Abstract
Objective
Evidence of therapy for dysfunctional coronary circulation in patients with ST-segment elevation myocardial infarction (STEMI) undergoing primary percutaneous coronary intervention (pPCI) is limited. This study was performed to compare the effects of atorvastatin and rosuvastatin on dysfunctional coronary circulation.
Methods
This retrospective study enrolled 597 consecutive patients with STEMI who underwent pPCI in 3 centers from June 2016 to December 2019. Dysfunctional coronary circulation was defined by the thrombolysis in myocardial infarction (TIMI) grade and the TIMI myocardial perfusion grade (TMPG). Logistic regression analysis was used to evaluate the impact of different statin types on dysfunctional coronary circulation.
Results
The incidence of TIMI no/slow reflow did not differ between the two groups, but the incidence of TMPG no/slow reflow was significantly lower in the atorvastatin than rosuvastatin group (44.58% vs. 57.69%, respectively). After multivariate adjustment, the odds ratio with 95% confidence interval of rosuvastatin was 1.72 (1.17–2.52) for after pretreatment TMPG no/slow reflow and 1.73 (1.16–2.58) for after stenting TMPG no/slow reflow. Atorvastatin and rosuvastatin showed no significant differences in clinical outcomes during hospitalization.
Conclusions
Compared with rosuvastatin, atorvastatin was associated with better coronary microcirculatory perfusion in patients with STEMI who underwent pPCI.
Keywords
Introduction
Dysfunctional coronary circulation occurs in nearly half of patients with ST-segment elevation myocardial infarction (STEMI) treated with primary percutaneous coronary intervention (pPCI), leading to an adverse clinical prognosis.1–3 On angiography, dysfunctional coronary circulation often presents as no/slow reflow. Distal embolization of atherosclerotic debris is a key event in the pathogenesis of dysfunctional coronary circulation after pPCI. 4 In patients with STEMI, especially those with a high thrombus burden (HTB), pPCI physically breaks the thrombus and plaque, generating lipid bubbles, microthrombi, and other substances that mechanically embolize the distal capillaries; this process results in dysfunctional coronary circulation. 5 In addition, ischemia–reperfusion injury induced by revascularization during pPCI is often accompanied by a high inflammatory response,6,7 which is also associated with a large infarct size and poor clinical outcomes.8,9 Many adjuvant therapies have been proposed to achieve adequate coronary microvascular perfusion. 3 There is evidence that in addition to lowering blood lipid levels, statins have multiple other protective effects such as protecting endothelial function, stabilizing atherosclerotic plaques, and reducing inflammation. 10 Previous studies have shown that pretreatment with statins may improve microvascular myocardial perfusion in patients with acute coronary syndrome (ACS), even in the setting of short-term preoperative administration.11–14
Atorvastatin and rosuvastatin, which are high-intensity statins commonly administered to patients with coronary artery disease, have pleiotropic effects beyond the lipid-lowing effect. 10 These pleiotropic effects may contribute to the prevention of dysfunctional coronary circulation after STEMI.15,16 In a real-world study, Rahhal et al. 17 reported that atorvastatin showed a cardioprotective effect similar to that of rosuvastatin in patients with ACS. Although a recent meta-analysis showed that atorvastatin significantly reduces major adverse cardiovascular and cerebrovascular events compared with rosuvastatin in patients with STEMI, 18 which statin is more effective in improving dysfunctional coronary circulation after pPCI in patients with STEMI remains unknown.
Few studies have compared the prognosis of patients with STEMI (coronary microvascular function and in-hospital adverse events) treated with different statins. In terms of pharmacokinetics, the time to peak plasma concentration of atorvastatin is about 1 hour, 19 and that of rosuvastatin is within 3 to 5 hours. 20 The aim of therapy with these drugs is to rapidly recover the coronary microcirculation. Therefore, this study was performed to compare the effects of atorvastatin and rosuvastatin on the coronary microcirculation and clinical outcomes in patients with STEMI who underwent pPCI.
