Abstract
Tobacco use has serious health consequences for both the user and the individuals in the user’s environment who are exposed to secondhand smoke. Children are the largest demographic group with chronic exposure to secondhand smoke, due almost exclusively to smoking by their parents and caregivers. 1 Secondhand smoke exposure among kids has been linked to premature death and a number of other illnesses, including otitis media, asthma, bronchitis, and pneumonia. 2 The surgeon general of the United States has concluded that there is no safe level of secondhand smoke exposure for children. 1 The most effective way to reduce children’s exposure to secondhand smoke is to intervene with the parent’s behavior to either cease smoking or make other efforts to minimize smoke in the child’s environment.
Effective behavioral and pharmaceutical smoking cessation interventions now exist for smokers, 3 but smokers must necessarily have some motivation and intention to change their smoking behavior to benefit from such interventions. Effective brief interventions are available to increase an individual’s motivational readiness to attempt cessation, including the 5 A’s (ask, advise, assess, assist, and arrange) and 5 R’s (relevance, risks, rewards, roadblocks, and repetition) that are recommended in the clinical practice guideline distributed by the US Department of Health and Human Services. 3 The guideline also recommends that smoking patients receive a brief intervention at each contact with a health care provider, focused on increasing motivational readiness to quit smoking.
Despite this recommendation, evidence suggests that such interventions occur at very low rates.4-6 A variety of roadblocks prevent frequent intervention in primary care settings, including workload demands (eg, high patient loads), lack of reimbursement, and personnel who have not received appropriate clinical training. The problem is compounded in the pediatric setting, where the identified patient is a child. However, the pediatric primary care setting presents an ideal opportunity to intervene. Many parents may or may not have access to health care themselves but may have frequent contacts with a pediatric clinic. Parents also typically have interest in the health of their children, and the pediatric clinic may therefore be an ideal opportunity to raise awareness about tobacco risks and enhance motivational readiness to quit. Tobacco clinical practice guidelines, 3 the American Academy of Pediatrics, 7 and the American Medical Association 8 all recommend that pediatricians intervene with parental smoking.
A small number of empirical studies have examined parental smoking cessation and attempts to reduce children’s exposure to secondhand smoke in outpatient pediatric health care settings.9-11 Each of these studies investigated the efficacy of a brief motivational intervention for reducing tobacco use and provided preliminary empirical support for these interventions with parents who smoke in outpatient pediatric health care settings. The purpose of the current study was to test the efficacy of an interactive web-based program in facilitating motivation and readiness to change by assessing and delivering personalized feedback to parents regarding their smoking behaviors and associated parental and child health risks. It was hypothesized that participants randomized to complete a web-based smoking cessation computer program would report a significant increase in motivational readiness to engage in smoking cessation compared to participants randomized to the control group.
Method
Participants and Setting
Participants were parents of children recruited from a pediatric outpatient medical clinic waiting room within a university hospital. Specifically, once parents checked in for their children’s appointment at the receptionist desk, they were approached by a member of the research team and asked a series of questions to determine eligibility and willingness to participate. Parents were deemed eligible to participate if they spoke English as their primary language and endorsed active, regular cigarette use. Parents who indicated actively attempting to quit smoking were excluded from the study. Additionally, candidates were asked to read aloud a short paragraph (from within the informed consent paperwork) constructed at a sixth-grade reading level using the readability statistics feature in Microsoft Word. Parents who read below the sixth-grade level were excluded from the study (n = 2). Informed consent was then obtained for each interested and eligible participant in accordance with the institutional review board.
Participants subsequently accompanied the research team member from the waiting room to a private exam room within the clinic to complete the study measures. Notably, the entire process (consents, baseline measures, and intervention) took less than 30 minutes to complete and occurred within a private clinic treatment room before the family’s appointment with the child’s physician. Last, participants were scheduled for a 1-month follow-up appointment and provided with a $5 gift card for participating. The 1-month follow-up appointment was completed during a clinic appointment or over the phone with a research assistant, and it consisted of the same measures completed at baseline. Participants were given or mailed a $10 gift card for participating in the follow-up.
Data collection occurred 2 to 3 days a week and continued for 10 months. A total of 113 parents provided consent and were randomized to the intervention or control group (see Figure 1). Forty-one participants failed to complete the follow-up component due to not attending the follow-up appointment or to being unreachable via phone. Seventy-two caregivers completed the follow-up component: 33 participants in the smoking cessation group and 39 people in the control group. On average, 45.1 ± 20.7 days lapsed between baseline and follow-up completion. For the purposes of the current study, only the participants who completed baseline and follow-up measures were included in subsequent analyses. Demographic and relevant smoking information are presented in Table 1.

Participant flow through trial.
Demographics and Study Characteristics

Test of between-group differences.
χ2 test.
t test.
