Abstract
Background
Post-traumatic headache (PTH) is a common secondary headache due to traumatic brain injury. In the past, significant research has been conducted to understand the pathophysiology and treatment options for PTH. However, PTH still lacks evidence-based treatment, and most of the management depends on the primary phenotype observed in the patient.
Objective
The main objective of this review is to provide a single reference that covers the current understanding of the pathophysiology and the treatment options available for PTH.
Methods
A detailed literature search on PubMed was performed, and a narrative review was prepared.
Results
The pathophysiology of PTH is multifactorial. Acute PTH may be attributed to increased peripheral pain sensitization with impaired pain inhibiting pathways. Chronic or persistent PTH may be due to a chronic inflammatory response and peripheral as well as central sensitization. The mechanism responsible for the transition of acute to persistent PTH is unknown. The migraine-like phenotype is reported to be the most prevalent headache type seen in PTH. New targets for preventive treatment have been identified in recent years, such as neuropeptides like calcitonin-gene-related peptide (CGRP), nitric oxide, and glutamate. The preventive pharmacological and non-pharmacological strategies employed for migraine (e.g. anti-CGRP monoclonal antibodies, onabotulinumtoxinA, physical therapy, cognitive and behavioral treatment, and neurostimulation techniques) have shown in preliminary studies that they are potentially efficacious, but large, randomized, double blind, placebo controlled trials are needed to further establish these as treatment options for PTH.
Conclusions
The lack of evidence-based treatment for PTH has created a need for future large trials to confirm the safety and efficacy of the currently employed treatments.
Keywords
Introduction
Post-traumatic headache (PTH) is defined as a secondary headache that starts within 7 days of a traumatic brain injury (TBI), per the International Classification of Headache Disorders (ICHD-III).1–3 Traumatic brain injury is often associated with post-traumatic stress disorder (PTSD). 2 Post-traumatic headache is further classified as either acute or persistent, depending on the duration of the headache. Acute PTH usually resolves within 3 months of its onset, while persistent PTH continues beyond 3 months.1,2 The TBI leading to PTH can be classified as mild TBI (mTBI, also referred to as concussion) or moderate-to-severe TBI, 1 based on the duration of the period of unconsciousness, Glasgow Coma Scale (GCS), post-traumatic amnesia (PTA), and Abbreviated Injury Severity Scale (AISS). Injuries with <30 min of loss of consciousness, GCS of 13-15, PTA <24 h, and AISS of 1-2 are classified under mTBI, whereas injuries with >30 min of loss of consciousness, GCS <13, PTA <24 h and AISS ≥3 are labeled as moderate-to-severe TBI.4,5 A greater proportion of mTBI patients develop PTH compared to moderate or severe TBI patients.6,7 The current review focusses on pathophysiology and management of PTH prevalent in mTBI patients.
Post-traumatic headache encompasses a broad range of headache phenotypes, including migraine-like headaches, tension-like headaches, trigeminal autonomic cephalalgias, cervicogenic headache, and cluster-like headache phenotypes.1,2,7 It can be complicated by medication overuse. The clinical manifestations often resemble migraine and include symptoms such as nausea, vomiting, pulsating pain, exacerbation of pain by light or sound, unilateral headaches, and worsening by physical activities. 7 Post-traumatic headache is often associated with disability, and reports have shown as many as 35% of patients being unable to return to work 3 months after the injury as a result of PTH. 1
Methods
An extensive literature search was conducted using PubMed and Google Scholar. Specific search terms related to traumatic brain injury, post-traumatic headache, post-concussion syndrome, chronic migraine, etc., were used. The articles retrieved were screened by title, abstract or full text accordingly. The included studies were only those relevant to the review, including (a) prevalence and risk factors associated with the development of PTH; (b) studies related to several theories of pathophysiologic mechanisms of PTH; (c) pharmacological and non-pharmacological management of PTH. Articles were excluded if they were found unrelated to the scope and aim of the review. Further, duplicate papers were also screened out.
