Abstract
Objective
To complete a systematic review of the literature examining neuroimaging findings unique to co-occurring syndromal depression in the setting of TBI.
Methods
A PRISMA compliant literature search was conducted in PubMed (MEDLINE), PsychINFO, EMBASE, and Scopus databases for articles published prior to April of 2022. The database query yielded 4447 unique articles. These articles were narrowed based on specific inclusion criteria (e.g., clear TBI definition, clear depression construct commenting on the syndrome of major depressive disorder, conducted empirical analyses comparing neuroimaging correlates in TBI subjects with depression versus TBI subjects without depression, controlled for the time interval between TBI occurrence and acquisition of neuroimaging).
Results
A final cohort of 10 articles resulted, comprising the findings from 423 civilians with brain injury, 129 of which developed post-TBI depression. Four articles studied mild TBI, three mild/moderate, one moderate/severe, and two all-comers, with nine articles focusing on single TBI and one including both single and recurrent injuries. Spatially convergent structural abnormalities in individuals with TBI and co-occurring syndromal depression were identified primarily in bilateral frontal regions, particularly in those with damage to the left frontal lobe and prefrontal cortices, as well as temporal regions including bilateral temporal lobes, the left superior temporal gyrus, and bilateral hippocampi. Various parietal regions and the nucleus accumbens were also implicated. EEG studies showed supporting evidence of functional changes in frontal regions.
Conclusion
Additional inquiry with attention to TBI without depression control groups, consistent TBI definitions, previous TBI, clinically diagnosed syndromal depression, imaging timing post-injury, acute prospective design, functional neuroimaging, and well-defined neuroanatomical regions of interest is crucial to extrapolating finer discrepancies between primary and TBI-related depression.
Introduction
The conceptualization of traumatic brain injury (TBI) as a disease process with potential for consequential downstream effects, rather than a singular isolated event, is becoming an increasingly validated viewpoint amongst clinical scientists. 1 TBI is an incompletely understood precursor to often under-reported neuropsychiatric disruption. It has been linked to a variety of new-onset psychiatric complications and is a risk factor for decompensation of previously well-controlled conditions. There may be utility in ascertaining how the clinical expression of psychiatric disorders in the context of TBI might compare to their idiopathic counterparts, especially as it relates to neuroanatomical underpinnings. Such ideas represent the premise of the current systematic review, which has the objective of synthesizing the existing literature that examines neuroimaging findings related to co-occurring syndromal depression in the setting of TBI.
Prevalence of depression following traumatic brain injury is quite common. It has been shown that 17−53% of TBI patients experience depressive symptoms during the first year post-injury.2–6 Studies have also found that 27 − 29% of TBI patients meet criteria for major depressive disorder.7–9 Studies that have followed individuals with TBI for one year or more post-injury found depression and anxiety to be the most commonly reported neuropsychiatric symptoms.10–12 There is also evidence that depression portends worse TBI recovery trajectories.13,14
Mood-related neuropsychiatric symptoms (NPS), including symptoms of depression, have not traditionally been at the center of TBI outcomes research. Instead, cognition-related NPS and functional ability have been the primary investigational foci. Despite this, NPS have a variety of characteristics that warrant their study in the setting of TBI. For example, the clinical profile of depression that occurs in patients with TBI is distinct from that of idiopathic depression. Individuals with TBI are at greater risk for developing more isolated depressive symptoms 8 characterized by increased apathy and irritability, 15 as well as greater social isolation, hostility, and more cognitive deficits. 16 The tendency for brain injury to manifest phenotypic expressions in a more isolated manner could provide key insights into the brain networks implicated in depression. TBI supplies an etiology, a key component of the disease triad, which is largely missing in idiopathic depression.
Neuroimaging studies on idiopathic depression have broadly suggested frontal lobe involvement including functional hypoactivation in the left dorsolateral PFC, 17 functional hyperactivity of the ventromedial PFC,18,19 and decreased volume in the PFC, hippocampi, amygdala, and basal ganglia. 20 Imaging correlates of idiopathic depression have received ample research attention, producing a multitude of systematic reviews and meta-analytic studies, with some even focusing on a single imaging modality.21–27 Imaging correlates of TBI-related depression, however, have not achieved such large-scale focus. Existing systematic reviews on post-TBI depression do not primarily focus on imaging findings.3,7,28–31
This systematic review summarizes the neuroimaging literature investigating syndromal depressive disorder in TBI. The aim of this review is to determine if there an area of the brain which when damaged, as demonstrated by neuroimaging, seems to be associated with the diagnosis of a depressive disorder after TBI. We posit that this effort may prove useful for future work regarding TBI screening and surveillance, bolstering preventative research efforts in neuropsychiatry, distinguishing specifiers in heterogeneous populations, and identifying neuroimaging markers for improvement in TBI patients.
Methods
Search strategy
A structured literature search strategy was designed to garner articles with neuroimaging and syndromal depression components in human TBI samples. Articles published up to April, 2022 were extracted from PubMed (MEDLINE), PsychINFO, EMBASE, and Scopus databases. Boolean searches were kept broad in the interest of reflecting all neuroimaging modalities and in order to capture broad domains of neuropsychiatric symptomatology. A more general approach was also necessitated by the current state of the TBI literature, which comprises many disparate approaches to definition, severity, population, and timing of assessment. We employed 41 imaging-, 35 neuropsychiatric symptom-, and 15 TBI-related keywords. Exact search phrases and MeSH search field qualifiers are outlined in Appendix 1.
