Abstract
Introduction
Diabetes mellitus (DM) is one of the burden diseases with dramatically increasing prevalence in both the developed and developing world. 1 In recent times, the Middle East region has seen some of the largest growth in DM in the world; 5 out of the top 10 countries with the highest diabetes prevalence are in the Middle East region and trends predict that the region will see more than 90% growth in the disease by 2030.1-3 These perturbing trends are a result of the dramatic changes in dietary and lifestyle habits in this rapidly economically developing region.4-6 In line with global trends, the most sociodemographically vulnerable populations in the region have been found to be the main victims of the diabetes epidemic, namely those of lower socioeconomic status, females, and the elderly.4-6 Diabetes is now commonly recognized as a coronary heart disease risk equivalent6-8 and believed to be one of the major factors accounting for the high percentage of deaths among diabetics due to cardiovascular disease (CVD). 9
Several studies7-11 showed that the differences in the lipid profile between diabetics (especially type 2 diabetics) and nondiabetics account for the increased CVD risk. Naturally, type 2 DM (T2DM) lipid profiles consist of elevations in triglyceride (TG) levels (>2 mmol/L) and reductions in high-density lipoprotein cholesterol (HDL-C). Statins are considered the first pharmacological line of treatment of dyslipidemia in diabetic patients. 12 However, various studies reported randomized trials among various ethnicities have documented that rosuvastatin is the most effective statin at reducing low-density lipoprotein cholesterol (LDL-C) and TG and at raising HDL-C levels.7,13-17 There are relatively few studies on the effects of statin treatment in altering lipid profiles in patients with the metabolic syndrome. 7 The Measuring Effective Reductions in Cholesterol Using Rosuvastatin Therapy (MERCURY) study was a large-scale randomized trial comparing low doses of rosuvastatin with commonly used doses of other statins in helping high-risk patients achieve the European and Adult Treatment Panel III (ATP III) LDL-C goals.7,13-15,18,19
The current study suggests that the metabolic syndrome increases the risk of coronary heart disease. Yet few data are available on the effects of statin treatment in improving lipid measures in patients with the metabolic syndrome (MetS). This analysis compares the effects of statin therapy on plasma LDL-C goal achievement and lipid levels in hypercholesterolemic patients with or without the metabolic syndrome. This is probably the only second or third article that has aimed at determining the efficacy of the 4 most commonly prescribed statins (rosuvastatin, atorvastatin, pravastatin, and simvastatin) for managing dyslipidemia among diabetic patients with and without metabolic syndrome (MetS).
Subjects and Methods
This was a cohort observational with routine care population-based study conducted at Hamad Medical Hospital and Primary Health Care (PHC) Centre. The study was based on 1542 consecutive diabetes patients who were diagnosed with dyslipidemia and prescribed any of the indicated statins to Hamad General Hospital and PHC, diabetic clinics, and underwent fasting blood test of lipid profile before initiation of statin treatment. The diagnosis and when to treat dyslipidemia in diabetics in Hamad General Hospital are established on the basis of the World Health Organization expert group 20 criteria and the World Health Organization recommends screening for lipid disorders at least annually in diabetic patients, and more often if needed to achieve goals. All lipid parameters were quantified on samples collected in the fasting state. Cholesterol and TG quantitation was determined by enzymatic assay. LDL-C was calculated using the Friedewald equation for patients with TG ≤400 mg/dL and measured by β-quantification for those with TG >400 mg/dL. Levels of non-HDL-C were calculated by subtraction of HDL-C from total cholesterol. Additionally, adults with low-risk lipid values (LDL-C <100 mg/dL [2.6 mmol/L], HDL-C >50 mg/dL [1.3 mmol/L], and TG <150 mg/dL [1.7 mmol/L]) may be screened every 2 years.