Results
Prevalence
Post-traumatic headache is the most prevalent consequence of mTBI. TBI can occur with motor vehicle accidents, falls, or military events. 8 In the young adolescent population, sports activities are a significant reason for TBI. 9 A prospective study in the United States (US) conducted among mTBI patients showed a cumulative incidence of 91% for persistent PTH over a period of one year, 10 while another study in the US reported the incidence to be 71% among TBI patients. 11 Further; a Dutch study reported that 51% of TBI patients developed acute PTH, while 40% developed persistent PTH. 12 The high incidence of PTH among mTBI patients makes it a significant health issue.
Migraine-like headaches are the most common phenotypes seen in PTH patients. In a prospective study conducted among 189 mTBI patients, 49% matched migraine-like and probable migraine-like headache, while up to 40% matched with the tension-like headache. 10 Similarly, in another study, 31% and 22% of patients reported migraine or probable migraine de novo headaches respectively, followed by tension-like headaches (21%) over the first year after TBI. 13 Moreover, a study was conducted among a cohort of 1567 soldiers (557 with and 1030 without mTBI) from the Warrior Strong Study to differentiate PTH from non-traumatic headaches. The migraine-like phenotype was reported to be more prevalent and severe in soldiers with mTBI and PTH (33%) compared to soldiers without mTBI (14%) and soldiers with mTBI but not PTH (20%). 14
Post-traumatic headache is one of the symptoms of post-concussion syndrome following mTBI. Other symptoms can include photophobia, phonophobia, fatigue, reduced memory, sleep disturbances, inability to focus, inattention, anxiety, depression, dizziness, or irritability. 15 Approximately 30% to 40% of the patients with persistent PTH (veterans or civilians) reported features of co-existent PTSD. These patients with comorbid PTSD reported having worse headaches than those without PTSD.16,17
Risk factors
The major risk factors for acute or persistent PTH after mTBI includes female gender, younger age at the time of the injury, pre-existing headaches, or early-onset headache shortly after the injury, and frequent TBI events.7,12,18 Further, the presence of psychiatric problems prior to TBI may or may not be a risk factor for PTH due to conflicting evidence.12,19 However, the development of persistent PTH has not been found to be related to the cause of TBI, alcohol abuse, loss of consciousness at the time of TBI, GCS scores, race, literacy level, occupation, or marital status.7,18 Moreover, some studies have found that individuals with concussion injuries may have a longer recovery time if any litigation process was involved after the mTBI.20–22
Patients exhibiting migraine or probable migraine as the phenotype of PTH present with more frequent and severe headaches.10,13 Additionally, patients with a family history of primary headaches or genetic predisposition to migraine may also be prone to develop PTH after TBI. 23
Pathophysiology
There are a number of mechanisms by which PTH may develop. These include structural or functional damage to cranial or central (e.g. spinothalamic, thalamocortical) structures, dysfunction of pain inhibiting mechanisms, and neurometabolic or neuroinflammatory changes [increased release of calcitonin gene-related peptide (CGRP)], leading to trigeminal sensory system activation and sensitization.1,24 The interplay between several overlapping mechanisms leads to PTH, which is depicted in Figure 1.1,25–28
