Abstract
As type 2 diabetes mellitus (T2DM) progresses, most patients will require escalating therapeutic interventions for disease control.1,2 To meet the challenges of caring for patients with T2DM, clinicians must be knowledgeable about, and adhere to, evidence-based guidelines. Performance improvement (PI) continuing medical education (CME) is an educational model for individualized improvement of clinical performance on validated measures known to enhance patient care and outcomes. 3 This 3-step process engages clinicians in a series of self-assessments, self-formulation and implementation of a plan for practice improvement, and reevaluation to assess the effect of adopted practice changes. Our original initiative was developed in 2008 and demonstrated significant changes in clinician performance and patient outcomes, but was not designed to show specific changes in therapeutic decision making.4,5 In order to more closely observe the effect of an educational activity on clinician decision making, as well as address continued needs for quality improvement, a follow-up activity was developed in 2011.
Participating US-based clinicians completed a practice patterns questionnaire in order to compare perceived with genuine performance. To evaluate actual performance, clinicians completed patient-chart abstraction on 25 random charts from patients with T2DM meeting the following inclusion criteria: (a) a diagnosis of T2DM, (b) age 18 to 75 years, and (c) negative pregnancy status. Assessments were based on validated performance measures and current practice guidelines.6-15 Clinicians received an individualized performance report and were asked to devise specific goals for self-improvement and a plan to achieve those goals. After a recommended period of 3 months, participants repeated the chart-review process. Participants received a second performance report comparing baseline and postintervention performance. As compensation, participants earned AMA PRA Category 1 CreditsTM after completion of each stage and upon completion of all 3 stages. The initiative was also approved by the American Board of Internal Medicine for Maintenance of Certification Part IV credit.
Because unique deidentified patient data were provided by participants, institutional review board oversight was not sought. 16 Chart data were unlinked and analyses did not attempt to adjust for clustering of patient data. Pearson’s χ2 tests and Fisher’s 2-tailed exact tests were employed.
A total of 561 participants completed the baseline assessment; of those, 102 participants attested to implementing their improvement plans and 64 participants completed the final chart abstraction (ie, “completers”). At registration, completers demonstrated practice deficiencies surrounding medical nutrition therapy and exercise counseling, with only 53% and 37% having systems for ensuring discussions of these topics, respectively.
On average, completers spent 100 days executing their plans. Participant-designed goals included creating/modifying note-taking templates within patient record systems, providing patient education, recruiting patient–care team members, improving the documentation of performance measures, and using treatment algorithms. One participant “[created] yellow stickers to place on the front of all our diabetic charts, which reminds physicians as well as staff to [perform annual foot exams]” and another “set up [a certified diabetes educator] in office for monthly group sessions [and provided] diet handouts for patients.”
After completers performed a total of 3200 chart reviews, significant changes in performance-based quality measures and suggested improvements in outcomes of patients with T2DM were observed (Table 1). Importantly, mean A1C values decreased and more patients had A1C values <7.0% postintervention. Additionally, the improvements in overall A1C testing, recommendations for self-monitoring blood glucose, and provision of sick-day instructions indicate that a focus on PI is an effective way to change clinician behavior and patient outcomes. We believe that the slightly higher frequency of hypoglycemia observed in the postintervention stage was likely related to improvements in the review of self-monitored blood glucose logs.
Performance of Type 2 Diabetes Mellitus Evidence-Based Measures.
