Abstract
Introduction
Many studies have found brain atrophy in patients with traumatic brain injury (TBI), but most of those studies examined patients with moderate or severe TBI. A few recent studies in patients with chronic mild or moderate TBI found abnormally large brain volume. Some of these studies used NeuroQuant®, FDA-cleared software for measuring MRI brain volume. It is not known if the abnormal enlargement occurs before or after injury. The purpose of the current study was to test the hypothesis that it occurs after injury.
Methods
55 patients with chronic mild or moderate TBI were compared to NeuroQuant® normal controls (n > 4000) with respect to MRI brain volume change from before injury (time 0 [t0], estimated volume) to after injury (t1, measured volume). A subset of 36 patients were compared to the normal controls with respect to longitudinal change of brain volume after injury from t1 to t2.
Results
The patients had abnormally fast increase of brain volume for multiple brain regions, including whole brain, cerebral cortical gray matter, and subcortical regions.
Discussion
This is the first report of extensive abnormal longitudinal brain volume enlargement in patients with TBI. In particular, the findings suggested that the previously reported findings of cross-sectional brain volume abnormal enlargement were due to longitudinal enlargement after, not before, injury. Abnormal longitudinal enlargement of the posterior cingulate cortex correlated with neuropathic headaches, partially replicating a previously reported finding that was associated with neuroinflammation.
Abbreviations
CWM-cerebral white matter. GM-cerebral cortical gray matter. ICC: intraclass correlations coefficient. IFT-infratentorial. MRI-magnetic resonance imaging. mTBI-mild TBI. NQ-NeuroQuant®. NeuroQuanted MRI-MRI that was used for subsequent NeuroQuant® brain volume analysis. SCN-subcortical nuclei. t0-time of injury. t1-time of first NeuroQuanted MRI scan after injury. t2-time of second NeuroQuanted MRI scan after injury. TBI-traumatic brain injury. VBR-ventricle-to-brain ratio. WBP-whole brain parenchyma.
Introduction
Decades of research have shown that traumatic brain injury (TBI) causes brain atrophy.1–3 However, most of this research was based on patients with moderate or severe TBI. Until recently, many investigators thought that brain volume was normal in patients with mild TBI, due in part to the normal appearance of MRI scans in those patients.4,5However, when MRI brain volume is measured, patients with chronic effects of mild or moderate TBI have been found to have volume abnormalities often6–19 but not always. 20
Although most studies found atrophy, a few studies found abnormal enlargement. To our knowledge, the first study to find abnormal enlargement was based on the volume estimation method, which involved estimating brain volume just before injury, measuring brain volume after injury, and comparing estimated brain volume change between patients and normal control participants. 12 Since then, other studies have found cross-sectional abnormal enlargement or increased cortical thickness.13,15,19 There has been only one previous study that found longitudinal enlargement. 15
It is not known if the abnormal enlargement occurs before or after injury. The purpose of the current study was to test the hypothesis that it occurs after injury. More generally, because longitudinal analyses are fundamentally different from cross-sectional analyses and may be more sensitive to detecting abnormalities, 21 the current study focused on longitudinal analyses. The methods were significantly improved from previous studies, including the following: (1) there was a greater number of patients (55 in the current study versus < 25 in most previous studies; see the Discussion section); (2) there was a greater number of brain regions analyzed (18 brain regions, including whole brain parenchyma, total cerebral cortical gray matter, cerebral white matter, multiple cortical gyral regions, and multiple subcortical regions); and (3) there was extensive use of NeuroQuant®, FDA-cleared software for measuring MRI brain volume. Unlike brain volumetric software typically used in university research settings, e.g. FreeSurfer or FSL, NeuroQuant® is readily available in clinical settings and includes age- and sex-matched data over the lifespan on over 4000 normal control participants (see the Methods section). Thus, research based on NeuroQuant® is more readily applicable in clinical situations than research confined to academic university settings. The current study provides an example of how to leverage that technology in order to better understand the effects of brain injury on TBI patients.
