Abstract
Alterations in the neurovasculature after traumatic brain injury (TBI) represents a significant sequelae. However, despite theoretical and empirical evidence supporting the near-ubiquity of vascular injury, its pathophysiology remains elusive. Although this has been shown for all grades of TBI, the vascular changes after injuries with the broad mild traumatic brain injuries (mTBI) classification, remain particularly difficult to describe. Our group has previously demonstrated hemodynamic alterations in mTBI by utilizing transcranial Doppler ultrasound and cerebrovascular reactivity in a cross-sectional study. That work identified a phasic progression of deviations over varying days post-injury. These phases were then characterized by a set of inverse models that provided a hypothetical process of hemodynamic dysfunction after mTBI. This model set provides a framework with the potential for guiding clinical treatment over the course of recovery. However, it is still unclear if individual patients will progress through the phases of dysfunction similar to that found at the population level. The work presented here explores six individual patients with high-density data collected during their post-injury recovery. Breath-hold index (BHI) was found to be the most robust feature related to mTBI longitudinally. All six subjects exhibited BHI recovery curves that followed the population model's progression. The changes in pulsatile features lacked the universality of BHI, but were present in subjects with higher self-reported symptom scores and longer periods of recovery. This work suggests neurovascular dysfunction after an mTBI may be a robust phenomenon. Additionally, the capabilities of TCD in capturing these changes highlights its potential for aiding clinicians in monitoring patient's recovery post mTBI.
Introduction
Monitoring the recovery of patients suffering from a mild Traumatic Brain Injury (mTBI) presents a challenge for clinicians. The broad heterogeneity of patients and their injuries, coupled with a reliance on cognitive or subjective measures of dysfunction, complicate diagnosis and treatment. This is further confounded by the delicate balance between rest and activity, both cognitive and physical, recommended by most consensus guidelines. 1 It has been suggested that 80%−90% of recurrent concussions occur within the first 7−10 days post-injury. 7 During this period of vulnerability, too much activity too soon after a concussion can affect outcome. 8 Conversely, waiting too long to resume light activity can also have a negative effect.2,9,10 In addition, complete cocoon therapy has been found to have either no benefit or to have negative consequences. 9 Similarly, there can be deleterious effects due to cognitive and executive over-exertion. 11 Ultimately, a staged recovery plan has been shown to offer the greatest benefit.1,8,14 However, clinicians are still reliant on patient compliance and subjective tests to assist in its implementation. An objective measure of concussion pathophysiology can help physicians as they monitor and guide patients through recovery. Alterations of the neurovasculature after mTBI present a unique target for the development of such biomarkers.
Measuring hemodynamic dysfunction provides direct insight into a patient's neurophysiological state that cognitive and other subjective measurements can not. In moderate and severe traumatic brain injuries (TBI), vascular alterations have been identified as a significant sequelae. 15 Additionally, this dysfunction has been shown to have a phasic progression.16,17 Similarly, distinct phases of hemodynamic alteration were observed with transcranial Doppler (TCD) ultrasonography in a population of subjects after a mTBI in Thibeault et al. (2018) 18