Materials and methods
The Ethics Committee of Guangdong Provincial People’s Hospital approved the study (approval number GDREC2018346H R2), which was performed in line with the principles of the Declaration of Helsinki. All participants provided verbal informed consent to participate in the study, and acquisition of this consent was supported by the Ethics Committee of Guangdong Provincial People’s Hospital. All patient details have been deidentified. The reporting of this study conforms to the STROBE guidelines. 21
Study population and data collection
This retrospective observational study involved consecutive patients diagnosed with STEMI who underwent pPCI from June 2016 to December 2019 at the following three centers: Jiexi People’s Hospital, Zhuhai Golden Bay Center Hospital, and Guangdong Provincial People’s Hospital. Figure 1 shows the flowchart of the study. All participants were required to meet the following inclusion criteria: (1) age of ≥18 years and diagnosis of STEMI, (2) onset of ischemia <12 hours before presentation or 12 to 48 hours before presentation with worsening ischemia symptoms, and (3) treatment by recommended pPCI and stent implantation. The exclusion criteria were (1) no statin therapy; (2) no stent implantation; (3) location of the culprit vessel at the left main artery; (4) arterial infarct due to in-stent occlusion/restenosis, coronary artery bypass graft occlusion/stenosis, or coronary artery dissection; and (5) allergy to contrast agent. The diagnosis of STEMI was based on satisfaction of at least two of the following three criteria: (1) typical signs of elevated cardiac biomarkers, including creatine kinase, creatine kinase-myocardial band (CK-MB), and troponin, which were proportionally increased; (2) typical signs of ST-segment elevation in two adjacent leads on an electrocardiogram; and (3) typical symptoms of chest pain. Atorvastatin (20 mg) or rosuvastatin (10 mg) was given immediately after diagnosis of STEMI in the emergency room. In total, 693 patients were initially enrolled; 27 patients treated with other types of statins or not taking statins and 69 patients without stent implantation were subsequently excluded.

Study flowchart.
Two authors were responsible for collecting all the data from the electronic medical record database.
Definitions and laboratory examination
Thrombolysis in myocardial infarction (TIMI) no/slow reflow (epicardial no/slow reflow) was defined as TIMI grade <3 flow after pretreatment (percutaneous transluminal coronary angioplasty [PTCA] with or without thrombus aspiration) or after stent implantation. 22 TIMI myocardial perfusion grade (TMPG) no/slow reflow (microvascular no/slow reflow) was considered as TMPG <3 after pretreatment (PTCA with or without thrombus aspiration) or after stent implantation. 23 HTB was defined as a thrombus score of >2 24 based on the definition provided by the TIMI study group.
Hypertension was diagnosed when the systolic blood pressure was ≥140 mmHg and/or diastolic blood pressure was ≥90 mmHg, which is equivalent to a self-monitoring blood pressure average of ≥135/85 mmHg at home twice a day for at least 3 days. 25 Type 2 diabetes mellitus (T2DM) was diagnosed based on a medical history of T2DM or a fasting plasma glucose level of ≥7.0 mmol/L (126 mg/dL) and hemoglobin A1c level of ≥6.5%/positive oral glucose tolerance test (2-hour plasma glucose of ≥11.1 mmol/L [200 mg/dL]). 26 Previous myocardial infarction and chronic kidney disease were diagnosed based on the patient’s medical history, and current smoking was defined by self-reporting. Major adverse cardiovascular events (MACEs) were defined as target vessel revascularization, ischemic stroke, recurrent myocardial infarction, and cardiac death. The levels of total cholesterol, triglyceride, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, and creatinine were measured using immunoturbidimetry or colorimetry. The CK-MB level was measured by an enzyme dynamic method. The levels of cardiac troponin I (cTNI), cardiac troponin T (cTNT), N-terminal pro-hormone brain natriuretic peptide (NT-proBNP), and brain natriuretic peptide (BNP) were detected using electrochemiluminescence. To allow for easy comparison, the cTNT and cTNI values were divided into those above or below 100 times the upper limit of detection, while the BNP and NT-proBNP values were divided into those above or below 10 times the upper limit of detection.