In this study, the inclusion criteria included type 1 and type 2 diabetes patients (fasting blood glucose ≥126 mg/dL (7.0 mmol/L) who were prescribed any of the indicated statins (rosuvastatin, atorvastatin, pravastatin, and simvastatin) during 2005-2011 (2 years within this period) to ensure at least 2 years of using the statin; aged ≥18 years, and had a total cholesterol level of ≥4 mmol/L, LDL-C ≥2.5 mmol/L, HDL-C ≤1 mmol/L in men and ≤1.2 mmol/L in women, and fasting triglycerides ≥1.7 mmol/l, obtained within 1 week before the first use of statins, which will be then compared at first and second year intervals.
Furthermore, this study excluded patients with genetic disorders, pregnant women, patients on other concurrent lipid-lowering agents such as bile acid sequestrants; patients with previous history of angina, severe vascular disease, or other life-threatening disease; patients with nephropathy and/or hypothyroidism or active liver disease and/or bile duct problems; patients with creatine kinase levels >10 × ULN; patients taking concurrent corticosteroids, cyclosporine, and/or hormone replacement therapy; patients who are physically inactive; and patients with a history of drug or alcohol abuse.
There are drug monitoring guidelines monitored by Physicians and Clinical Pharmasists. The choice of statins at the Hamad Medical Corporation is based on the drug monitoring procedure and doctors’ knowledge and experience. If a drug has side effects, the physician is eligible to change to another statin medication. In fact, clinical check-up at 2-month intervals were performed to monitor the patient compliance with regard to statins.
The study was approved by the Hamad General Hospital, Hamad Medical Corporation. All human studies have been approved by the Institutional Review Board, Research Ethics Committee HMC RP# 11212/11 and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Patients Data Collection Instrument
A total of 1542 consecutive diabetes patients who were diagnosed with dyslipidemia and prescribed any of the indicated statins between January 2007 and September 2012 were included in the study. Patients were identified as statin users (rosuvastatin, atorvastatin, pravastatin, and simvastatin) during the study period by using the pharmacy database. Data collection sheets were designed by the researcher for each patient. Patients were included in the study for each statin dose, and these had to have been on the same dose for at least 2 years between the periods indicated above. The data for these patients were obtained from 2 sources, that is, the electronic medical database called (EMR-viewer) and the patients’ files from the Medical Records Department of Hamad Medical Corporation (HMC) and PHC Centre. The prescribed doses in HMC and PHC Center for diabetic patients with dyslipidemia are as follows:
Atorvastatin 10 mg (n = 150), 20 mg (n = 200), and 40 mg (n = 140); N = 490
Pravastatin 20 mg (n = 1740 and 40 mg (n = 160); N = 274
Rosuvastatin 10 mg (n = 300) and 20 mg (n = 222); N = 522
Simvastatin 20 mg; N = 196
In order to be classified as having MetS in this analysis,5,7,18 patients had to meet 3 or more of the following characteristics: body mass index >30 kg/m2, TG ≥1.69 mmol/L (150 mg/dL), HDL-C <1.03 mmol/L (40 mg/dL) in men or <1.29 mmol/L (50 mg/dL) in women, blood pressure ≥130/≥85 mm Hg or current antihypertensive medication use, and fasting blood glucose ≥6.1 mmol/L (110 mg/dL). These criteria are identical to those listed in the ATP III.7,8 In a retrospective analysis, achievement of LDL-C goals specified by ATP III guidelines, in addition to changes in lipid parameters, was assessed in patients with and without MetS in each treatment group. ATP III LDL-C goals are <4.1 mmol/L (160 mg/dL) in patients with no or 1 risk factor and no CHD, <3.4 mmol/L (130 mg/dL) for patients with 2 or more risk factors and 10-year CHD risk of ≤20%, and <2.6 mmol/L (100 mg/dL) for patients with CHD or CHD equivalents, that is, other atherosclerotic diseases, diabetes, or multiple risk factors and a 10-year CHD risk of >20%.7,8 Baseline values for LDL-C and other lipid measures were calculated by averaging measurements obtained at weeks −2, −1, and 0. Lipids were subsequently measured at 8 weeks. The patients are monitored regularly with the pharmacy database with the number of tablets prescribed. Meanwhile, the diabetic patients with MetS enrolled in the study were chosen based on their blood sugar levels, HbA1C, (glycated hemoglobin), and antidiabetic drugs (oral, or insulin or antihyperglycemic agents). Therefore, HbA1C has been considered as indicator for controlled and uncontrolled patients, that is, HbA1C ≤7 controlled and HbA1c >7 uncontrolled.