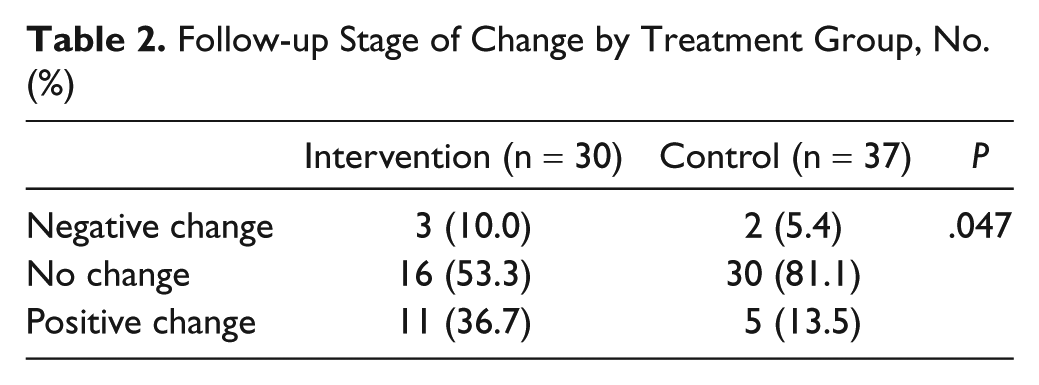
Follow-up Stage of Change by Treatment Group, No. (%)
Design
The current study used a 2-group randomized trial design. Upon completion of baseline assessment measures, participants were randomly assigned to either the smoking cessation group or the control group using a computer-generated block randomization procedure.
Intervention group
Participants in the intervention group completed an interactive web-based smoking cessation program that assessed participants’ smoking behavior and provided personalized feedback aimed at developing a discrepancy between the participant’s current behavior and his or her goals, values, and/or self-image. Specifically, the interactive program provided assessment and personalized feedback on the participants’ level of nicotine dependence, expired breath carbon monoxide, daily cigarette consumption, money spent on cigarettes, behavioral consequences of smoking, individual medical consequences of smoking, and family members’ medical consequences of secondhand smoke. Feedback was provided in the form of text and graphics, as well as information regarding the health benefits of quitting smoking, smoking cessation tips, and smoking cessation resources (see Figure 2 for an example of personalized feedback).
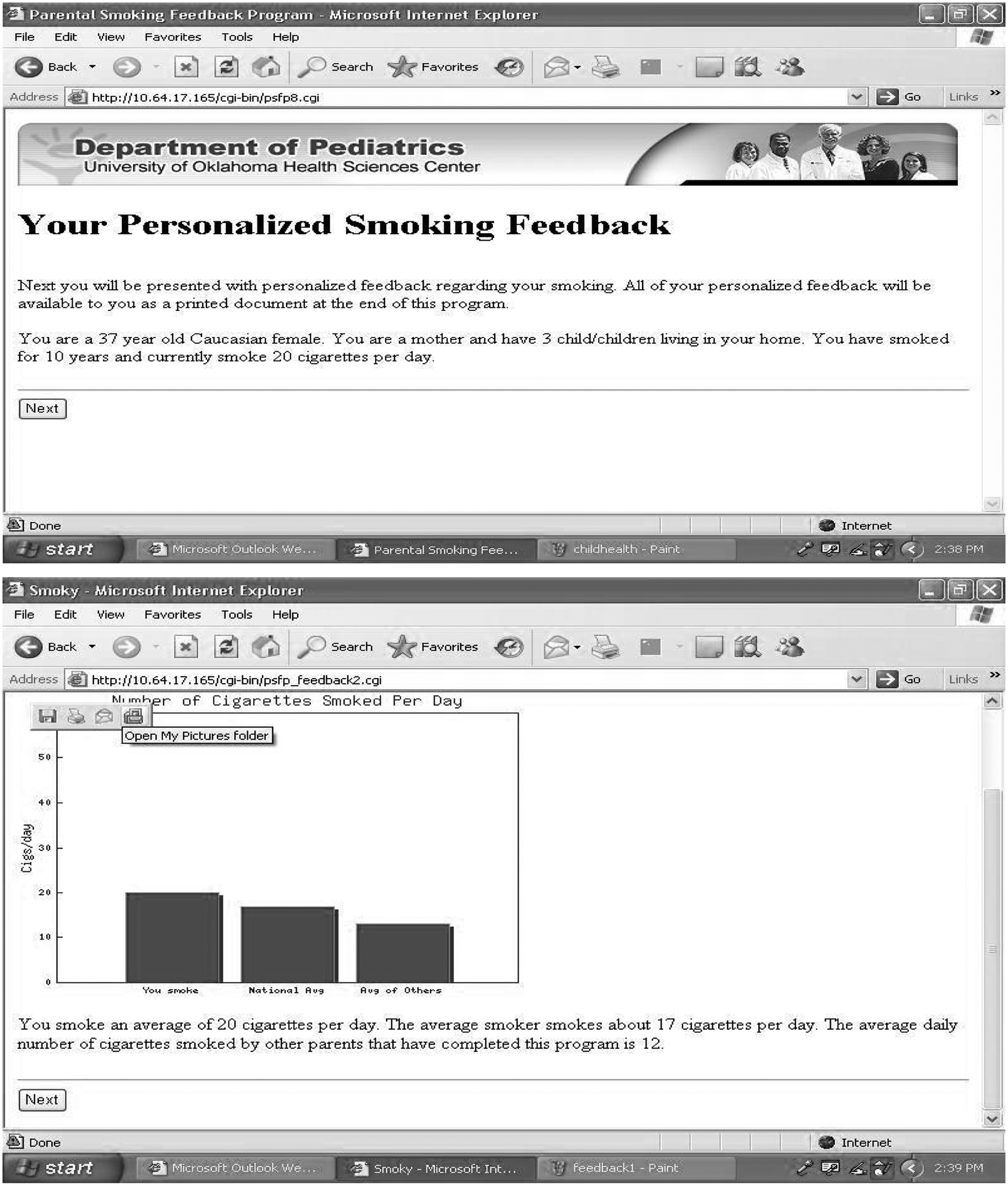
Examples of personalized feedback.