Coronary angiography and percutaneous coronary intervention
Coronary angiography was performed using the Judkins technique with a radial or femoral approach. The use of thrombus aspiration and/or PTCA for pretreatment and the use of glycoprotein IIb/IIIa inhibitors (GPIs) was at the operator’s discretion. After pretreatment (PTCA with or without thrombus aspiration) and after stent angiography, TIMI/TMPG blood flow data were collected. Severe stenosis was defined as the presence of ≥50% stenosis in the left main coronary artery and/or ≥70% stenosis in the right coronary artery, left circumflex branch artery, or left anterior descending branch artery. Multivessel disease was defined as >70% stenosis of one or two other coronary arteries in addition to the culprit vessel.
Quantitative coronary angiography analysis
Two experienced observers from the Angiographic Laboratory of Guangdong Provincial People’s Hospital were responsible for anonymous analysis of offline angiography image data. Interobserver errors were determined by a third observer. QAngio XA 7.2 software (Medis Medical Imaging Systems, Leiden, Netherlands) was used on the Create Life workstation to conduct quantitative coronary angiographic evaluation of all lesions at the best diastole lesion exposure site. We used standard techniques to measure the proximal reference, distal reference, minimum lumen diameter, reference diameter, minimum lumen area, percentage area stenosis, percentage diameter stenosis, and lesion length. 27
Echocardiographic analysis
Experienced sonographers performed transthoracic echocardiography using a GE Vivid E95 system (GE Healthcare, Milwaukee, WI, USA), which was connected to 2.5- to 3.5-MHz phased-array probes. The left ventricular ejection fraction was measured using Simpson’s method. Systolic dysfunction was defined as a left ventricular ejection fraction of <54% for women and <52% for men.
Statistical analysis
Statistical analysis was performed in three steps. First, independent-sample t-tests were performed for normally distributed data, the chi-square test or Fisher’s exact test was used to identify significant differences in categorical variables between groups, and the Mann–Whitney U test was performed for skewed data. Second, multivariate logistic regression was used to evaluate the association between the type of statin used and no/slow reflow, and 95% confidence intervals (CIs) and odds ratios (ORs) were computed. Potential confounding factors that were statistically or clinically significant in the univariate analysis were included in the multivariate model. Third, considering the effect of unbalanced sample sizes between the two groups, a propensity score analysis was executed to reduce the selection bias. The propensity score-matching (PSM) ratios were 1:1 and 1:2 (rosuvastatin vs. atorvastatin) with a caliper width equal to 0.1. Subgroup analysis was conducted to examine the effect of the two statins on TMPG no/slow reflow among various subgroups, including age (<60 vs. ≥60 years), sex (male vs. female), hypertension (no vs. yes), T2DM (no vs. yes), currently smoking (no vs. yes), dual antiplatelet therapy (aspirin plus clopidogrel vs. aspirin plus ticagrelor), onset-to-door time (<12 vs. ≥12 hours), onset-to-balloon time (<12 vs. 12–24 vs. >24 hours), Killip class (I vs. II–IV), HTB (no vs. yes), multivessel disease (no vs. yes), and thrombus aspiration (no vs. yes). We tested the interactions and modifications of the subgroups with the likelihood-ratio test. We calculated the required sample size (1:1) for detecting a difference in the incidence of TMPG no/slow reflow between the two groups at α = 0.05 and 70% power, and the result was 179 patients in each group. The proportion of missing data in the sample for analysis was not greater than 5%; missing data were interpolated using the mean or median value. Two-sided p-values of <0.05 were defined as statistically significant. Stata version 15.0 (StataCorp LP, College Station, TX, USA), R version 3.4.3 (R Foundation for Statistical Computing, Vienna, Austria), and EmpowerStats (http://www.empowerstats.com) were used to perform the statistical analyses.
Results
Baseline characteristics
The baseline information of the two groups before and after PSM is shown in Table 1 and Supplementary Table 1, respectively. This study included 597 patients (83.4% male), among whom 415 (69.5%) used atorvastatin and 182 (30.5%) used rosuvastatin. Patients in the atorvastatin group more often used aspirin plus clopidogrel (84.34% vs. 75.82%) and had a higher BNP/NT-proBNP level (114 [27.47%] vs. 32 [17.58%], with BNP/NT-proBNP being >10-fold higher) than patients in the rosuvastatin group. The rosuvastatin group was more often treated with GPIs (68.68% vs. 59.04%) and had a higher cTNI/cTNT level (145 [79.67%] vs. 289 [69.64%], with cTNI/cTNT being >10-fold higher). There were no statistically significant differences in age, sex, T2DM, hypertension, current smoking, previous statin use, onset-to-door time, previous myocardial infarction, culprit vessel distribution, peak CK-MB level, or lesion characteristics between the atorvastatin and rosuvastatin groups.