Review protocol
This review adhered to PRISMA 32 guidelines for implementation and reporting of systematic reviews. A summary of the review protocol, including the number of articles included and excluded in each step, can be found in Figure 1. In the first level of the screening process, titles and abstracts were reviewed in parallel for determination of inclusion or exclusion. Individuals in dyads, each with at least one senior TBI researcher, were blind to each other's determinations. An identical data extraction sheet was utilized by all reviewers (authors LNR, BRB, AK, MJCB, TP, SJ, NTT, JA, MBJ, AB, DAS, CR, ELG, LM, EBA, AP, DJL, ML, MEP). Discrepancies and cases in which a reviewer was unsure were routed to a third-party reviewer for a final decision. All included articles were then subjected to a full-text review by dyads, again followed by a reappraisal if necessary. The resulting article cohort was then split up into six neuropsychiatric domains: depression, anxiety, post-traumatic stress disorder, sleep disturbance, behavior/personality change, and psychosis. The present review focuses on syndromal depression. A series of subsequent reviews focused on the other neuropsychiatric domains will be published from these same efforts.
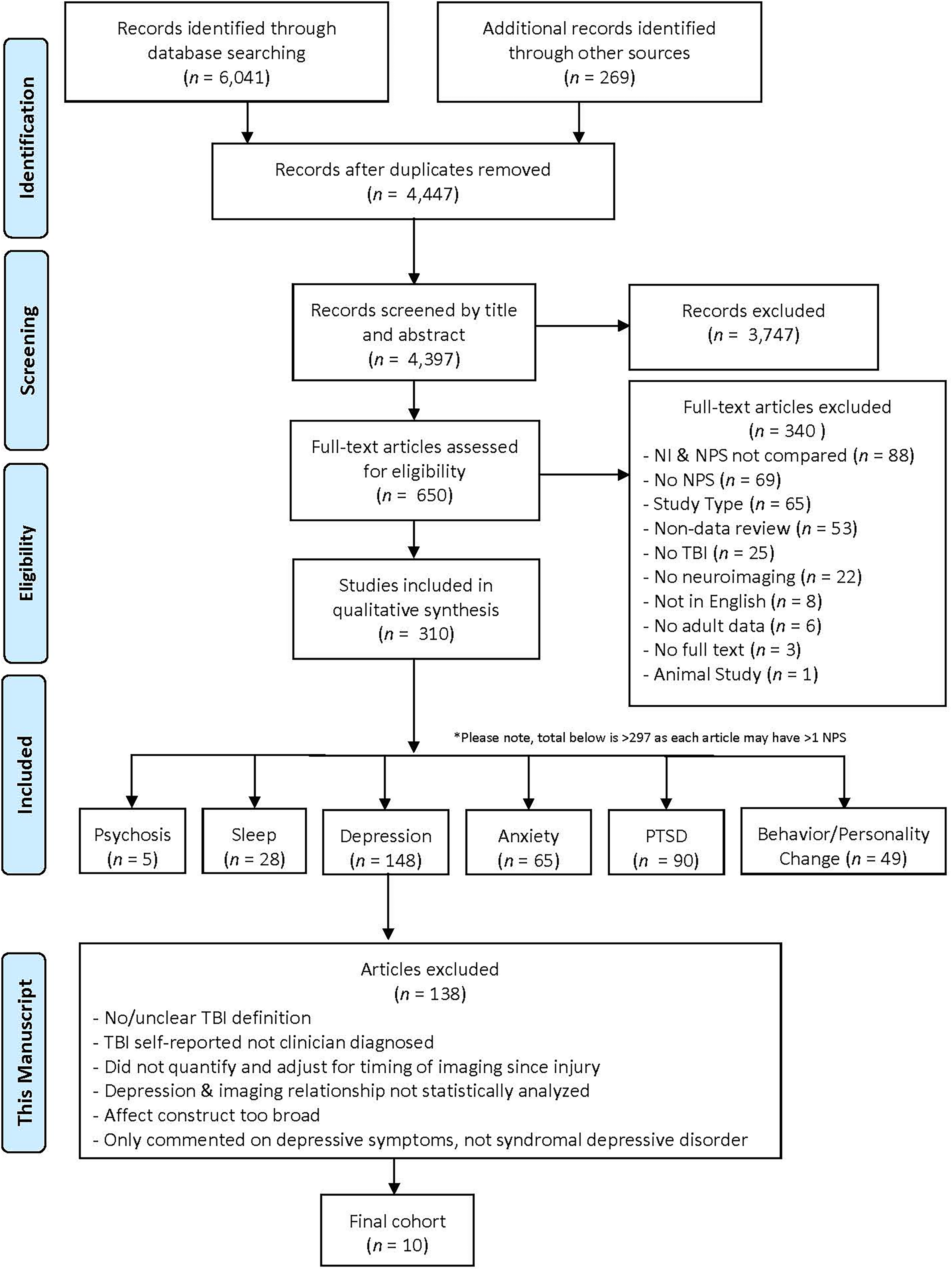
Article selection process
Inclusion and exclusion criteria
For both title/abstract and full-text reviews, a standardized set of inclusion and exclusion criteria were applied. Articles were excluded if they: (1) Lacked any one of the three key elements (i.e., neuroimaging, NPS, and TBI); (2) Were of an undesirable study type (i.e., case reports/case series with n < 5, editorials, commentary letters, replies to editor, book reviews, non-peer-reviewed articles, conference proceedings, poster abstracts, dissertations); (3) Were not written in English; and/or (4) The study population had no human subjects or adult data (≤18 years). Articles were not excluded on the basis of TBI severity, singularity or reoccurrence of TBI, acuity or chronicity of neuropsychiatric symptoms, neuroimaging modality, or if neuroimaging was conducted in the acute (≤ 48 h), sub-acute (2 days − 2 weeks), intermediate (2 weeks − 6 months), or chronic (> 6 months) time-span post-TBI. This information was, however, collected on all articles.
