Abstract
Objectives
The objective of this study was to investigate the time to recovery after an SRC comparing riboflavin 400mg daily to placebo in a group of elite level athletes from multiple sports.
Methods
The study was a double-blind randomized placebo-controlled trial with intention to treat conducted from 2016–2020 at two different academic institutions. The study enrolled varsity student-athletes (SA) at each institution. The investigators and participants were blinded to treatment allocation. The treatment group received 14 capsules of either riboflavin 400mg or placebo to take daily until completed. The team physician made the diagnosis of an SRC within 24 h of the injury.
Results
A total of sixty participants enrolled in the study. Fifty-two participants completed the study. Subjects in the riboflavin group had a statistically significant lower number of average days to recovery of 9.92 days (CI ± 2.8) compared to placebo of 22.2 days (CI ± 11.5) (P < 0.05). Subjects matched for age, gender, history of SRCs, attention deficit hyperactivity disorder and Periodic Health Questionnaire-9 scores.
Conclusions
This is one of the first studies that documents an effective treatment option for an SRC at the time of injury. Riboflavin is safe, inexpensive, and readily available making it an ideal treatment.
Introduction
Sport-related concussion (SRC) is a frequent injury in sports affecting 1.6–3.8 million athletes annually at all levels from recreational teams to professional leagues. 1 Most athletes are highly motivated to return to sport, however there are no therapies athletes can take at the time of injury to shorten the duration of an SRC. Current treatment guidelines emphasize cognitive and physical rest in the first days of an SRC.2–4 Subthreshold aerobic exercise is a valuable therapy for SRCs, but current research on this topic limits treatment to starting no sooner than 48 h after an SRC.5,6 A reliable treatment option for SRCs at time of injury that shortens recovery time could profoundly impact how health care providers manage concussions.
SRC is defined as a complex pathophysiological process affecting the brain, induced by biomechanical forces with alteration in mental status. 4 The mechanical forces such as a helmet to helmet hit in American football causes major cellular dysfunction and several pathophysiological pathways are activated.7,8 Shortly after injury there is a large neurotransmitter release of potassium, calcium and glutamate causing a cycle of continued activation that stimulates downstream cascades leading to cell damage.7,8 This leads to a metabolic mismatch with increasing demand for ATP the local environment cannot sustain. As the increased energy demands accelerate, the cells are forced to utilize glycolysis to create ATP which then produces lactic acid. This metabolic mismatch stimulates the neuroinflammatory cascade releasing cytokine mediators, proteases, and reactive oxygen species.7,9 The metabolic pathways of neurotransmitter release, energy imbalance and inflammation largely leads to the pathology of SRC and the resulting symptoms exhibited by patients. 9
Research into the treatment of SRCs has taken several different routes with dietary supplements being a major focus. Most published dietary supplement evidence to date is based on animal studies.10–17 Among the supplements, studies have investigated B vitamins due to their modifying effects on the metabolic pathways impacted in an SRC.10,17–22 Vitamin B2 or riboflavin is important in the mitochondrial respiratory chain to produce ATP and helps to maintain a major antioxidant enzyme, glutathione.14,20–22 Riboflavin's physiologic actions as an energy producer and antioxidant are naturally tailored to the metabolic mismatch found at the cellular level in SRCs. 23 Because of this, several animal studies have reported on the impact of riboflavin in concussed rat models showing an improvement in behavioral function, a reduction in brain edema and a decrease in glial fibrillary acidic protein around the brain contusion site.14,24
Previous research has shown that sports medicine providers are prescribing nutraceuticals including riboflavin to speed recovery based on these animal studies. 25 Unfortunately, there is limited clinical data to support their use. However, there is research that supports riboflavin's use in another clinically similar disease entity, migraines. Migraines and SRCs have associated symptomatology.18,25–27 Migraines have also shown a dysfunction in brain energy metabolism. 28 As such there may be similar pathophysiologic pathways between SRCs and migraines that riboflavin modulates. These previous migraine studies used a dose of riboflavin 400 mg daily and showed riboflavin to be well tolerated and safe. 29 While the safety of riboflavin is known, its efficacy as a treatment for SRCs is unclear. The objective of this study is to investigate the time to recovery after an SRC comparing riboflavin 400 mg daily to placebo in a group of elite level athletes from multiple sports.
