Abstract
Exosomes are extracellular vesicles with a diameter of 30–100 nm, which are released into the extracellular space by fusion of multivesicular and plasma membranes. These vesicles actually play a distinct role in cell communication, although they were considered as membrane debris in the past. The endosomal sorting complex required for transport (ESCRT)-dependent and ESCRT-independent mechanisms are currently considered to be involved in the sorting of exosomes, and the release of exosomes is related to the members of Rab protein family and SNARE family. In recent years, the therapeutic potential of exosomes has become apparent. For example, via the direct transplantation of exosomes, the ischemic area after stroke is reduced, and the neurological function is improved significantly. Furthermore, they can be used as effective drug delivery vehicles due to their unique characteristics such as low immunogenicity and nanometer size. In conclusion, exosomes provide a cell-free treatment for ischemic stroke.
Keywords
1 Introduction
Communication between cells has always been considered as the basis for the completion of life activities. Exosomes were originally treated as waste products of cells [1], but gradually discovered to be involved in the release mechanisms, which fully affirmed them as important inter-cell communication substances [2]. However, until today the mechanism of exosome fusion has not been fully studied, so it is worth exploring their biogenesis.
Exosomes, in addition to being secreted by different types of cells [3], are also present in body fluids, including cerebrospinal fluid and semen. Many substances from parental cells, including miRNAs and proteins [4], are contained in exosomes, participating in the formation and development of many diseases [5]. For example, the development of Parkinson’s disease is associated with the spread of α-synucleins, which can be found in exosomes [6].
Exosomes promote drug delivery system reforming as natural carriers. They can not only be exploited directly as some potential agents, but essentially, they can also carry other substances such as small molecule drugs into recipient cells [7]. Exosomes are promising in the treatment of neurological diseases such as post-stroke ischemic areas, because they have been shown to cross the blood–brain barrier and peripheral organ retention [8]. In this review, we will use stroke treatment as an example to describe the therapeutic potential of exosomes.
2 Biology of exosomes
2.1 Exosomes composition
Intercellular communication is an essential hallmark of multicellular organisms, and can be mediated through cell–cell contact directly or transfer of secreted molecules [2]. Exosomes are endogenous nanovesicles whose interaction between cells plays a key role by transmitting functional genetic information and proteins [9]. Exosomes were first discovered in 1983, but were originally thought to be wastes released by stripping the serosa [4]. However, there has been a surge of interests in exosomes in the past few decades, and after long-term exploration the exosomes contents are found to be lipids, proteins, nucleic acids [mRNA, microRNA (miRNA), long non-coding RNA (lncRNA), and rare DNA] [10]. In addition, mounting evidence has shown that these vesicles are important messengers between cells, because they can store and transmit cellular signals [10]. In recent years, these vesicles have been shown to affect the physiological functions of adjacent recipient cells in different ways. For example, extracellular vesicles can provide new genes for recipient cells, giving them new properties [3].
Exosome is a type of extracellular vesicles, which includes apoptotic bodies and microvesicle [11]. Both exosomes and microvesicles are vesicles enclosed by a lipid bilayer, but their membrane components are not identical because of their origins [1]. Exosomes are derived from the endocytic pathway, but microvesicles are directly budding from the plasma (Fig. 1). In addition to the secretory pathway, the size is usually different between them. Microvesicles are 100 nm to 1 μm in diameter, while exosomes are 30 nm to 100 nm [12]. Moreover, exosomes come from a wide range of sources, and can be found in physiological fluids such as urine, lymph, amniotic fluid, malignant pleural effusion, milk, semen, saliva, and plasma [13]. Almost all mammalian cell types, including primary cells of the immune, stem cells, nervous systems, and many cancer cell lines, can secrete exosomes. Importantly, apart from mammalian cells, exosomes have also been identified in lower eukaryotes and prokaryotes [1].
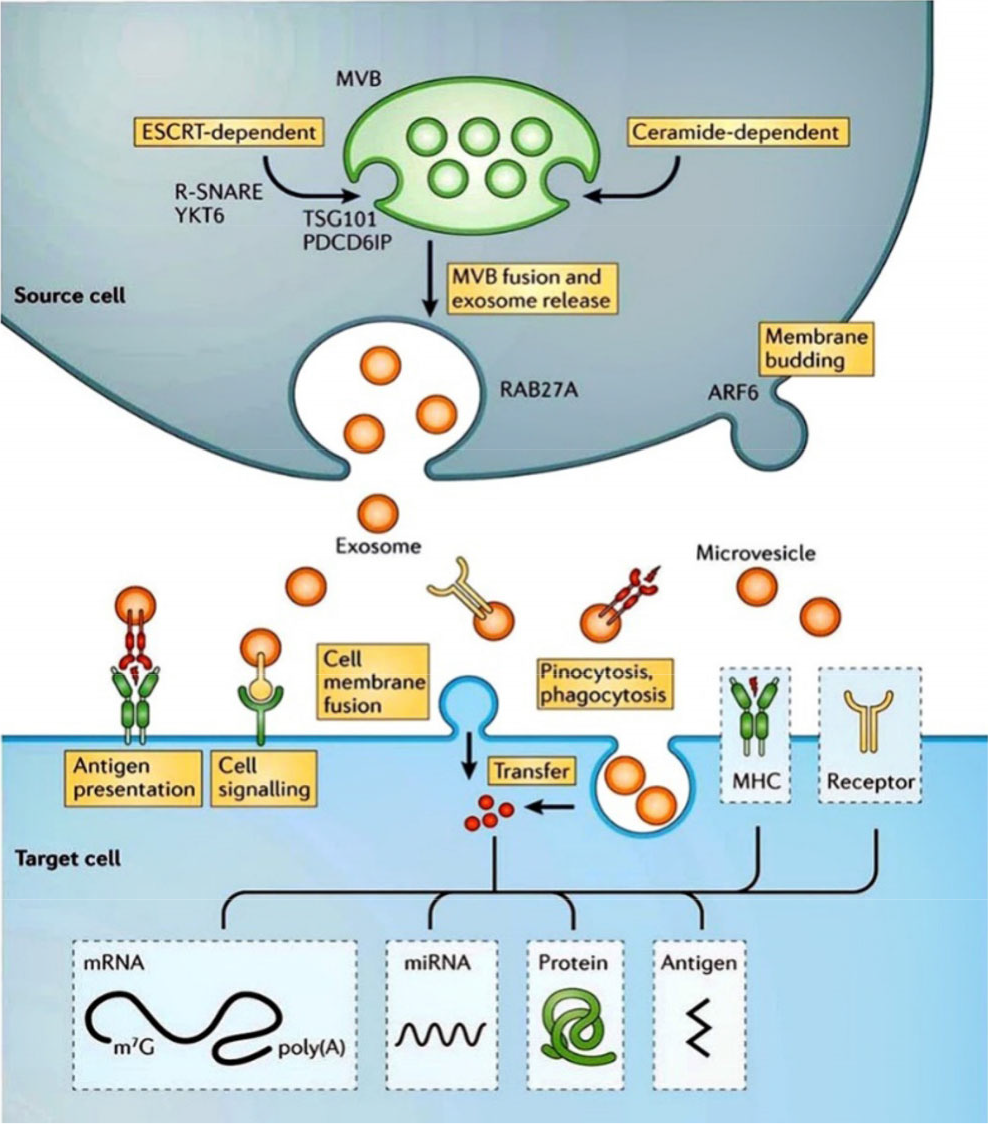