Final articles selected for the present syndromal depression-centric review include those that met all of the following additional criteria: (1) Conducted empirical analyses comparing neuroimaging correlates in TBI subjects with depression versus TBI subjects without depression; (2) Had a clear TBI definition for participants included in the study (i.e., formalized or study-specific criteria with any combination of Glasgow Coma Scale score, loss/alteration in consciousness, and/or post-traumatic amnesia); (3) TBI was clinician diagnosed, (e.g., a hospitalization-requiring injury that yielded confirmatory medical records and was not based on self-report); (4) Quantified and controlled for the time interval between TBI occurrence and acquisition of neuroimaging; (5) Had a clear depression construct (i.e., used DSM criteria/a structured diagnostic interview to comment on syndromal depressive disorder, not only depressive symptoms; was not broadly studying “affect”, which could include mood pathology other than depression, such as anxiety or mania).
Article quality review
Included articles were reviewed for methodological quality and sources of potential bias (Supplemental Figures 1a and 1b) using the Newcastle-Ottawa Scale. 33 Broadly, this scale evaluates the risk of selection bias, comparability of comparison groups (e.g., risk of potential confounds), and the validity of outcome/exposure ascertainment for observational, non-randomized investigations (including distinct evaluation criteria for case-control and cohort studies). Articles were sorted based on the appropriate study type (i.e., case-control or cohort), and the corresponding Newcastle-Ottawa schematic was applied. Articles were determined to be case-control or cohort based on the outcome of interest to the systematic review, not necessarily the articles’ primary outcome. Each article was analyzed by a dyad of reviewers (LNR & BRB or AK & MJCB) followed by a consensus process, during which any discrepancies were addressed. Quality assessment data are presented, however, articles were not excluded from the systematic review based on quality outcomes.
Brain mapping
Approximate regions of interest (ROIs) reported by each study as having statistically significant associations with depression were extracted from a brain parcellation map in the Montreal National Institute (MNI) space 34 (JHU MNI Type II) 35 using MATLAB 36 (MATLAB R2021b, The MathWorks). In JHU MNI type II parcellation map, the entire gray matter and the white matter are parcellated. Color overlays of segmented brain structures were displayed on a representative T1-weighted MP-RAGE image (Figure 2(a)) using MRIcron visualization 37 software. Subsequently, a statistical analysis tool in MRIcron was used to extract region overlaps across studies. Finally, color overlays representing the extracted overlaps of 2 and 3 studies were displayed on a T1-weighted MP-RAGE image (Figure 2(b)).
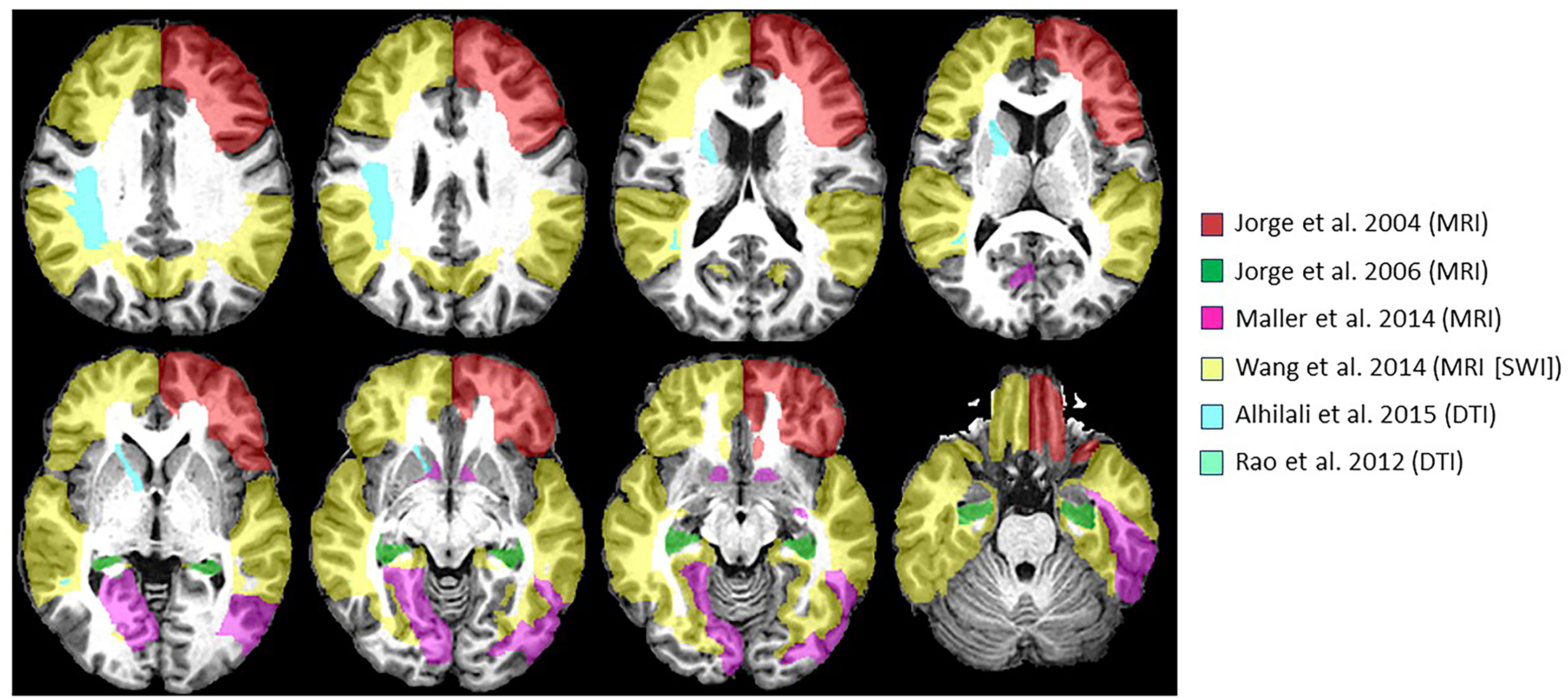
Brain mapping by article of approximate structural ROI overlays implicated in TBI-related depression.