Methods
Participants
Patients
Selection criteria. The sample of patients was expanded from that studied and described in detail previously. 19 In brief, patients included in this study were adult outpatients consecutively admitted to the Virginia Institute of Neuropsychiatry. They were diagnosed with traumatic brain injury according to the criteria of Menon et al. 22 and had a mild or moderate level of brain injury according to the criteria of Silver et al. 23 Each patient had at least one “NeuroQuanted MRI” brain scan, i.e. an MRI that was used for subsequent NeuroQuant® brain volume analysis. They had no pre-injury history of brain disorder that could affect brain volume measurements. The majority of the patients included in the study had mood disorders (e.g. depression, generalized anxiety or irritability) and posttraumatic stress disorder due to the accident or injury. This study was approved by the Sterling Institutional Review Board (approval number 6450) and satisfied the requirements of the Code of Ethics of the World Medical Association (Declaration of Helsinki) for human research. Each patient agreed to be in the study and signed the informed consent form.
Description of patient sample. 55 patients met the selection criteria. Demographic characteristics were as follows: 29 men and 26 women; mean number of years of education was 14.3 (SD 3.0; range 10–21); mean age in years at the time of the injury (t0) was 45.7 (SD 12.3; range 16.9–80.2); mean interval between time of injury and time of first MRI (t0-t1) was 1.47 years (SD 1.11; range 0.11–5.68); mean interval between t1 and t2 MRIs (t1-t2) was 0.62 years (SD 0.27; range 0.22–1.59).
Because all 55 patients had at least 1 MRI brain scan, t0-t1 brain volume analyses (see below for details) were performed for all of them. A subset of patients had 2 MRI brain scans available (N = 36) allowing for t1-t2 longitudinal analyses.
Causes of injury included motor vehicle accident (n = 44), train accident (n = 4), hit to head with object (n = 4), fall down steps (n = 1), mining accident (n = 1), and motor vehicle vs. pedestrian accident (n = 1).
48 patients had mild TBI and 7 patients had moderate TBI. The mean GCS score was 14.7, median 15.0, range 11–15. The mean duration of loss of consciousness was 2.4 minutes, median 0, range 0–30 minutes. The median duration of posttraumatic amnesia was 0.33 hours, range 0–408 hours.
Regarding other neuropsychiatric symptoms due to the brain injury, in general, the sample of patients had a wide range of chronic symptoms including impaired cognition, impaired mood, impaired sleep and wakefulness, posttraumatic stress disorder, and pain, which caused them to seek treatment at a TBI specialty outpatient clinic. The mean score on the Glasgow Outcome Scale-Extended version (GOS-E) 24 was 5.5 (SD = 0.6). The vast majority of patients had either GOS-E = 5 (consistent with Lower Moderate Disability [LMD]) or 6 (consistent with Upper Moderate Disability[UMD]). The main difference between patients with LMD and UMD was that most patients with LMD were unable to work and most patients with UMD were able to work despite some impairment in work ability. 52.9% of the patients were able to return to work (although almost always with difficulty), and 47.1% of the patients were unable to return to work.
Two sets of normal control data
Two sets of normal control data were used for this study: one based on NeuroQuant® software (https://www.cortechslabs.com/products/neuroquant) and the other based on NeuroGage® software (http://www.neurogage.com). These software programs are described in the Brain imaging and volume measurement section, “Brain imaging and volume measurement.” The normal control data associated with them are described in next two subsequent sections, just below. These two sets of data were used for their complementary strengths, described below in the Brain imaging and volume measurement section.
NeuroQuant® normal control data. NeuroQuant® normal control data came from the NeuroQuant® software produced by CorTechs Labs, Inc. (https://www.cortechslabs.com) (San Diego, CA, USA). NeuroQuant® is FDA-cleared software for measuring MRI brain volume and assessing for abnormal brain volume (for further details, see the Brain imaging and volume measurement section, “Brain imaging and volume measurement”).
An advantage of the NeuroQuant® normal control data over most other normal control data sets was that it included a very large number (over 4000) of normal control participants (that number was based on personal communication with Weidong Luo, Senior Scientist, CorTechs Labs Inc., March 23, 2020) collected from a variety of sources. The ages of the NeuroQuant® normal controls ranged from 3 to 100 years. The NeuroQuant® software was designed to compare the MRI brain volume data for each subject to that of a large database of normal controls, matched for age and sex. The large number of normal controls and range of ages allowed for more accurate estimates of normal brain volume throughout the lifespan than most other normal control datasets. In the current study, these features of NeuroQuant® allowed for accurate between-group analyses of brain volume while minimizing the effects of age and sex.