Biogenesis of extracellular vesicles and their interactions with recipient cells. Reproduced from Ref. [1] with permission of Springer Nature.
2.2 Exosome cargoes
Studies have shown that some certain proteins or protein families are expressed in exosomes of different cell origins, but the specific function of these conserved proteins is unknown at present, which may be related to cell origin. These ubiquitous proteins include multivesicular-associated proteins [e.g. ALG-2 interacting protein X (ALIX), tumor susceptibility gene 101 (TSG101), endosomal sorting complex required for transport (ESCRT)], metabolic enzymes [e.g. glyceraldehyde-3-phosphate dehydrogenase (GAPDH), peroxidase, pyruvate kinase, lactate dehydrogenase], signal transduction proteins (e.g. protein kinases, heterotrimeric G proteins), and membrane transport proteins (e.g. Rab protein, annexin). Exosomes also contai molecular chaperones. Heat shock protein (Hsp) 70 and Hsp90 are thought to be involved in antigen presentation, because they can bind antigenic peptides and participate in loading peptide onto major histocompatibility complex (MHC) molecules [14]. Meanwhile, tetraspanins [15] [e.g. cluster of differentiation (CD) 9, CD63, CD81, CD82] existing in exosomes that are secreted by almost all types of cells can mediate cell migration, fusion, adhesion, and signal transduction.
Some proteins with special functions are also present in exosomes except for ubiquitous proteins. These specific proteins contained in exosomes can reveal which mother cells they are from. For example, exosomes derived from intestinal epithelial cells contain various metabolic enzymes [16]; exosomes derived from T cells have T cell surface receptor proteins; and the ones from dendritic cells contain a large number of proteins associated with antigen-presenting cells, such as MHC class I molecules, MHC class II molecules, and costimulatory molecules CD80 and CD86 [17]. These proteins can be used as markers to provide a basis for early diagnosis of disease, such as tumors. In addition, the production of specific proteins is associated with the secretory pathway of exosomes. The secretion of general proteins in the cells is carried out through Golgi complex, which is, the secretions are synthesized and stored in the Golgi complex through the endoplasmic reticulum, and the cells are stimulated by the Golgi complex membrane and the cell membrane after receiving the external signal. However, unlike the general secretory pathway of intracellular proteins, exosomes are small vesicles that are released by fusion of multivesicular membrane with plasma membrane. The surface of the exosomes has special protein compositions that come from different secretory pathways, but it lacks certain membrane proteins with high expression on the surface of the cell membrane, due to the endoplasmic reticulum which does not contain proteins. For example, dendritic cells express high levels of Fc receptors, but the surface of the exosomes from dendritic cells does not.
Lipid, including phosphatidylserine, sphingomyelin, and cholesterol, is an important component of exosomes [18]. The lipid molecules contained in the exosomes not only maintain the shape of exosomes, but also can be used as signaling molecules in many biological processes, such as passing intermediate signaling molecules like prostaglandins, phosphate kinase C and D [19]. The membrane structure of the exosomes is similar to the cell membrane, and the phospholipid bilayer can protect the exosomes from degradation by enzymes in the extracellular environment, and further enhance their ability to adsorb target cells.
Exosomes also contain numerous functional RNA molecules (e.g. miRNAs, lncRNAs) that reflect the physiological and pathological of the cells from which they originate [18]. These genetic substances are incorporated into the target cells via the fusion of exosomes with the target cell membranes, thereby exerting the function of genetic information exchange [20, 21]. For example, a kind of miRNA can regulate gene expression by being bound to the 30 untranslated regions of the target mRNA, resulting in translational inhibition and/or mRNA degradation. Like miRNAs, lncRNAs do not encode proteins, but are expressed specifically at different tissues and developmental stages, indicating that they have important biological implications. Exosomes do not contain mitochondria, lysosomes, or other organelles and nucleuses, because exosomes are budded from the capsular sag and encapsulated in processing lipids, proteins, and genetic materials in the cytoplasm. Studies have shown that tumor cells secrete microparticles containing genomic DNA, c-Myc single-stranded DNA, and cDNA.
