Abstract
Introduction:
Neuropsychiatric systemic lupus erythematosus is often clinically challenging to diagnose, treat and monitor. Although brain magnetic resonance imaging is frequently performed before lumbar puncture in neuropsychiatric systemic lupus erythematosus, it is not clear from the literature whether specific brain magnetic resonance imaging findings are associated with distinct clinical features of neuropsychiatric systemic lupus erythematosus.
Methods:
We conducted a systematic review and meta-analysis on published studies of neuropsychiatric systemic lupus erythematosus including brain magnetic resonance imaging and the 1999 American College of Rheumatology-defined clinical neuropsychiatric systemic lupus erythematosus syndromes to determine their relationship. Pooled prevalence and risk ratio for distinct neuropsychiatric systemic lupus erythematosus associations were determined with 95% confidence intervals.
Results:
Of 821 studies screened, 21 fulfilled inclusion criteria. A total of 818 participants were evaluated (91% female) with 1064 neuropsychiatric systemic lupus erythematosus episodes assessed. Neuropsychiatric systemic lupus erythematosus features included headache (24%), seizures (19%), cerebrovascular disease (18%), cognitive dysfunction (15%) and acute confusional state (14%). Normal magnetic resonance imaging was significant for anxiety disorder (risk ratio: 9.00; 95% confidence interval: 2.40, 33.79), autonomic disorder (risk ratio: 7.00; 95% confidence interval: 0.51, 96.06) and plexopathy (risk ratio: 5.00; 95% confidence interval: 0.81, 31.00). Highest risk ratio of neuropsychiatric systemic lupus erythematosus syndrome with abnormal magnetic resonance imaging was observed for cerebrovascular disease (risk ratio: 0.15; 95% confidence interval: 0.10, 0.24) and demyelination (risk ratio: 0.11; 95% confidence interval: 0.02, 0.72).
Conclusion:
Normal magnetic resonance imaging in neuropsychiatric systemic lupus erythematosus was the most significant correlate from our meta-analysis for psychological symptoms including anxiety and peripheral nerve features of autonomic disorder and plexopathy. The main abnormal brain magnetic resonance imaging correlates included cerebrovascular disease and demyelination. Brain magnetic resonance imaging correlates poorly with neuropsychiatric systemic lupus erythematosus features, and specific clinical symptoms should be the main determinants of performing magnetic resonance imaging rather than presence of neuropsychiatric systemic lupus erythematosus per se.
Introduction
Systemic lupus erythematosus (SLE) is a chronic auto-immune disorder that affects multiple organ systems with inflammation, immune complex deposition and vasculopathy as the primary pathological findings. The disease prevalence of SLE varies worldwide from 13 to 51 cases per 100,000 and has been reported as increasing due to improvements in survival.1,2 The condition shows a marked female predominance, being approximately nine times as common in women than in men.3–5
Neuropsychiatric systemic lupus erythematosus (NPSLE) is characterised by diverse syndromes affecting both the central and peripheral nervous systems (CNS and PNS). Features can range from mild headaches to more severe manifestations, such as epilepsy, cerebrovascular disease, psychiatric disorders, cranial and peripheral neuropathies.6,7 NPSLE features can be difficult to distinguish from other diseases, while the heterogeneity of the condition can make the conduct of research challenging. Objective and subjective criteria have been used for inclusion in studies, while variations in baseline patient characteristics and duration of observation may add to the inconsistency of the reported data.6,8,9 The prevalence of neuropsychiatric NPSLE varies from 31% to 91%. 9
Several processes have been implicated in the pathogenesis of NPSLE. Local release of inflammatory cytokines, autoantibody-mediated response to CNS components and formation of immune complexes, resulting in cerebrovasculopathic changes and neuronal injury may all be key.10–13 The presence of antiphospholipid antibodies may exacerbate the disease, increasing the risk of thrombotic arterial or venous disease and accelerated atherosclerosis. 14
Diagnosis and management options are dependent on the underlying syndrome. The adoption of cerebrospinal fluid (CSF) analysis, electroencephalography (EEG) and neuroimaging techniques has been recommended by the European League Against Rheumatism (EULAR) task force for the diagnosis of NPSLE or to exclude concomitant illnesses, infection or drug side effects. 15 Despite this, correct attribution of neuropsychiatric events to NPSLE remains a clinical challenge as there is no gold standard diagnostic test. Glucocorticoids, immune-modulatory drugs and anticoagulants/anti-platelets have been used empirically in the treatment of inflammatory and vasculopathic causes of NPSLE. However, clinical trial data on these interventions remain sparse.
Although the prognosis of SLE has improved considerably in recent times, NPSLE still remains a major cause of morbidity and mortality. A recent study reported a 19% mortality rate, with infection and NPSLE attributed as the leading causes of death in SLE patients. 16 Despite the frequent occurrence of brain involvement in SLE, our knowledge of NPSLE syndromes and their association with imaging findings remains speculative. The American College of Rheumatology (ACR) published a consensus for the classification, nomenclature and case definitions for 19 distinct neuropsychiatric syndromes in 1999, with the aim of facilitating research. 7 The criteria have been subject to debate in several studies, some of which have questioned the low specificity for diffuse syndromes such as headache, cognitive dysfunction and minor psychiatric disorders commonly reported in chronic diseases generally.8,17,18
In a meta-analysis conducted in 2011, headache (12%) was determined as the most prevalent syndrome, followed by mood disorder (7%), seizure disorder (7%), cognitive dysfunction (7%) and cerebrovascular disease (CVD) (5%). PNS syndromes such as autonomic disorder and Guillain–Barré syndrome (GBS) (<0.1%) were rarely reported. 9 Unterman et al. 9 reported that Asians (72%), Caucasians (16%) and Hispanics (4%) were the most frequently affected ethnicities.
Brain magnetic resonance imaging (MRI) remains the most commonly used imaging technique to assist in the diagnosis of NPSLE due to its wide availability. Recently, more advanced imaging tools, such as the MRI methods of magnetisation transfer imaging (MTI), diffusion-weighted imaging (DWI) and diffusion tensor imaging (DTI), and methods using radioactive tracers, such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT), have been increasingly studied.15,19 Furthermore, the EULAR task force has recommended the use of advanced imaging in cases of normal MRI findings in patients with NPSLE, providing further rationale to specifically assess MRI. Table 1 lists the different imaging modalities, their descriptions and common findings. SPECT studies have demonstrated a higher sensitivity in patients with diffuse neurological involvement compared with MRI.19,26,27 However, the cost and limited availability of SPECT and PET and the need for better standardisation and validation for interpretation of advanced quantitative MRI protocols have prevented their establishment in routine clinical practice thus far.15,28,29 For this reason, the focus of our study was conventional MRI.