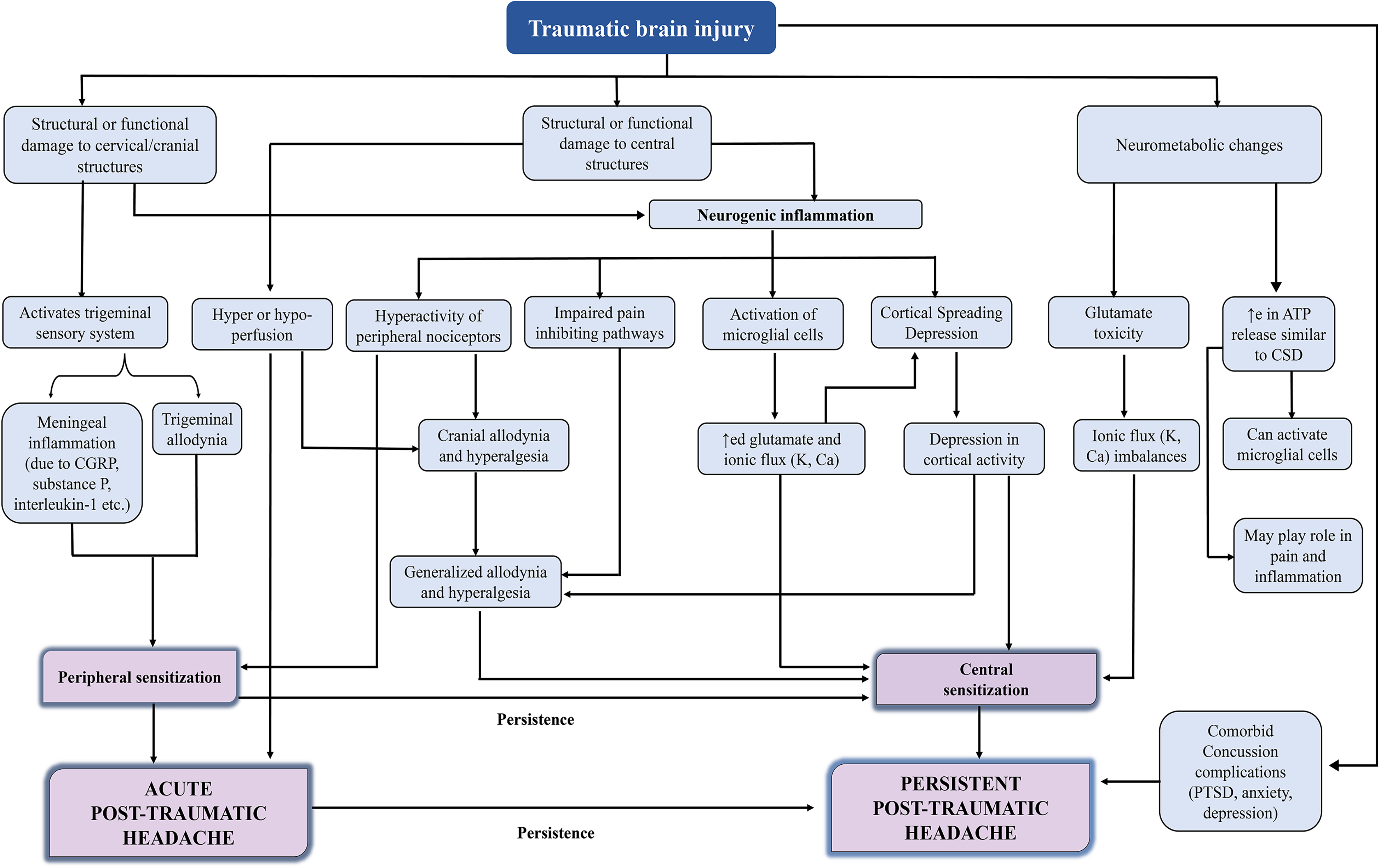
Proposed pathophysiologic mechanism leading to post-traumatic headache. PTH: Post-traumatic headache; PTSD: Post-traumatic stress disorder; CGRP: Calcitonin gene related peptide; CSD: Cortical spreading depression; Adapted from Defrin 201426, Conidi 201725, Kamins and Charles, 201827, Ashina et al., 20191, Mares et al., 201928.
The pathophysiology of acute transforming to persistent PTH is a continuous spectrum of sensitization. The etiology of acute PTH is multifactorial and related to increased peripheral pain sensitization with impaired pain inhibiting pathways. Persistent PTH may develop due to chronic inflammatory response along with peripheral and central sensitization. Furthermore, the mechanism behind the transition from acute to persistent PTH remains a research challenge that may involve inflammatory mechanisms and sensitization theories. 28
The major pathophysiologic mechanism of acute PTH may involve structural and functional damage to the neck or cranial structures due to mTBI. This damage initiates a cascade of events such as axonal injury, impaired descending pain neuromodulation, imbalance in ionic flux (calcium influx and potassium efflux), changes in cerebral blood flow as well as meningeal inflammation via inflammatory mediators like CGRP, nitric oxide, pituitary adenylate-cyclase-activating polypeptide (PACAP), and substance P.1,25,27–29 The increased neuroinflammation (increased release of neuropeptides like CGRP, PACAP, etc.) during TBI excites the central nervous system, peripheral nociceptors, and the trigeminal sensory system.1,27,28,30 The hyper-responsiveness of the trigeminal sensory system (involving trigeminal ganglion, peripheral trigeminal afferents, or spinal trigeminal nucleus), also termed as trigeminal allodynia may lead to peripheral sensitization in acute PTH patients, similar to the pathophysiology seen in migraine.1,25,28,30,31
Persistent PTH may have peripheral or central origin or both. 26 It may occur as a result of damage to central structures (such as spinothalamic or thalamocortical) and neurometabolic changes during TBI. 28 This central damage can lead to changes in cerebral blood flow responsible for the headache symptoms. It may, however, be prolonged in some persistent PTH cases, resulting in altered cerebrovascular function. 27 Additionally, increased neuroinflammation during TBI can lead to hyperactivity of peripheral nociceptors and impair the pain inhibiting pathways. These inflammatory mechanisms may lead to the development of cranial allodynia, which can progress to generalized allodynia over time among persistent PTH patients. 26 Further, the inflammatory response activates astrocytes and glial cells along with the release of nociceptive neuropeptides (e.g. CGRP), causing glutamate excitotoxicity, ultimately resulting in neuronal and vascular damage.1,27,28 Persistent PTH may be linked to cortical spreading depression (disrupts cortical function for minutes to hours).1,28 This cortical spreading depression may be triggered as a result of increased production of excitatory neurotransmitters (e.g. glutamate) and imbalances in ionic flux (increased calcium influx and potassium efflux)..1,25 The raised intracellular calcium levels further disrupt the axonal structure and can cause neuronal damage.1,28
