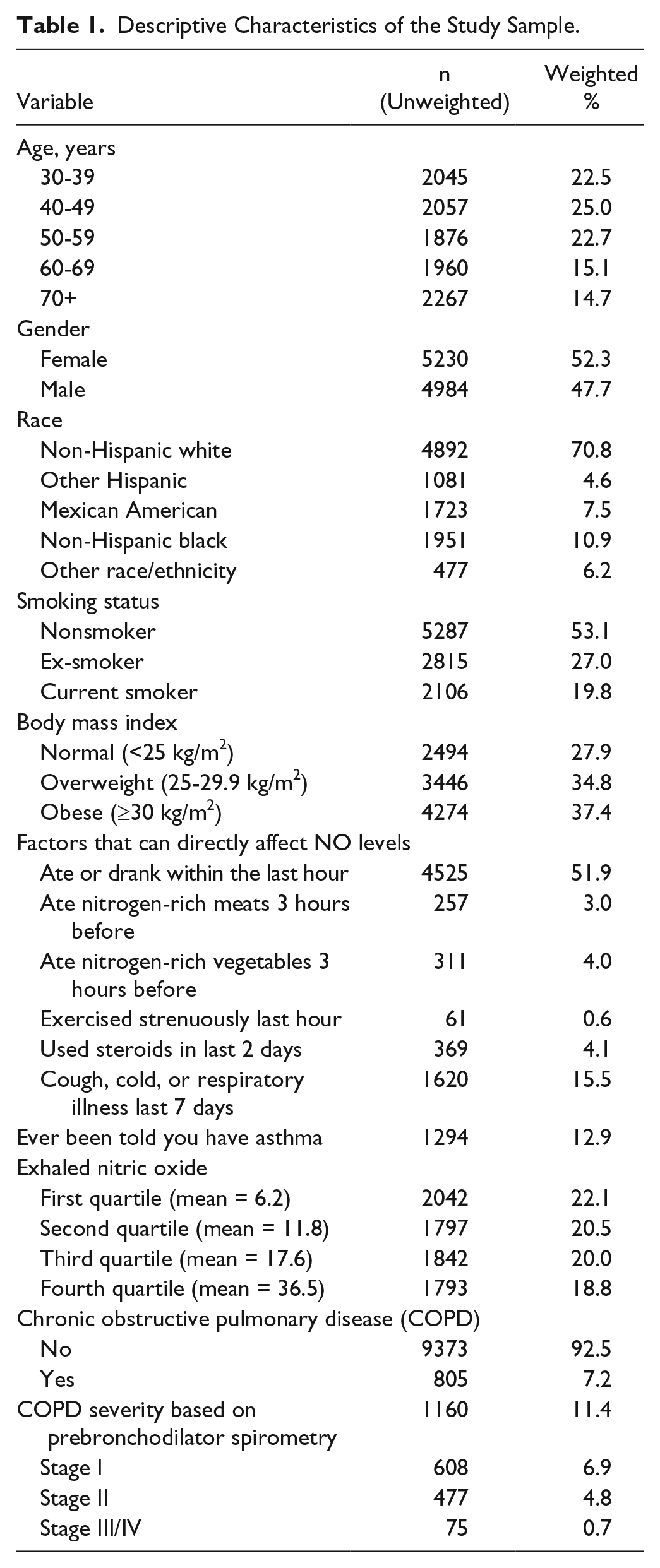
Exhaled NO data were provided by 81% (n = 7474) of the sample. These results were grouped into quartiles: quartile 1 ranged from 3.5 to 9.5 ppb (mean = 6.3), quartile 2 from 9.5 to 14.5 ppb (mean = 11.8), quartile 3 from 14.5 to 22.5 ppb (mean = 17.6), and quartile 4 ranged from 22.5 to 301 ppb (mean = 36.5). Frequency distribution of factors that could directly affect eNO levels ranged from as low as 0.6% for those reporting exercising strenuously in the last hour to as high as 52% for those reporting eating or drinking within the last 3 hours before beginning the examination (Table 1).
The prevalence of COPD, based on self-reported response to questions on chronic bronchitis and emplysema, was 7.2% (n = 805). Whereas, based on the prebronchodilator spirometry analysis, the prevalence of COPD was 11.4% (n=1160). The prevalence rates for different stages of COPD severity were 6.9% for stage I, 4.8% for stage II, and 0.7% for stage III/IV (Table1).