Methods
The authors’ institutional review boards approved this study. The study was a double-blind randomized placebo-controlled trial with intention to treat conducted from 2016−2020 at two different academic institutions. One of the institutions stopped enrolling subjects after one year because the principal investigator who worked at that institution left for another job. The study was registered at ClinicalTrials.gov ID. An institutional new drug request was registered with the Food and Drug Administration.
The study enrolled varsity student-athletes (SA) at each institution. Table 1 lists the exclusion criteria. We recorded demographic data to include those participants with a diagnosis of attention deficit disorder. We also recorded the number of historical concussions each subject reported or had been diagnosed with. Participants completed a periodic health assessment (PHQ9) questionnaire within 24 h of their SRC to document depression symptoms.
Exclusion criteria.

The study randomly assigned subjects to either the treatment or the placebo group. A local pharmacy labeled both the riboflavin and the placebo capsules in sequential order based on a randomized list generated online (www.random-ize.com). The investigators and participants were blinded to treatment allocation. The treatment group received 14 capsules of riboflavin 400 mg with instructions to take one capsule daily until completed. The placebo group received 14 placebo capsules with instructions to take one daily until completed. The study purchased both the riboflavin and the placebo from Biotech Pharmacal (Fayetteville, AR, USA). Because riboflavin is highly colored, Biotech Pharmacal used opaque capsules to conceal the riboflavin from placebo. With the help of athletic department nutritionists, we limited use of other nutraceuticals including omega-3 polyunsaturated fatty acids, magnesium, multivitamins and other B vitamins.
We obtained consent prior to the injury, typically in the preseason. All athletes had a supervising athletic trainer and sports medicine fellowship trained primary care team physician. This medical team made the diagnosis of an SRC within 24 h of the injury. Once the subject was diagnosed, the team physician provided the subject with one sequentially numbered bottle with instructions to take 1 tablet once daily to completion. The SRC diagnosis was based on each institution's concussion management protocol. Both institutions had similar protocols which included using the Sport Concussion Assessment Tool (SCAT) as the primary tool to diagnose a concussion. 30 Each institution did baseline testing in the preseason recording results from the SCAT, the modified balance error scoring system (mBESS), sensory organization test (SOT) and a computer-based neurocognitive test - Immediate Post-Concussion Assessment and Cognitive Testing (ImPACT). The SOT was only available at one institution resulting in the institution without the SOT using the mBESS as their balance test According to protocol, once the subject became symptom free or returned to their baseline symptom score, they then took the ImPACT and each institution's respective balance test The study period ended when the subject's ImPACT and balance test returned to their baseline values.
The study's primary outcome investigated if riboflavin shortens the duration of an SRC in the number of days to recovery. We defined recovery as symptoms returning to baseline followed by ImPACT and balance test results returning to baseline.
The authors’ pilot study resulted in an average time to recovery from an SRC of approximately 10 days taking riboflavin 400 mg compared to historical controls of 13 days with a standard deviation of 6.5. As such, a sample size of 75 subjects in each group would be needed to attain a statistical power of 80% and an alpha error level of 5%. We tested all outcome variables for assumptions of normality. When assumptions were met, we performed a paired t-test For the independent variable of treatment, the Levene's test for equality of variance was significant so we corrected for the violation of the homogeneity of variance using the Welch-Satterwaite method. We used SPSS 26. (IBM SPSS Statistics for Windows, Version 26.0; Armonk, NY, USA: IBM Corp)
Results
A total of sixty participants enrolled in the study. Three of the participants were determined later to have had an SRC within the year previous to the study SRC and were excluded. We excluded five subjects because more than 24-h had elapsed since their SRC occurred. Fifty-two participants completed the study and met all inclusion and exclusion criteria with twenty-five in the riboflavin group and twenty-seven in the placebo group (Figure 1). No subjects dropped out due to harm or unintended effects. Subjects in the riboflavin group had a statistically significant lower number of average days to recovery of 9.92 days (CI ± 2.8) compared to placebo of 22.2 days (CI ± -11.5) (P < 0.041) (Figure 2).