2.3 Isolation and identification of exosomes
Obtaining high-quality exosomes is the key to better analyzing their contents and functions. The separation and purification of exosomes have been a hindrance for the study of their characters, however, efficient methods for separating exosomes are rapidly improving. Ultracentrifugation is the most commonly used method for purification of exosomes, and vesicle particles of the desired size can be obtained by increasing the speed of centrifugation. Johnstone et al. originally initiated the centrifugation method for the separation of exosomes in reticulocyte tissue culture fluid [22], and then they improved the method on that basis. The tissue supernatant was centrifuged at 300 × g, 1200 × g, and 10,000 × g for 5, 20, and 30 minutes respectively to remove the doped cell debris, dead cells, and detached bodies. Finally, exosomes were isolated by high-speed ultracentrifugation (110,000 × g) for 1 h, the purity was increased twice [23]. For the production of a large number of exosomes, the steps involving 1200 × g, 10,000 × g of centrifugation can be replaced by filtration on 0.22 μm in order to eliminate large fragments [23]. The sucrose density gradient centrifugation method is an improved method based on ultracentrifugation to further improve the purity of exosomes, because the ultracentrifugation does not clearly distinguish exosomes from other small vesicles or large protein aggregates. Ultracentrifugation is followed by linear or non-linear sucrose gradient centrifugation depending on the density of the exosomes ranging from 1.13 g/mL to 1.19 g/mL, then the contaminating materials, including nucleosome fragments that are released by apoptotic cells or protein aggregates, can be separated easily from exosomes by flotation on sucrose gradients [24 –26]. The re-suspended pellet was wrapped with a sucrose gradient, and then centrifuged at 100,000 × g for 5 h. The layers were collected, and the expression of exosome marker molecules such as CD63 and CD8l in the solution was analyzed by Western blotting. In this case, the density layer of exosomes can be determined. In recent years, researchers have also described a PEG-based ExtraPEG technology that accumulates exosomes rapidly and inexpensively by small volume ultracentrifugation after centrifugation of large volumes of culture medium. ExoQuick, Pure-Exo and Total Exosome Isolation kits can be used to isolate exosomes [27 –29]. In addition, integrated on-chip isolation and in situ electrochemical analysis of exosomes from serum, a two-stage microfluidic platform (exoPCD-chip) [30] can promote the capture efficiency of exosomes without any surface modifications via an improved staggered Y-shaped micropillars mixing pattern (Fig. 2).
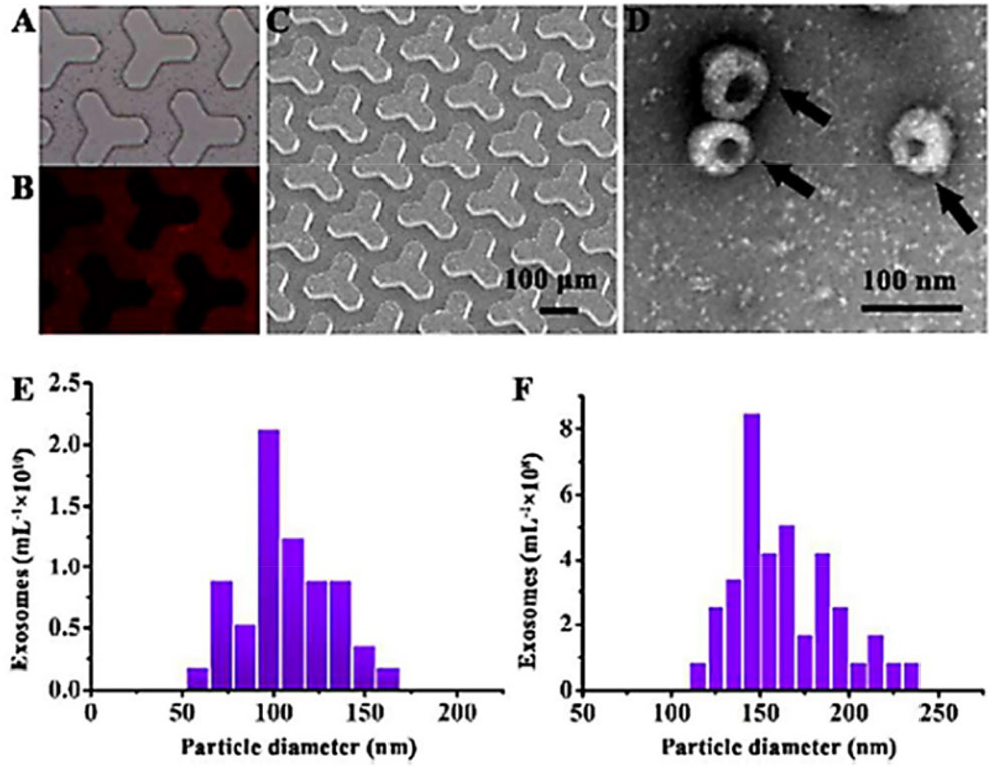
(A) Bright-field microscope image of Tim4 beads manipulated in the microfluidic channel. (B) Fluorescence images of the DiI labeled exosomes in the microfluidic channel. (C) SEM image of the array of Y-shaped PDMS micropillars. (D) TEM image of exosomes captured by the ExoPCD-chip. qNano analysis of the size distribution of exosomes purified by the ExoPCD-chip (E) and commercial exoEasy Kit (F). Reproduced from Ref. [30] with permission of The American Chemical Society.