Summary of imaging modalities, their descriptions and common findings in NPSLE.
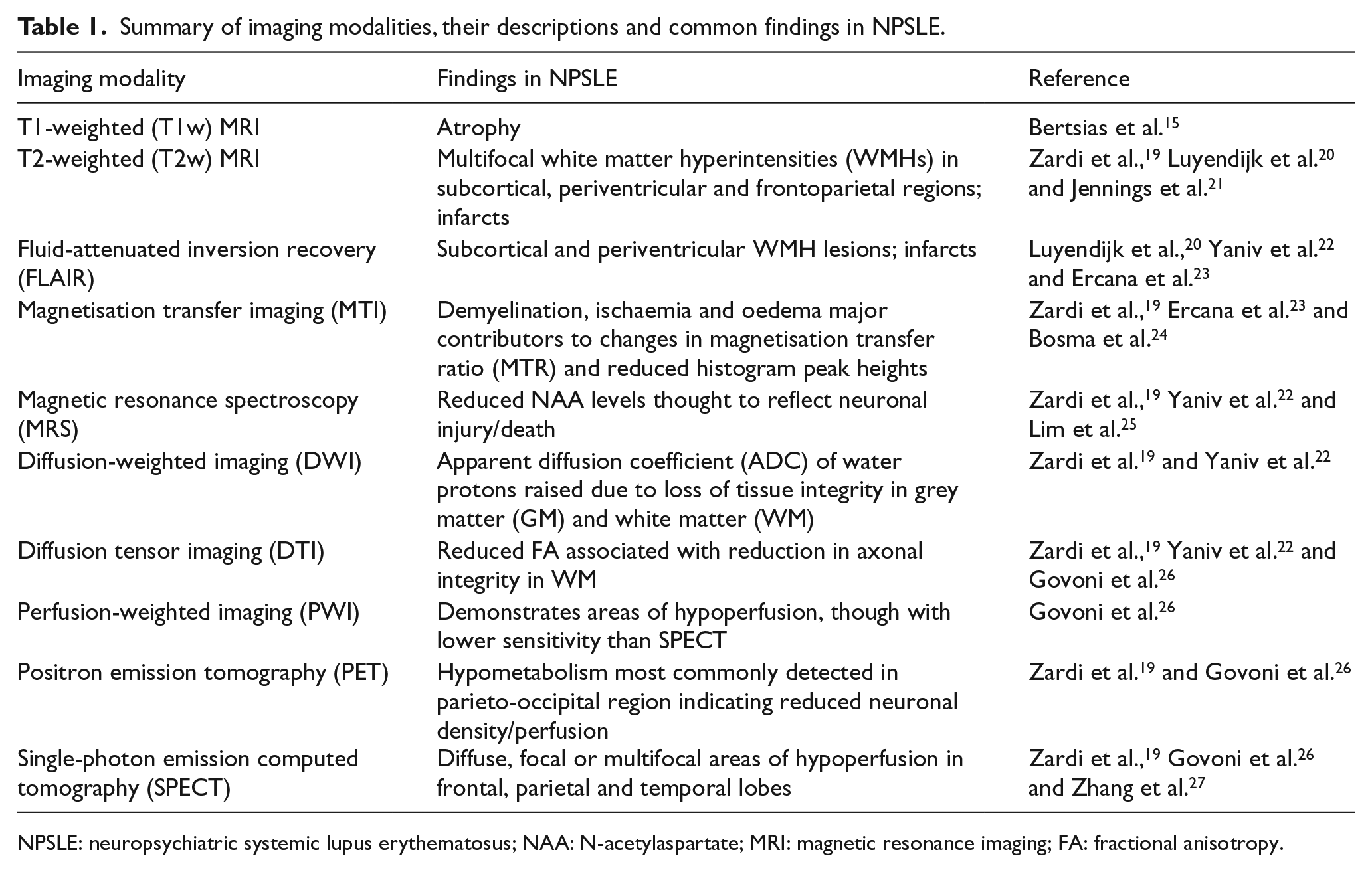
NPSLE: neuropsychiatric systemic lupus erythematosus; NAA: N-acetylaspartate; MRI: magnetic resonance imaging; FA: fractional anisotropy.
The aim of this systematic review and meta-analysis was to assess whether the clinical NPSLE syndromes defined by the 1999 ACR criteria are linked to distinct brain MRI abnormality. This is important as any brain MRI association with specific NPSLE features could potentially aid clinicians in diagnosing NPSLE with more confidence and link individual NPSLE syndromes with structural or pathologic changes such as thrombosis and CVD.
Materials and methods
Search strategy
The electronic databases PubMed, MEDLINE (via Ovid), Embase (via Ovid) and the Cochrane Library were searched from their inception to November 2015. The grey literature and trial registries OpenGrey, WHO International Clinical Trials Registry Platform, Current Controlled Trials and the United States National Institute of Health Trials Registry were also searched from inception to November 2015 for any additional papers omitted from the principle search. The terms used for the MEDLINE search were as follows: systemic lupus erythematosus, neuropsychiatry/neurologic disease, lupus erythematosus, neuropsychiatric systemic lupus erythematosus, lupus vasculitis, CNS, nuclear magnetic resonance imaging, magnetic resonance imaging and MRI.
