Abstract
Introduction
The arena of mental health and primary care is moving from consideration of single conditions and their outcomes to more real-world, complex-care paradigms. 1,2 However, to date, no synthesis has been done of the evidence on practice-based interventions that accounts for the primary care patient with multiple chronic conditions 3,4 and examines both mental health and chronic medical outcomes simultaneously. Repeated evidence reviews show the benefits of integrated and collaborative care models, as compared with usual care, on depression outcomes in the general health setting, but it remains unclear whether better treatment of depression in primary care can also improve chronic medical outcomes, such as for diabetes. 5 -7
Half of all Americans live with a chronic medical condition 8 and up to one quarter of people living with chronic medical conditions have limitations in daily activity. 8 Living with chronic disease also takes a personal and emotional toll on patients and their families, owing to significant reductions in quality of life. 8 Depression is common in patients with chronic medical conditions, including arthritis (13% to 20%), 9,10 heart disease (10% to 47%), 11 diabetes (11% to 31%), 12,13 asthma and chronic obstructive pulmonary disease (27% for each), 14,15 and cancer (9% to 50%). 16,17 Depression among people with chronic medical conditions has also been linked to an increase in health care utilization, disability, and work absenteeism when compared with those without depression, even after controlling for the varying burden of the chronic medical condition. 18,19
For the Agency for Healthcare Research and Quality, we conducted a systematic review and meta-analysis to summarize the body of evidence that examines the effectiveness of practice-based interventions aimed at improving depression or both depression and chronic medical conditions in adult primary care patients with depression and chronic medical condition(s). 20 We were interested in interventions that targeted the care process within a system of care, such as coordinated care, integrated care, or collaborative care; medication-only studies or psychotherapy-only studies were not the focus of this report.
Methods
We developed and followed a standard protocol. A technical report that details methods and includes search strategies and additional evidence tables is available at http://www.effectivehealthcare.ahrq.gov/reports/final.cfm. This article reports on mental health outcomes, chronic medical outcomes, and harms.
Data Sources and Searches
We searched MEDLINE, Embase, the Cochrane Library, CINAHL, and PsycINFO from the inception of each database (1948, 1947, 1898, 1937, and 1880, respectively) through June 11, 2012, limited to English-language articles. We used Medical Subject Headings as search terms when available and key words when appropriate, focusing on terms to describe the relevant population and interventions. We consulted with experts in the field and manually searched reference lists of pertinent reviews, included trials, and background articles to look for any relevant citations that our searches might have missed.
Study Selection
We developed inclusion and exclusion criteria with respect to populations, interventions, comparators, outcomes, timing, settings, and study designs. 21,22 We included controlled studies of at least 6 months’ duration in adults (age 18 years or older) with depression and one or more chronic medical conditions defined as priority conditions by the Agency for Healthcare Research and Quality 23 and the Institute of Medicine 24 : arthritis; diabetes; asthma or chronic obstructive pulmonary disease; cancer; chronic pain; stroke; HIV/AIDS; heart disease, heart failure, myocardial ischemia, coronary artery bypass graft, postmyocardial infarction, and coronary artery disease; “complex” patients with multiple comorbidities; and frailty due to old age. We also searched for systematic reviews of such studies.
Depression was defined as meeting criteria for a disorder as determined by valid and reliable measures with established cut points but not necessarily confirmed by gold standard interview; we excluded subthreshold symptoms and minor depression. Included studies must have used practice-based interventions aimed at improving the mental health condition or both the mental health and chronic medical condition(s). We defined “practice-based” as any intervention that (a) targets the care process within a system of care and (b) aims to improve depression or both depression and chronic medical conditions. Examples of practice-based interventions include but are not limited to coordinated care, integrated care, and collaborative care; they often involve a care manager. Each of these terms has varying, and possibly overlapping definitions and is not specifically defined for the purposes of this report. In general, we perceive them broadly to mean primary care providers and mental health providers working together to address the comprehensive needs of the patient. Eligible controls were other practice-based interventions or usual care.