The existing separation techniques are mainly based on the size and structure of exosomes, so it is difficult to completely distinguish them from other vesicles or macromolecular protein complexes, and requires identification [2]. The exosomes can be identified by nanometer particle tracer analysis (NTA) to measure particle size [31], by Western blotting and flow cytometry to analyze signature proteins (CD9, CD63, CD81, HSP90, etc.). Electron microscope can directly observe the morphology and particle size of exosomes, but the testing has higher requirements on the preparation of the sample, sample separation method and sample properties, which may affect the the results. In the research field of exosomes, NTA technology has been used as one of the methods for characterizing exosomes. Compared with other characterization methods, the sample processing of NTA technology is simpler, which can ensure the original state of exosomes, and the operation is quicklier. In addition, western blotting is used to detect exosome surface proteins (e.g. CD63, CD8, TSG101, flotillin-1, ALIX, CD9, CD81, CD82) to improve the reliability of the exosome characterization.
3 Exosomes biogenesis
3.1 ESCRT-independent and -dependent mechanisms
In the process of maturation into multivesicular endosome (MVE), exosomes are produced as intraluminal vesicles (ILVs) in the lumen of the endosomes, a process involving a specific “sorting machine”. In recent years, ESCRT on exosomes formation has received increasing attention. A device consisted of four main compounds and several auxiliary components is positioned in the cytoplasm side of the endocytosis, and its main role is to form the precursor of exosomes by sorting specific components into the ILVs. ESCRT contains four complexes (ESCRT-0, I, II, III) and accessory proteins (VPS4, Vta1, ALIX, etc.), with each component playing different roles. ESCRT-0 identifies and separates the ubiquitination of endocytic membrane transmembrane proteins; ESCRT-I synergizes with ESCRT-II to encapsulate endosomal membrane with specific contents through inward bud; the ESCRT-III is responsible for cutting the vesicles [14]; finally, the ATPase vacuolar protein sorting 4 (VPS4) was recruited in bud necks by interacting with the ESCRT III subunit protein, and it can hydrolyze ATP under the regulation of positive regulators Vta1, negative regulators IST1 [32], Did2 and VPS60 to obtain energy to depolymerize ESCRT from the endocytic membrane and enter the cytoplasm [33]. Meanwhile, the cycle acts on a new round of multivesicular formation and protein sorting processes. In addition, the study found that the ESCRT-0, I, II and the Alix protein, all of which seem to be involved in the disruption of the plasma membrane, but they actually only play a role in recruiting ESCRT-III complex.
Interestingly, some researchers have found that CD63 markers associated with exosomes still exist despite knocking out ESCRT complexes, suggesting that exosomes can also be formed in an ESCRT-independent manner. The ceramides, transmembrane 4 superfamily tetraspanins and heat shock proteins assisted ILVs and multivesicular body (MVB) with their formation [34]. Neutral sphingomyelinase hydrolyzes sphingomyelin to form ceramide, and both the ceramide production and the exosomes were significantly reduced when neutral sphingomyelinase is inhibited by the enzyme inhibitor GW4869 [35]. The constant supply of S1P catalyzed by SphK2 allows the S1P receptor to remain active on MVE, and this continuous activation of the S1P receptor is required for cargo sorting into the ILV, while the intervention of FTY720 leads to inhibition of the S1P signal on the MVE, and prevents the maturation of exosomes [36]. As a regulator of ILV germination and exosome production, the small G protein ADP ribosylation factor 6 (ARF6) on the endocytic membrane activates phospholipase D2, while phospholipase D2 can invade the cytoplasmic membrane and produce MVB and ILVs [34]. In addition, tetraspanins are also frequently found to play an important role in the formation of LVs and exosomes [37].
3.2 Mechanism of secretion of exosomes to the extracellular
The fusion process of the vesicles produced by the donor membrane with the receptor membrane is an important step in the vesicle transport pathway, which includes two processes: the vesicle specifically recognizes the target membrane, and the vesicle fuses with the target membrane to release the contents. SNAREs have been found to be involved in this step, which includes two contents: the vesicular SNAREs (VAMPs) that is localized in the donor membrane, and the target SNAREs (t-SNAREs) that is localized in the acceptor compartment [38]. Syntaxin 3 (STX3) plays a role in the recruitment of exosomes, the STX3-5r mutation as a major dominant negative inhibitor is effective in reducing the number of Human cytomegalovirus (HCMV) particles in polarized epithelial cells. These results indicate that STX3 plays a role in the transport of vesicle contents [39]. Studies by Gross et al. showed that in HEK293 cells, the SNARE protein component YKT6 promoted the production of exosomes loaded with WNT3A morphogens [40].
Secretion of exosomes involves the machineries that separate microvesicles (MVs) from plasma membranes, the movement of secreted MVs to the periphery of the cell, and the fusion of vesicles with the cell surface. The above processes are related to the cytoskeleton, molecular switches, molecular motors, and fusion mechanisms [2, 41]. The Rab protein family is the largest subfamily of the Ras superfamily, and includes small GTP-binding proteins. As a molecular switch for vesicle trafficking, Rab proteins control the transport of intracellular vesicles (Fig. 3), such as vesicle formation, transportaton, adhesion, anchoring, and fusion, suggesting that Rab may be involved in the secretion of exosomes [42]. The expression of Rab11 regulates exosome secretion by reticulocyte cell lines, which proves that Rab GTPases plays a role in exosomes secretion.