Baseline characteristics.
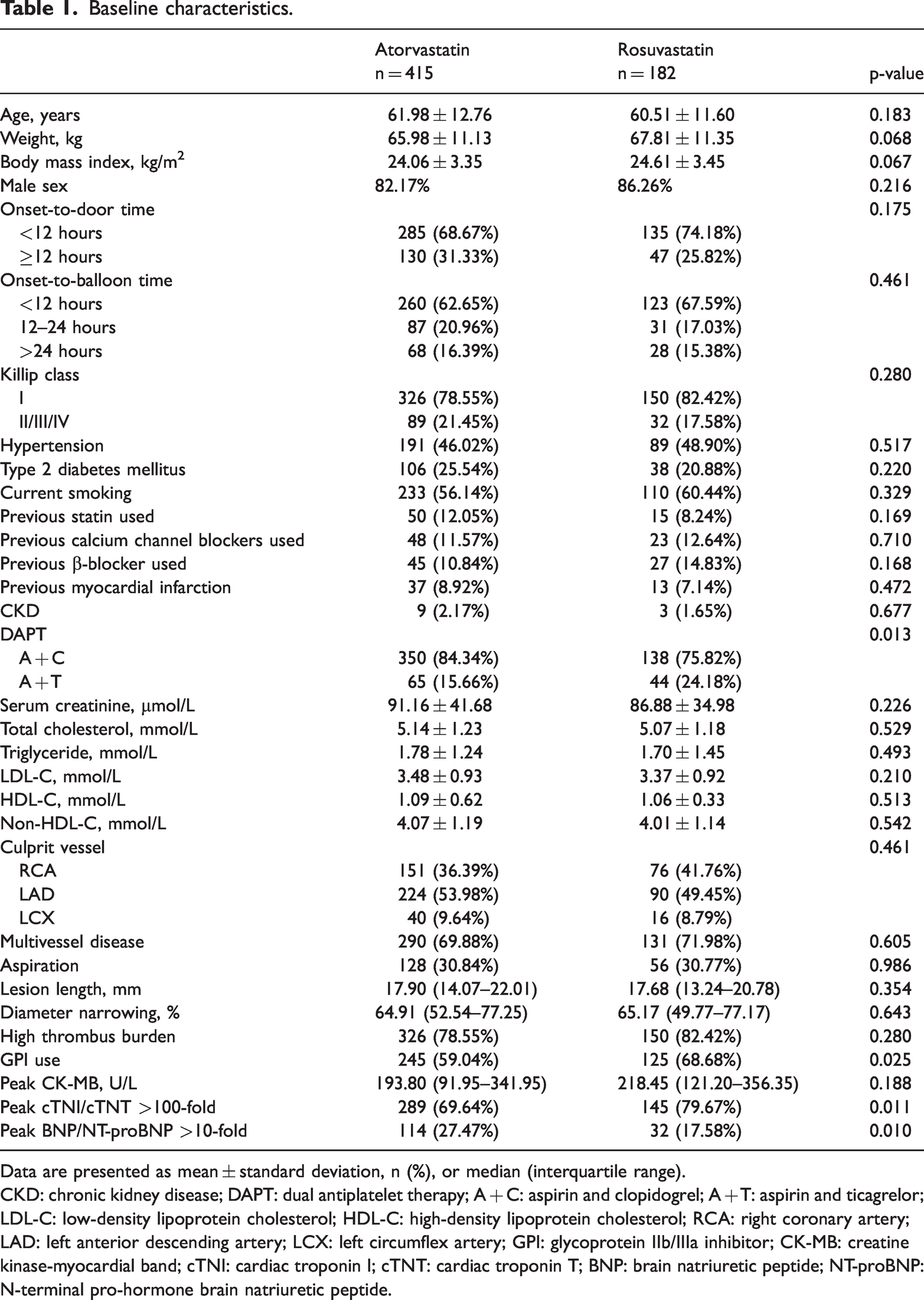
Data are presented as mean ± standard deviation, n (%), or median (interquartile range).