The changes described at the population level illustrated two distinct phases of dysfunction followed by a return to basal levels. The current hypothesis is that the initial phase is initiated by the mechanical insult resulting from the injury. The transition into phase II is likely a result of autonomic imbalance and the well-established neurometabolic cascade. 19 This response is a direct illustration of the theory that not all neuronal damage occurs at the time of injury. 15
These population results were subsequently extended, in Thibeault et al. (2018), 20 into a population model that generalized the time course of hemodynamic changes. In that, an inverse model was fit to the data to define the changes in the dominant TCD features. In this study, the utility of that model is explored when applied to individual mTBI subjects with longitudinally collected post-injury exams. This can elucidate more about how that generalized theory of hemodynamic changes will describe individual subjects. In addition, tracking the hemodynamic changes of a subject, and their deviations from the population, may offer clinicians complementary information to existing tests -- aiding in return to activity assessments.
Materials and methods
Participants
For the overall population study, the participants consisted of adolescents between the ages of 14 and 19 years-old who were either asymptomatic controls (109 subjects), with no recent head injuries reported, or cases (70 subjects), clinically diagnosed with a concussion by independent physicians. When possible multiple scans over the course of recovery were collected for the case subjects, resulting in 187 total scans. For the analysis here, subjects with a scan conducted within the first 48-h post-injury and at least 5 total scans over the course of their recovery were selected. Of the 70 total subjects, 13 had 5 or more scans. However, only 6 of those had scans within the first 48 h post-injury. Those 6 are presented here. The study was approved by Western Institutional Review Board (IRB #20141111).
Data collection
The cerebral blood flow velocity (CBFV) was collected by trained ultrasound technicians with the subjects seated upright. A monitoring headset with 2 MHz probes was utilized to insonate the middle cerebral arteries (depth was restricted to 45−60 mm) through the transtemporal windows bilaterally. The end-tidal CO2 was collected through a nasal cannula with a Nonin Respsense capnometer. These data streams and the patient symptom information were collected with a custom software package developed in Python. 18
Protocol
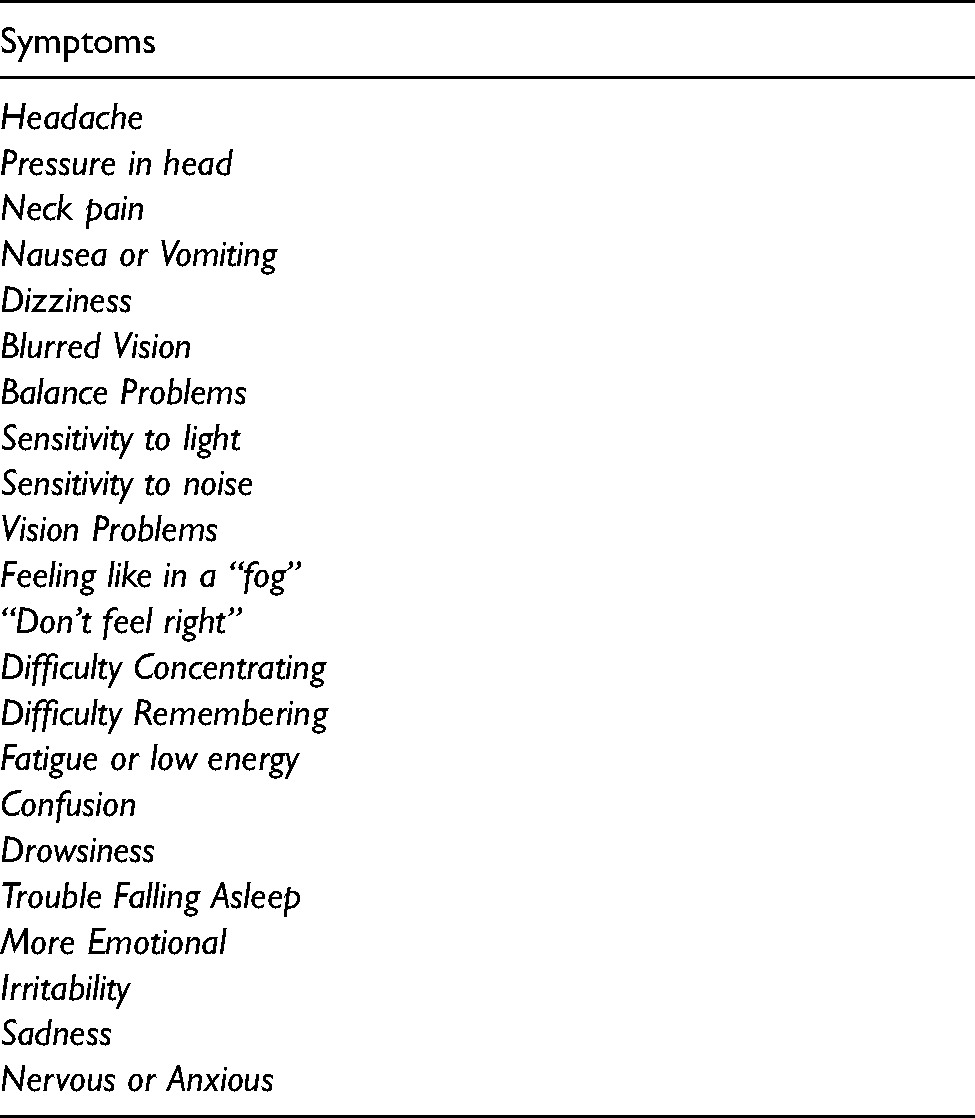
At the start of each scan subjects were asked to rate their current symptomatic state based on a series of questions similar to those in the graded symptom scale, 21 see Table 1. These are presented individually as well as summed together as an estimate of symptom severity. Some subjects were also evaluated with the Standardized Concussion Assessment Tool version 2 (SCAT2) or version 3 (SCAT3). 22 Alternatively, the Standardized Assessment of Concussion (SAC)22,23 was included for some subjects. This was dependent on the clinician responsible for the subject's care. The experimental procedure, illustrated in Figure 1(a), consisted of a 5-min resting baseline and four breath-holding challenges. The challenges were composed of a 25-s period of breath-holding, after a normal inspiration, followed by a 35-s rest period of normal breathing.
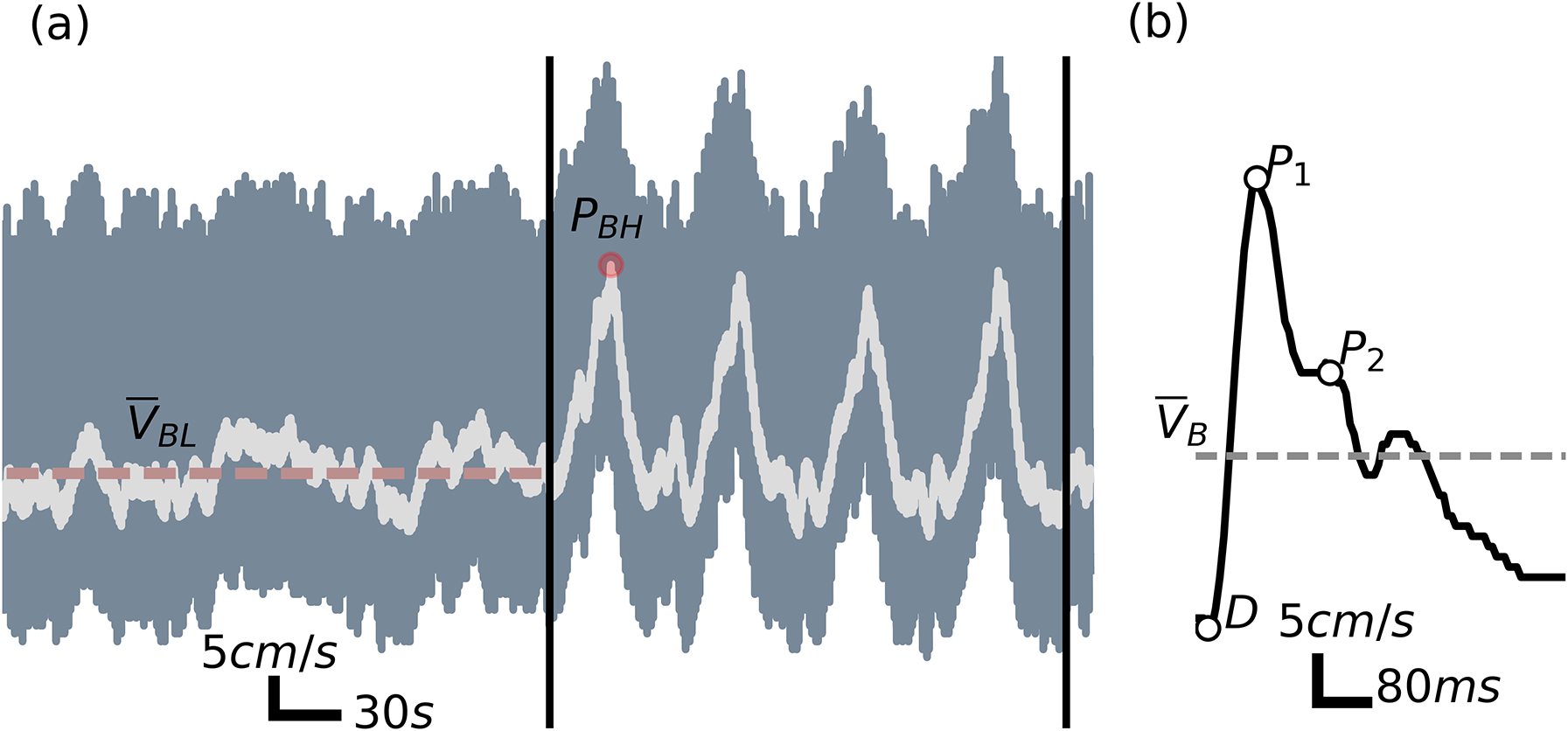
Experimental protocol. (a) The representative waveform illustrates the breath-holding protocol. An initial five-minute baseline is followed by the four breath-holding challenges. The CBFV waveform is filtered to extract the low-frequency component. The BHI is then computed using the baseline mean velocity (VBL, dashed line) and the largest peak velocity (PBH). (b) The individual pulses are extracted at the diastolic valleys (D). Then the systolic peak (P1), second peak (P2), and the mean velocity (VB), are identified.
Subjects rated themselves on the following symptoms based on how they felt at the time of the exam – none (0), mild (1,2), moderate (3,4), severe (5,6).