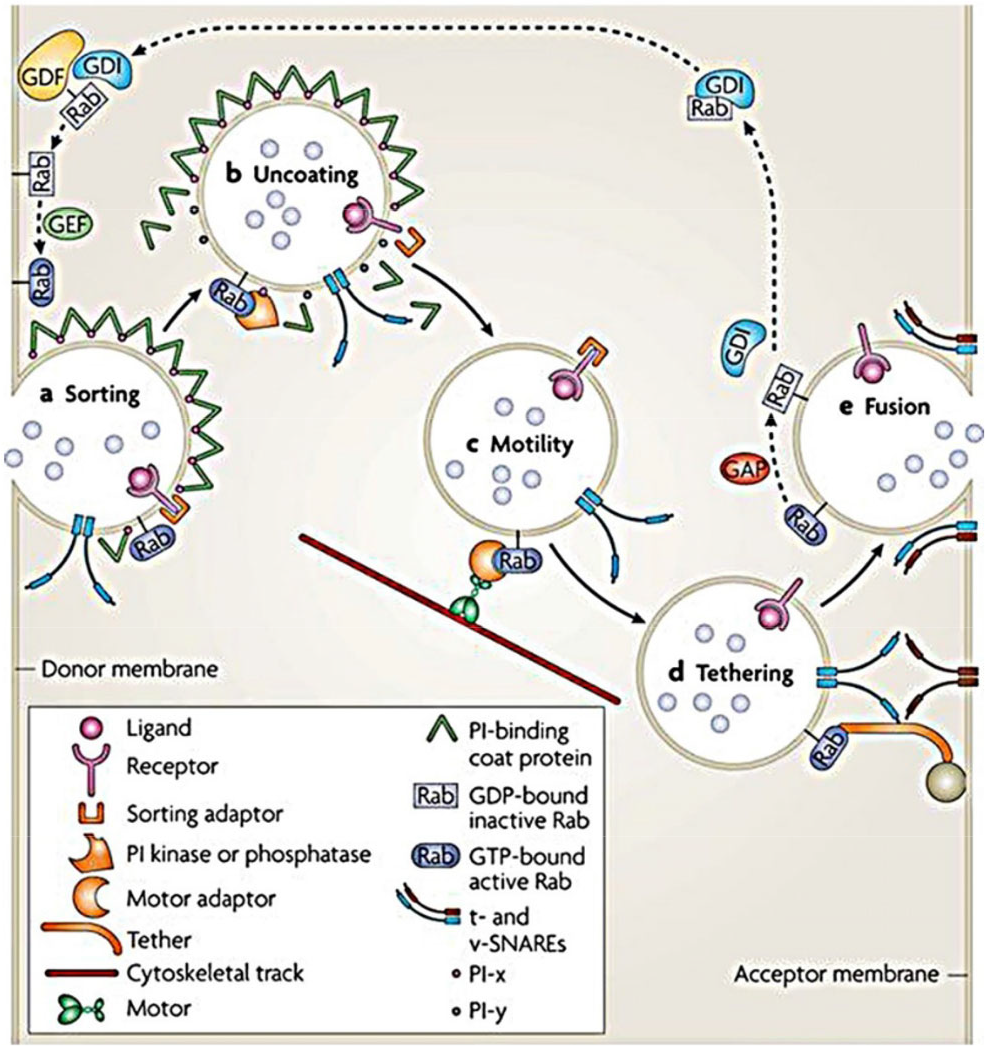
Abrami et al. demonstrated that SNX3, TSG101, and ALIX are involved in the biosynthesis of LF-loaded exosomes in human retinal pigmented epithelial 1 (RPE1) cells, and the Rab11 and Rab35 actually act on exosomes release rather than Rab27, thus anthrax toxins through the secretory pathway spread to distant cells [43]. Studies have found that Rab27a and Rab27b can promote the distribution of MVEs to the periphery of cells, and play a role in the docking of MVEs and plasma membrane. In addition, knockdown of Rab27a and Rab27b significantly reduced the number of HeLa cell exosomes; MVEs in total internal reflection fluorescence (TIRF) microscope imaging showed that silencing of Rab27b significantly reduced MVEs density [44]. In primary oligodendrocytes, silencing of Rab35 can interfere with glutamate-dependent release of proteolipid protein (PLP) and ALIX [45]. Notably, studies have shown that the effects of GTPases can be complementary, for instance, the release of exosomes is only partially affected by selective inactivation of Rab11, Rab27, and Rab35.
4 Therapeutic potential
Ischemic stroke is the leading cause of chronic disability worldwide [46]. Recombinant tissue-type plasminogen activator (alteplase) was approved by the US Food and Drug Administration (FDA) in 1996. So far, it still remains the only medication proven to affect outcomes when given in the hyperacute time frame after ischemic stroke [47, 48]. The regression line indicated that the treatment would be effective if patients are treated with TPA within 4.5 hours after stroke, while it would be ineffective after 4.5 hours, even harmful after 6 hours [49]. “Time is the brain”, but the rigorous 4.5-hour time window often prevents this treatment from being fully utilized [50]. Intravascular thrombectomy can extend the treatment window and reduce the risk of intracranial bleeding. However, due to vessel tortuosity, the stenosis of the arteries, and the inaccessibility of blood clots, the mechanical recombination device also showed limitations [10]. Therefore, it is crucial to find a safer and more effective treatment for stroke.
4.1 Exosomes transplantation therapy directly
The core objective of regenerative medicine is to repair or regenerate various tissues and organs, and to address tissue and organ functional disorders caused by diseases, trauma, aging, or genetic factors [51]. The functional recovery of cell therapy in regenerative medicine plays an extremely important role, for example, the inflammation can be effectively controlled, and the nerve function is restored after the transplantation of cells into damaged region. Stem cells have become the primary choice for transplantation therapy because of their “infinite” proliferation and multi-prospective differentiation potential. Moreover, stem cell transplantation therapy has been shown to promote the recovery of damaged tissues [52, 53], including spinal cord, heart, brain, and liver. Previous researches have shown that cell proliferation can be found in subventricular zone in rat model disposed by the middle cerebral artery occlusion (MCAO), suggesting that endogenous recovery process is started after stroke [54 –57]. However, endogenous resilience may be limited due to hostile immune microenvironment over time [56, 58]. In addition, the delayed inflammatory response to stroke leads to secondary neurological injury [52, 58]. Insufficient endogenous stem cells after stroke can result in weak regeneration of nerve cells in the damaged area and slowdown in tissue function recovery. It is an effective treatment to solve the problem of endogenous neuronal defects after stroke by directly injecting exogenous stem cells into the ischemic area [58 –60].