CKD: chronic kidney disease; DAPT: dual antiplatelet therapy; A + C: aspirin and clopidogrel; A + T: aspirin and ticagrelor; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; RCA: right coronary artery; LAD: left anterior descending artery; LCX: left circumflex artery; GPI: glycoprotein IIb/IIIa inhibitor; CK-MB: creatine kinase-myocardial band; cTNI: cardiac troponin I; cTNT: cardiac troponin T; BNP: brain natriuretic peptide; NT-proBNP: N-terminal pro-hormone brain natriuretic peptide.
Comparison of coronary microvascular function between atorvastatin and rosuvastatin groups
As shown in Figure 2, although TIMI blood flow did not differ between the groups, the incidence of both after pretreatment no/slow reflow (40.48% vs. 53.85%, p = 0.002) (Supplementary Table 2) and after stenting TMPG no/slow reflow (26.75% vs. 39.56%, p = 0.002) (Supplementary Table 2) was significantly higher in the rosuvastatin group than in the atorvastatin group. Table 2 presents the association of TMPG no/slow reflow and statin type among the patients. In unadjusted Model 1, the OR (95% CI) of rosuvastatin was 1.71 (1.21–2.44, p = 0.003) for after pretreatment TMPG no/slow reflow and 1.79 (1.24–2.59, p = 0.002) for after stenting TMPG no/slow reflow. After adjusting for demographic, clinical, procedural, and biochemical characteristics in Model 2, rosuvastatin was found to be significantly correlated with TMPG no/slow reflow after pretreatment (OR [95% CI]: 1.72 [1.17–2.52], p = 0.006) and after stenting (OR [95% CI]: 1.73 [1.16–2.58], p = 0.008). In the subgroup analysis, we noted that in the aspirin plus clopidogrel subgroup, more TMPG no-reflow (OR [95% CI]: 1.96 [1.31–2.92]) was also found in the rosuvastatin group than in the atorvastatin group, and the same tendency (OR [95% CI]: 1.07 [0.49–2.29]) was found in the aspirin plus ticagrelor subgroup (Table 3).

Angiographic outcomes.
Association of TMPG no/slow reflow and statin type (atorvastatin as reference).

Model 1: Unadjusted.
Model 2: Adjusted for age, sex, weight, hypertension, type 2 diabetes mellitus, previous myocardial infarction, chronic kidney disease, smoking, onset-to-door time, onset-to-balloon time, Killip class, dual antiplatelet therapy, low-density lipoprotein cholesterol level, culprit vessel, multivessel disease, lesion length, diameter narrowing, high thrombus burden, thrombus aspiration, and use of glycoprotein IIb/IIIa inhibitor.
Model 3: Propensity score-matching analysis (1:1).
Model 4: Propensity score-matching analysis (1:2).
TIMI: thrombolysis in myocardial infarction; TMPG: TIMI myocardial perfusion grade; DF: degrees of freedom; OR: odds ratio; CI: confidence interval.
Subgroup analysis for TMPG no/slow reflow group.

TMPG: TIMI myocardial perfusion grade; OR: odds ratio; CI: confidence interval; DAPT: dual antiplatelet therapy; A + C: aspirin and clopidogrel; A + T: aspirin and ticagrelor.
Comparison of clinical outcomes between atorvastatin and rosuvastatin groups
There were no significant differences in the left ventricular ejection fraction (53% vs. 52%) or MACE (3.61% vs. 2.75%) during hospitalization between the atorvastatin and rosuvastatin groups (Supplementary Table 3). No significant differences were observed between the atorvastatin and rosuvastatin groups in all-cause death (1.20% vs. 1.10%) or acute heart failure (4.10% vs. 4.40%) (Supplementary Table 3).
PSM analyses
After 1:1 and 1:2 PSM to match the characteristics between the groups (Supplementary Figure 1), we conducted a sensitivity analysis. The demographic, clinical, and procedural characteristics were not significantly different between the two groups (Supplementary Table 1). The PSM analysis also showed that rosuvastatin was significantly correlated with TMPG no/slow reflow (Table 2).