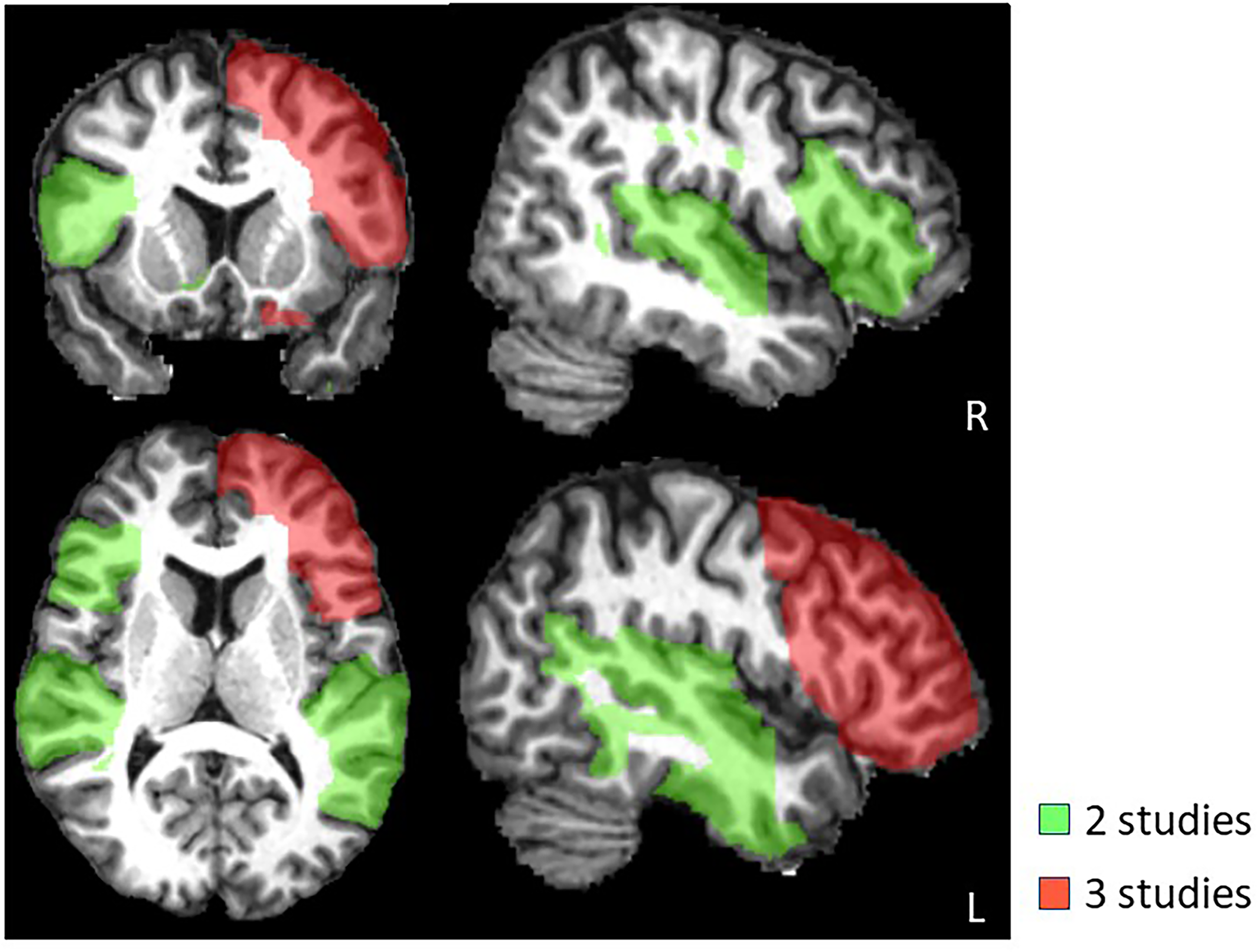
Brain map representing the approximate locations of replicated structural neuroimaging findings in TBI-related depression.