Model fitting
The population models were previously developed and described in Thibeault et al. (2018). 18 These results are summarized in Figure 2(a to c). For this work those population level models were fit to the selected individual longitudinal subjects to qualitatively explore how their recovery compared to the population. This was accomplished with the Levenberg-Marquardt nonlinear least squares minimization method implemented in the lmfit library. 24
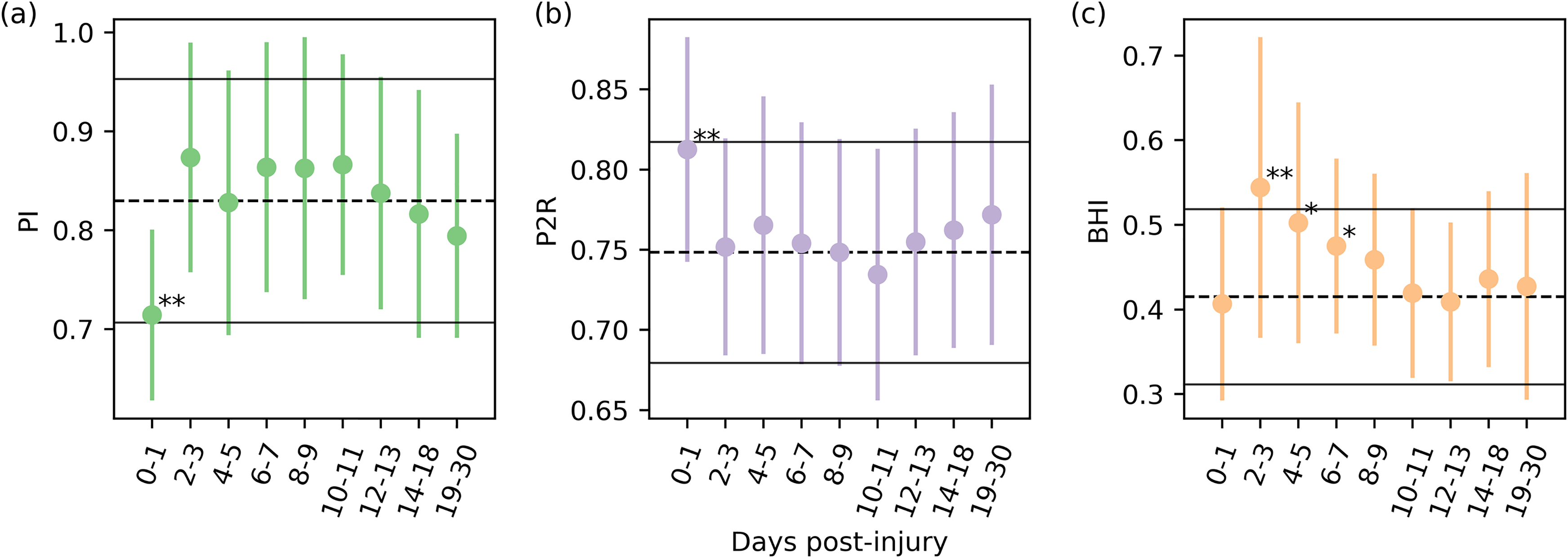
Population results grouped by days post-injury. (a) PI. (b) P2R (c) BHI. *P < 0.05, **P < 0.01 as compared to the control population. Error bars delineate the standard deviation.
Analysis
Population results
For each subject the hemisphere with the highest baseline mean velocity was selected for analysis. Cerebrovascular reactivity was then estimated using the Breath Hold Index.25,26 This was computed by low-pass filtering the TCD waveform and selecting the breath-hold period with the highest peak, Figure 1(a). From the baseline section the mean velocity, 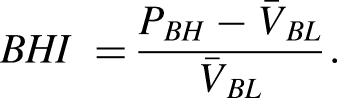
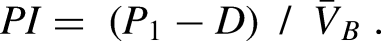
The second pulsatile feature, P2R, is the ratio of the first two peaks (P1 and P2) of a beat -- see Figure 1(b). It has been suggested that this may be related to distal bed compliance.
28
It is computed by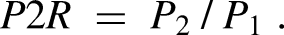
Population models
Using these features, and the population results from Thibeault et al. (2018), 18 recovery models were developed that describe the phases of hemodynamic recovery after a concussion in Thibeault et al. (2019). 20
For BHI this took the form of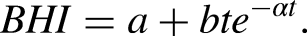
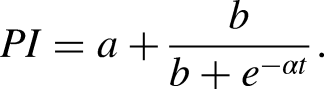
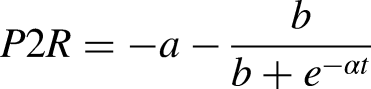
For these,
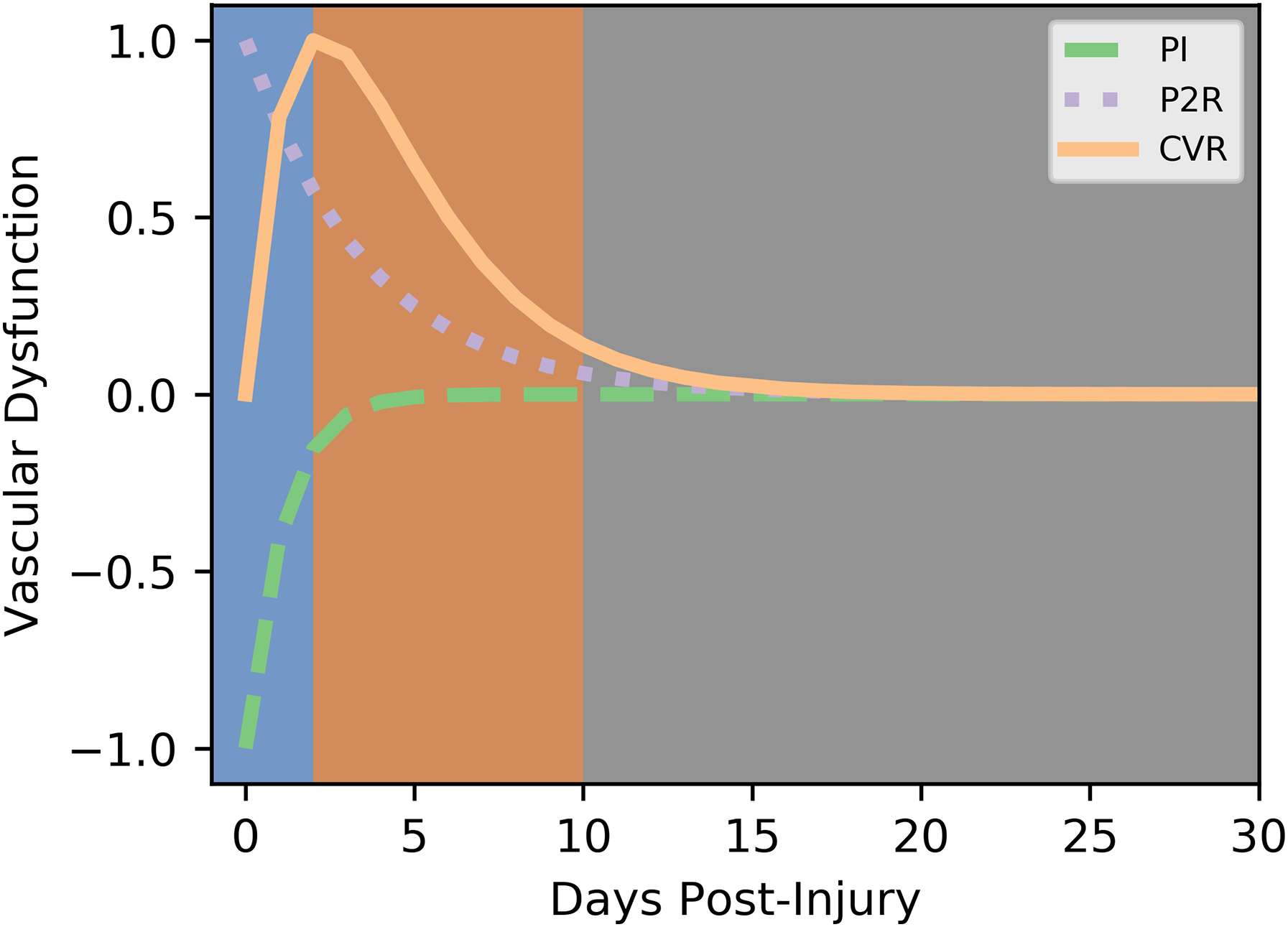
Normalized population models. The models developed in Thibeault et al. (2019)20 defined three periods of hemodynamic recovery. The curves are recreated here, normalized, with 1.0 representing the maximum level of dysfunction for BHI and P2R and −1.0 for PI. The generalized temporal separation of the three hemodynamic phases of recovery are illustrated by the different blocks along the x-axis.