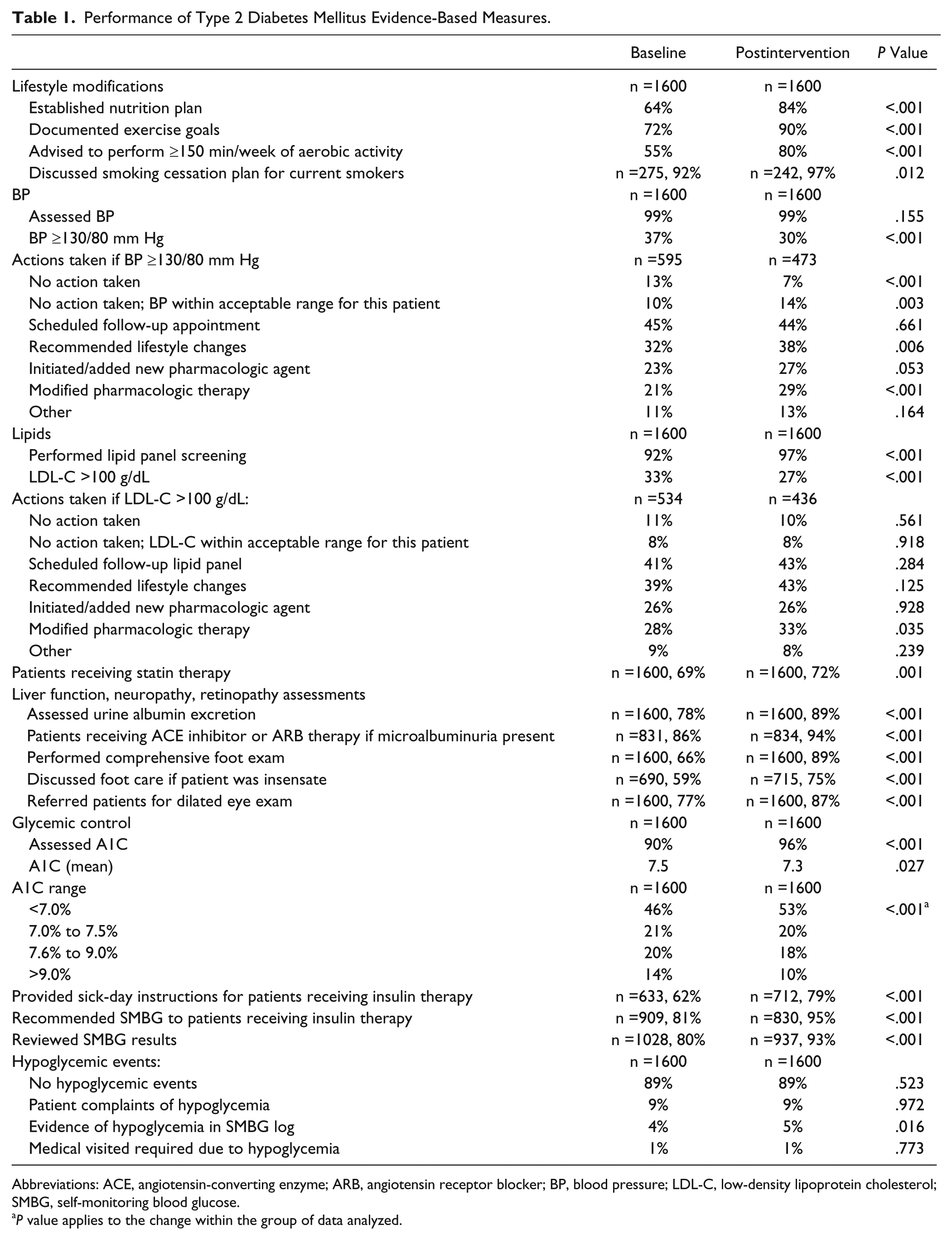
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BP, blood pressure; LDL-C, low-density lipoprotein cholesterol; SMBG, self-monitoring blood glucose.
P value applies to the change within the group of data analyzed.
We also observed important changes in both clinician performance and patient outcomes in key components of preventing and managing diabetes-related complications (Table 1). From baseline, there was a decrease in the percentage of patients with high blood pressure and low-density lipoprotein cholesterol (LDL-C) values. Furthermore, greater percentages of patients with blood pressure and LDL-C values above goal received adjustments to their pharmacologic therapies in the postintervention assessment.
At registration, participants’ reports showed low levels of adherence to measures related to counseling regarding appropriate lifestyle modifications. Given the prevalence of obesity, it is encouraging to see significant improvements in the adoption of diabetes self-management recommendations (Table 1).
It is difficult to conclude with certainty that the changes observed were a direct result of participation in the absence of a control group of clinicians. However, another study retrospectively examined outcomes of patients treated by participants in an earlier PI initiative. 5 Those data show clinically important improvements in patient outcomes. Thus, it is plausible that changes in clinician performance in this initiative correlate with changes in patient outcomes. In addition, Stowell et al 5 found significant changes in patient outcomes for patients whose clinicians completed only the first chart abstraction and implemented their improvement plans, and these changes were not different than those seen in patients treated by clinicians who completed the entire PI initiative. This suggests that patients cared for by the noncompleter clinician group in the initiative reported herein were likely to have had correspondingly similar improvements in patient outcomes as those treated by completer participants.
We recognize several limitations to our study. A larger data pool would capture more meaningful observations; however, requiring more patient charts may have further deterred participation because of time- and resource-related barriers. 17 Data were self-reported and charting biases may have occurred. Because the analysis did not control for potential clustering effects, some effects may be slightly overstated because of restricted variability. Finally, to account for the need for personalized care, participants were given the option to note whether values were within an acceptable range for individual patients. However, we did not assess whether participants were correct in those assessments.
As the diabetes epidemic grows, it is critical that clinicians evaluate individualized self-performance of evidence-based clinical care that directly affects patient outcomes. We are encouraged to report clinically meaningful changes in health care professional practice, and the suggestion of changes in patient outcomes, as a result of participation in a PI CME initiative.
Footnotes
Acknowledgements
The authors thank Dr Silvio Inzucchi for assistance with the development of the performance improvement continuing medical education activity; Martha Inglis-Legall and Whitney Stevens for project management; LaWanda Abernathy Stone for participant recruitment; Mary Catherine Downes, Kristin Hartman, Kenny Khoo, Samantha Roberts, and Amy Sison and for outcomes management; Marika Bonaguidi for IT support; Sara Metzger for production assistance; Kim Keaton for assistance with data analysis; and Lisa Rinehart for editorial assistance.
Authors’ Note
Howard B. A. Baum, MD, is currently at Division of Diabetes, Endocrinology, and Metabolism, Vanderbilt University Medical Center, Nashville, Tennessee. The funding source has no role in the execution, analysis, or development of the article associated with this initiative.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Baum discloses that he has received consulting fees from Novo Nordisk and Dr Cagliero discloses he has received consulting fees from AstraZeneca. The remaining authors have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The initiative was supported by an unrestricted educational grant from Lilly USA, LLC.