Two investigators independently reviewed titles and abstracts, and then another 2 investigators independently reviewed the full text of all articles marked for possible inclusion to determine final inclusion/exclusion. Disagreements were resolved by discussion and consensus among all investigators, as facilitated by a senior member of the team most experienced in comparative effectiveness research methods.
Data Extraction and Quality Assessment
We designed and used structured forms to extract pertinent information from each article, including information about the methods and populations, interventions, comparators, outcomes, timing, settings, and study designs. All data extractions were reviewed for completeness and accuracy by a second team member.
To assess the quality (internal validity) of studies, we used predefined criteria based on those developed by the United States Preventive Services Task Force (ratings: good, fair, or poor) 25 and the University of York Centre for Reviews and Dissemination. 26 These included assessment of the adequacy of randomization, allocation concealment, similarity of groups at baseline, masking, attrition, and whether intention-to-treat analysis was used. Two independent reviewers assigned quality ratings for each study. Disagreements were resolved by consulting an experienced member of the team. Studies with significant risk of bias (e.g., stemming from serious errors in design or analysis) that may invalidate their results were rated “poor” and excluded from this review (N = 2).
Data Synthesis and Analysis
Quantitative analyses were conducted of outcomes reported by a sufficient number of studies that were homogeneous enough for us to justify combining their results. We used random-effects models to estimate pooled effects. 27 For continuous outcomes, we used the weighted mean difference as the effect measure; if the measurement scale differed among trials, we calculated the standardized mean difference. For most dichotomous outcomes, we report risk differences. Sensitivity analyses were conducted for all analyses in which considerable heterogeneity was present (ie, I 2 statistic greater than 75%).
When quantitative analyses were not appropriate (e.g., because of heterogeneity, insufficient numbers of similar studies, or insufficiency or variation in outcome reporting), we synthesized the data qualitatively.
We graded the strength of evidence (SOE) based on the guidance established for the Evidence-Based Practice Center Program. 28 Two reviewers assessed each domain for each key outcome, and differences were resolved by consensus.
Results
Twenty-four published articles reported data from 12 studies (9 randomized controlled trials and 3 preplanned subgroup analyses from a separate randomized controlled trial; Figure 1).

PRISMA flow diagram.
Results are organized by main outcomes and grouped by medical condition(s) when possible. Our results pertain to the general adult population; no studies that met our inclusion criteria reported on young adults or pregnant women. Regarding older adults, one study selectively recruited participants aged 60 years or older 29 -33 ; however, participants across all studies in this review tended to be middle aged or older (mean age = 59 years; range of means = 47-72), so we do not report results for older adults separately. Several studies reported on traditionally underrepresented populations, including women, 34 -36 Spanish speakers, 34 -37 and predominantly African American male veterans with HIV; 38 we report these results in the context of overall results by medical condition, not in separate categories (Table 1).
Characteristics of Included Studies, by Chronic Medical Condition.

Abbreviations: ADAPt-C = Alleviating Depression Among Patients with Cancer; DSM-IV = Diagnostic and Statistical Manual of Mental Disorders, fourth edition; MDD, major depressive disorder; HADS = Hospital Anxiety and Depression Scale; HITIDES = HIV Implementation of Translating Initiatives for Depression into Effective Solutions; HRSD = Hamilton Rating Scale for Depression; IMPACT = Improving Mood—Promoting Access to Collaborative Treatment; MDDP = Multifaceted Diabetes and Depression Program; MODP = Multifaceted Oncology Depression Program; NR = not reported; PC = primary care; PHQ = Patient Health Questionnaire; PRIME-MD = Primary Care Evaluation of Mental Disorders; SCI = structured clinical interview; SCL = Symptoms Checklist; SMaRT = Symptom Management Research Trial in Oncology; UK = United Kingdom; US = United States.
Overall mean as reported, range of means for treatment groups, or overall mean calculated using mean age from each treatment group.
These criteria assess for biases including appropriate masking/blinding, attrition, and intent-to-treat analyses. In general terms, a “good” study has the least risk of bias and its results are considered to be valid. A “fair” study is susceptible to some bias but probably not sufficient to invalidate its results.