Moreover, comorbid conditions (PTSD, anxiety, depression) and individual risk factors (genetic predisposition, gender, previous episodes of headaches) may account for the development of persistent PTH. 28
Management
A multidisciplinary approach is needed to manage PTH, including both pharmacological and non-pharmacological therapy. At present, there is no specific treatment approved by Food and Drug Administration (FDA) for PTH.7,32,33 Primarily, PTH is managed by the conventional treatments used for primary headaches, predominantly those used to treat migraine. However, this approach may not be completely efficacious because of comorbid conditions, such as PTSD, dizziness, depression, anxiety, cognitive problems, or sleep disturbances (Refer Box 1). Non-pharmacological therapy combined with pharmacological therapy can be tailored to address PTH as well as comorbid conditions, including PTSD.7,34,35
Key recommendations for clinical management for PTH
Successful management of PTH involves a tailored approach via a combination of pharmacological and non-pharmacological therapy.
Onabotulinumtoxin A, CGRP monoclonal antibodies, and nerve blocks with local anesthetics are more suitable for PTH treatment than traditional oral preventive medications since they do not cause cognitive changes or fatigue.
Non-pharmacological strategies in combination with pharmacological treatment can help improve compliance, reduce the severity and frequency of headaches, and manage comorbid conditions.
The pharmacological treatment for PTH involves using limited doses of acute medications to reduce the severity of the headache and preventive medications to reduce the frequency of headaches.32,35,36 Acute medication use needs to be limited to prevent the development of medication overuse. Pharmacological agents like Onabotulinumtoxin A, CGRP monoclonal antibodies, and nerve blocks with local anesthetics do not cause cognitive changes or fatigue, making them more suitable for PTH treatment than many traditional oral preventive medications. The non-pharmacological modalities include physical therapy, vestibular therapy, neuromodulation, cognitive-behavioral therapy, biofeedback, neurofeedback, education, and lifestyle modifications.33,34,37 The existing acute and preventive pharmacological therapy and non-pharmacological modalities are reviewed below.
Acute pharmacological therapy for PTH:
Migraine-like or probable migraine-like headaches are commonly found in PTH patients. Analgesics, non-steroidal anti-inflammatory agents (NSAIDs), triptans, ditans, and gepants are prescribed for the acute treatment of migraine and can be used to treat migraine-like PTH.35,38 Other agents for acute treatment include diphenhydramine, methylprednisolone, and anti-emetic agents (metoclopramide, ondansetron, promethazine, prochlorperazine).7,39,40 The use of all these acute medications, aside from the gepants, should be limited to prevent the development of medication overdose headache (rebound headaches).35,38
The pharmacological treatment for tension-like headaches in PTH patients includes similar agents used to treat migraine-like headaches, such as NSAIDs and muscle relaxers. Of note, triptans, gepants, and ditans are not the preferred choices for this particular phenotype.7,24,41 Opioids and narcotics should be avoided in all cases.