In order to take advantage of these features of NeuroQuant®, each patient and each NeuroGage® normal control (for explanation of “NeuroGage® normal control” see the next section) was analyzed using the NeuroQuant® Triage Brain Atrophy analysis (https://www.cortechslabs.com/products/neuroquant/tba), which created a normative percentile for each brain region after matching the patient’s age and sex to the normal control database, and adjusting for intracranial volume. The Triage Brain Atrophy report includes dozens of brain regions, including cortical gray matter, cerebral white matter, multiple cortical gyral regions, basal ganglia and other subcortical regions, brainstem and cerebellum.
For the current study, the normative percentiles were converted to Z scores in order to allow the use of parametric statistics. Because the normative percentiles--and in turn, the Z scores--were based on a procedure that minimized the effects of age, sex and intracranial volume on brain volume (as described in the NeuroQuant® cross-sectional analyses section below), it was not necessary to compare patient and normal control volume data--or further adjust the volume data--with respect to those variables.
NeuroQuant® does not provide information about level of education for its normal controls. Regarding potential effects of education on brain volume, the mean years of education of the patients (n = 55) was 14.3 (SD = 3.0), which was similar to the mean years of education for the U.S.A. population in 2012 (13.3 years; http://data.uis.unesco.org/index.aspx?queryid = 242), which was considered to be the best estimate of the mean years of education of the NeuroQuant® normal control group.
NeuroGage® normal control data. Despite the multiple advantages of NeuroQuant’s normal control data, it did not provide data on variability (SD) of longitudinal volume change. Therefore, a second set of normal control data, based on NeuroGage® software, was used to measure normal variability of volume change. The NeuroGage® normal control participants are described in this section, and the use of NeuroGage® normal control data to measure variability is described in the Brain imaging and volume measurement section, “Brain imaging and volume measurement.”
The NeuroGage® 2.0 normal control participants were described previously. 25 The group included 80 normal control participants whose MRI brain scans were obtained from a larger group previously studied as part of the Alzheimer’s Disease Neuroimaging Initiative (ADNI).26–28 The ADNI normal control subjects were selected to be healthy and free of cognitive and other medical problems that would affect brain volume measurement. The ADNI MRI data have been used in numerous peer-reviewed publications; a search of the term “ADNI” in PubMed returned 1534 publications. For more details about ADNI, see the cited references.26–28
The NeuroGage® normal control group consisted of 80 participants (N = 40 women and 40 men; mean age (in years) = 68.4, SD 3.2, median 70, range 60–72. The NeuroGage® normal controls were significantly older (mean = 68.4 years, SD = 3.2) than the patients (mean = 47.2, SD = 12.3)(t = -12.56, df = 59.2, p < .0001). Because the NeuroGage® normal control data were used only to measure variance of change of brain volume, and since that was likely to increase with age, the older age of the normals controls likely led to less ability to find statistically significant differences between the patient and normal control groups, a conservative limitation. The mean interval between t1 and t2 MRIs (t1-t2) was 1.06 years (SD 0.07; range 0.82–1.29).
Brain imaging and volume measurement
Magnetic resonance imaging
Each participant had one or more MRI brain scans at one of various radiology centers using the scanning protocol recommended for allowing later NeuroQuant® analysis; this protocol is described in detail on the NeuroQuant® website (http://www.cortechs.net/products/neuroquant.php). In addition to the general requirements for having an MRI (e.g. having no magnetic metal in the body), the NeuroQuant® protocol required, at a minimum, the following:
MRI scanner which supported the NeuroQuant® scanning protocol MRI scanning protocol based on the ADNI scanning protocol T1-weighted timing sequence Non-contrast Sagittal 3D
All patients had 3.0 Tesla scans, instead of 1.5 Tesla scans, in order to have higher scan resolution for clinical purposes. Normal control participants from the NeuroQuant® and NeuroGage® normal control groups were scanned with either 1.5 T or 3.0 T scanners.
NeuroQuant® is FDA-cleared to be used on a variety of scanners, using 1.5 T or 3.0 T magnets, indicating good reliablity between scanners and magnet strengths for the volume measurements (https://www.cortechslabs.com/resources/technical-information/recommended-scanner-settings).
For longitudinal analyses, patients and NeuroGage® normal controls had a second MRI brain scan performed on the same scanner as their first scan in order to eliminate within-subject interscanner differences.