Study eligibility and identification
Papers were eligible if they provided data on the prevalence of clinical syndromes for NPSLE and presented corresponding brain MRI imaging data to assess the prevalence for individual clinical syndromes. Papers were excluded if they were reviews, case reports, letters to the editor or studies not published in English or assessing human subjects. Studies were included irrespective of the number of cases of NPSLE evaluated in individual studies due to the relative rarity of NPSLE1,2 and to minimise the risk of small sample size publication bias from impacting on the review’s analysis. The results of the search strategy were reviewed by two authors (O.A., A.K.). Each title and abstract was assessed against the eligibility criteria. The full texts were obtained for those papers which were thought to be potentially eligible. The same two reviewers assessed the eligibility of each full text, and if they met the criteria, they were included in the review. In our search, we were unable to include comparisons of brain MRI changes in patients without SLE and without neuropsychiatric manifestations, as we were unable to find studies of this kind and we recognise this as a limitation. We were also unable to compare MRI findings between NPSLE versus SLE patients or SLE patients versus healthy controls, as we were unable to find any studies outlining these specified comparisons. Instead, we classified SLE patients based on the outcomes, that is, having normal versus abnormal MRI scans and then compare each group based on individual NPSLE syndrome. There are two issues with this approach. First, the definition of normal MRI scan is more difficult to establish and more subjective. Second, there is no causal relationship or correlation between NPSLE syndrome and MRI finding which can be concluded. Third, there is a chance that some of the NPSLE syndromes may (e.g. anxiety) develop later despite a normal structural MRI scan. Our results therefore need to be interpreted with these limitations in mind.
Data extraction and appraisal
Data were extracted from each included paper by two reviewers (O.A., A.K.). This was performed independently, with the results verified by each other. Data which were extracted onto our data extraction form included the following: country of study, study design, MRI imaging sequence, number of patients MRI assessed, number of syndromes detected, mean and range of ages of subjects, gender of subjects, duration of NPSLE, age of onset, disease activity, clinical syndromes detected, prevalence of syndromes within the cohort, prevalence of normal and abnormal MRI findings and prevalence of specific MRI findings. The MRI findings extracted included the following: white matter hyperintensities, grey matter hyperintensities, parenchymal defects, cerebral atrophy, large vessel disease, small vessel disease, inflammatory lesions, microbleeds, recent infarct, lacunes, vascular signs, intracranial haemorrhage, focal oedema, diffuse cerebral oedema, punctuate focal lesions, periventricular lesion and microcalcification.
Two reviewers (O.A., A.K.) independently assessed the quality of each included paper using a modified Critical Appraisal Skills Programme (CASP) 30 checklist. This was chosen as it is a valid tool which consists of a set of questions designed to evaluate the internal and external validities of clinical studies. The results from each reviewer were evaluated to gain a consensus on the final quality assessment judgement.
Data analysis
An assessment of study heterogeneity was derived through assessment of the data extraction table. Where population, study design, data collection and MRI investigation were considered comparable, a pooled assessment of prevalence of clinical syndromes in NPSLE was made. Similarly, an assessment of pooled prevalence of MRI features for each specific clinical syndrome was made. The risk ratio (RR) was determined of detecting an abnormal MRI result compared to normal MRI for people with NPSLE for each individual clinical syndrome. For each calculation, pooled prevalence was presented as a percentage with 95% confidence intervals (CIs). Similarly, RR was assessed with 95% CIs using a random effects model analysis and statistical heterogeneity using I2 and χ2 statistical tests. Sensitivity analyses were conducted, where appropriate assessing outcomes, when there were 100 participants or more to minimise the risk of small study effects from impacting on the outcome. 54 Small sample size publication bias was assessed using a funnel plot of the most frequently reported NPSLE feature (seizures). All analyses were conducted on RevMan (Review Manager version 5.1; Copenhagen: The Nordic Cochrane Centre (the Cochrane Collaboration, 2011)) and Stata version 12.0 (StataCorp LP, College Station, TX, USA).
Results
Search strategy
A summary of the search results is presented in Figure 1. A total of 821 citations were identified from the search strategy. Of these, 21 were deemed eligible and included in the analysis based on the search criteria.
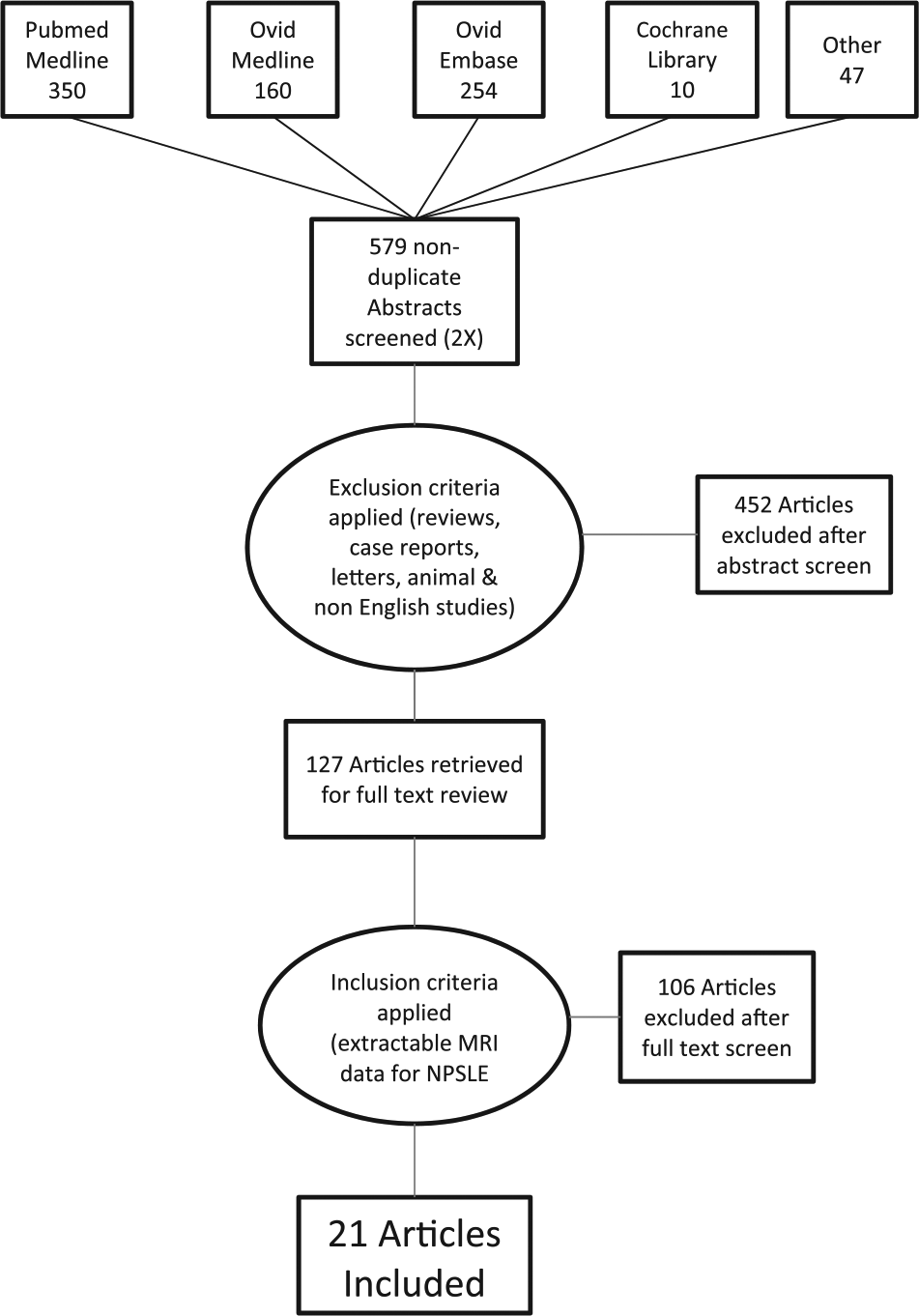
Flow chart summarising the results of the search strategy and the process of study inclusion/exclusion.