Preventive pharmacological therapy for PTH
The consensus guidelines for preventive therapy for migraine are that a preventive treatment should be started if patients have more than 4 disabling headache days in a month or the headache affects the patient's quality of life (QoL).41–43 There are no guidelines for the treatment approach for PTH; however, early and aggressive preventive treatment should be considered due to the risk of sensitization and potential medication overuse. 41 Starting preventive treatment early in the course may prevent sensitization and an overall decrease in disease burden. Antidepressants (tricyclic antidepressants [TCA] like amitriptyline, nortriptyline), anti-epileptics (topiramate, valproic acid, gabapentin, zonisamide), β-blockers (propranolol, timolol, metoprolol), calcium channel blockers (flunarizine)7,32,39,42 and ACE inhibitors (Candesartan) 30 can all be tried. However, the choice of preventive agents in PTH patients should be tailored depending on the presence of comorbidities or contraindications. For example, TCAs like amitriptyline are preferred for patients with insomnia, whereas gabapentin may be preferred for patients with anxiety.7,42 However, these medications may cause fatigue with a negative impact on cognition. 30 As a result, medications that do not cause side effects of fatigue and cognitive slowing are preferred. Furthermore, oral preventive medications often have adherence issues owing to their tolerability or inefficiency. 44
If acute medications are needed routinely to manage the headache severity of PTH, then preventive medications should be initiated to prevent medication overuse. Nutraceuticals like herbals (butterbur, feverfew), vitamins (vitamin B2, coenzyme Q10), and mineral (magnesium) supplements have shown some benefit in migraine.7,24 Certain preventive medications under development that have the potential to be used in PTH therapy are discussed below:
Monoclonal antibodies targeting CGRP:
Based on the role of CGRP in the pathogenesis of PTH, anti-CGRP monoclonal antibodies may show benefit in persistent PTH. CGRP is implicated in migraine attacks 45 and anti-CGRP antibodies have been developed to prevent migraine. These include eptinezumab, erenumab, fremanezumab, and galcanezumab. These are all FDA-approved for clinical use in migraine. The adverse event profile of these agents was found similar to placebo except for injection site reactions, constipation, and muscle spasms. 46 However, at present, only erenumab and fremanezumab have been studied in patients with PTH.
Erenumab, the first completely humanized anti-CGRP monoclonal antibody, targets the CGRP receptor, unlike other monoclonal antibodies of this class. 46 In an observational study conducted among seven PTH patients, a single dose of 140 mg subcutaneous erenumab reported a significant reduction in headache days with no relapse till 6 weeks. 47 A case series involving 5 women with poorly managed PTH reported benefits from erenumab administration. 48 Further, a non-randomized open-label study conducted among 89 patients with persistent PTH also demonstrated a mean reduction of 2.8 moderate-to-severe headache days per month. Further, no serious adverse events were reported. The most prevalent side effects observed were constipation and injection site reactions. 49
Fremanezumab has proven efficacy as a preventive treatment for migraine as a quarterly or monthly regimen. 50 Since it has proved efficacious in migraine, a Phase 2 randomized, placebo-controlled, interventional trial (NCT03347188) tested its efficacy in PTH patients. However, the effect of fremanezumab in PTH patients was not significantly different from placebo. 51 In contrast, a retrospective analysis of 63 PTH patients who received fremanezumab, galcanezumab, and erenumab observed improved headache severity and frequency. Patients who switched between anti-CGRP monoclonal antibodies also benefited. Moreover, no significant adverse events were observed except constipation, throat pain, or nausea. 52 Given the limitations of these studies, there is a need for large, randomized, placebo-controlled trials to establish the efficacy of anti-CGRP monoclonal antibodies in PTH patients. 46
Onabotulinumtoxina
OnabotulinumtoxinA (Botox) is FDA approved for the prevention of chronic migraine since 2010. 53 The injections are given as fixed-dose (minimum dose: 155 U; maximum dose:195 U), fixed-site, intramuscular injections across 7 specific muscle areas (corrugator, procerus, frontalis, temporalis, occipitalis, cervical paraspinal, and trapezius).54–56 OnabotulinumtoxinA inhibits the peripheral nociceptive pathways in migraine and mediators like CGRP, nitric oxide, substance P, and glutamate. It also reduces the central sensory processes in the central nervous system. 57 Several clinical trials have been conducted to show its efficacy and safety in chronic migraine (Supplemental Table 1). Based on the efficacy in chronic migraine, it has also been evaluated in PTH patients. Studies conducted to date are listed in Supplemental Table 1. Overall, these studies provide clinical evidence of the beneficial role of OnabotulinumtoxinA in persistent PTH patients. Further, it is found to be well-tolerated except for some mild and transient adverse events such as injection site reactions, neck pain, eyelid ptosis, musculoskeletal stiffness, etc.53,55 However, large, controlled trials are warranted to confirm its role in PTH patients.