NeuroQuant® software was used for cross-sectional brain volume analysis
All cross-sectional MRI brain volumes were measured using NeuroQuant®, FDA-cleared software for measuring MRI brain volume in humans. In addition to its FDA clearance, the reliability and validity of NeuroQuant® has been supported by over 91 peer-reviewed publications (supplemental online file “All NQ pubs_COPY” https://www.dropbox.com/sh/n96hsfrgu4lg0iv/AAC-rgKKrMdibBxSfT7KE4cGa?dl = 0). These include multiple studies that found that NeuroQuant® is reliable for measuring brain volume in normal subjects, patients with TBI, and other neuropsychiatric patients8,29–35 and valid for assessing brain volume in patients with TBI.4,5,8,12,16,19,25,33,35–41
Neuroquant® automated brain MRI segmentation. The brain MRI grayscale images for each patient and NeuroGage® normal control® were uploaded to the NeuroQuant® server, which processed and analyzed the brain imaging data. The output of the NeuroQuant® computer-automated analysis included one or more reports that contained volumetric information, and a set of segmented (colored) brain images that were visually inspected for errors by one of the authors (D.E.R), in order to ensure accurate identification of brain regions by the software, using our previously described quality control procedure. 42 For the normal controls, there were no segmentation errors. For the patients, 1.5% of brain regions were judged to have been identified inaccurately by NeuroQuant®, and therefore those data were omitted from subsequent analyses. Therefore, 98.5% of brain volume data were included for the patients, including the large majority of brain volume data for each patient.
NeuroQuant® cross-sectional analyses. All patients were analyzed using the NeuroQuant® 2.3 Triage Brain Atrophy report (https://www.cortechslabs.com/products/neuroquant/tba), which included 44 brain regions, including cortical gray matter, cerebral white matter, basal ganglia, infratentorial regions, and numerous cortical gray matter regions. These reports provided normative percentiles for each cross-sectional brain volume after adjusting the volumes for intracranial volume (by dividing by intracranial volume), matching for age (by comparing the subject’s volume to normal controls of the same age) and matching for sex (by comparing the volume to normal controls of the same sex). The volume data were averaged over left and right-sided counterpart brain regions.
All NeuroGage® normal controls were analyzed using the NeuroQuant® 2.0 Triage Brain Atrophy report. The reliability between the NeuroQuant® 2.0 and 2.3 (used for patients) software versions were assessed and found to be excellent for all brain volumes (see Reliability section). Therefore, results from the two software versions were considered to be interchangeable and both were used for the current study.
NeuroGage® longitudinal brain volume analyses
NeuroGage® is software designed to extend the utility of NeuroQuant® in several ways, including by measuring volume change over time and estimating volume just before injury.
Brain volume longitudinal analyses were performed using NeuroGage® software per our previously published procedures.5,8,12,25,38 These procedures are briefly summarized below, and updated aspects of the procedures are explained in detail.
Two types of longitudinal brain volume analyses were performed: t0-t1 and t1-t2 analyses, where t0 = time just before injury, t1 = time of first NeuroQuanted MRI, and t2 = time of second NeuroQuanted MRI.
t0-t1 longitudinal analyses. For t0-t1 analyses, brain volume was estimated at t0 using our previously published brain volume estimation method,12,16 and volume was measured at t1. The volume estimation method is based on 2 previous findings by many investigators, as follows: 1) brain volume changes at predictable rates over the lifespan; and 2) intracranial volume does not change during adulthood (for a summary of this literature, see 12 ). Based on these two principles, normal brain volume at any point during adult life is a predictable percentage of intracranial volume and therefore can be estimated reliably by using the subject’s age at that time and their intracranial volume measured at any point during adulthood.
For the current study, brain volume was estimated at t0 by using the patient’s intracranial volume at t1 (measured with NeuroQuant®), age at t0, sex, and known normal relationships between these variables over the lifespan. In contrast to our previous study,12,16 in order to predict brain volume at a given age, we used the NeuroQuant® normal control data instead of the normal control data from Hedman and colleagues 43 for the following reasons: 1) unlike the Hedman data, the NeuroQuant® data were FDA-cleared; and 2) the Hedman data were not measured with NeuroQuant®, and therefore it was a more consistent approach to use NeuroQuant® for the volume estimation method in addition to the other aspects of the current study. The NeuroQuant® normal control data that were used to predict brain volume over the lifespan were obtained by digitizing graphs of volume versus age obtained from NeuroQuant® reports. An example of the volume estimation method applied to a single patient for t0-t1 calculations is depicted in Figure 1.