Study quality assessment
Full assessment of the studies with their total scores is shown in Table 2. The results of the CASP appraisal highlighted several methodological limitations. First, while 90% defined the study population and the recruitment process, only 57% of studies used the 1999 ACR guidelines for NPSLE case definitions, with research published prior to 1999 relying on clinicians for assessment and definitions used. Only 29% enrolled control groups for comparison, with either healthy individuals or SLE participants with no brain involvement serving as controls. Just 24% of the studies described blinding of the neuroradiologist to minimise bias. The quality of the evidence base was further weakened, as 57% of the studies did not mention any confounding factors. A total of 38% of studies carried out a statistical analysis, and 71% described their methods section in sufficient detail.
Table showing study characteristics and critical appraisal assessment scores using a modified CASP tool.
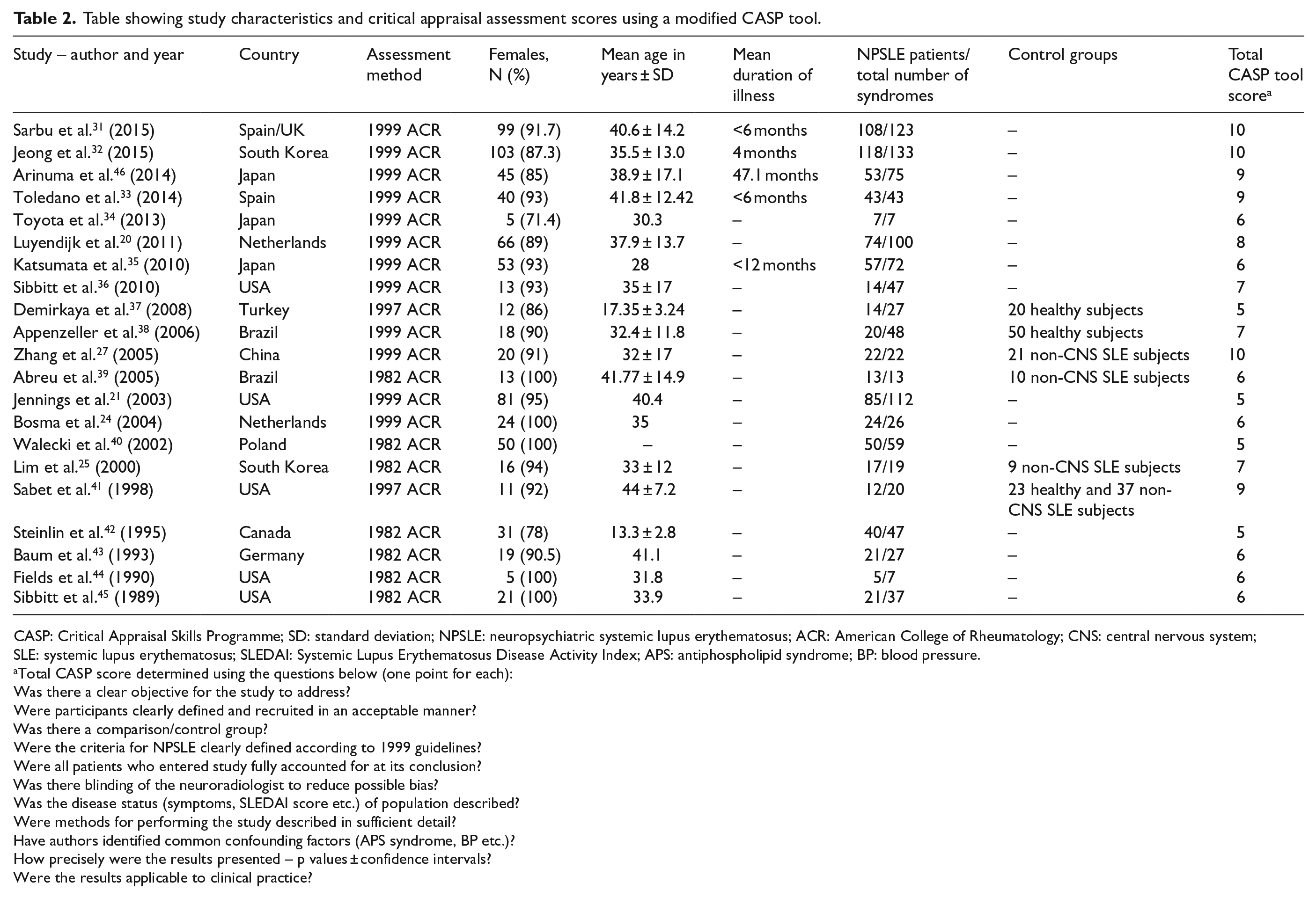
CASP: Critical Appraisal Skills Programme; SD: standard deviation; NPSLE: neuropsychiatric systemic lupus erythematosus; ACR: American College of Rheumatology; CNS: central nervous system; SLE: systemic lupus erythematosus; SLEDAI: Systemic Lupus Erythematosus Disease Activity Index; APS: antiphospholipid syndrome; BP: blood pressure.
Total CASP score determined using the questions below (one point for each):
Was there a clear objective for the study to address?
Were participants clearly defined and recruited in an acceptable manner?
Was there a comparison/control group?
Were the criteria for NPSLE clearly defined according to 1999 guidelines?
Were all patients who entered study fully accounted for at its conclusion?