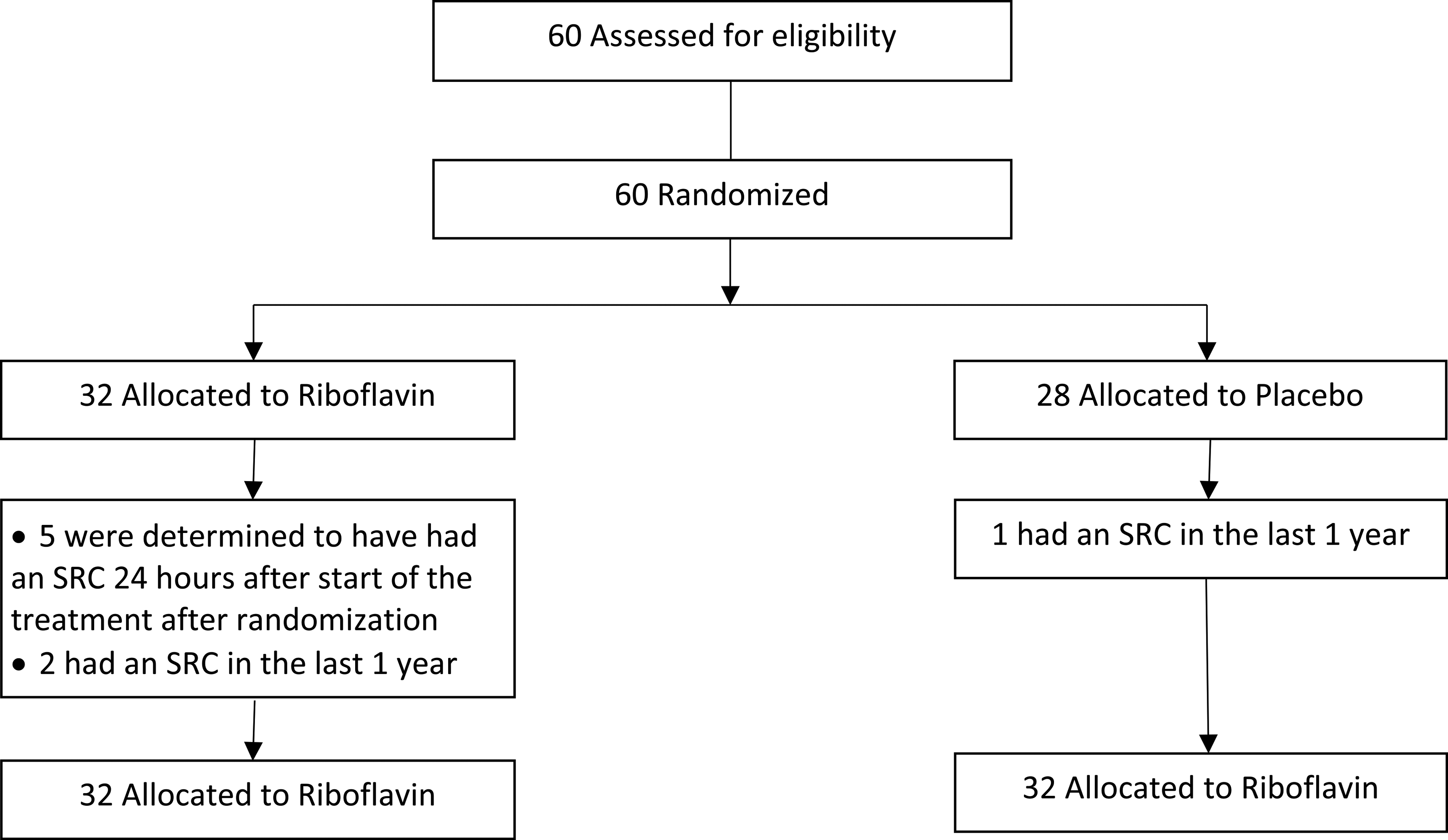
CONSORT flow diagram. SRC indicates sport-related concussion.