A combination of onabotulinumtoxinA with anti-CGRP monoclonal antibodies can be a strategy for preventing headaches in migraine patients. The effectiveness of this combination has been reported in some clinical studies.58–60 Two retrospective reviews among chronic migraine patients evaluated combination therapy of OnabotulinumtoxinA and anti-CGRP monoclonal antibodies (erenumab, fremanezumab, and glacanezumab). The results reported better clinical outcomes in terms of headache or migraine days per month for the combination treatment than onabotulinumtoxinA treatment alone. Also, the combination therapy demonstrated a similar safety profile as individual therapy of onabotulinumtoxinA or anti-CGRP monoclonal antibodies.59,60 However, further mechanistic, and prospective clinical studies are required to confirm the applicability of this combination in terms of additional benefits and safety. A similar combination may be effective in PTH patients, but needs further clinical evidence.
Nerve blocks and trigger point injections
Peripheral nerve blocks are also reported as an interventional treatment strategy for PTH patients. Applying local anesthetics (e.g. lidocaine or bupivacaine injections) to peripheral nerves involved in the pain pathway could reduce the transmission to the trigeminal nucleus and thus reduce the pain sensation. The common nerve sites include occipital (greater and lesser), auriculotemporal, supraorbital, and/or supratrochlear nerves, and sphenopalatine ganglion. A retrospective case series conducted among 28 patients (< 18 years of age) with PTH evaluated the pain-relieving efficacy of peripheral nerve block. This study reported 71% of patients with complete pain relief after nerve block. 61 Another case series also reported the beneficial effect of occipital nerve block in 64% of 15 adolescent PTH patients. 62 Further, steroids can also be combined with local anesthetics to induce unilateral or bilateral nerve blocks.7,63 However, no significant differences were observed in a study comparing local anesthetics (lidocaine) vs. a combination of local anesthetic with steroid (lidocaine and triamcinolone) in migraine patients. 64
Intranasal block of a sphenopalatine ganglion can also treat PTH, owing to its efficacy in chronic migraine patients. 65 However, to date, only a single case of persistent PTH after a sport-related injury has been reported to be managed by an intranasal block of the sphenopalatine ganglion. 66
Patients with whiplash injuries often have trigger points in the neck region, and these can contribute to the headache. Trigger point injections of local anesthetics given in the following muscles: trapezius, levator scapulae, semispinalis capitis, splenius, and sternocleidomastoid may be helpful. 67 However, no studies have been reported on the use of trigger point injections in PTH patients to date.7,63
Epidural steroid injections and radiofrequency ablation
Certain experimental interventional treatment strategies for PTH patients include cervical epidural steroid injections and radiofrequency ablation, especially for the cervicogenic-like phenotype. Cervical epidural steroid injections combined with local anesthetics have preliminarily shown to be effective for short- and long-term pain relief in cervicogenic headache patients with radicular pain, especially those who have exhausted the conservative treatments. 68 Furthermore, radiofrequency ablation is another experimental interventional technique being tested for cervicogenic headache. Ablating the afferent nerve supply by an alternating current in the radiofrequency range could destroy the pain generator and help relieve pain. However, the effectiveness of RFA in cervicogenic headache patients is limited as only one high-quality randomized trial (but insufficient patient numbers) supports it. 69 High-quality randomized controlled studies are lacking for these techniques to prove their efficacy for cervicogenic headache patients. These techniques are also not yet tested among PTH patients.