However, paracrine factors (especially exosomes) appear to play a major therapeutic role in recent studies, although the therapeutic effects of cell transplantation can be directly and indirectly explained by growth factors and cytokines [61 –63]. Moreover, Xin et al. have demonstrated increased generation of posterior nerve cells and oligodendrocytes in stroke-injured areas after intravenous injection of exosomes containing miRNA-17–92 cluster (Fig. 4) [59]. Symptoms can be effectively relieved in other systemic diseases after exosomes are transferred to the site of injury. For example, cardiac-derived exosomes have been shown to have cardio-protective effects, stimulate regenerative signals, and participate in repair after myocardial infarction [64]. The above examples demonstrate that exosomes-transplantation therapy could play a practical role in the treatment of disease. Notably, Kalani found that stem cellderived exosomes are not only as effective as stem cell transplants, but also overcome the limitations of ischemic stem cell therapy compared to the previous cell therapies [7].
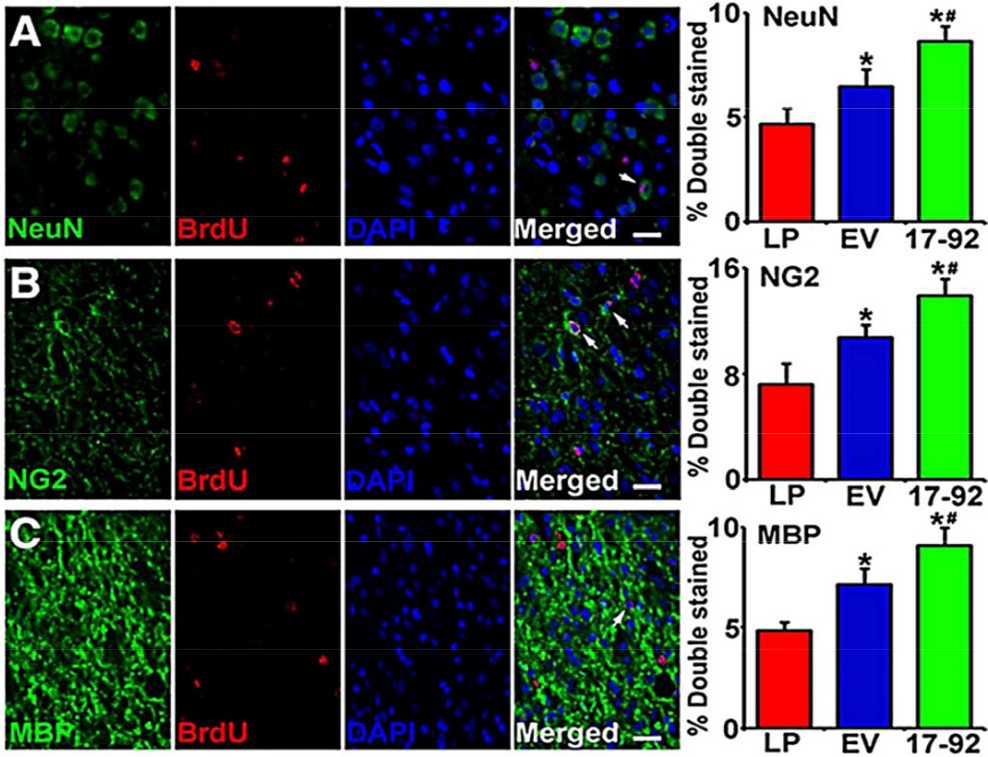
miRNA-17–92 cluster-elevated multipotent mesenchymal stromal cell (MSC) exosomes increase neurogenesis and oligodendrogenesis in the ischemic boundary zone (IBZ). Representative micrographs show the double stained cells with neuron (NeuN) and 5-bromodeoxyuridine (BrdU) (A), progenitor (NG2) and BrdU (B
4.2 Cell source and delivery path of exosome transplantation
4.2.1 Cell source
The cell source and injection path of the exosomes are also critical for the treatment of stroke. Studies have shown that exosomes released from stem cells may activate the self-regenerating process of injured cells by providing stem cell-like phenotypes, providing therapeutic potential for a variety of diseases [66]. For instance, mesenchymal stem cell (MSC)-derived exosomes have numerous benefits against stroke by promoting functional recovery, neurovascular plasticity, neuroprotection, nerve regeneration, and regulation of peripheral immune responses. Therefore, the source of stem cells for exosomes used for stroke treatment needs to be considered carefully. Embryonic stem cells (ESCs) have the characteristics of infinite proliferation, self-renewal and multi-directional differentiation in vitro, and it can be induced to differentiate into almost all cell types both in vitro and in vivo. However, the research on exosomes derived from embryonic stem cells indicated that the embryos need to be damaged, and found that some embryonic exosomes carry infectious particles, which limits its application [11]. Induced pluripotent stem cells (IPSCs) can avoid similar problems as a source of exosomes, but the application of iPSCs has also encountered obstacles that may be involved in tumorigenesis [67]. In recent years, exosomes derived from adult stem cells have attracted much attention because they have no significant side effects on recipient cells. MSCs come from a wide range of sources, not only in bone marrow, bone, muscle, or adipose tissue, but also from tissues isolated from the brain, kidney, liver, and spleen [68]. Moreover, MSCs are abundantly productive compared to other cells known to produce exosomes [69]. The exosomes secreted by endothelial progenitor cells (EPCs) are rich in miRNA-126 and miRNA-296, and have an effect of promoting angiogenesis and antiapoptosis. In addition, neural stem cells produce all major cell types of the central nervous system including neurons, astrocytes, and oligodendrocytes, and their secreted exosomes are bound to enhance functional recovery after stroke. Chen et al. found that human neural stem cells-derived exosomes produced under hypoxia preconditioning can promote the treatment of stroke [70]. Neural stem cells (NPCs) have stronger differentiation potential and renewal ability than neural progenitor cells. It is also a prolific producer of exosomes similar to mesenchymal stem cells. Adult stem cells may be a good source of cells for exosomes.