The data collection form included sociodemographic characteristics such as age, gender, nationality, height, weight, consanguineous marriage, physical activity, lifestyle habits like smoking and alcohol, type of statin prescribed for the patient for at least 2 years continuously and dose of statin, type of DM, its duration and presence of hypertension, clinical and biochemistry laboratory investigations such as fasting blood glucose, HbA1C, total cholesterol, HDL, LDL cholesterol levels, TG, creatine kinase level, serum creatinine, bilirubin, liver function test, gamma-glutamyl transferase, serum albumin, and so on, and adverse events like microalbuminuria and macroalbuminuria. Finally, the study included the initiation of antihypertensive medications (angiotensin receptor blockers and/or angiotensin-converting enzyme inhibitors). Since the consanguinity rate was very high (52%) among Qatari population, this ethnic group is very important.
Student’s t test was used to ascertain the significance of differences between mean values of 2 continuous variables and confirmed by nonparametric Mann–Whitney test. Chi-square test and Fisher’s exact test were performed to test for differences in proportions of categorical variables between 2 or more groups. A nonparametric statistical method, the Kruskal–Wallis one-way analysis of variance, was performed to evaluate differences among different characteristic groups. The level P < .05 was considered as the cutoff value for significance.
Results
Out of total 1542 subjects, 562 (36.4%) were diagnosed with MetS using the international Diabetes Federation criteria. Among those with MetS, 125 were prescribed with atorvastatin, 162 pravastatin, 177 rosuvastatin, and 98 simvastatin. In contrast, among those without MetS, 365 used atorvastatin, 172 pravastatin, 345 rosuvastatin, and 98 simvastatin. Table 1 shows the baseline