Results
The following results are presented for each of the individual subjects. These are not displayed in any particular order and the numerical designations are arbitrary. The clinical histories for these subjects were collected by different clinicians involved in the study. As such, the clinical reports are not standardized and are reported here to give context to the injury and the subject's recovery. The extracted features for each of the subject plots are overlaid on the three phases outlined in Figure 3. The individual symptoms greater than zero are illustrated by the intensity based color-coding, with the colors darkening as the value increases, as well as the numerical response.
Subject 1
Subject 1, a seventeen-year-old male American football player who was injured after colliding with an opposing player's knee during a game. At the time of injury, the subject was diagnosed with a concussion by the team physician.
The first study session occurred 60-min after injury. As shown in Figure 4 (a-c), the subject displayed depressed PI but unremarkable P2R or BHI. There were also a high number of self-reported symptoms, Figure 4 (d-e). The subject subsequently reported to a specialist four days post-injury. At that time, the subject reported mainly dizziness and headaches to the physician. School was restricted as needed, however no assignments or tests were allowed by the physician. In addition, no sports-related activity was permitted. On day six post-injury the subject participated in a second study session. At this point PI had increased and P2R was largely unchanged. However, a spike in BHI index was observed. Finally, the self-reported symptoms were present but reduced when compared to the values observed at the time of injury. At day 11 post-injury, PI, and P2R appeared to have stabilized and BHI had dropped down to a level close to that observed initially. However, the subject had a number of self-reported symptoms. This was similarly observed on day 13. However, at day 12 the subject reported no symptoms to the physician and was put on a 48-h graduated return to activity plan. The subject participated in two additional follow-up sessions with generally stable metrics, as illustrated in Figure 4, and participation was concluded at day 27. However, it was later revealed in the clinical notes that the subject had a relapse at 30 days post-injury and was diagnosed with post-concussive syndrome (PCS).
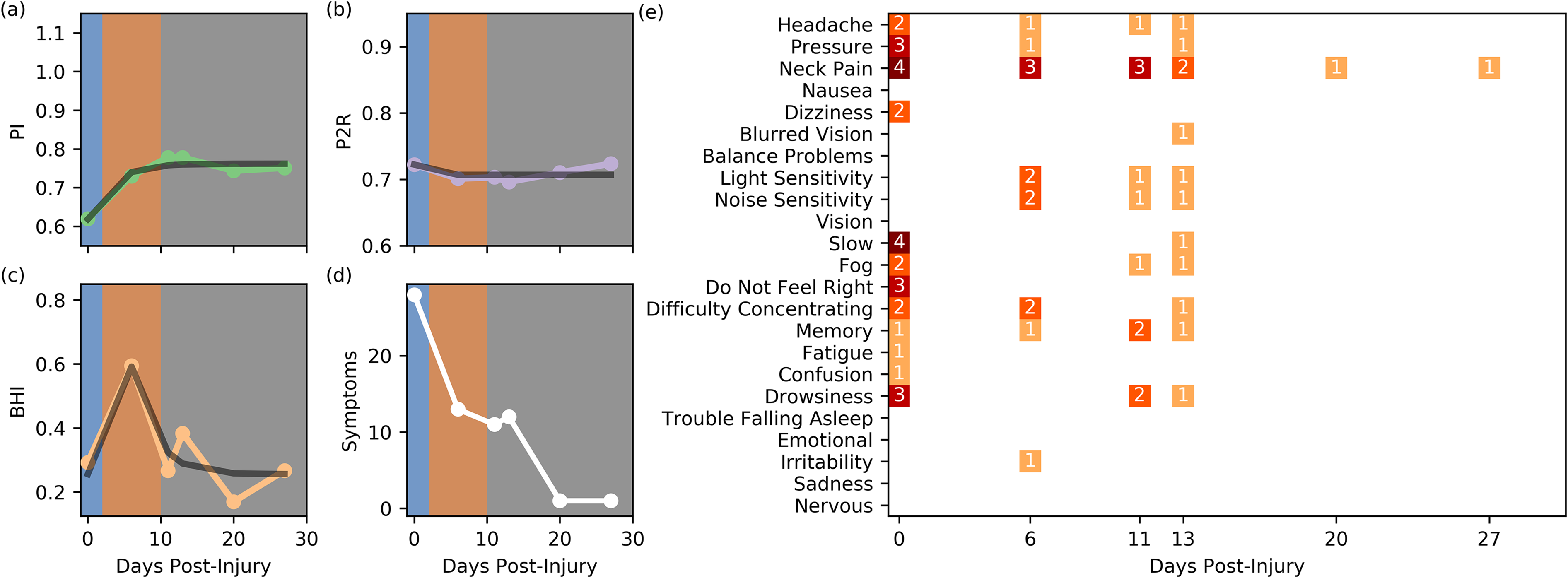
Subject 1, sixteen-year-old male American football player. (a) PI. (b) P2R. (C) BHI. (d) Summed Symptoms. (e) Individual Symptoms. The black lines indicate the model fits for this subject.
The model fits overlaid in Figure 4(a to c), illustrate that this subject did fit the proposed model of hemodynamic recovery for PI and BHI. Although P2R did not show a similar level of dysfunction as that found in the overall population, it is not particularly surprising given that PI and P2R were largely uncorrelated during this time period as reported in Thibeault et al. (2018). 18 The missing data between days 0 and 6 presents a difficult problem. In the population model, BHI peaks around day 2 and gradually returns to a steady-state value. In this case, that period of time is missing, however the hyperreactivity is still present on day six. This suggests one of two things. The first is that the time course of vasoreactivity recovery was elongated in this subject -- the return to steady-state was delayed. The other is that the peak in hyperreactivity was delayed -- this would suggest a longer period of time in phase I. Regardless, the subject’s hemodynamic dysfunction and recovery exhibits the phased progression predicted by the model.