Patients were recruited before hospital discharge; intervention took place over the telephone.
Diabetes and/or heart disease.
Sample sizes ranged from 55 to 1001, and study duration ranged from 6 to 60 months. Most trials were conducted in the United States (including one in Puerto Rico 37 ); one took place in Scotland. 40 All included studies characterized their respective intervention as a form of collaborative care, not another form of a practice-based intervention (such as integrated care). Three studies described in 5 publications 29 -33 are secondary analyses from the Improving Mood—Promoting Access to Collaborative Treatment (IMPACT) trial 51 ; it tested a collaborative care depression intervention in older adult primary care patients, including preplanned subgroups of patients with arthritis, cancer, and diabetes. For ease of interpretation, we consider each subgroup a unique “study.” Consequently, our results include data from 12 studies (9 stand-alone randomized controlled trials and 3 IMPACT subgroups). The chronic medical conditions included arthritis, 30,33 cancer, 29,34,36,40 diabetes, 5,7,35,43 -46 heart disease, 47 and HIV. 38 Two studies selected patients with one or more active chronic medical conditions. 37,48 Each study compared the intervention with usual or enhanced usual care (Table 2).
Characteristics of Interventions, by Chronic Medical Condition.

Abbreviations: ADAPt-C, Alleviating Depression Among Patients with Cancer; CHD, coronary heart disease; DM, diabetes mellitus; HITIDES, HIV Implementation of Translating Initiatives for Depression into Effective Solutions; IMPACT, Improving Mood—Promoting Access to Collaborative Treatment; MDDP, Multifaceted Diabetes and Depression Program; MODP, Multifaceted Oncology Depression Program; PCP = primary care provider.
Usual care consisted of informing patients of their depression status and advising them to share this information with their primary care provider. Enhanced usual care usually included some degree of additional communication between the research staff or diabetes care manager and the patient’s primary care provider and/or family about the patient’s depression status.
Diabetes and/or heart disease.
Mental Health Outcomes
We summarize the mental health-related outcomes and SOE in Table 3. These results were consistent across medical conditions and reflect clinically meaningful changes on well-accepted measures of depression (the Patient Health Questionnaire–9, the Symptom Checklist–20, the Symptom Checklist–90, and the Hamilton Rating Scale for Depression–17, all using valid and reliable cut-points). Figure 2 shows forest plots for the comparison between collaborative care interventions and usual care on the outcome of depression response (at least 50% reduction in depression score) at 6 and 12 months. The evidence showed that 5 patients would need to be treated to achieve one more depression response than would be seen with usual care at 6 months, with a number needed to treat of 6 patients at 12 months.
Summary of Results for Collaborative Care Interventions Compared With Controls for People With Depression and One or More Chronic Medical Conditions: Mental Health Outcomes.

Abbreviations: CI = confidence interval; HITIDES = HIV Implementation of Translating Initiatives for Depression into Effective Solutions; MH = mental health; NA = not applicable; NNT = number needed to treat; RCT = randomized controlled trial; RD = risk difference; SMD = standardized mean difference; WMD = weighted mean difference.
Results are from meta-analysis of the 4 trials that reported satisfaction for both intervention and control arms. Two additional trials reported treatment satisfaction for the intervention arm but not the usual care arm.
Results of the meta-analysis excluding the HITIDES data, which was an outlier and accounted for significant heterogeneity.