sociodemographic, anthropometric, and lifestyle characteristics in patients with and without MetS. There was no statistically significant difference in terms of gender, consanguinity, and smoking status, mild and vigorous physical activity across different types of statin therapies in patients with and without MetS. On the other hand, the proportion of patients with Qatari nationality was significantly higher in atorvastatin therapy group than other therapies among both groups (with and without MetS; P < .05).
Baseline Sociodemographic, Anthropometric, and Lifestyle Characteristics in Patients With and Without Metabolic Syndrome (N = 1542). a
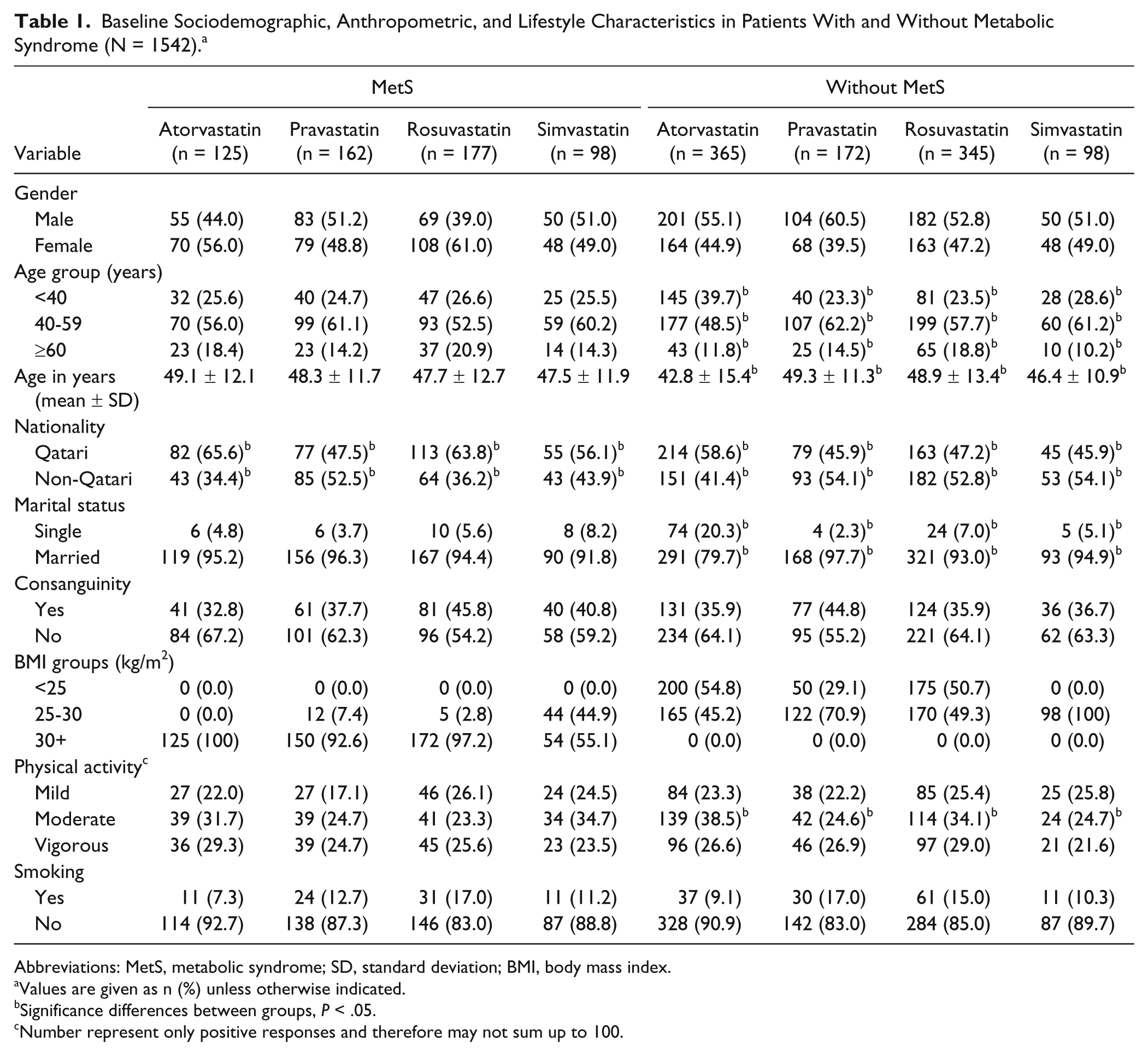
Abbreviations: MetS, metabolic syndrome; SD, standard deviation; BMI, body mass index.
Values are given as n (%) unless otherwise indicated.
Significance differences between groups, P < .05.
Number represent only positive responses and therefore may not sum up to 100.
Table 2 shows comparison of baseline comorbidities, biochemistry, and lipid levels among patients with and without MetS. The proportion of comorbidities such as type 1 diabetes, type 2 diabetes, and hypertension remained similar across different types of statins among patients with MetS. However, among those without MetS, the proportion of type 1 diabetes was significantly higher in atorvastatin group and proportion of type 2 diabetes was higher in simvastatin group than the other statin therapies (P < .05). There was also significant difference in terms of the duration of diabetes across different statin therapies among patients without MetS. Nevertheless, average values of total cholesterol, LDL-C, HDL-C, and TG remained similar across different statin therapies in both patient categories with and without MetS.
Baseline Comorbidities, Biochemistry, and Lipid Levels in Patients With and Without Metabolic Syndrome (N = 1542).