Results
Application of inclusion and exclusion criteria produced a final cohort of 10 articles (Table 2). Not including duplicate sample populations, these articles comprised the findings of 129 civilian subjects who developed post-TBI depression, and 294 who did not develop post-TBI depression. No studies with military or sport populations survived to the final article cohort. Two articles did not exclude for pre-TBI depressive disorders,12,38 three articles did not exclude for co-occurring anxiety disorders,39–41 and two articles excluded for previous TBIs.39,42 Only one article conducted a formal power analysis 43 and three studies reported effect size statistics.43–45 For details regarding article population, TBI severity, TBI occurrence, timing of imaging post-TBI, and neuroimaging modality, refer to Table 1. For limitations of the literature base and recommendations based on these limitations, refer to Table 3. For technical data on imaging parameters, see Supplemental Table 1.
Summary of article characteristics (n = 10)
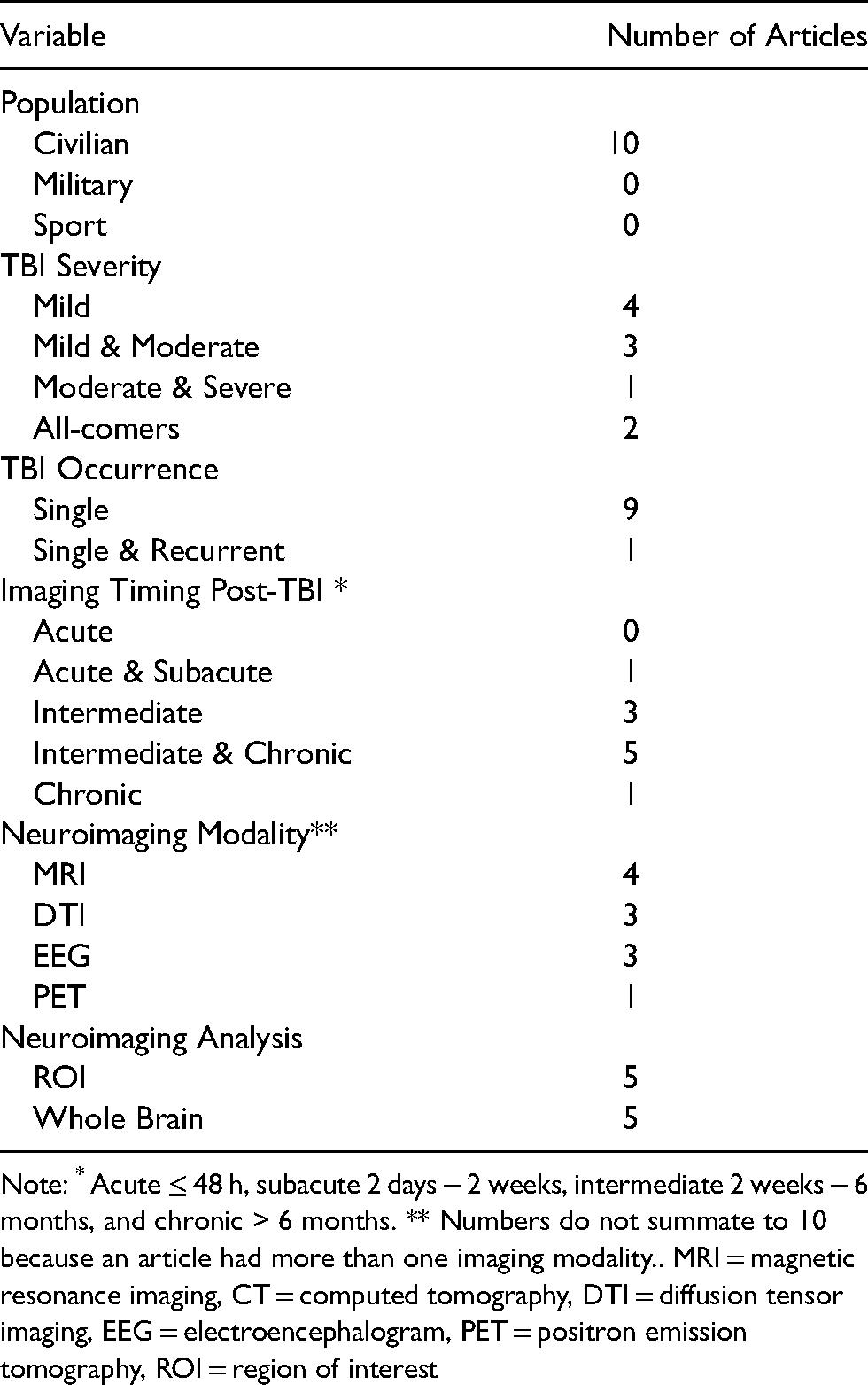
Note: * Acute ≤ 48 h, subacute 2 days − 2 weeks, intermediate 2 weeks − 6 months, and chronic > 6 months. ** Numbers do not summate to 10 because an article had more than one imaging modality.. MRI = magnetic resonance imaging, CT = computed tomography, DTI = diffusion tensor imaging, EEG = electroencephalogram, PET = positron emission tomography, ROI = region of interest
Articles with neuroimaging findings comparing traumatic brain injury patients with and without syndromal depression (n = 10)
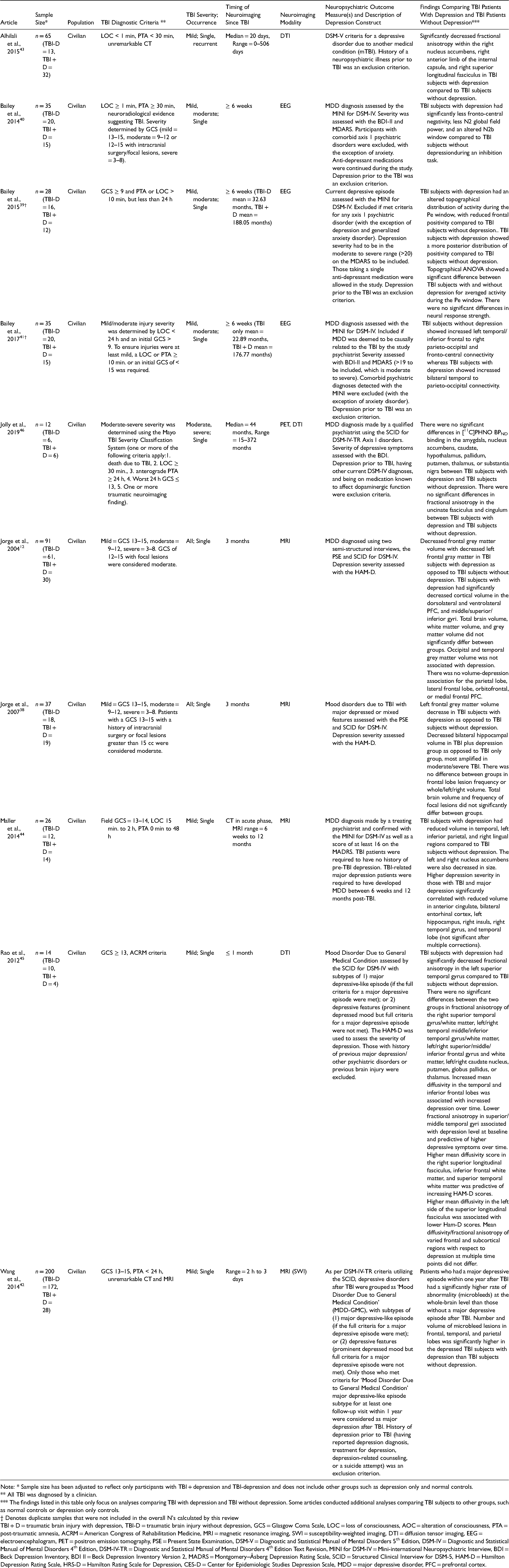
Note: * Sample size has been adjusted to reflect only participants with TBI + depression and TBI-depression and does not include other groups such as depression only and normal controls.
** All TBI was diagnosed by a clinician.
*** The findings listed in this table only focus on analyses comparing TBI with depression and TBI without depression. Some articles conducted additional analyses comparing TBI subjects to other groups, such as normal controls or depression only controls.
† Denotes duplicate samples that were not included in the overall N's calculated by this review
TBI + D = traumatic brain injury with depression, TBI-D = traumatic brain injury without depression, GCS = Glasgow Coma Scale, LOC = loss of consciousness, AOC = alteration of consciousness, PTA = post-traumatic amnesia, ACRM = American Congress of Rehabilitation Medicine, MRI = magnetic resonance imaging, SWI = susceptibility-weighted imaging, DTI = diffusion tensor imaging, EEG = electroencephalogram, PET = positron emission tomography, PSE = Present State Examination, DSM-V = Diagnostic and Statistical Manual of Mental Disorders 5th Edition, DSM-IV = Diagnostic and Statistical Manual of Mental