The clinical history revealed two interesting aspects of this subject’s recovery after injury. The first is the discrepancy in symptoms reported to the physician as compared to those reported a day later to the study personnel. The lack of reported symptoms allowed the clinician to begin the subject’s return to activity period. This highlights the difficulty clinicians face when relying on subjective information alone when treating concussive injuries. The second is the subject's ultimate decline into PCS. Unfortunately, the subject’s participation had concluded but subsequent studies are being developed to allow for extended periods of subject participation to determine if there were physiological predictors of PCS in the early phases of recovery.
Subject 2
Subject 2, A fifteen-year-old male American football player who was injured during a helmet to helmet hit during a game. The subject was placed on the concussion protocol despite reporting no symptoms after 2 h.
The baseline SAC score was 29/30 at the time of injury. The initial study session was completed at this time with unremarkable results, Figure 5(a and b). This was the only time the subject provided non-zero values for the self-reported symptoms, Figure 5(d and e). Over days one through three post-injury, the subject completed individual study sessions. During this period, no symptoms were reported and the results for PI and P2R did not present any alterations. However, during this time BHI revealed the hallmarked rise in reactivity characterized by the population model, Figure 5(c). At the end of this period, day three post-injury, the subject reported no symptoms to the clinician, SAC 30/30, and was cleared resume school work but with limited screen time. They were not cleared for sport. On day five post-injury the subject had a SAC of 25/30 and was cleared for activity. The study session revealed that BHI had begun to decrease but was still elevated as compared to the initial measurement, Figure 5(c). Two more study sessions were completed before the subject withdrew where the BHI can be observed decreasing as predicted by the model, Figure 5(c).
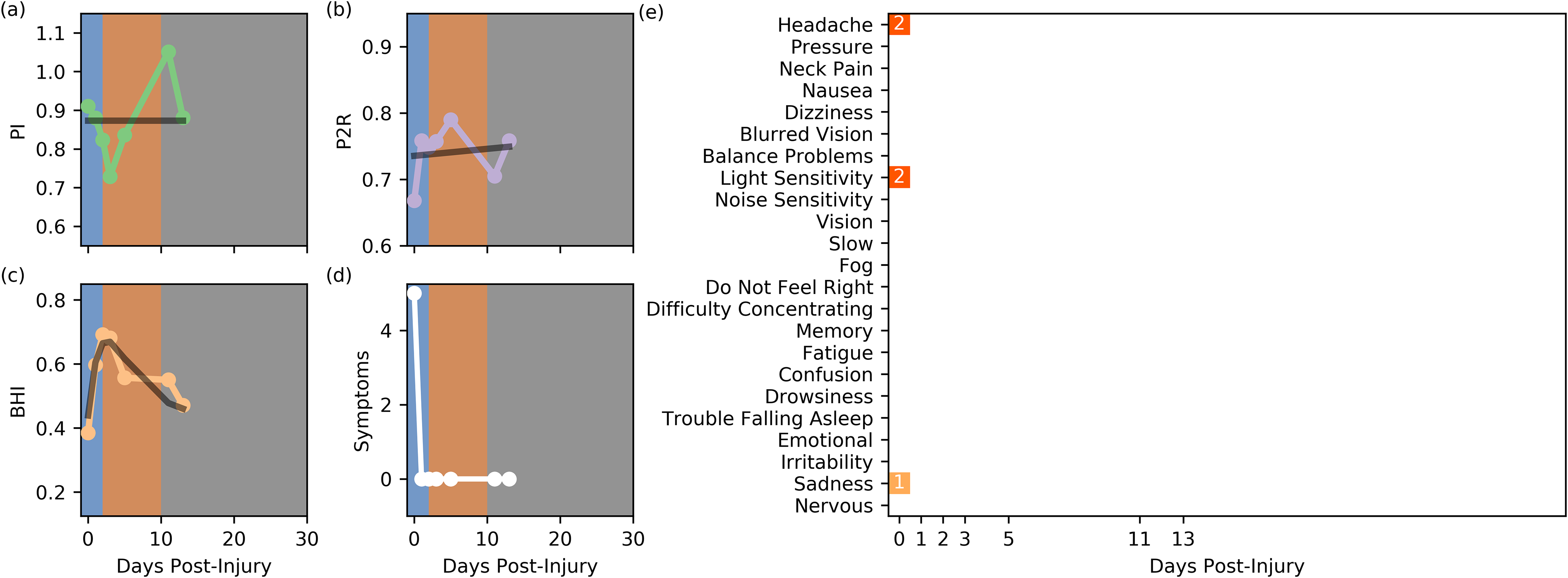
Subject 2, fifteen-year-old male American football player. (a) PI. (b) P2R. (C) BHI. (d) Summed Symptoms. (e) Individual Symptoms. The black lines indicate the model fits for this subject.
In this instance the subject did not exhibit the characteristic responses of PI and P2R, Figure 5(a and b). BHI however, followed the predicted trend closely, Figure 5(c). This is a case where all clinical measures of dysfunction indicated a complete recovery, Figure 5 (d and (e)), but the physiological perturbation measured with BHI seems to suggest that an underlying hemodynamic dysfunction remained. This period of hyperreactivity may reveal an unobserved state of vulnerability.
Subject 3
Subject 3, a sixteen-year-old female soccer player was injured after a collision with another player when attempting to head the ball during a game.
The subject reported to the physician one day post-injury. They had no history of concussions, headaches, or mood disorders. Score of 25/30 on the SAC and 52 on the sport concussion assessment tool 2 (SCAT2) symptom severity score were reported. The patient reported significant symptoms, Figure 6(d and e), in addition to lowered PI and raised P2R as illustrated in Figure 6(a and b). The study session on day two post-injury revealed the expected spike in BHI, as well as increased PI, and decreased P2R Figure 6(a to c). The self-reported symptoms were slightly reduced from day one post-injury but remained high. Over days four to six post-injury, two additional scans were completed. These revealed physiological measurements that visually matched those predicted by the model. At the study session at day seven post-injury a spike in PI was observed, delineated by the star in Fig (a). This was accompanied by a spike in symptoms as well. Interestingly this session was collected at the start of the subjects menstruation cycle. The subject's follow-up exam occurred on day eight post-injury. A SCAT2 symptom severity score of 62 was reported and a return to play protocol was initiated as well as academic concessions. At this stage the subject's TCD scans revealed a hemodynamic progression as predicted by the model. However, on days 16 and 28 there is a noticeable increase in PI as well as decrease in P2R, Figure 6(a and b). In addition, the subject had significantly increased self-reported symptoms Figure 6(d and e). Visits to the clinician on days 22 and 29 post-injury revealed that the subject was still too symptomatic to return to activity. It wasn't until day 57 post-injury before the clinician reported that symptoms had subsided.