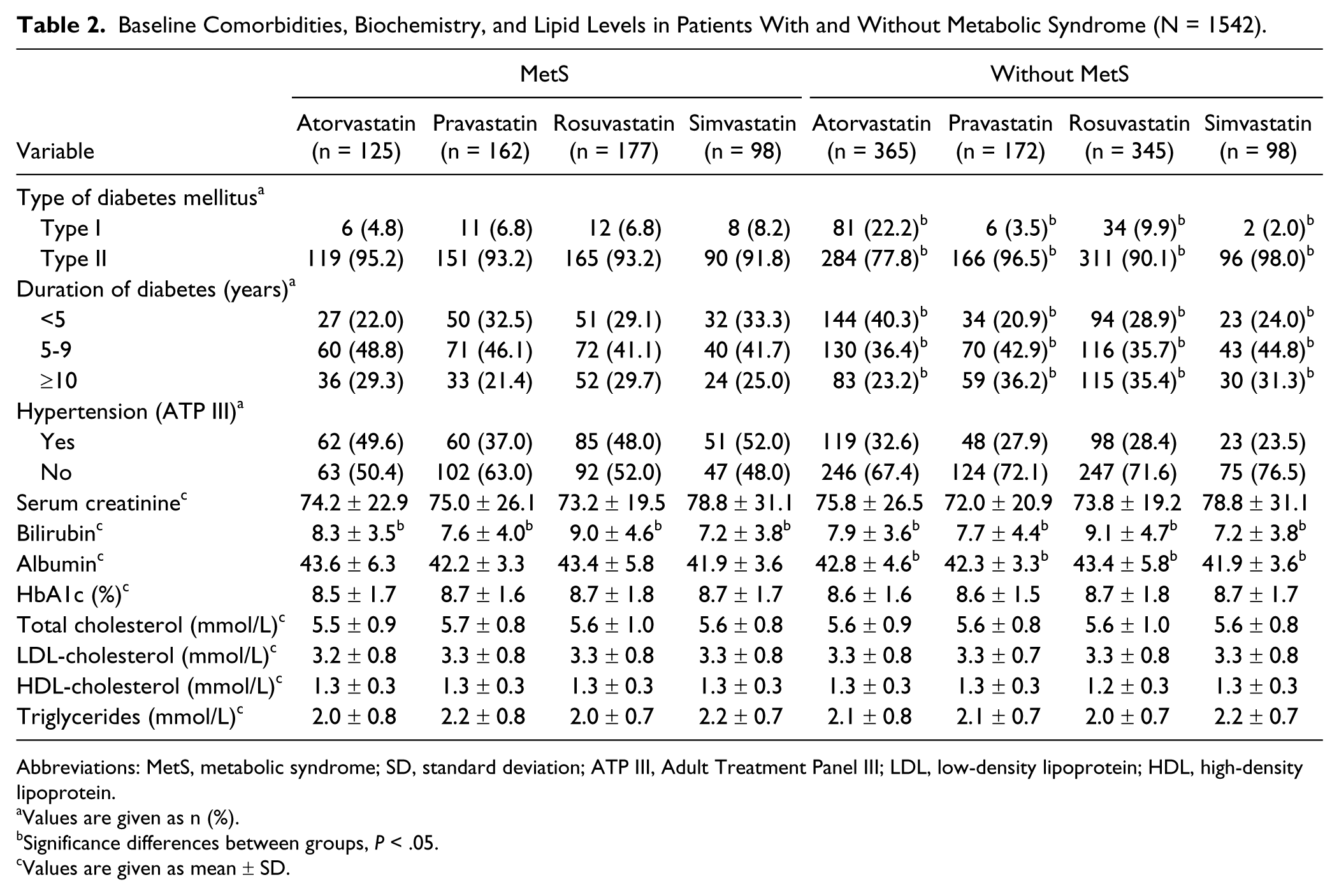
Abbreviations: MetS, metabolic syndrome; SD, standard deviation; ATP III, Adult Treatment Panel III; LDL, low-density lipoprotein; HDL, high-density lipoprotein.
Values are given as n (%).
Significance differences between groups, P < .05.
Values are given as mean ± SD.
Table 3 presents the comparison of mean percentage change after 2 years of therapy from the baseline in lipid measures between patients with and without MetS. Among patients with MetS, rosuvastatin therapy resulted in significantly higher LDL-C and total cholestrol reduction (23%, P = .006; and 20.3%, P = .015, respectively) as compared with other statins. Rosuvastatin therapy was closely followed by pravastatin in reducing LDL and total cholesterol; however, atorvastatin was found to be the least effective in reducing both LDL and total cholesterol as compared with other statins among patients with MetS. Similarly, significantly higher percentage of patients receiving rosuvastatin therapy were successful in achieving the target of total cholesterol <4 mmol/L and TG <1.7mmol/L after 2 years (38.4%, P = .012; and 67.2%, P = .010, respectively) as compared with other therapies. In contrast, among patients without MetS, Rosuvastatin therapy resulted in highest percentage drop in total cholesterol (20.1%, P = .016) than other statins. Unlike patients with MetS, among those without MetS, rosuvastatin was closely followed by atorvastatin therapy in reducing lipid measures and simvastatin was found to be the least effective therapy in reducing lipids.