Control group
Participants assigned to the control condition did not receive personalized smoking feedback, thus constituting an assessment-only control. This group received treatment as usual for the outpatient clinic.
Measures
Stages of change
The Stages of Change Algorithm,12,13 based on Prochaska and DiClemente’s transtheoretical model of stages of behavior change, was used to categorize participants’ individual stage of change in regard to smoking cessation. The algorithm is a commonly used measure in smoking cessation research and has good concurrent and predictive validity; for example, DiClemente et al found that differences in smokers’ stage of change as categorized by the algorithm predicted attempts to quit smoking and cessation success at 1- and 6-month follow-up. 13 The algorithm consists of a single item, specifically, “Are you thinking of quitting smoking?” and provides the following answer options (with the identified stage of change in parentheses): “No, I am not seriously thinking of quitting within the next 6 months” (precontemplation), “Yes, I am seriously thinking of quitting within the next 30 days” (contemplation), or “Yes, I am seriously thinking about quitting within the next 6 months” (preparation).
Treatment fidelity
The Treatment Fidelity Questionnaire is a 13-item true-false questionnaire (possible scores of 0-13) designed for this project, with higher scores indicating agreement that the intervention was received. The reliability of the questionnaire was excellent (Cronbach α = 0.97).
Results
Overview of Analyses
Preliminary analyses were conducted to determine if there were significant group differences between individuals who did or did not complete follow-up measures and between the treatment and treatment-as-usual groups. Next, primary analyses were conducted from a stages-of-change framework. All analyses were conducted using a framework of positive change, no change, and negative change corresponding to whether a participant endorsed higher, lower, or the same stage of change at follow-up. Chi-square and Fisher exact test were utilized for the primary analyses with a significance level of .05.
Analyses
Analyses revealed no significant differences between those that completed the follow-up and those that did not, for race, sex, average number of cigarettes smoked each day, income, or stage of change in regard to smoking cessation. Furthermore, no differences were found for any demographic variables between treatment and treatment-as-usual groups in the analyzed sample.
Treatment fidelity and satisfaction
Participants in the intervention group reported high scores on the Treatment Fidelity Questionnaire (12.3 ± 2.3), indicating good exposure to the intervention. At follow-up, participants in the intervention group reported that the feedback was presented in a clear manner (100%), was useful (97%), was a good use of time (100%), and would be recommended to a friend (100%).
Effects on readiness to change
Analyses demonstrated a significant association between experimental groups on the Stages of Change Algorithm at follow-up, χ2(2, N = 72) = 6.05, P = .047. Specifically, 37% of parents in the intervention group endorsed a positive increase in readiness to quit smoking compared to 14% in the treatment-as-usual group.
When analyzed by baseline stage-of-change group, this effect was observed among only those in the contemplation stage at baseline (n = 42, P = .034, Fisher exact test), not precontemplation or preparation.
Discussion
This study investigated a newly developed web-based intervention that takes relatively little time and staff to implement. The goal of the intervention was not necessarily behavior change but raising awareness and enhancing motivation as an important precursor to behavior change. The self-administered intervention tested in this study was found to be effective at increasing readiness to change parental smoking behavior. Specifically, exposure to the intervention did result in increased motivational readiness to quit smoking soon among those who were already contemplating change, but it did not increase contemplation of change among those with little preexisting readiness.
The overall effects of this intervention were relatively small and did not include differences in smoking behavior itself. Such findings would be unacceptable for costly, labor-intensive interventions. However, brief, self-administered, and web-based interventions such as this can be disseminated and deployed at relatively little cost or burden to existing practices, which makes small effects more meaningful and justifiable. By increasing readiness to quit smoking among patients exposed to this intervention, it should make subsequent intervention attempts by clinical staff more fruitful and a better use of staff resources.
While we find the results of this study encouraging, the study does have a number of limitations. Although the study does utilize a clinical sample in a practice setting, it is relatively small, and the findings are considered preliminary. The study relied on self-reports of parents and did not include biochemical measures of smoking behavior. Future investigations should investigate this intervention with larger samples and with expanded measures of parent smoking behavior.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work supported in part by a grant from the University of Oklahoma College of Medicine Alumni Association.