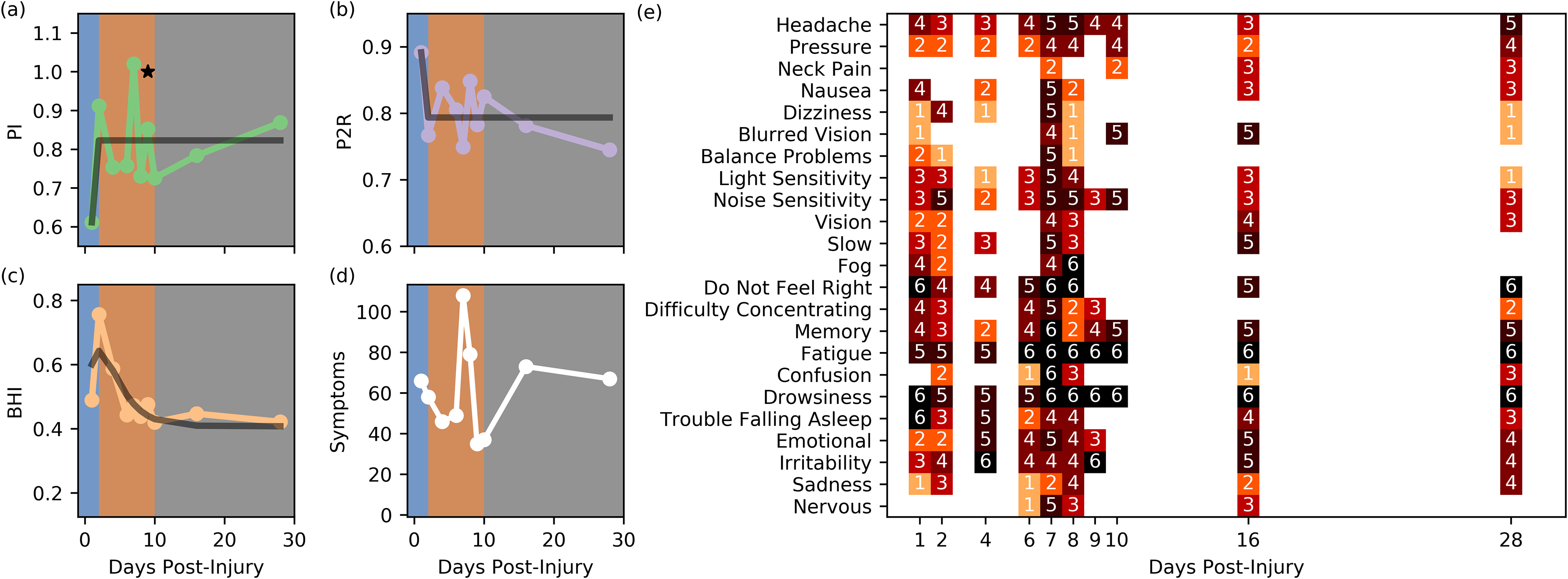
Subject 3 sixteen-year-old female soccer player. (a) PI. (b) P2R. (C) BHI. (d) Summed Symptoms. (e) Individual Symptoms. The black lines indicate the model fits for this subject.
Other than the significant spike in PI, Figure 6(a), the TCD measured physiological measurements all followed those of the model of hemodynamic changes in the early stages, Figure 6(b and c). It is speculative to suggest that the changes in PI and P2R towards the end of the study were related to the persistent symptoms and correspondingly extended period of recovery. However, this is something that will be explored in future studies.
Subject 4
Subject 4, an eighteen-year-old female was involved in a car accident where both front and side airbags were deployed.
On day 1 post-injury the subject reported minor symptoms to the physician. The subject had a SCAT3 symptom severity score of 54. The study session revealed unremarkable BHI and PI measurements but an elevated P2R, Figure 7(a to c). The subject was cleared by the physician to return to activity on day 10 post-injury after no symptoms were reported and a SCAT3 symptom severity score of 0.
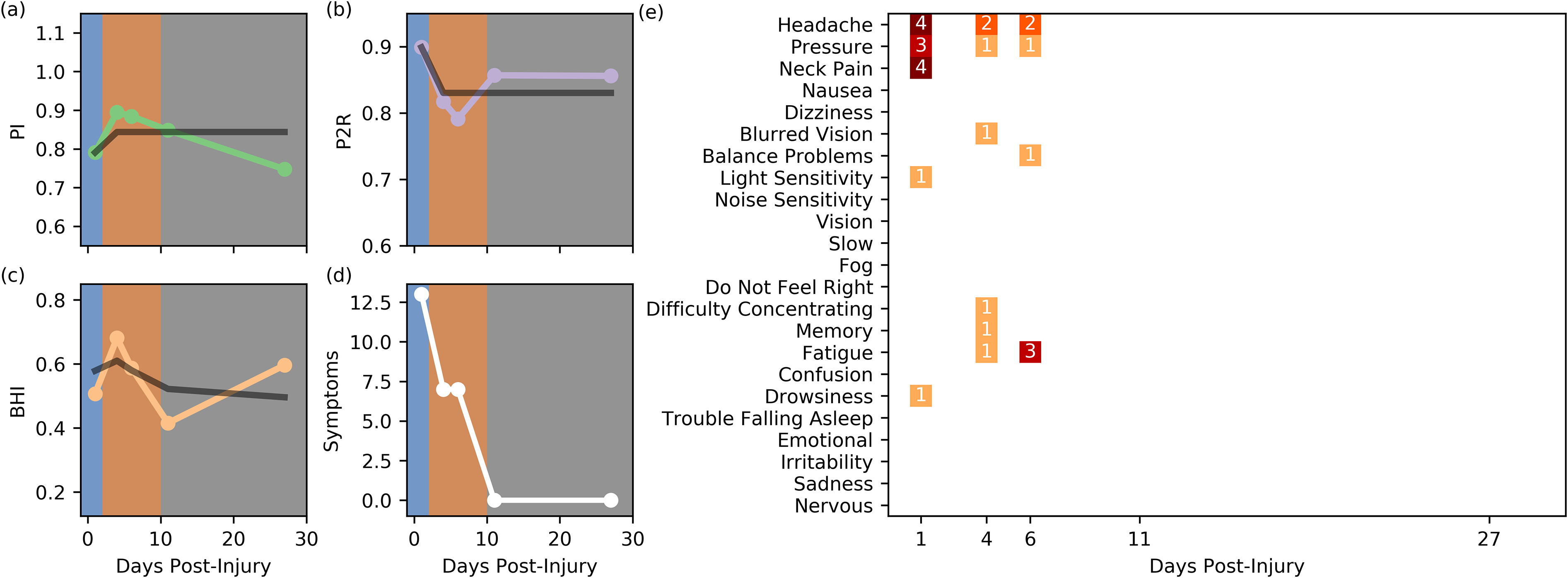
Subject 4, eighteen-year-old female. (a) PI. (b) P2R. (C) BHI. (d) Summed Symptoms. (e) Individual Symptoms. The black lines indicate the model fits for this subject.