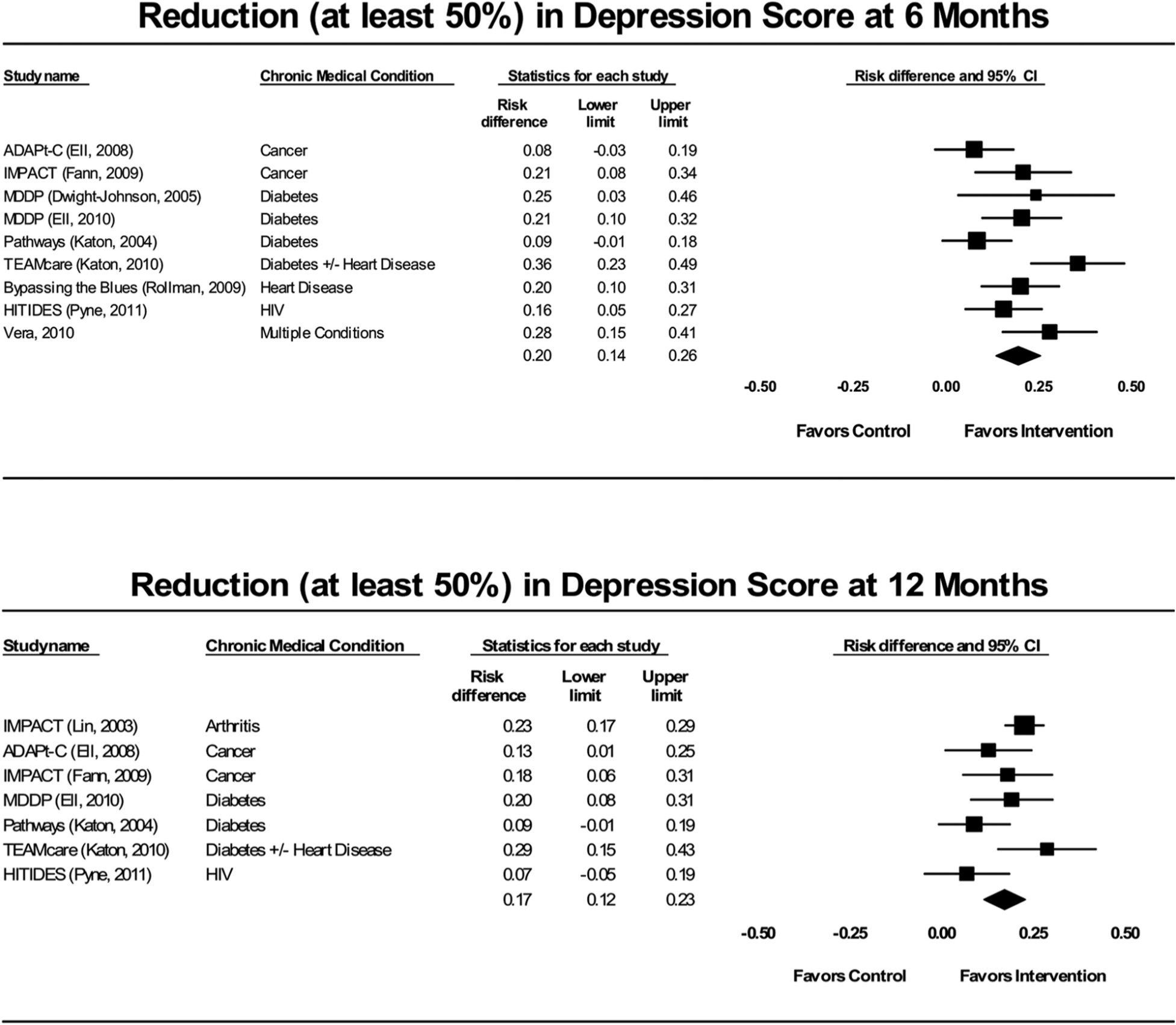
Forest plots for depression score response (at least 50% reduction) for practice-based interventions compared with controls.
Meta-analysis of data from 4 studies 32,35,36,38 showed greater mental health–related quality of life for patients in collaborative care intervention arms than usual care at 6 and 12 months using the mental component of the Medical Outcomes Study Short Form. One suicide was reported in the usual care arm of a cancer trial. 40 No data were available on sick days or employment stability (insufficient SOE).
Chronic Medical Outcomes
We summarize the effects and SOE of collaborative care interventions on outcomes for the specified chronic medical condition(s) in Table 4. For most chronic medical conditions of interest, we found just one study. We found multiple studies of people with diabetes and depression.
Summary of Results for Collaborative Care Interventions Compared With Controls for People With Depression and One or More Chronic Medical Conditions: Chronic Medical Outcomes.