4.2.2 Delivery path
Various forms of exosome delivery route are used to treat ischemic stroke, such as intravenous/intra-arterial infusion, intracerebral route, and intracranial injection. Intracranial injection is a classic method of injection, which involves injecting exosomes directly into the injured area. However, direct injection is traumatic and single exosome injection can’t do much, otherwise it will cause an intracranial lesion, and has certain complications such as subdural hematoma, epilepsy. Intravenous injection has attracted much attention due to its low damage and convenience as a common transplant approach. However, high blood volume in the blood vessels and extremely fast blood flow rate will lead to insufficient intracerebral cell concentration. In addition, exosomes that are not specially treated can be quickly eliminated by phagocytosis in the reticuloendothelial system (RES), filtered by the kidneysor the bile excretion as the blood flows through the body organs, thereby resulting in a decreased concentration of exosomes in the brain. This is similar to the result of most stem cells staying in peripheral organs after intravenous transplantation. For example, Pluchino found that after intravenous injection into the experimental autoimmune encephalomyelitis (EAE) mice model, only about 3% of the NPCs migrate back to the central nervous system after 24 hours, whereas most of the cells are distributed throughout the body tissues [71]. In addition, for stroke, intranasal delivery of exosomes is an alternative choice because of its simple operation and noninvasiveness. The uniqueness of intranasal route is that it is a non-invasive method, and transmission of the material can bypass the blood–brain barrier, and the transfer medium are nasal nerves, such as trigeminal nerve, cerebrospinal fluid [72 –74]. Zhuang found that exosomes carrying curcumin and activator of transcription 3 (Stat3) inhibitor can delay the EAE disease, and inhibit the growth of tumors in vivo by intranasal injection [75]. This experiment demonstrates that exosomes can effectively reach the central nervous system through the intranasal route.
4.3 Exosomes as carriers
Drug therapy is a key part of the treatment routine of ischemic stroke. However, conventional drug treatment often has some problems, such as poor water solubility, low cell penetration, and dispersion in the body. These problems will lead to increase of drug dosage by limiting the efficacy of the drug and increasing the likelihood of side effects, or wasting medical resources. Thus, in addition to the further development of drugs, a reasonable drug delivery system can solve the current problems, and enable drugs to reach the injured site safely and effectively. As a natural nano-scale particulate material, exosomes have the potential of being material transport carriers because of their biocompatibility, low immunogenicity, and stable circulation in the blood [76]. In addition, exosomes have significant advantages over existing drug carriers (such as artificial liposomes): some proteins on the surface of exosomes from different cell sources allow the exosomes to selectively bind to the receptor cells. Meanwhile, the specific surface molecules of exosomes can avoid interaction with antibodies and clotting factors in specific cells [such as immature dendritic cells (DCs)], avoiding the human immune response as much as possible. Studies have found that sustained activation of immune effectors in some patients help maintain longterm disease stability after receiving autologously derived exosomes [77, 78].
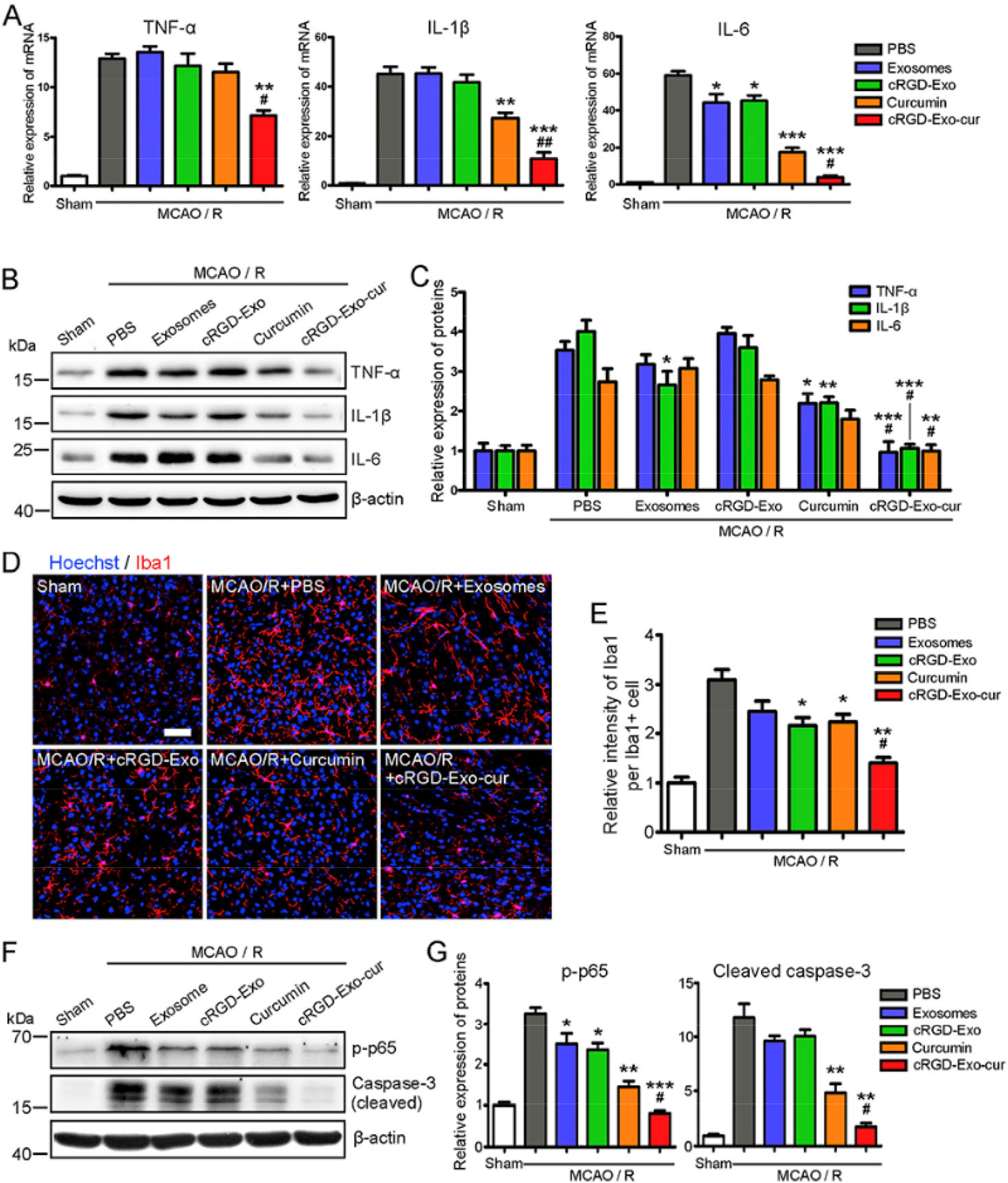