Over the course of the study the subject exhibited the expected changes in BHI and P2R but not necessarily PI, Figure 7(a to c). Similar to Subject 1, this subject did not have scans between days 1 and 4 post-injury, therefore, the peak BHI value may be due to the reasons noted before. Although the subject reported some symptoms during the first three visits, Figure 7(d and e), they resolved after day 10 post-injury.
Subject 5
Subject 5, a seventeen-year-old female water polo player who was hit on the right side of the head during a match.
The subject visited the clinician on day 2 post-injury and reported sensitivity to light and noise as well as a headache induced during driving. A SCAT3 symptom severity score of 42 was measured. During the study session, PI and P2R were unremarkable but there was an increased BHI, Figure 8(a to c). Over days 3 and 4 post-injury, the BHI dropped significantly, as expected by the model, Figure 8(c). The subject reported a relapse of symptoms on day 6 post-injury that was resolved on day 11 post-injury, Figure 8d and e). At 18 days post-injury the subject reported no symptoms, Figure 8(d and e), SCAT3 symptom severity score of 0, and was cleared for activity by the physician.
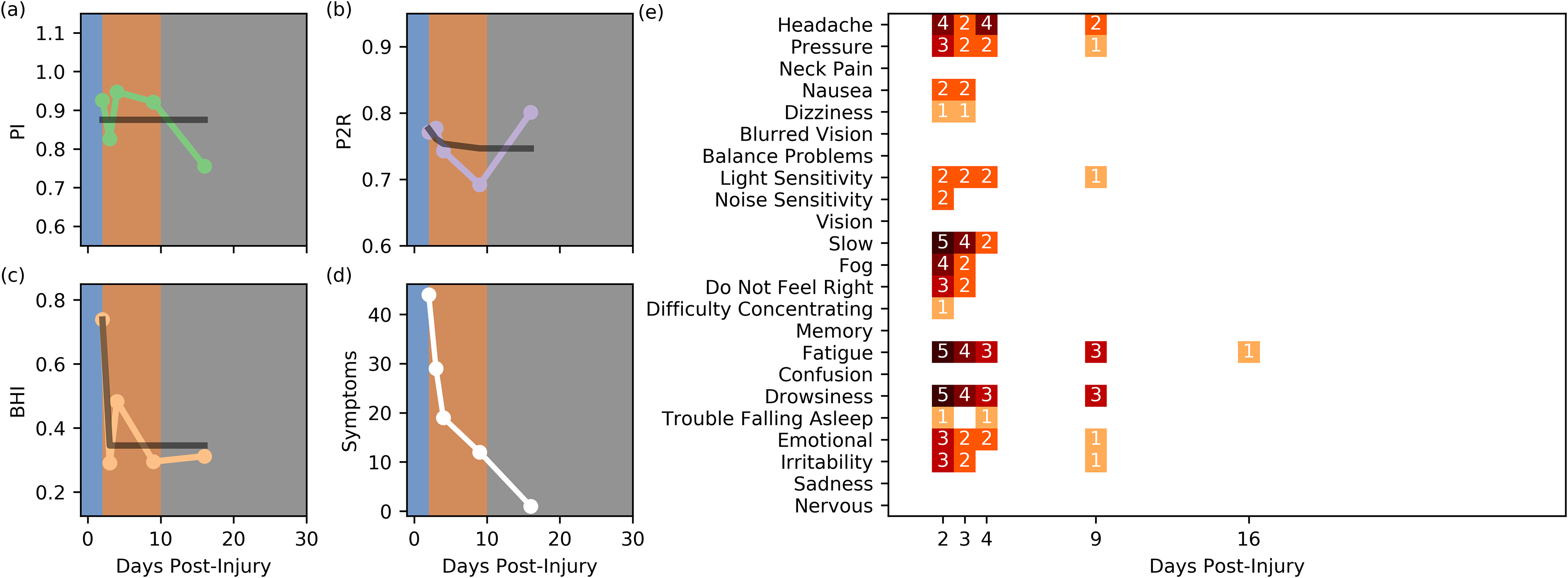
Subject 5, seventeen-year-old female water polo player. (a) PI. (b) P2R. (C) BHI. (d) Summed Symptoms. (e) Individual Symptoms. The black lines indicate the model fits for this subject.
The absence of early scans means the subject's phase I alterations were unobserved. However, the peak at day 2 in the BHI measurement matches that predicted by the model.
Subject 6
Subject 6, a seventeen-year-old male fell while snowboarding. A loss of consciousness was reported by the subject.
At day 1 post-injury the subject has a SCAT2 symptom severity score of 13. This increased to a 22 on day 2. Over this period, days 1 and 2 post-injury, both PI and P2R illustrated dysfunction similar to that predicted by the model, Figure 9(a and b). However, this was delayed, extending into day 3 post-injury. During this period a high number of symptoms were reported, Figure 6(d and e). Over days 4 and 5 post-injury, PI and P2R both appeared to reach a steady-state, Figure 6(a and b). Similarly, the BHI measurements began to decrease, Figure 9(c), as would be expected based on the model. There was a significant spike in BHI on day 8 that lasted into day 9, Figure 9(c). Interestingly, this was preceded by the subject reporting significant symptoms on day 7 post-injury -- severe enough that the subject could not look at the screen long enough to answer the symptom questions. This was extended into day 8 post-injury where the subject had a parent fill out the forms. At day 9 post-injury however, the subject only measured a 9 on the SCAT2 symptom severity score. Sixteen days post-injury the subject was still reporting symptoms to the physician. School in particular was an issue. Finally, on day 49 post-injury the subject was asymptomatic and returned to activity.