Disorders 4th Edition, DSM-IV-TR = Diagnostic and Statistical Manual of Mental Disorders 4th Edition Text Revision, MINI for DSM-IV = Mini-international Neuropsychiatric Interview, BDI = Beck Depression Inventory, BDI II = Beck Depression Inventory Version 2, MADRS = Montgomery–Åsberg Depression Rating Scale, SCID = Structured Clinical Interview for DSM-5, HAM-D = Hamilton Depression Rating Scale, HRS-D = Hamilton Rating Scale for Depression, CES-D = Center for Epidemiologic Studies Depression Scale, MDD = major depressive disorder, PFC = prefrontal cortex.
Limitations existing among the included studies in the current systematic review and recommendations for future research

Findings by imaging modality
MRI volumetrics
Structural findings visualized by neuroanatomical region of interest (ROI) via brain mapping are displayed in Figure 2(a) and 2(b). Of the three articles with structural findings related to MRI volumetrics, all had significant findings associating onset of depression with decreased volume/cortical thinning in subjects who developed post-TBI depression compared to TBI subjects who did not.12,38,44 Areas implicated were left frontal grey matter, dorsolateral PFC, ventrolateral PFC, middle/superior/inferior gyri, 12 left frontal grey matter, bilateral hippocampal volume, 38 temporal, left inferior parietal, and right lingual regions as well as left and right nucleus accumbens. 44
Susceptibility-weighted imaging
There was one article that utilized susceptibility-weighted imaging 42 which found the number and volume of microbleed lesions to be higher in the depressed TBI group when compared to the non-depressed TBI group referencing the frontal lobe, temporal lobe, parietal lobe, and whole brain. 16
Diffuse tensor imaging (DTI)
Three articles conducted diffusion tensor imaging (DTI) analyses. Two of these articles found decreased fractional anisotropy to be associated with presence of depression in subjects with mild TBI when compared to mild TBI with absence of depression.43,45 These findings corresponded to the right nucleus accumbens, right anterior limb of the internal capsule and right superior longitudinal fasciculus, as well as the left superior temporal gyrus. The third article did not find significant differences in fractional anisotropy of the uncinate fasciculus and cingulum between TBI subjects with depression and TBI subjects without depression. 46
Positron emission tomography (PET)
One article featured the PET modality, specifically focusing on dopamine D2/D3 receptor availability following TBI. 46 There were no significant differences in [11C]PHNO BPND binding in the amygdala, nucleus accumbens, caudate, hypothalamus, pallidum, putamen, thalamus, or substantia nigra between TBI subjects with depression and TBI subjects without depression.