Studies have shown that small molecule drugs have a positive effect on the treatment of stroke. For instance, salidroside promotes the expression of M2 markers in microglia after stroke, and protects neurons by promoting polarization of M2 microglia [79]. Moreover, it can reduce the brain infarct size, and improve neurological function in animal models. It is an alternative choice for therapeutic molecules to be delivered to the injury sites by exosomes. Gao has demonstrated that curcumin-loaded cRGD-Exo lead to a strong inhibition of inflammatory response in the lesion area of ischemic mice (Fig. 5) [80]. Blood–brain barrier, however, is the major obstacle to traditional methods of transmission, although damaged barrier cells caused by stroke will slowly repair over time. Because of the blood–brain barrier, only one drug among the top ten molecules is a neurological treatment drug according to the statistics of the world’s top prescription drugs in 2014 [81]. Exosomes were found to be able to cross the blood–brain barrier [82], and exosomes can be used for drug delivery in the nervous system that requires efficient, non-toxic drugs. Interestingly, the exosomes are lipid bimolecular membranes, but they have a hydrophilic core, which is very important for the delivery of soluble drugs [83]. At present, there are two methods for binding molecular drugs to exosomes: 1. Bind drugs to donor cells, and separate exosomes containing therapeutic molecules from cells. 2. The exosomes are first purified from the cells, and the therapeutic molecules are loaded into the exosomes by specific methods. These methods include co-incubation of the exosomes with the drug, electroporation applied to open a transient pore in the exosomes, during which time the small molecule drugs were inhaled into the exosomes [76], and direct chemical transfection of the exosomes. In addition, exosome donor cells can be selected from immature DCs because exosomes derived from immature DCs only cause a weaker immune response [84].
Genes can be loaded by exosomes. The phospholipid bilayer structure of the exosome surface ensures that the genes present in the exosomes are protected from degradation during long-term cycling, and the stability of the contents is highly maintained to synergize with the recipient cells. Exosomes can also act on recipient cells by loading foreign genes. Brain-derived neurotrophic factor (BDNF) is a protein synthesized in the brain that is widely distributed in the central nervous system. It plays a key role in the survival, differentiation, and growth of neurons during the development of the central nervous system. Studies have shown that BDNF can prevent acute ischemic injury, increase brain tissue repair, and promote synaptic plasticity [85]. Chen et al. found that BDNF has neuroprotective effects on MCAO-treated mice, thus the binding of BDNF to the modified exosomes can be introduced into the injured tissues to support the nerves in the damaged area [86]. Transferring these genes into donor cells by certain means (e.g. genetic engineering) can improve tissue ischemia in the intracranial area by producing exosomes to induce regeneration of blood vessels.
5 Conclusion
In summary, we briefly discussed the physiology, origin, disease-related mechanisms, and therapeutic potential of exosomes. However, many problems related to exosomes still exist. For example, it is unclear what the molecular mechanism is, by which MVB fusion sites are defined in the plasma membrane, how receptor cells specifically select exosomes, and what the mechanisms of fusion is. Although the differential centrifugation operation is simple, and more vesicles can be obtained to meet the experimental needs, some problems haven’t been solved. For example, the centrifugation process is timeconsuming, and the recovery rate is unstable. In addition, repeated centrifugation operations can also result in damage to the vesicles, which can degrade quality. Commercial kits capture more exosomes from the cell supernatant, but the purity is questioned. Exosomes treatment offers new insights into stroke treatment by minimizing adverse effects, which may replace cell-based therapies. However, it is likely, as cell-based therapies, most of the exosomes are distributed in other organs such as liver. How to effectively improve the targeting of exosomes will be the key to therapeutic applications of exosomes.
Footnotes
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81671819).
Conflict of interests
The authors declare no conflict of interests.