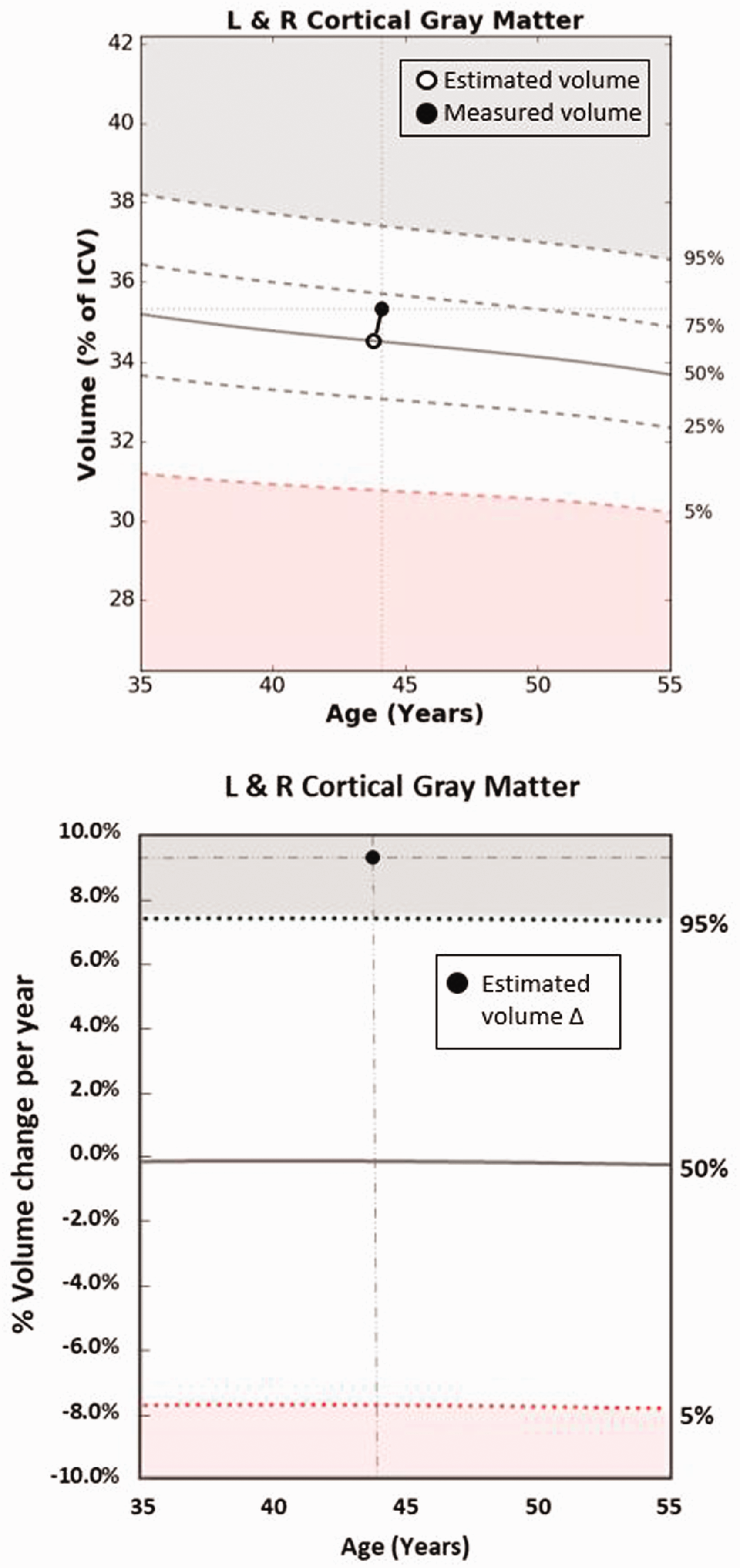
Example of application of t0-t1 brain volume estimation method in female patient who had TBI at 43.9 years of age. Top: At t0 (time of injury, indicated by open circle), her cortical gray matter volume was estimated to be equal to the mean of normal women her age (based on the NeuroQuant® normal control data). At t1 (time of first NeuroQuanted MRI, indicated by solid circle), she was 44.1 years old, and her volume was measured and found to be increased from the pre-injury estimate. This top figure is based on the NeuroQuant® report, except for the open circle and boxed key, which was added by the authors. Bottom: NeuroGage® analysis showed that her estimated rate of brain volume increase was 9.4% per year from t0-t1 (solid circle, plotted at the midpoint of the interscan interval, age 44.0). The normal mean % change of volume per year (50th normative %tile function in this bottom figure) was determined by differentiating the mean volume (50th normative %tile function in the top figure) with respect to time and was found to be −0.26%/year at age 44.0 years. The normal standard deviation of % volume change per year was calculated to be 4.61% per year, based on the NeuroGage® normal control data. The 95th and 5th normative percentiles were calculated as the mean % volume change per year +/− 1.6449 standard deviations respectively. A Z-score was calculated as the patient’s data (9.4%/year) minus the normal mean (−0.26%/year), the quantity divided by the normal SD (4.61%/year), which equalled 2.1, corresponding to the 98.3 normative %tile. Her Z score of 2.1 was used in the group analyses.