The odds of COPD decreased as the amount of eNO increased from as high as 0.68 (95% CI = 0.51-0.92) for quartile 2 to as low as 0.53 (95% CI = 0.39-0.73) for quartile 4. When adjusted for age, gender, race, and smoking status, all odds ratios became statistically nonsignificant (model 2). The odds ratio of COPD for the second and the third quartiles of eNO exposure remained statistically nonsignificant for models 3 and 4, and only reached statistical significance for the fourth quartile with a 40% decline in the odds of COPD observed for both models (Table 2).
Univariable and Multivariable Association Between Exhaled Nitric Oxide (eNO) and Chronic Obstructive Pulmonary Disease. a

Model 1: Unadjusted odds ratio (OR) and 95% confidence interval (CI). Model 2: Model adjusted for age, gender, race, and smoking. Model 3: Model adjusted for factors that can directly affect nitrogen oxide levels (“Ate or drank within last hour,” “Ate nitrogen-rich foods within the last 3 hours,” “Ate nitrogen-rich vegetables within last 3 hours,” “Smoked within the last hour,” “Someone smokes in the home,” “Exercised strenuously last hour,” “Used steroids within the last 2 days,” “Cough, cold or other respiratory illness in last 7 days,” and “Ever been told you had asthma.” Model 4: Model adjusted for age, gender, race, smoking, body mass index, “Ate or drank within last hour,” “Ate nitrogen-rich foods within the last 3 hours,” “Ate nitrogen-rich vegetables within last 3 hours,” “Smoked within the last hour,” “Someone smokes in the home,” “Exercised strenuously last hour,” “Used steroids within the last 2 days,” “Cough, cold or other respiratory illness in last 7 days,” and “Ever been told you had asthma.”
Table 3 illustrates association of eNO quartiles with COPD severity stages. A 48% decline in COPD severity was observed for exposure to third quartile of eNO in the unadjusted analysis only (unadjusted OR=0.52, 95%CI=0.34-0.80) (Table 3).
Association Between Exhaled Nitric Oxide (eNO) and Severity of Chronic Obstructive Pulmonary Disease. a
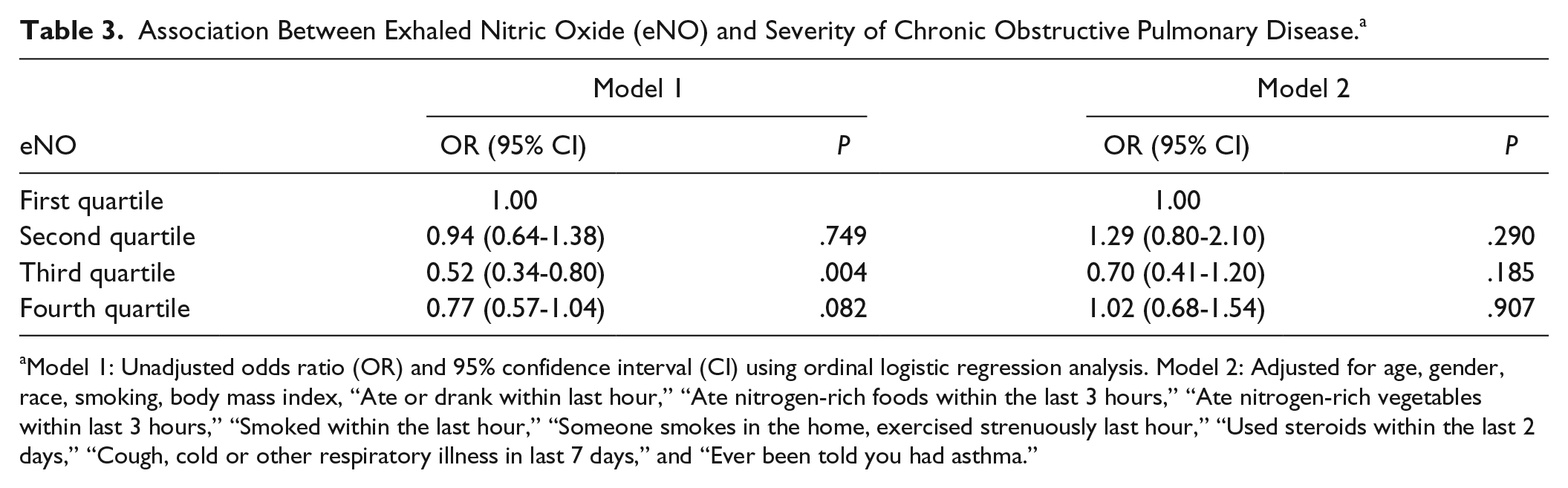
Model 1: Unadjusted odds ratio (OR) and 95% confidence interval (CI) using ordinal logistic regression analysis. Model 2: Adjusted for age, gender, race, smoking, body mass index, “Ate or drank within last hour,” “Ate nitrogen-rich foods within the last 3 hours,” “Ate nitrogen-rich vegetables within last 3 hours,” “Smoked within the last hour,” “Someone smokes in the home, exercised strenuously last hour,” “Used steroids within the last 2 days,” “Cough, cold or other respiratory illness in last 7 days,” and “Ever been told you had asthma.”