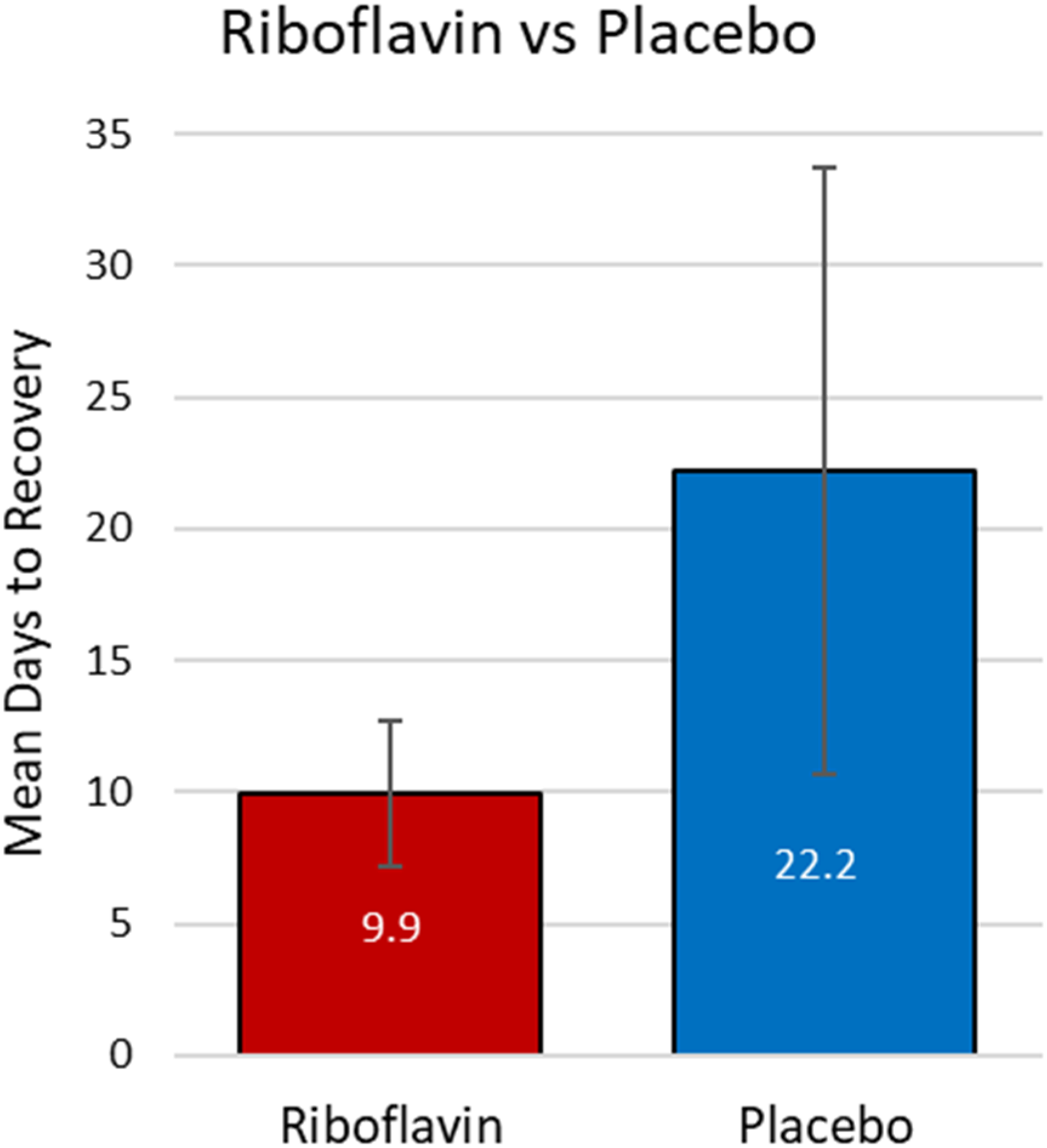
Mean days to recovery after SRC comparing riboflavin versus placebo. The riboflavin group took 9.9 days (SD ± -2.8) compared to the placebo group which took 22.2 days (CI ± 11.5) (P = 0.041).