Was there blinding of the neuroradiologist to reduce possible bias?
Was the disease status (symptoms, SLEDAI score etc.) of population described?
Were methods for performing the study described in sufficient detail?
Have authors identified common confounding factors (APS syndrome, BP etc.)?
How precisely were the results presented – p values ± confidence intervals?
Were the results applicable to clinical practice?
Characteristics of included studies
In total, 818 individuals (745 females/73 males) with 1064 NPSLE clinical syndromes were included and analysed. The study sample’s ages ranged from 13.3 42 to 44.0 years. 41 The duration from NPSLE diagnosis to MRI was not specified in 16 papers. In three papers, the duration from NPSLE diagnosis to MRI was less than 3 months,31,32,33 it was up to 1 year in one study, 35 while it was a mean 47.1 months in another study. 46 Disease activity was defined and reported in eight studies. This ranged from 4.7 Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) 38 to 34 SLEDAI 36 and 14.2 British Isles Lupus Activity Group (BILAG). 46
A variety of MRI investigation descriptions were provided. In 10 papers, T1, T2 and fluid-attenuated inversion recovery (FLAIR) were used;20,21,31,33–36,46,39,40 four studies simply stated that MRI was undertaken.37,43–45 Imaging was described in individual studies as follows: T1, T2, FLAIR and DWI; 32 T1, T2, voxel-based morphometry; 38 T1, T2, FLAIR, SPECT; 27 T1, T2, FLAIR, MTI; 24 T1, T2, magnetic resonance spectroscopy (MRS); 25 and MRI, SPECT. 42
Five studies were undertaken in the United States,21,36,41,44,45 three in Japan,34,35,46 two in South Korea,25,32 two from the Netherlands20,24 and two from Brazil.38,39 Single studies were undertaken in Turkey, 37 China, 27 Poland, 40 Canada, 42 Germany 43 and Spain, 33 while one study was conducted in both Spain and the United Kingdom. 31 Nine papers reported retrospective studies, while seven were prospective studies. It was unclear whether the studies were retrospective or prospective in five instances.
Publication bias
Small sample size publication bias was assessed using a funnel plot. As represented in Figure 2, for the measure with the largest data set (seizures), this produced a broadly symmetric funnel plot suggesting low risk of publication bias.
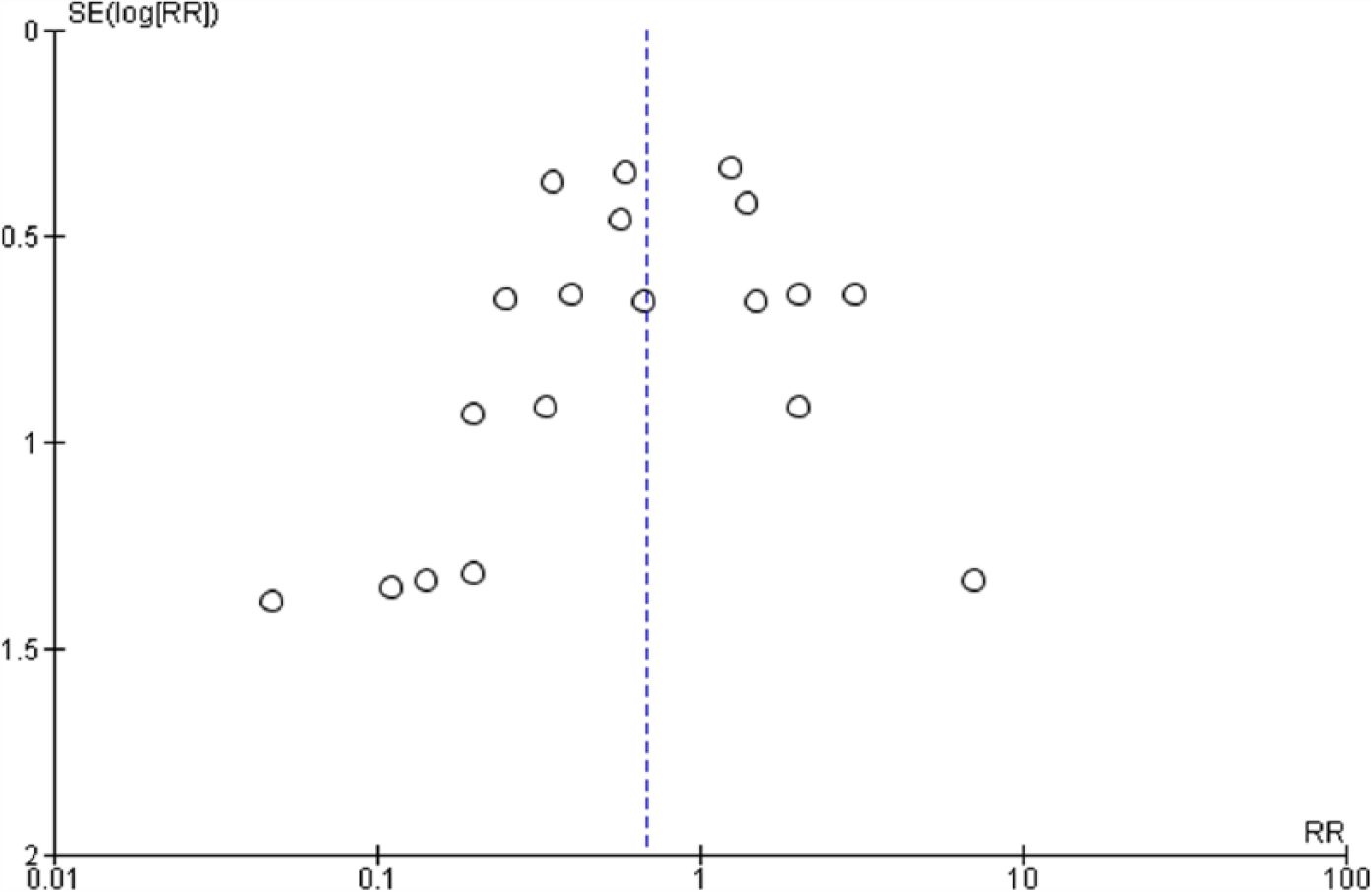
Funnel plot of small sample publication bias for the assessment of seizures with NPSLE and abnormal compared versus normal MRI findings.