Abbreviations: OR, odds ratio; CI, confidence interval; HbA1c = hemoglobin A1c; mm Hg = millimeters of mercury; RCT = randomized controlled trial; SBP = systolic blood pressure; WMD = weighted mean difference.
Five studies 32,35,36,38,47 measured self-reported quality of life using the physical component of SF-1232,35,36,38 or SF-36. 47 Four studies 29,33,35,48 used the Sheehan Disability Scale of Functional Impairment. 52 One study 38 used the Quality of Well-Being Self-Administered Scale (QWB-SA). In 3 studies, 29,30,32,33 patients were asked to self-rate on a scale that ranged from 0 to 10.
HbA1c was reported as a measure of response in 4 trials described in 5 publications 5,32,35,41,48 of people with diabetes; baseline HbA1c ranged from 7.28% to 9.03%. Our meta-analyses of the 3 trials (4 publications) with usable data 32,35,41,48 found no significant differences between intervention and control groups at either 6 or 12 months (Figure 3); findings were somewhat inconsistent and lacked precision (low SOE). However, the only study to use HbA1c as a predefined outcome measure, the TEAMcare study, 48 reported significant differences in HbA1c. The values were as follows for intervention versus control groups: 8.14 versus 8.04 at baseline; 7.42 versus 7.87 at 6 months; and 7.33 versus 7.81 at 12 months (overall P < .001).

Forest plots for reduction in HbA1c for practice-based interventions compared with controls among adults with diabetes.
Meta-analysis of data from 6 studies 29,33,34,36,38,39,49 found that patients receiving collaborative care interventions generally reported significantly greater physical health quality of life than control patients at 6 and 12 months. Six trials, 31,35,36,40,43,48 using various methods, reported costs of the intervention. Using the reported per-patient intervention costs and accounting for variability in study duration, we estimated the average cost of the intervention per patient was $705.00. Individual studies measured other aspects of cost but were not amenable to pooling.
Harms
Very few data were reported on harms, leaving insufficient evidence to draw conclusions. Only the TEAMcare study, in patients with depression and comorbid diabetes and/or heart disease, 48 defined adverse events; the investigators reported higher rates of mild (e.g., medication side effects) and of moderate (e.g., falls) adverse events in the intervention arm.
Discussion
Our findings reinforce the evidence for the effectiveness of collaborative care interventions for treating depression in primary care. 53 Moreover, they add a level of detail that had previously not been systematically reviewed. We selected trials that required the diagnosis of one or more chronic medical conditions (rather than generic primary care samples), and we reported on both the depression and the chronic medical outcomes. This review also extended the parameters of primary care to include settings in which certain patients with chronic disease receive the majority of their care. We found that recipients of collaborative care had significantly greater improvement in depression outcomes as compared with patients receiving usual care, for people with arthritis, cancer, diabetes, heart disease, and HIV.
Although the relationship between depression and chronic disease is established, 13,54,55 the extent to which successful treatment of depression improves chronic medical conditions remains unknown. As we move to consider shared savings programs, such as accountable care organizations 56 and the patient-centered medical home, 57 consumers and payers are eager to identify interventions and processes that can streamline care for multiple conditions and improve the quality and efficiency of care. Our review shows that investigators are beginning to examine these outcomes, particularly in diabetes, although largely as secondary outcomes and with negative or inconclusive findings at present. We excluded some relevant studies because of duration of follow-up less than 6 months 58 or because the treatment occurred outside the purview of a primary care–like setting. 59 -61 However, our inability to answer the basic question posed by a primary care provider, “Will treating my patient’s depression (with an evidence-based collaborative care program) improve his or her medical conditions?” was both surprising and disappointing.
One study in the review, TEAMcare, 48 is unique because it identified markers of disease risk for multiple conditions as primary outcomes. Using a guideline-based “treat-to-target” approach delivered by a medically trained nurse, the investigators targeted patients with poorly controlled diabetes, coronary artery disease, or both and coexisting depression; their aim was to reduce overall risk factors. This approach differs from the traditional model, in which the focus is on collaborative care of depression, presumably in the hope that treating depression will improve overall health. Perhaps partly because of the benefits of having an integrated health care system, TEAMcare recipients showed clear improvements not only in depression but also in reducing HbA1c and systolic blood pressure to target goals.