Discussion
In the present study, the use of atorvastatin was associated with better recovery of coronary microvascular function than was rosuvastatin in patients with STEMI who underwent pPCI, but there was no significant difference in the clinical prognosis. Preoperative administration of atorvastatin may contribute to the alleviation of dysfunctional coronary circulation during pPCI in patients with STEMI.
Catheterization laboratory-derived coronary blood flow is a common and direct way to assess coronary microvascular function during pPCI for patients with STEMI. Prolonged contrast elution has been considered a classic angiographic marker of coronary microvascular damage, introducing the concept of TMPG. 23 Jang et al. 28 conducted a study evaluating coronary microvascular dysfunction in patients with STEMI and found that TMPG was more sensitive than TIMI for screening out microvascular dysfunction. Jia et al. 29 adopted TMPG of the intervened vessel to evaluate the benefits of high-dose statins on improving coronary microcirculation in patients with ACS. Although a previous meta-analysis showed that atorvastatin reduces MACE compared with rosuvastatin, 18 few studies have compared the effect of two statins on dysfunctional coronary circulation after pPCI in patients with STEMI. In our study, although the incidence of TIMI no/slow reflow did not differ between the groups, TMPG blood flow was significantly higher in the atorvastatin than rosuvastatin group, and the results remained consistent after multivariate adjustment. We also considered the effect of statins on coronary microvascular function beyond antiplatelet drugs by performing a subgroup analysis. In the aspirin plus clopidogrel group, TMPG no-reflow was more often observed in the rosuvastatin than atorvastatin group. However, the result was not significant in the aspirin plus ticagrelor subgroup, which may have been due to the relatively small sample size. Among the enrolled patients, most of the culprit lesions were accompanied by an HTB, which led to an increased incidence of dysfunctional coronary circulation and allowed for better comparison of the effects of the two statins. In addition, we observed higher GPI use and a higher peak cTNI/cTNT level in the rosuvastatin group, which was consistent with this group’s higher incidence of dysfunctional coronary circulation and severe myocardial injury. Based on these findings, our study suggests that preoperative administration of atorvastatin, rather than rosuvastatin, may prevent dysfunctional coronary circulation during pPCI to some extent.
The pathophysiology of periprocedural dysfunctional coronary circulation includes embolism of atheromatous or thrombotic debris, endothelial injury, and inflammation. 30 Atorvastatin has been shown to diminish microcirculatory impairment in patients undergoing PCI.31–33 Although the mechanism for this is not fully clear, it may result from ameliorated endothelial function, 34 improved stability of atherosclerotic plaques, 35 inhibition of the thrombogenic response, and decreased oxidative stress and vascular inflammation. 36 In the context of ACS, dysfunctional coronary circulation reflects the multifaceted pathophysiology of myocardial reperfusion deficits. 3 In the acute phase of myocardial injury, the myocardial protective and anti-inflammatory effects of statins go beyond the lipid-lowering effect. 14 In the acute phase of STEMI, statins provide cardioprotection by upregulating endothelial nitric oxide synthase to increase the level of endothelial nitric oxide, which is important for vasodilation, endothelium–leukocyte interactions, vascular smooth muscle proliferation, and platelet aggregation.37–39 Given the established relationship between coronary atherosclerosis and inflammation, statins express anti-inflammatory effects by reducing inflammatory cytokines such as interleukin, thus inhibiting proinflammatory biological processes such as monocyte chemotaxis and nuclear factor-κB activation.38,39 The pleiotropic effects of statins improve coronary microcirculation through multiple mechanisms.15,16 We found that pretreatment with atorvastatin reduced the occurrence of dysfunctional coronary circulation in patients with STEMI, highlighting the benefit of atorvastatin in the management of dysfunctional coronary circulation with short-term administration. This benefit may be multifaceted, covering common mechanisms of reperfusion disorder. Moreover, impaired TMPG is associated with a higher thrombus burden and larger infarct size than is normal TMPG. 5 In the present study, most patients had HTB, indicating an active tendency to develop distal thrombosis and severe inflammation, and the effectiveness of atorvastatin also made this therapy an advantage in HTB treatment.