Non-pharmacological treatment
Cervicogenic-like headaches, psychiatric symptoms, PTSD, and cognitive complaints are commonly observed in mTBI patients, besides migraine-like and tension-like headaches. A multidisciplinary approach with a combination of pharmacological and non-pharmacological treatment strategies can help improve compliance, reduce the severity and frequency of headaches, and manage comorbid conditions.7,34 Several non-pharmacological strategies are currently being used for different phenotypes of PTH, such as physical therapy, behavioral treatment, neuromodulation, acupuncture, or even lifestyle modifications.
Physical treatment and rehabilitation:
Physical therapy is preferred for PTH patients with cervicogenic headache phenotype. Studies have shown that PTH patients (both acute or persistent) who underwent complementary physical therapy showed improved headache intensity or neck pain compared to patients without such therapy.33,63 Different modalities such as physiotherapy, massage therapy, spinal manipulation, and spinal mobilization are preferred to reduce headache intensity, frequency, or duration. It is reported that cervical spine mobilization techniques are safer than manipulation techniques since manipulation involves high-velocity force that may lead to disc herniation or arterial dissection.7,63 Further, a study comparing spinal mobilization technique versus massage therapy reported significant improvement in headache variables with spine mobilization as compared to massage therapy. 70 Further, aerobic exercise therapy during rehabilitation after a TBI was found to benefit some patients (especially athletes) with persistent symptoms. 71
Moreover, balance disorders have also been observed in post-concussion patients, including those experiencing PTH.37,72 In a randomized controlled trial of 31 sport-related post-concussion patients, vestibular therapy combined with cervical physiotherapy was found to be effective in reducing the time to return to sport. 73 A systematic review has also suggested vestibular therapy to be useful for mTBI patients who have persistent symptoms. 37 However, evidence is limited and more insights into the implementation of this therapy in PTH patients are required.
Behavioral treatment
The PTH usually coexists with other post-concussion symptoms such as PTSD, depression, anxiety, cognitive complaints, insomnia, or even reduced QoL. These coexisting symptoms can be managed by psychological or cognitive-behavioral treatment (CBT). Different CBT approaches (relaxation therapy, psychotherapy, psychoeducation, and techniques like learning to cope with the trigger) have benefitted PTH patients with their headache intensity, frequency, and emotional well-being.33,74 However, patients with coexisting PTSD require more CBT sessions than those without comorbid PTSD. 33 CBT can be combined with other therapies such as bio- and neurofeedback (e.g. Flexyx Neurotherapy System), or psychoeducation.33,74
Neuromodulation:
Refractory PTH patients who do not respond to preventive medications (e.g. antidepressants, anti-epileptics, β-blockers, etc.) may benefit from peripheral neurostimulation (especially, greater occipital or supraorbital nerve) by a reduction in headache days. 63 Further, repetitive transcranial magnetic stimulation (non-invasive technique of neurostimulation) has been found to improve headache severity, persistent post-concussion symptoms, and QoL among PTH patients. This was evidenced in a study conducted among 20 persistent PTH patients. Large clinical trials are, however, warranted to provide robust clinical evidence for the effectiveness of such neurostimulation techniques. 75
Acupuncture:
Acupuncture may serve as an adjunct therapy for PTH patients. 33 It can help relieve the cervicogenic pain associated with tension-like PTH following TBI. 76 However, more studies are required to confirm this recommendation. 33
Several ongoing clinical trials are currently evaluating therapeutic or preventive medications as well as non-pharmacological strategies for the management of PTH. The details of these clinical trials are depicted in Supplemental Table 2.
Gaps in knowledge and future directions
Several gaps still persist in the acute and preventive treatment of PTH (Refer Box 2). The pathophysiology of PTH is complex and needs more in-depth studies. There is a knowledge gap in the mechanism responsible for the shift from acute to persistent PTH. Detailed insight into the pathophysiologic mechanisms may help identify the unknown mechanisms and discover newer targets for specific treatment of persistent PTH. Most of the therapies currently being employed are based on the primary phenotype of PTH. It is essential to accumulate scientific evidence and increase the reliability of such treatments. Further, large, randomized controlled, prospective trials are warranted for newer therapies such as onabotulinumtoxinA, a combination of anti-CGRP monoclonal antibodies and onabotulinumtoxinA, gepants, and ditans. These therapies have proven clinical benefits in migraine-like PTH. However, future extensive trials are needed to confirm the results.
future directions for research in PTH
More studies are needed for the understanding of pathophysiologic mechanisms responsible for the shift of acute to persistent PTH.