The paucity of data on harms in the included studies could be attributed to increased rates of medication adjustment related to the collaborative care intervention. Additionally, patients in the intervention arm had more frequent contacts with the care manager and thus had more opportunities to report adverse events, so findings might be the result of detection bias.
Applicability
Our findings are generally applicable to primary care patients with depression and at least one chronic medical condition, but they may not apply to patients with multiple chronic conditions. The average age across studies was 59 years, an age-group likely to have chronic disease. For that reason, we cannot speak directly to the relevance of these results to young adults with chronic disease. People of Hispanic origin (predominantly female) 35,36 and male veterans 38 were represented and appeared to respond similarly across outcomes, but there were too few data to analyze separately. Reported studies used clinically meaningful measures and had study durations (at least 6 months) that provided a real-world context.
Although these trials represented several settings, including primary care–like cancer and HIV clinics, they all had in common a care manager who directed the intervention. The mental health outcomes achieved might, therefore, apply only to settings that can accommodate and afford to provide such services.
Limitations
Defining the interventions of interest was challenging because we wanted to be inclusive of the widest possible range of service level interventions that occurred in the primary care setting. With input from our Technical Expert Panel, we arrived at the term practice-based to differentiate interventions relative to this review from person-level interventions such as medications or stand-alone psychotherapy. We did not find the term practice-based in the literature, but we used other eligibility criteria, a general intervention term and known interventions to inform our searches. Even though we also added the terms collaborative care, integrated care, and telemedicine to guide our search, we may have missed relevant interventions that are not indexed in these categories.
We chose to exclude studies without comparison groups because of the potential risk of bias in such studies (especially the risk of selection bias and confounding). Important and innovative systems efforts in the fields of mental health and primary care 62 may be overlooked using these methods. However, the purpose of this review was not to uncover hypothesis-generating information but rather to find evidence with sufficiently low risk of bias to provide more definitive answers to the key questions.
Few relevant trials reported medical outcomes as a primary interest; only 1 of 12 studies 48 was specifically designed to assess medical outcomes. The remainder aimed to look at mental health outcomes in patients with different medical conditions. Finally, studies did not necessarily screen for mental health comorbidities (such as substance abuse), which may have negatively influenced medical outcomes, particularly related to self-care activities, and biased toward the null. A completely unexplored area is personality disorders, which are pervasive by nature and can prove a barrier to achieving therapeutic goals. 63
Research Gaps
Although it is clear that collaborative care works to improve depression in primary care, we do not know if it outperforms other practice-based interventions (such as integrated care or co-location of mental health services) because no head-to-head studies exist. We also do not know whether the effective treatment of depression in the primary care setting can alter the course of chronic disease. To determine the relative benefit of implementing collaborative care programs for depression (or other mental health conditions) on overall health, we need studies designed to measure the effectiveness of practice-based interventions on medical outcomes. The TEAMcare approach offers an example, in which treatment goals included targets for all relevant diseases and individualized approaches to reach these targets. Designing, implementing, and sustaining such approaches will not be without considerable challenge, and studies will require larger sample sizes, longer time frames, and, optimally, higher levels of joint funding from multiple institutes more used to focusing on one disease.
Conclusions
Collaborative care interventions improved outcomes for depression and quality of life in primary care patients with varying medical conditions. Few data were available on medical outcomes. Future studies of concomitant depression and chronic medical conditions should be designed to consider measures of medical outcomes as primary outcomes.
Footnotes
Acknowledgements
The authors gratefully acknowledge the contributions of Carol Woodell (Project Manager), Tania Wilkins (biostatistician), Sonia Tyutyulkova and Beth Collins-Sharp (AHRQ Task Order Officers), and Megan Van Noord (literature search).
Authors’ Note
The views expressed in this article do not represent and should not be construed to represent a determination or policy of the Agency for Healthcare Research and Quality or the US Department of Health and Human Services.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Agency for Healthcare Research and Quality, Contract No. 290 2007 10056 I.
Author Biographies