Comparison of Mean Percentage Change (SE) From Baseline in Lipid Measures Between Patients With and Without Metabolic Syndrome (N = 1542).

Abbreviations: MetS, metabolic syndrome; LDL, low-density lipoprotein; HDL, high-density lipoprotein; TG, triglycerides.
LDL cholesterol <2.5 mmol/L (100 mg/dL) for patients with cardiovascular disease and/or type 2 diabetes.
Figure 1 shows the percentage of patients across different statin therapies in achieving European LDL-C goal (LDL-C <2.5 mmol/L for patients with CVD and/or diabetes) among those with and without MetS. Rosuvastatin therapy in patients, both with and without MetS, showed the highest proportion of those achieving the European LDL-C goal.

Percentage of patients with and without metabolic syndrome meeting European low-density lipoprotein cholestrol (LDL-C) goal of <4.0 mmol/L (for patients with diabetes and/or cardiovascular disease [CVD]), according to different types of statin therapy.
Furthermore, the cost-effectiveness of statin drugs in Qatar were also investigated. The yearly acquisition costs were lower for rosuvastatin 10 mg compared with atorvastatin 20 mg (€758 vs €962) in the State of Qatar, and this is consistent and confirmative with the Western countries (United States, $959.95 vs $1204.50 and the United Kingdom, £235.03 vs £321.20). 19
Discussion
The State of Qatar in the Middle East region is predicted to have one of the highest prevalences of T2DM in the world. However, data are lacking regarding the efficacy and safety of the 4 most commonly prescribed statins (rosuvastatin, atorvastatin, pravastatin, and simvastatin) for managing dyslipidemia among diabetic patients. 12 In the current study, rosuvastatin was found to be the most effective statin at reducing LDL-C when compared with atorvastatin and simvastatin. The findings of this study are consistent with the previous reported studies10-13,15,18,19 in that rosuvastatin at its lowest dose in this study (10 mg) was more effective in reducing LDL-C levels than atorvastatin and pravastatin at their highest doses (40 mg). Indeed, it should be noted that rosuvastatin, which is the latest statin to receive approved labeling by the Food and Drug Administration, 15 has been consistently found to be the most effective at reducing LDL-C levels in the most recent studies comparing its efficacy with other statins. 19
The STELLAR trial,13,15,18 MERCURY trial,7,19 and PULSAR 19 are the major open-label, randomized, multicenter trials to compare rosuvastatin (10, 20, 40, or 80 mg) with atorvastatin (10, 20, 40, or 80 mg), pravastatin (10, 20, or 40 mg), and simvastatin (10, 20, 40, or 80 mg) across dose ranges for reduction of LDL-C. 15 The results of the STELLAR trial revealed that rosuvastatin was consistently, across all doses, the most effective at reducing LDL-C levels in comparison to all of the other statins. The MERCURY7,19 and STELLAR studies13,15,18 reported that rosuvastatin therapy is effective in achieving LDL-C goal and in improving the lipid profile in hypercholesterolemic and high-risk diabetic patients. This is consistent with the current study performed in Qatar.