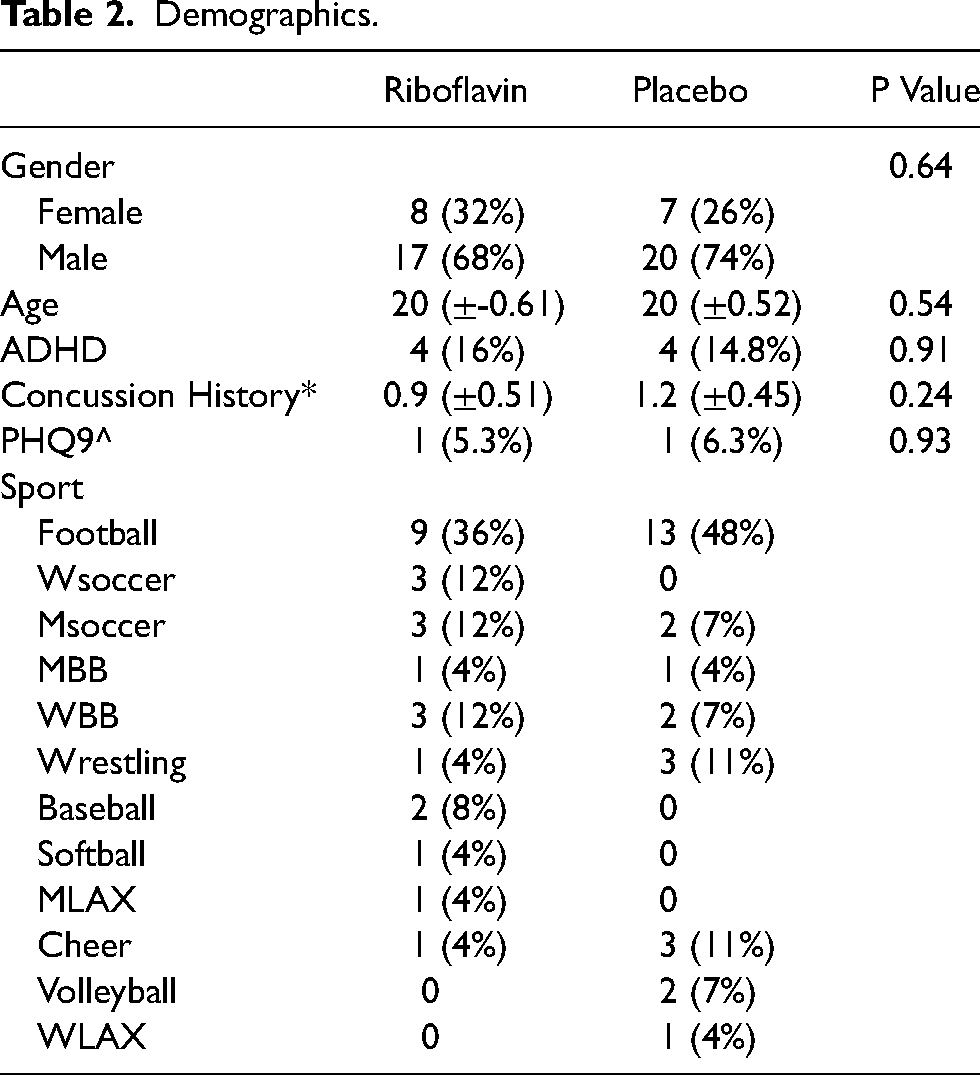
Demographic data is detailed in Table 1. Regarding the Periodic Health Questionnaire-9 (PHQ9), one riboflavin subject and one placebo subject reported a score greater than four which is equivalent to mild depression or greater. All other subjects reported a score of zero on the PHQ9. We were missing PHQ9 data from seven of the treatment subjects and eleven of the placebo subjects. Each group had nearly equivalent participants diagnosed with attention deficit disorder. We did not have the concussion history for one of the treatment subjects and one of the placebo subjects. Between group comparisons for gender, age, attention deficit hyperactivity disorder (ADHD), concussion history and PHQ9 were not significant (Table 2).
Demographics.
Discussion
There are limited treatment options for SRCs in the initial stages of management to accelerate recovery. For an athlete during the season, one day or even twelve hours could mean the difference between competing in a game versus sitting out. An ideal treatment for an SRC would be one that the athlete can take at the time of injury, is safe and inexpensive. This study reports on the use of riboflavin to shorten the duration of an SRC. In a group of elite level athletes, this study showed a significantly shorter duration of an SRC compared to placebo. This is one of the first studies to show an effective treatment option at the time of injury to speed recovery.