Few studies to date have compared the efficacy of short-term application of atorvastatin and rosuvastatin in patients with STEMI. However, early administration of statins is associated with better clinical outcomes. 40 In our study, the onset-to-door time was similar in the two groups, indicating that there was no significant difference in the time of statin administration. Therefore, the drug characteristics of the two statins may be the main explanation for their discordant effects on dysfunctional coronary circulation. Because the molecular structures of statins are different, their mechanisms of action are also different. 38 On the one hand, the time to peak plasma concentration of atorvastatin is less than that of rosuvastatin; therefore, we assume that atorvastatin may rapidly improve the coronary microcirculation during pPCI.19,20 On the other hand, there is evidence suggesting that lipophilic statins (atorvastatin) have greater efficacy for reducing inflammation and improving short-term cardiovascular outcomes than hydrophilic statins (rosuvastatin).39,41 These differences might explain the superior effects of atorvastatin on coronary microcirculation in patients with STEMI.
Although we observed benefits of atorvastatin in improving dysfunctional coronary circulation after pPCI in patients with STEMI in terms of rapid recovery of coronary microvascular function, we did not observe significant differences in MACE or other in-hospital incidents between different statin types. This finding is similar to the results of previous studies.39,42 Therefore, the influence of rapid coronary microvascular recovery on clinical outcomes requires further investigation.
Our study had several limitations. First, although we performed multivariate and PSM analyses to minimize the effects of the inherent limitations of observational studies, residual selection bias or confounders may have still been present. The results of this study should be used to generate hypotheses. Second, the sample size was limited; however, we calculated the power of the result and it reached 86%. Third, the patients included in our study were enrolled from June 2016 to December 2019 when high-dose statins were not routinely used. Therefore, we only explored the different drug effects of atorvastatin and rosuvastatin on dysfunctional coronary circulation at a moderate dose level. Previous research has suggested that in patients with STEMI who have a low baseline low-density lipoprotein cholesterol level (<3.6 mmol/L), double-dose statin therapy does not provide additional clinical benefits over moderate doses of statins. 43 Because a high-dose statin regimen is now highly recommended in patients with STEMI, the effects of different statin doses on the coronary microcirculation and the mechanisms of efficacy of different statins require further evaluation. Fourth, the reliability of the results would be enhanced by measuring the coronary microcirculation with more precise indicators (such as fractional flow reserve or the microvascular resistance index) and more imaging evaluation methods (such as positron emission tomography–computed tomography, cardiac magnetic resonance imaging, and myocardial contrast echocardiography). Finally, more studies are needed to explore the efficiency of different doses of statins on the coronary microcirculation in patients with STEMI undergoing pPCI.
Conclusion
Compared with rosuvastatin, atorvastatin was associated with better coronary microcirculation and less dysfunctional coronary circulation in patients with STEMI undergoing pPCI; however, these effects did not lead to improved in-hospital outcomes in our study. Therefore, whether different types of statins affect outcomes in patients with STEMI undergoing pPCI needs to be further evaluated.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231182547 - Supplemental material for Effects of atorvastatin and rosuvastatin on dysfunctional coronary circulation in patients with ST-segment elevation myocardial infarction
Supplemental material, sj-pdf-1-imr-10.1177_03000605231182547 for Effects of atorvastatin and rosuvastatin on dysfunctional coronary circulation in patients with ST-segment elevation myocardial infarction by Langping Zhou, Xiangming Hu, Haotian Zhang, Haoyu Lu, Yan Lin, Weimian Wang, Bingyan Yu, Wensheng Liang, Yingling Zhou, Guang Li and Haojian Dong in Journal of International Medical Research
Footnotes
Acknowledgements
Author contributions
LPZ and XMH prepared and wrote the manuscript. XMH and YL analyzed the data. HTZ, HYL, WMW, BYY, and WSL acquired the data. GL and HJD reviewed and critically revised the manuscript. YLZ, GL, and HJD contributed to the ideas and approved the final version of the manuscript. All authors read and approved the final manuscript.
Data availability statement
The data analyzed in this study can be obtained from the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Key Research and Development Program of China (No. 2016YFC1301202), the Science and Technology Project of Tibet Autonomous Region (No. XZ202201ZY0051G), and the Guangzhou Municipal Science and Technology Bureau (No. 202102080466).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