Meta-analysis
Prevalence of clinical syndromes in NPSLE
Pooled prevalence of clinical syndrome in NPSLE was headache, with a prevalence of 23.8% (95% CI: 21.1%–26.9%; N = 818). This was followed by, in order of frequency, seizures (prevalence: 18.9%; 95% CI: 16.3%–21.7%; N = 818), CVD (prevalence: 17.6%; 95% CI: 15.1%–20.4%; N = 818) and cognitive dysfunction (prevalence: 15.3%; 95% CI: 13.0%–17.9%; N = 818). The least prevalent clinical syndromes were myasthenia gravis (prevalence: 0%; 95% CI: 0.0%–0.01%; N = 712), GBS (prevalence: 0.3%; 95% CI: 0.08%–1.1%; N = 659), plexopathy (prevalence: 0.9%; 95% CI: 0.4%–2.0%; N = 659) and polyneuropathy (prevalence: 1.4%; 95% CI: 0.1%–2.6%; N = 659).
RR of an abnormal MRI finding for individual clinical syndromes in NPSLE
A summary of the pooled prevalence of normal and abnormal MRI brain findings for each clinical syndrome with NPSLE is presented in Table 3. There was variable statistical heterogeneity for each of these analyses (Supplementary Table 1). The highest prevalence of normal MRI brain features for individual clinical syndromes in the NPSLE cohort was for anxiety (prevalence: 90%; 95% CI: 69.9%–97.2%; N = 20), followed by plexopathy (prevalence 83.3%; 95% CI: 43.7%–97.0%: N = 6), cranial neuropathy (prevalence: 64.0; 95% CI: 44.5%–80.0%; N = 25) and aseptic meningitis (prevalence: 55.0%; 95% CI: 34.2%–74.2%; N = 20). When solely assessing analyses of over 100 subjects, the highest prevalence of normal MRI brain features was for headache (prevalence: 48.9%; 95% CI: 41.8%–56.1%; N = 184), seizures (prevalence: 37.7%; 95% CI: 30.6%–45.5%; N = 159) and cognitive dysfunction (prevalence: 26.7%; 95% CI: 19.1%–35.8%; N = 105). Seven clinical syndromes demonstrated statistically significant association when assessed with RR. CVD, seizures, cognitive dysfunction, acute confusional state (ACS), demyelinating syndrome and movement disorders all demonstrated a greater risk of demonstrating abnormal MRI brain findings in people with NPSLE. Only anxiety was shown to have a lower risk of an abnormal MRI brain finding in NPSLE (RR: 9.00; 95% CI: 2.40–33.79; N = 20) (Figure 3).
Pooled prevalence of MRI findings by individual clinical feature for people with NPSLE.
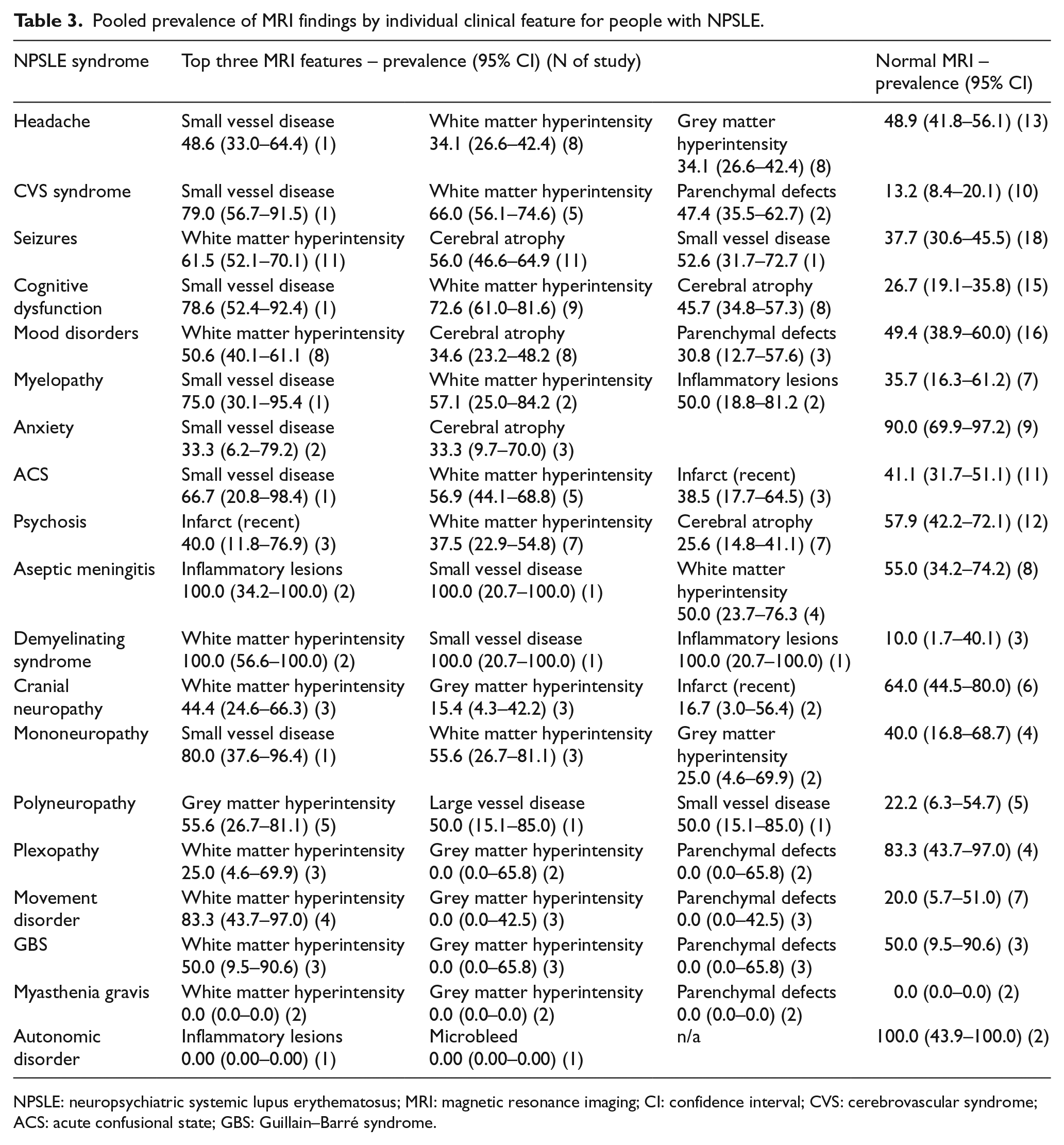
NPSLE: neuropsychiatric systemic lupus erythematosus; MRI: magnetic resonance imaging; CI: confidence interval; CVS: cerebrovascular syndrome; ACS: acute confusional state; GBS: Guillain–Barré syndrome.