Electroencephalography (EEG)
Three articles contained EEG analyses, all of which were in the chronic/intermediate time frame for when imaging was conducted relative to injury occurrence. Two studies focused on frontal regions and found less fronto-central negativity, less N2 global field power, an altered N2b window, reduced frontal positivity, a more posterior distribution of positivity, and an altered pattern of averaged activity during the Pe window in depressed TBI subjects compared to non-depressed TBI subjects.39,40 The third study found that TBI subjects without depression showed increased left temporal/inferior frontal to right parieto-occipital and fronto-central connectivity whereas TBI subjects with depression showed increased bilateral temporal to parieto-occipital connectivity. 41 All three of the EEG modality articles were out of the same research group.
Article quality
Bias amongst cohort studies (n = 3) solely resulted from comparability of cohorts on the basis of design or analysis (Supplemental Figure 1a). Bias in case-control studies (n = 7) was mainly due to definition of controls, comparability of cases and controls on the basis of design or analysis, representativeness of the cases, and to a lesser degree selection of controls (Supplemental Figure 1b). Additionally, assessment of article quality revealed a widespread absence of methodologically controlling for previous TBI(s).
Discussion
This systematic review summarized the neuroimaging literature reporting on structural and functional changes associated with syndromal depressive disorder in the setting of TBI. Existing systematic reviews address the imaging correlates of idiopathic depression, and separately the imaging correlates of TBI, however this review aimed to synthesize the imaging findings that integrate depression and TBI. From a final cohort of 10 articles representing the findings from 423 total participants with TBI, we ascertained that syndromal depression developed after a TBI is generally associated with decreased volume or cortical thinning,12,38,44 decreased functional connectivity,39–41 decreases in white matter fractional anisotropy,43,45 and higher number/volume of microbleeds 42 when compared to TBI subjects who did not develop depression. Regionally, TBI-related depression was associated with a variety of bilateral frontal regions,12,39,40,42 particularly in those with damage to the left frontal gray matter,12,38 including the dorsolateral and ventrolateral prefrontal cortex, and middle/superior/inferior gyri.. 12 Temporal regions were also well-represented, including bilateral temporal lobes,41,42,44 the left superior temporal gyrus, 45 and bilateral hippocampi. 38 Other regions were the parietal lobes (bilateral, left inferior lobe, lingual gyrus)41,42,44 and nucleus accumbens.43,44 Taken together, these findings suggest that TBI-related depression is associated with diffuse changes throughout the brain, accompanied by a host of specific, regional alterations.
The findings reviewed herein identified spatially convergent structural abnormalities in individuals with chronic TBI and co-occurring syndromal depression in the bilateral frontal regions, particularly in those with damage to the left frontal lobe, including the prefrontal, orbitofrontal, and anterior cingulate cortices. Functional findings from EEG studies in this review further supported the beforementioned structural emphasis on frontal regions. Those regions have been consistently reported to be part of the biological pathways underpinning heterogeneous mental disorders like major depression.26,47,48 Nevertheless, TBI is more likely to affect frontal areas due to the anatomy of the brain within the skull in the context of TBI mechanics. 49 Thus, there is a degree of uncertainty regarding the precise pathways which could be influenced by those structural products with relevance to the underlying neurobiology of subsets of individuals who develop depression following TBI. More likely, it cannot be fully attributed to a specific frontal brain lesion but should be related to the whole frontal cortex and its interconnections.
Localized frontal lobe findings could be categorized into the three frontal-subcortical circuits implicated in TBI: the dorsolateral prefrontal circuit, orbitomedial frontal circuit, and anterior cingulate circuit. 50 These circuits are implicated in corresponding clinical syndromes (i.e., dysexecutive, disinhibition, and apathy syndrome, respectively). 51 In the current review, regarding differences between TBI subjects with and without depression, the two circuits that were most implicated were the dorsolateral prefrontal circuit (see Jorge et al., 2004 regarding the dorsolateral prefrontal cortex 12 ) and orbitomedial frontal circuit (see Maller et al., 2014 and Alhilali et al., 2015, which both involve the nucleus accumbens43,44). The anterior cingulate circuit did not have as many findings when comparing TBI subjects with and without depression, though Maller et al., 2014 found that higher depression severity in those with TBI and depression significantly correlated with reduced volume in the anterior cingulate. 44 Many articles in the current review focused on functional activity of the frontal lobes more globally,40,41 volumetrics of total frontal white/gray matter, 12 and microbleeds in frontal regions. 42
Amongst the included articles, frontal regions were a main focus and temporal regions were also well represented. The parietal lobes, including occipital/lingual regions, and right nucleus accumbens, are also worthy of mention. Regions of lesser emphasis were the right anterior limb of the internal capsule and right superior longitudinal fasciculus. Global volumetric brain analyses on MRI did not reach significance, which is consistent with systematic review results in idiopathic depression. 24 In contrast, significant findings from whole-brain analyses have been identified in a systematic review of TBI alone. 52 It is therefore possible that some of the less consistent findings in this review are related to TBI itself rather than the co-presenting depressive syndrome.