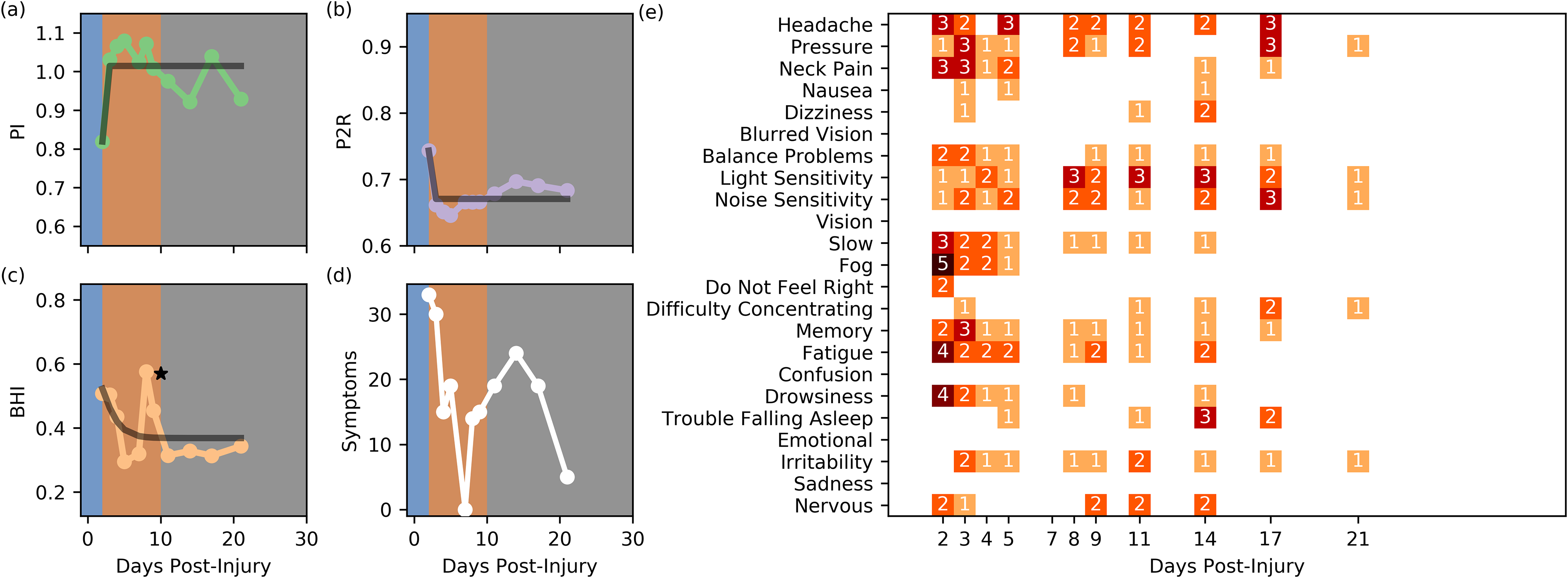
Subject 6 seventeen-year-old male snowboarder. (a) PI. (b) P2R. (C) BHI. (d) Summed Symptoms. (e) Individual Symptoms. The black lines indicate the model fits for this subject.
With the exception of the increase in BHI during recovery, Subject 6 presented physiological symptoms that very closely followed the models -- albeit with a phase delay. The difficulty in fully participating in the study mid recovery is interesting in light of the subject's extended recovery time. Although it is difficult to claim these are related, it provides an interesting aspect of hemodynamics that will need to be explored in future studies.
Discussion
As expected, people and the injuries they sustain are different. Although everyone here followed some aspect of the proposed hemodynamic changes, not everyone exhibited each of the proposed alterations, or in exactly the same time course. Within the overall population these subjects represent 8.5% of all subjects and 11% of all scans. There is the risk that their contribution to the cross-sectional results could have been disproportionately large. However, when revisiting the mixed effects models from Thibeault et al. (2019), 29 the results were not affected after removing the six subjects presented here. In those models, subjects were included as a random effect, with both days post-injury and sex included as fixed effects. This further supports the hypothesis of neurovascular dysfunction following mTBI in an adolescent population.
Pulsatile features
The absence of alterations in PI and P2R in Subjects 2 and 5 was an unexpected finding. In Thibeault et al. (2018), 18 PI and P2R were uncorrelated over the first 48 h. Based on that, it was expected that some individual subjects would display dysfunction in one or the other -- as demonstrated by Subjects 1 and 4. However, Subjects 2 and 5 did not exhibit recovery curves predicted by the model for either metric. From the medical records and self-reported symptoms, Subject 2 appeared to have a particularly mild injury. It is possible that there was not enough of a mechanical insult to affect measures of macroscopic function like PI and P2R. Similarly, Subject 5 was not measured until day 1 post-injury. A change in either PI or P2R may have been missed by the time the first measurement had been collected. Only Subjects 3 and 6 displayed the predicted alterations for both PI and P2R. Interestingly, both of these subjects had prolonged recovery periods, 57 and 49 days respectively. The robustness of these features will need to be established in future studies with a larger population.
Cerebrovascular reactivity
The most reliable feature of this study was BHI. All six subjects exhibited recovery curves that fit the model prediction. Although not all of them followed the exact time course, each displayed a period of hyperreactivity with a corresponding drop to steady-state. This suggests that alterations in cerebrovascular reactivity may be a robust phenomenon of mTBI.
Deviations From The model
Two subjects, 3 and 6, had anomalous values during their recovery that corresponded with external events or symptoms. The increase in BHI of Subject 6 was preceded by a severe relapse in self-reported symptoms, whereas Subject 3 exhibited a sharp increase in PI that coincided with the start of their menstrual cycle, as reported by the subject. Hormones have been shown to have a unique role in TBI. A recent study from Gallagher et al. (2018), 30 found that female mTBI patients who took hormonal contraceptives reported lower symptom severity. In rats, estrogen has been shown to have an adverse effect in females but a protective one in males. 31 Although speculative, it is possible that Subject 3 may have experienced a hormonal imbalance that exacerbated the existing neurometabolic dysfunction. However, subject 3 did not report an increase in activity, it is also possible that this spike was due to non-compliance to the recovery protocol. As mentioned above these subjects both had prolonged recovery periods. Future studies will explore the prognostic utility that deviations from the predicted recovery curve may have.
Clinical potential of hemodynamic assessment
Currently, hemodynamic assessment is not part of most clinical standards of care when treating mTBI patients. However, evidence of dysfunction has been found with a number of different modalities -- including MRI,32,40 TCD,18,41,46 and NIRS. 47 Incorporating vascular measurements into clinical practice during both the acute and recovery stages would complement existing diagnostic practices. Using currently available tools, clinicians could monitor changes from a baseline scan in the acute phase. Similarly, during the recovery phase, deviations from successive scans -- the procedure demonstrated here -- can elucidate the hemodynamic recovery of the subject. As research in the field progresses, the different factors that influence that recovery will be revealed -- providing more guidance in that tracking. In addition, diagnostic capabilities will continue to evolve for these different modalities. This will give clinicians a more quantitative assessment of hemodynamic dysfunction after an mTBI.
Conclusion
At the population level, the changes in these three parameters over different time points post-injury provided evidence for a general hypothesis of hemodynamic changes after a mTBI. 18 The models generated from this data then offered additional insight into the proposed phases of alteration--as well as a first step in providing prognostic capabilities. 20 The results here further strengthen the theory of a phasic progression of vascular dysfunction after mTBI by demonstrating how that model is also applicable to individual subjects. As more subjects with high-density data are collected in future studies, a more complete understanding of these phases and alterations can be developed.
Footnotes
Author contributions
Corey Thibeault had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: at the time that this research was conducted, Corey M. Thibeault, Amber Y. Dorn, Shankar Radhakrishnan and Robert B. Hamilton, were employees of, and either hold stock or stock options in NovaSignal Corporation.
Ethics statement
This study was carried out in accordance with the recommendations of Western Institutional Review Board (IRB #20141111), with written informed consent from all subjects. All subjects gave written informed consent in accordance with the Declaration of Helsinki. The protocol was approved by the Western Institutional Review Board.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Neurological Disorders and Stroke (grant number 1R43NS092209-01, 2R44NS092209-02).
Role of funder/sponsor
The NIH had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