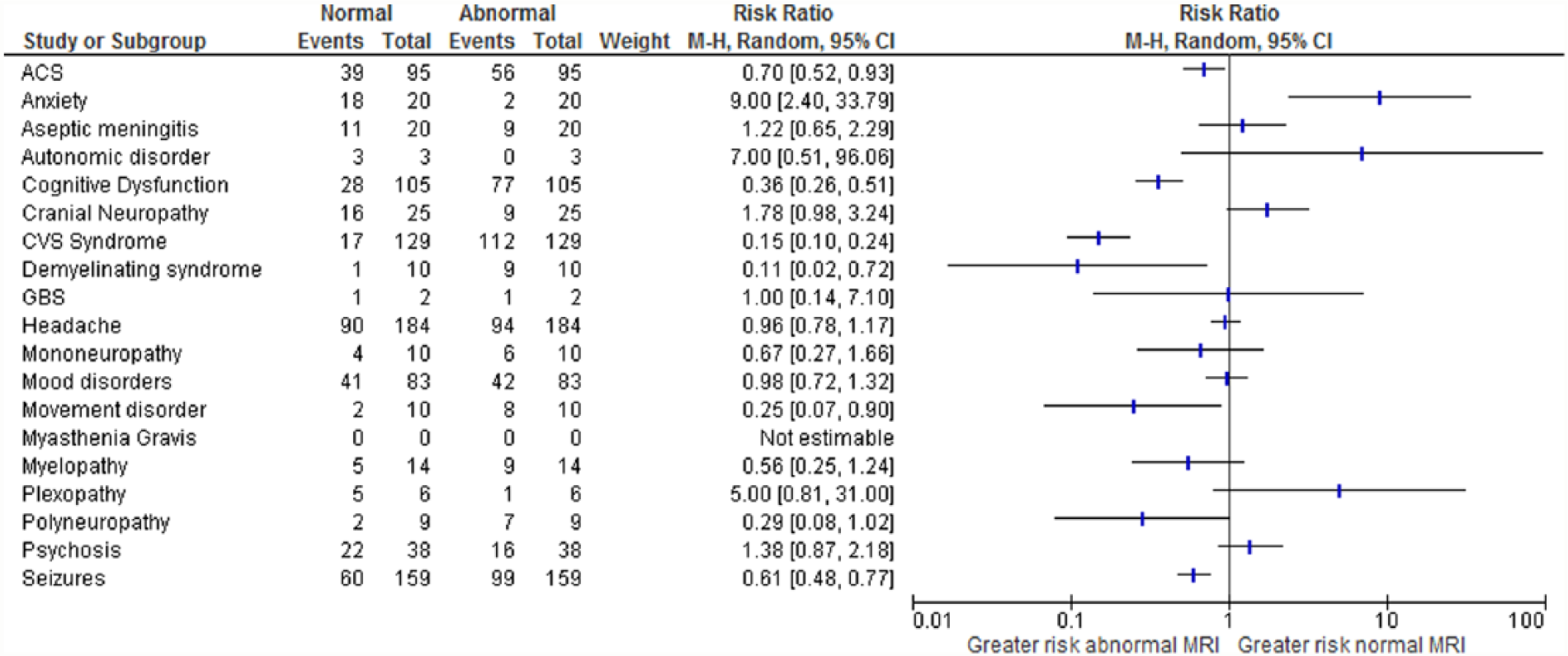
Forest plot of risk ratio of an abnormal compared to normal MRI findings for 19 clinical features of NPSLE.
Prevalence of MRI features for individual clinical syndromes in NPSLE
The prevalence data for each individual abnormal MRI brain feature and the clinical syndrome are presented in Table 3. The most prevalent MRI features were white matter hyperintensities (WMHs) (34.1%), grey matter hyperintensities (GMHs) (22.9%), parenchymal defects (6.5%), cerebral atrophy (16.9%) and small vessel disease (48.6%). On assessment of the 10 distinct brain MRI changes that we evaluated, CVD showed positive MRI findings in all categories, the most prevalent of which were white matter hyperintensity (66%), small vessel disease (79.0%), grey matter hyperintensity (44.8%) and parenchymal defects (47.4%). Headache demonstrated positive MRI features in 6 of the 10 categories assessed, with the most frequent findings being small vessel disease (48.6%) and white matter hyperintensity (34.1%). All the clinical syndromes were assessed for nine MRI abnormalities; in several, MRI findings were not observed, including myasthenia gravis features (100% MRI scans did not show abnormalities), plexopathy (88.9%), movement disorder (88.9%), GBS (88.9%), myelopathy (44.4% MRI changes not seen), anxiety (66.7%) and mood disorders (33.3%).
Discussion
Despite the presence of NPSLE syndromes with varying disease severity, a normal imaging outcome on conventional MRI was frequently observed in our study. The most common NPSLE syndromes in patients undergoing MRI evaluation in our selected studies were headache (23.8%), seizures (18.9%), CVD (17.6%), cognitive dysfunction (15.3%), ACS (13.8%) and mood disorders (11.1%). CNS manifestations were more frequent than PNS ones (91.3% vs 8.7%).
The most significant findings from our meta-analysis were the observation of normal brain MRI in a variety of clinical syndromes, including psychiatric syndromes such as anxiety (RR: 9.00) and PNS features such as autonomic disorder (RR: 7.00) and plexopathy (RR: 5.00). Associations between abnormal brain MRI and clinical NPSLE syndromes were observed in a broad range of clinical features including CVD (RR: 0.15), demyelination (RR: 0.11) and seizures (RR: 0.61).
Brain MRI in NPSLE is often conducted as part of the clinical workup, especially before performing a lumbar puncture. We found a low detection rate of distinct clinical NPSLE features correlating with specific brain MRI changes. Nevertheless, it is important to take into account that patients with objective syndromes such as seizures and CVD present with overt symptoms may undergo MRI scans sooner, while milder manifestations of subjective syndromes like cognitive dysfunction, mood or anxiety disorder may not be referred for MRI investigation until reaching a clinically severe stage. This reduces the reliability of the prevalence values with this possibility leading to an under- or over-estimation of some features. Interestingly, the EULAR task force recommended MRI investigation more urgently in selected syndromes. 15 Notably, our meta-analysis included studies which did not investigate all 19 NPSLE syndromes. For example, Toyota et al. 34 included only seizure patients in NPSLE. This may have therefore led to selection bias when estimating prevalence values.