SRCs pathophysiology is based on a metabolic mismatch after injury. The local environment is unable to meet the demands due to an increased need for ATP and traumatic activation of downstream inflammatory mediators. As a generator of ATP and the caretaker of the antioxidant of enzyme, glutathione, riboflavin is perfectly positioned to modify this mismatch. Riboflavin is also known to cross the blood brain barrier enabling it to act directly within the chaotic brain ecosystem after an SRC. 31
Riboflavin is safe and at the study dose, readily available over-the-counter. This makes it an ideal treatment option for an SRC. Additionally, athletes at risk for SRCs may gain an added benefit by taking riboflavin prophylactically. Preinjury use may further mitigate the detrimental cellular dysfunction after an SRC if riboflavin is immediately available for the brain's defense. With that said, there is no evidence for this. Further studies could investigate the combination of nutritional supplements. Since the pathophysiological process has many downstream mediators, supplements such docosahexaenoic acid (DHA), lipoic acid or magnesium could be combined.10–17 Each one modifying a different branch point along the cascade to mitigate the detrimental effects of an SRC.
Questions remain on riboflavin's mechanism for a quicker recovery. Since riboflavin has also been shown to lessen migraines, its impact on a concussed athlete may modify other unknown headache pathways. Another consideration is that riboflavin's potential to improve isolated headache symptoms could improve SRC symptoms scores without modifying the underlying SRC. However, it would seem unlikely that riboflavin would be able to modify neurocognitive tests or balance tests as was necessary in this study without modifying other pathologic components of an SRC.
There were several limitations in this study. The study did not reach its projected enrollment based on our initial calculations. Two events led to this. Our enrollment slowed after one of the institutions withdrew from the study. Based on the enrollment trend it would have taken four to five more years to reach our projected numbers. The second event was the COVID-19 pandemic which caused the cancellation of all athletics at the study site with an unclear restart date. With a pause in research due to COVID-19, we took a close look at our data and found a positive significance. The combination of positive results, an unknown restart date and the potential for a prolonged enrollment prompted us to present the data as it currently is. Despite not meeting our enrollment, we did reach significance. Sample size and study power is more important when no significance is found. Another limitation is the placebo group generated a higher average days to recovery than our previous historical controls. Two aspects that may have played a part in this included sport variation and initial symptom severity. Not all sports were equally distributed in the study which could have led to bias. Regarding initial symptom severity, the original design of the study did not account for recording initial symptom severity and so we were missing total symptoms severity score data from a third of the subjects. The study was unable to control for riboflavin's yellow discoloration of the urine as the placebo pill did not cause a urine discoloration. This known interaction was stated in the written consent, but we did not verbally counsel them on this otherwise benign specific issue. Regardless, the yellow urine could have led to a bias. Since this was a small selective group of athletes there could be sampling bias and may not be generalizable to a more diverse group of people.
While the results of this study are positive, due to this study's limitations more research into the impact of riboflavin on SRCs is needed. A multicenter study with several arms investigating riboflavin and/or a combination of nutraceuticals would provide more answers.
Conclusion
This study reports on the impact of riboflavin in a group of elite level athletes diagnosed with an SRC. This randomized placebo-controlled trial showed the riboflavin group had a statistically significant shorter days to recovery compared to the placebo group. This is one of the first studies that documents an effective treatment option for an SRC at the time of injury. Riboflavin is safe, inexpensive, and readily available making it an ideal treatment.
Footnotes
Acknowledgements:
The authors would like to thank Bio Tech Pharmacal and Kurt Doege for his work in acquiring the riboflavin and placebo pills. We also thank MediCap pharmacy in Harrisonburg, Virginia for their work in bottling and labeling the pills. And finally, we would like to thank Randy Bird, MS, RD for his nutritional expertise.
Competing interests:
There are no conflicts of interest
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval statements that refer to your institution:
The University of Virginia Institutional Review Board Approved this stuy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American Medical Society for Sports Medicine, (grant number Young Investigator's Grant)