Regarding imaging modality, amongst articles finding significant differences between TBI patients with and without depression, there were four articles on structural MRI concerned with cortical findings (one susceptibility weighted), three articles on DTI concerned with white matter and subcortical findings, and three articles that focused on connectivity with EEG. A recent systematic review was conducted on the characterization of chronic traumatic encephalopathy/TBI using various imaging modalities. 52 By comparison, there was a far greater proportion of articles in the chronic traumatic encephalopathy/TBI review focused on structural imaging (21 of 25 articles) and fewer that utilized functional imaging (6 of 25 articles). This may suggest discordant approaches to studying emotional syndromes (focus on brain function) vs. brain trauma itself (focus on structure).
It is also an important confound to consider that certain connectivity measures, namely those used in DTI, have demonstrated non-linear brain changes over time post-injury. Two of the three DTI articles included in this review found significantly decreased fractional anisotropy in acute and subacute TBI compared to controls,43,45 which is consistent with the literature regarding connectivity measures in TBI longitudinally. 53 The third article in this review, which looked at chronic TBI, did not note significant differences in fractional anisotropy. 46
This review identified notable limitations in the literature base and areas of focus for future research, which are displayed in Table 3. There are also several methodological limitations to consider with the reported findings of this review. The timing of a patient's depression as it related to their TBI, as well as the timing of imaging data acquisition, was variable across articles and therefore not restricted. Also included in this review were several articles by the same authors that utilized some of the same participants in both studies. This issue of duplicate samples was factored in when calculating the sample size of this review. Three EEG articles were included in this review; the conclusions we can draw from those articles and the comparisons that can be made to MRI, DTI, and PET studies are limited by the spatial localization limitations that are inherent to the EEG modality. Additionally, because publication dates of the final article cohort ranged from 2004 to 2019, articles used both DSM-IV and DSM-5 criteria with their respective structured clinical interviews.
A systematic review was performed to examine neuroimaging correlates of co-occurring syndromal depression in TBI with the aim of utilizing neuropsychiatric illness to improve our understanding of what neuroanatomy to investigate in idiopathic depression. Research examining the nexus of TBI and depression represents an emerging field; additional inquiry with attention to TBI-only control groups, consistent TBI definitions, previous TBI, clinically diagnosed syndromal depression, co-occurring anxiety disorders, imaging timing post-injury, acute prospective design, functional neuroimaging, and well-defined neuroanatomical regions of interest is crucial to extrapolating finer discrepancies between the pathophysiology of idiopathic and TBI-related depression. The impact of TBI on brain abnormalities in individuals with depression requires further study, with continued research focus on structural, functional, and metabolic correlates of the prefrontal cortex, anterior cingulate, and other selected regions.
Supplemental Material
sj-jpg-1-ccn-10.1177_20597002221133183 - Supplemental material for Neuroimaging correlates of syndromal depression following traumatic brain injury: A systematic review of the literature
Supplemental material, sj-jpg-1-ccn-10.1177_20597002221133183 for Neuroimaging correlates of syndromal depression following traumatic brain injury: A systematic review of the literature by Lisa N. Richey, Barry R. Bryant, Akshay Krieg, Michael J. C. Bray, Aaron I. Esagoff, Tejus Pradeep, Sahar Jahed, Licia P. Luna, Nicholas T. Trapp, Jaxon Adkins, Melissa B. Jones, Andrew Bledsoe, Daniel A. Stevens, Carrie Roper, Eric L. Goldwaser, LiAnn Morris, Emily Berich-Anastasio, Alexandra Pletnikova, Katie Lobner, Daniel J. Lee, Margo Lauterbach, Simon Ducharme, Haris I. Sair and Matthew E. Peters in Journal of Concussion
Supplemental Material
sj-jpg-2-ccn-10.1177_20597002221133183 - Supplemental material for Neuroimaging correlates of syndromal depression following traumatic brain injury: A systematic review of the literature
Supplemental material, sj-jpg-2-ccn-10.1177_20597002221133183 for Neuroimaging correlates of syndromal depression following traumatic brain injury: A systematic review of the literature by Lisa N. Richey, Barry R. Bryant, Akshay Krieg, Michael J. C. Bray, Aaron I. Esagoff, Tejus Pradeep, Sahar Jahed, Licia P. Luna, Nicholas T. Trapp, Jaxon Adkins, Melissa B. Jones, Andrew Bledsoe, Daniel A. Stevens, Carrie Roper, Eric L. Goldwaser, LiAnn Morris, Emily Berich-Anastasio, Alexandra Pletnikova, Katie Lobner, Daniel J. Lee, Margo Lauterbach, Simon Ducharme, Haris I. Sair and Matthew E. Peters in Journal of Concussion
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
Ethical approval
Not applicable. This study is exempt from IRB review.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