Newer targets need to be identified from the pathophysiologic mechanisms to treat persistent PTH.
Extensive, properly designed, randomized, controlled, double-blind, prospective trials among PTH patients are needed to prove the effectiveness of newer therapies like onabotulinumtoxinA, anti-CGRP monoclonal antibodies, gepants, and ditans that are employed for migraine headaches.
Moreover, there are sparse clinical studies with appropriate study designs evaluating the efficacy and safety of the new modalities treating persistent PTH. Hence, future trials should be designed as double-blind, randomized controlled trials that can lay the foundation for evidence-based medicine in the acute and preventive treatment of persistent PTH rather than the current expert-opinion-based therapy.
Further, there are some inherent challenges for designing trials for studying the intervention's outcomes in PTH patients. Some of these challenges include the timing of treatment in a study, various mechanisms of injury for different individuals, different outcomes (e.g. the severity of headache and QoL may be more significant than the number of headache days in some patients), etc. Thus, outcome measurements in such trials may differ from those in traditional headache trials.
Additionally, the treatment outline varies for different phenotypes of PTH, and thus, the ICHD diagnostic criteria should include phenotype-based classification of PTH. This classification would help in patient stratification and develop more specific therapies. A secondary diagnosis of medication overuse headache will need to be considered if there is continued high use of acute medications. Future randomized controlled trials should target improvement in overall quality of life as secondary endpoints. This could help define better long-term therapies for persistent PTH patients.
Conclusions
Persistent PTH is the most prevalent consequence of mTBI with a complex pathophysiology. Inflammation and sensitization theories are proposed to be responsible for the acute to persistent PTH transition. Furthermore, there is a lack of treatment guidelines specific to PTH. Thus, the therapy is mainly based on the treatment guidelines of the primary phenotype observed in the PTH patient. The typical phenotype observed in PTH patients is migraine-like; thus, medications used for treating migraine headaches are employed for acute and preventive treatments of PTH. However, the reliability and efficacy of the newer therapies (e.g. anti-CGRP monoclonal antibodies, onabotulinumtoxinA, etc.) need to be established through rigorous, blinded, randomized controlled prospective trials. Moreover, the non-pharmacological strategies employed for PTH treatment also require further studies to confirm their role in PTH therapy. Future studies are needed to develop treatment guidelines for PTH that include comorbid conditions such as depression, PTSD, sleep disturbances, and dizziness.
Supplemental Material
sj-docx-1-ccn-10.1177_20597002221093478 - Supplemental material for Post-traumatic headache: Pathophysiology and management - A review
Supplemental material, sj-docx-1-ccn-10.1177_20597002221093478 for Post-traumatic headache: Pathophysiology and management - A review by Andrew Blumenfeld, Jennifer McVige and Kerry Knievel in Journal of Concussion
Footnotes
Acknowledgements
Declaration of Conflicting Interests
Dr. Andrew Blumenfeld has served on advisory boards for AbbVie (Allergan), Aeon, Alder, Amgen, Axsome, Biohaven, Impel, Lundbeck, Lilly, Novartis, Revance, Teva, Theranica, and Zoscano, and has received funding for speaking from Allergan, Abbvie, Amgen, Biohaven, Lundbeck, Lilly, and Teva. He holds patents for onabotulinumtoxinA in migraine assigned to Allergan. He has provided his consultancy services at Allergan, Abbvie, Alder, Amgen, Biohaven, Lilly, Lundbeck, Novartis, Teva, and Theranica. Dr Jennifer McVige has received an honorarium for speaking for AbbVie (Allergan), Amgen/Novartis, Eli Lilly, and Company, Avanir and Teva. Her institute has received clinical trial support from Amgen/Novartis, Impax, Teva, Eli Lilly and Company, Depomed and Theranica. She has received funding for investigator-initiated trials from the Dent Family Foundation. She is a member of the American Academy of Neurology, the American Headache Society and the American Society of Neuroimaging. She serves on the Board of Directors for the American Society of Neuroimaging. She has received honorarium for speaking at American Academy of Neurology courses.
Funding
The author(s) disclosed receipt of the following financial support for the publication (writing and editorial support) of this article: This work was supported by the Miotox, LLC, (grant number NA).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