Despite the proven benefits of LDL-C reduction, lipid management is suboptimal and many patients fail to achieve recommended LDL-C goals.21-23 Perhaps the most likely reason for this is the use of agents with a poor efficacy for LDL-C lowering and suboptimal dose titration. The most effective statin at the lowest dose would represent a simple, effective treatment strategy, enabling more patients to achieve goals without the need for dose titration. 19 The effective statin at the lowest dose in current study is consistent with the previous reported studies.19,21-23
It has been reported by several studies9,14,18 that the lowering of TG is another important goal in reducing CVD risk among diabetic patients. In the current study, the greatest reduction in TG was −20.6% and was achieved by patients taking rosuvastatin. However, it is important to note that atorvastatin both achieved the second highest reduction in TG among those with and without MetS. These findings are similar to the majority of studies in the literature, which have shown a slightly higher reduction in TG in patients taking rosuvastatin in comparison with atorvastatin.7,19,13,15,18,19 It thus appears that in relation to this factor (TG) that both rosuvastatin and atorvastatin are effective at reducing TG.
Additionally, in most cases raising HDL-C levels is another major factor known to reduce CVD risk as reported by some studies. 11 In the current study, all the statins appear to have reduced rather than raised HDL-C levels. Rosuvastatin had the least reduction of and would thus be regarded as the most effective; however, none of the values for the statins were significant. The current study is consistent with the previous studies and trials such as the MERCURY trial,7,19 STELLAR trial,13,15,18 and PULSAR, 19 which investigated starting doses of rosuvastatin and atorvastatin and found that the increase in HDL-C was significantly greater statistically with low dosage rosuvastatin than with high dose of atorvastatin.
Furthermore, more recently, the VOYAGER Database study 11 investigated the effects of different statins on HDL-C levels, relationships between changes in HDL-C and changes in LDL-C, and meta-analysis of 32 258 dyslipidemic patients included in 37 randomized studies using rosuvastatin, atorvastatin, and simvastatin. The HDL-C raising ability of rosuvastatin and simvastatin was comparable, with both being superior to atorvastatin. Increases in HDL-C were positively related to statin dose with rosuvastatin and simvastatin but inversely related to dose with atorvastatin. The analysis also revealed that the HDL-C raising achieved by all 3 statins was totally independent of the reduction in LDL-C. And finally, it has been found that baseline concentrations of HDL-C and plasma TG and the presence of diabetes are robust, independent predictors of statin-induced elevations of HDL-C.11,14
More recently, various studies7,18 reported that patients with MetS had greater reductions in TG and somewhat greater percentage increases in HDL-C with statin treatment, as expected. The comparisons between statin treatment groups showed consistent advantages of rosuvastatin treatment, compared with atorvastatin, simvastatin, and pravastatin, in LDL-C goal achievement and in LDL-C, total cholesterol, and non-HDL-C reduction. As in the main study analysis, rosuvastatin 10 mg provided benefits comparable to a higher dose of atorvastatin in the MetS population. It is worth noting that a pharmacoeconomic analysis of the primary MERCURY results7,18 showed that treatment with rosuvastatin 10 mg was more cost-effective compared with equivalent or higher doses of atorvastatin, simvastatin, and pravastatin, and that switching patients from a comparator statin to rosuvastatin improved LDL-C goal attainment at relatively little additional cost, with equivalent (or lower) associated drug costs.7,18 Thus, rosuvastatin may have pharmacoeconomic advantages, compared with atorvastatin, while providing comparable efficacy.
Finally, rosuvastatin at a low dose has demonstrated high efficacy for LDL-C lowering, enabling patients with hypercholesterolemia to achieve their lipid goals.13,18 In addition, rosuvastatin has beneficial effects on other components of the lipid profile, including HDL-C,13,24-27 which is a major, independent risk factor for CVD.8,25 Safety data from several large-scale clinical and pharmacoepidemiologic studies have shown that the safety of rosuvastatin25-27 and results from the current recent study also support these findings. 19
Conclusion
The present study confirmed that rosuvastatin therapy in commonly prescribed doses is the most effective statin for LDL-C goal achievement and for improving the lipid profile in hypercholesterolemic diabetic patients with and without MetS.
Footnotes
Acknowledgements
The authors would like to thank Hamad Medical Corporation, Qatar Diabetes Association, and Qatar Foundation for their support and ethical approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was generously supported and funded by the Hamad Medical Corporation (Grant No. HMC and IRB RP# 11212/11, RP# 10262-1027A/2011), Qatar Diabetes Association, and Qatar Foundation.