Discussion
The prevalence of COPD in this study was 7.2% based on physician diagnoses. However, based on prebronchodilator spirometry analysis, 11.4% of the study population was diagnosed with some form of COPD. The prevalence of COPD severity was 6.9% for stage I, 4.8% for stage II, and 0.7% for stage III. These results are similar to the ones reported by Lindberg et al, 17 who reported the prevalence of mild, moderate, and severe GOLD-COPD as 8.2%, 5.3%, and 0.7%, respectively. The findings from this study further supports the argument made by earlier researchers that COPD is often under- or misdiagnosed in the clinical setting.5,6,18 Routine use of spirometry testing in outpatient settings should be encouraged to diagnose, treat, and manage COPD cases early in the disease process to prevent the disease from progressing to a later, more severe stage.
When examining the unadjusted analysis of eNO and COPD, all eNO quartile P values were statistically significant (quartile 2 P = .013, quartile 3 P = .010, and quartile 4 P <.001). ORs decreased from 0.68 in quartile 2 to 0.53 in quartile 4. However, when adjusted for factors directly affecting eNO (model 3) and for all variables (model 4), only quartile 4 remained statistically significant at P < .05. These results demonstrate that only higher concentrations of eNO are associated with lower odds of COPD. The inverse relationship observed in the current study could possibly be because only 4% of COPD patients were reportedly using steroids prior to the spirometry testing during the NHANES examination and were likely to be clinically stable; eNO levels rise during acute exacerbations, especially in patients hospitalized with severe COPD. 19 Similar findings were also reported by Beg et al 9 who found a negative correlation between FEV1/FVC ratio and eNO concentration in patients with COPD (P = .028). In a small clinical study of 32 Dutch subjects (16 with COPD and 16 controls), Rutgers et al 20 reported no significant difference in eNO concentrations between subjects with and without COPD consistent with the results from the current study.
The current study did not find any association between eNO and severity of COPD, as determined by the GOLD criteria. Inflammation in COPD is mostly present in small airways and lung parenchyma. Brindicci et al 11 found that COPD severity correlated with an increase in the alveolar fraction of exhaled NO, CalvNO. However, their results were not confirmed by Lehouck et al 6 and Bazeghi et al 10 who found no correlation between eNO, CalvNO, and COPD severity, consistent with the results of the current study. Hence, the findings of this current study confirm previous data and weaken the prospect of using eNO as a biomarker for COPD.
There were some limitations to this study. In NHANES, participants were not diagnosed with COPD using objective clinical measures. Hence, misclassification bias cannot be ruled out. However, any misclassification is likely to be nondifferential resulting in underestimation of ORs. This was evident from the fact that prevalence of self-report COPD was underestimated when compared with the prevalence based on spirometry data. We were not able to estimate the prevalence of GOLD stage IV (very severe) category since data for GOLD stage IV severity were few and was combine with stage III category to provide stable prevalence estimates. Also, we were not able to use postbronchodilator data due to small numbers. Asthma and COPD can coexist, and sometimes it is difficult to separate them clinically.21,22 There were 366 subjects in the current study who had both asthma and COPD. Removing asthmatics from the analyses did not change the results. Therefore, to maintain the appropriate power, we chose to leave the asthma variable in multiple logistic regression analyses as a potential confounder. Although the current study included a large number of potential confounding variables, residual confounding due to unmeasured variables could not be ruled out. Lastly, because of the cross-sectional nature of the data, reverse causation cannot be ruled out.
In conclusion, this study found no association of eNO with COPD and COPD severity. Use of eNO as a noninvasive biomarker of COPD or COPD severity is not supported by our findings. However, our results suggest that COPD is underdiagnosed in clinical settings. Therefore, routine use of spirometry should be encouraged for diagnosis and management of COPD in outpatient settings. Future studies should also explore the role of high levels of eNO in excluding the diagnosis of COPD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