The 21 included studies reported inconsistent details of MRI lesions in terms of their size, number and locations. The radiographic terms used to describe and list these into specific groups often differed, as no standard guidelines were adhered to. Hence, the resulting wide heterogeneity of lesion descriptions in the studies made cross-comparisons difficult. It was therefore decided to group them into themes of the most recurring terms used, which in our selected studies were WMH, GMH and atrophy. The ‘vascular lesion’ group consisted of descriptions, which could be confidently assigned into this particular category which included ‘microbleeds’, ‘infarcts’, ‘lacunar lesions’ and ‘intracranial haemorrhage’. Any terms recorded, where a degree of uncertainty existed over which category they would fit into best, were categorised as ‘other’. We found that WMHs were consistently the most frequently reported abnormality (33.1%–53.3%) among the eight most prevalent syndromes, followed by vascular lesions (6.7%–31.7%), although a considerable overlap between the two is likely. Atrophy was also a common feature (6.7%–35.7%), while GMHs were less frequent (2.8%–14.4%).
The precise role of distinct MRI lesions in the pathophysiology of NPSLE remains uncertain. Focal WMH lesions have been linked to various non-specific changes such as necrosis, reduced neuronal density, inflammatory infiltrates and demyelination. An underlying acute infarct has been suggested in areas where reduced diffusion was observed secondary to cytotoxic oedema, while bilateral WMHs are thought to be a sign of chronic hypoperfusion. 20 Common sites of WMH lesions are in the frontoparietal region, periventricular and subcortical white matter, and their presence has been recorded in both active and inactive NPSLE.29,47–49 Although evidence still persists that WMHs are not specific to NPSLE, as it was shown in SLE without overt neuropsychiatric involvement, they were found to be higher in quantity and total volume in active and past NPSLE when compared with non-NPSLE patient groups. 50 Specifically, a correlation between cognitive dysfunction, cerebrovascular syndrome and WMHs has been shown in previous studies.31,47
Atrophy is another non-specific radiographic finding in NPSLE patients, and many factors have been associated to contribute towards it, including disease duration, corticosteroid use, advancing age and presence of antiphospholipid syndrome (APS). 47 However, a recent study found that participants with NPSLE had significantly pronounced atrophy compared to SLE with no brain involvement and healthy controls, after adjusting for age. 51 Histopathologic correlations for atrophy vary, having been observed in patients with diffuse and focal brain injury.36,52,53 Atrophy has been found to be significantly associated with cognitive dysfunction, seizure disorder and CVD in another study. 47
MRI results in other diffuse syndromes such as ACS, psychosis and anxiety disorder were investigated by Arinuma et al. 46 and found to be normal in 52.8% of the cases. Another study pointed out that the rate of abnormal lesions detected on conventional MRI in diffuse syndromes was not significantly different from SLE patients with no neuropsychiatric involvement. 50 They concluded that while MRI remains sensitive for focal lesions, a combined approach of morphological and functional imaging, such as SPECT, would be more helpful in excluding brain abnormalities in diffuse syndromes with normal MRI. Our findings suggest that although small in sample size, patients with SLE with anxiety or peripheral nerve symptoms are significantly and mostly likely to have normal structural MRI scans. The findings from our study are preliminary, and more MRI research needs to be done on SLE patients with anxiety, autonomic disorder, plexopathy and polyneuropathy.
Our study presents with a small number of important limitations. First, only a small number of papers reported disease activity measure or the time of disease onset to MRI evaluation. Similarly, details on treatment were rarely presented which could have introduced a bias since patients may have been given effective treatment before undergoing MRI scanning. Another source of bias could have arisen from the patient selection in the individual studies. As not all NPSLE patients are referred for MRI investigation, it is likely that the patients in our analysis may be different in terms of disease severity from patients where MRI referral was not warranted. There were also limited data provided on ethnicity of participants. Other potential sources of bias to limit generalisability included the lack of blinding of the neuroradiologist reporting the MRI images. Only 26% of the studies explicitly stated that appropriate measures were taken to ensure minimal bias in this regard. Variations between MRI scanner resolutions across the studies may have led to inconsistencies in evaluating imaging findings. This is difficult to prevent in retrospective designs, which in our case consisted of studies published between 1989 and 2014. Additionally, the introduction of FLAIR imaging techniques, which were used only in studies from 2002 onwards in our study, could have added to a difference in the reporting of MRI findings. A potential pitfall in interpreting MRI outcomes includes the occurrence of overlapping syndromes for patients presenting with more than one NPSLE syndrome. In these cases, it is difficult, if not impossible, to reliably ascribe MRI outcomes to a particular syndrome with certainty. In our study, it was not possible to combine quantitative neuroimaging data due to the broad nature of the studies involved. Although the conventional way is voxel or co-ordinate-based meta-analyses of neuroimaging data which will identify neuroanatomical areas affected by NPSLE, it was not possible to do such analyses from the studies we identified. We therefore recognise that assigning an MRI as either ‘normal’ or ‘abnormal’ has its limitations but was necessary in our study due to the wide variability in the data extracted. Finally, our review did not exclude studies and analyses based on the number of cases of NPSLE evaluated in individual studies. This minimised the risk of small sample size publication bias from impacting on the review’s findings. However, the consequence of this was that for a number of features, such as autonomic disorders, demyelinating syndrome and GBS, there were very few events included in the analysis, thereby presenting under-powered results. As the evidence base develops, it is anticipated that further analyses will be undertaken to better understand NPSLE.
Conclusion
The results of this article suggest that – despite being the current imaging modality of choice in NPSLE – MRI should not be used in isolation to make a diagnosis due to its inability in some circumstances to differentiate changes from non-NPSLE presentations. In the future, the development of morphological and metabolic or functional imaging tools could be more useful in early monitoring of cerebral involvement, thereby allowing earlier effective treatment and improved survival for this population.
Footnotes
Acknowledgements
The authors would like to thank Judith Scammell for assistance with literature search.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