In our earlier study, 12 which was based on the first generation of NeuroQuant® software, the reliability of the volume estimation method was found to be good for 4 relatively large brain regions: 1) whole brain parenchyma (WBP), 2) cerebral cortical gray matter (GM), 3) cerebral white matter (CWM), and 4) subcortical nuclei (including basal ganglia, thalamus, hippocampus and amygdala) + infratentorial regions (including brainstem and cerebellum) (SCN+IFT) (for illustration of the 4 brain regions, see p. 571, Figure 6 of 12 ). The volume of GM + CWM + SCN+IFT equalled the volume of WBP. In that earlier study, reliability was tested in the NeuroGage® normal control group (N = 20) by comparing measured brain volumes to estimated brain volumes using intraclass correlation coefficients (ICCs).
In the current study, reliability of the volume estimation method was tested in a similar fashion but instead using the current 80 NeuroGage® normal controls (for details of statistical methods for testing reliability, see the next section). Reliability again was found to be good for the 4 brain regions (WBP, GM, CWM, and SCN+IFT) (for details, see the Reliability section).
For brain volume change from t0-t1, for both patients and normal controls, brain volume was estimated at t0 and measured at t1 (for normal controls, because they were not injured, there actually was no t0 data; so their t1 data served as a proxy for t0 date of injury data, and their t2 data served as a proxy for t1 data regarding date of injury and volume measurement). For details about this procedure see. 16
Accordingly, brain volume change was estimated from time 0 (t0) to time 1 (t1). Annualized percentage change of brain volume was calculated using the standard approach as follows: 1) calculate Volume Change as the change from t0 to t1 = [(Volume at t0)-(Volume at t1)]; 2) calculate Proportion Change = (Volume Change/Volume at t0); 3) calculate Percentage Change = (Proportion Change × 100%); and 4) calculate Percentage Change per year = [Percentage Change/(t0-t1 interval measured in years)].
For each patient, in order to adjust for the effects of age and sex on annualized percentage change of brain volume, each patient’s volume change was compared to the corresponding age- and sex-matched NeuroQuant® normal control group data. This method involved converting each patient’s data to a Z score as follows: 1) Subtract the patient’s value from the mean of the age-matched and sex-matched NeuroQuant® normal control group data; and 2) divide the result by the standard deviation (SD) of t0-t1 volume change values based on the NeuroGage® normal control data. Regarding the latter step, the SD of t0-t1 values based on the NeuroGage® normal control data were used as an estimate of the SD of t0-t1 values based on the NeuroQuant® normal control data, since the latter were not available; this was similar to our previously published approach. 12
t1-t2 analyses. For the t1-t2 longitudinal analyses, brain volume change and Z score calculations were performed using the same methods that were used for the t0-t1 analyses, except that volume was measured--not estimated—at both time points (t1 and t2). 18 brain regions had NeuroQuant® normal control data available throughout the adult life span allowing t1-t2 analyses for those regions. An example of the longitudinal analysis method applied to a single patient for t1-t2 calculations is depicted in Figure 2.
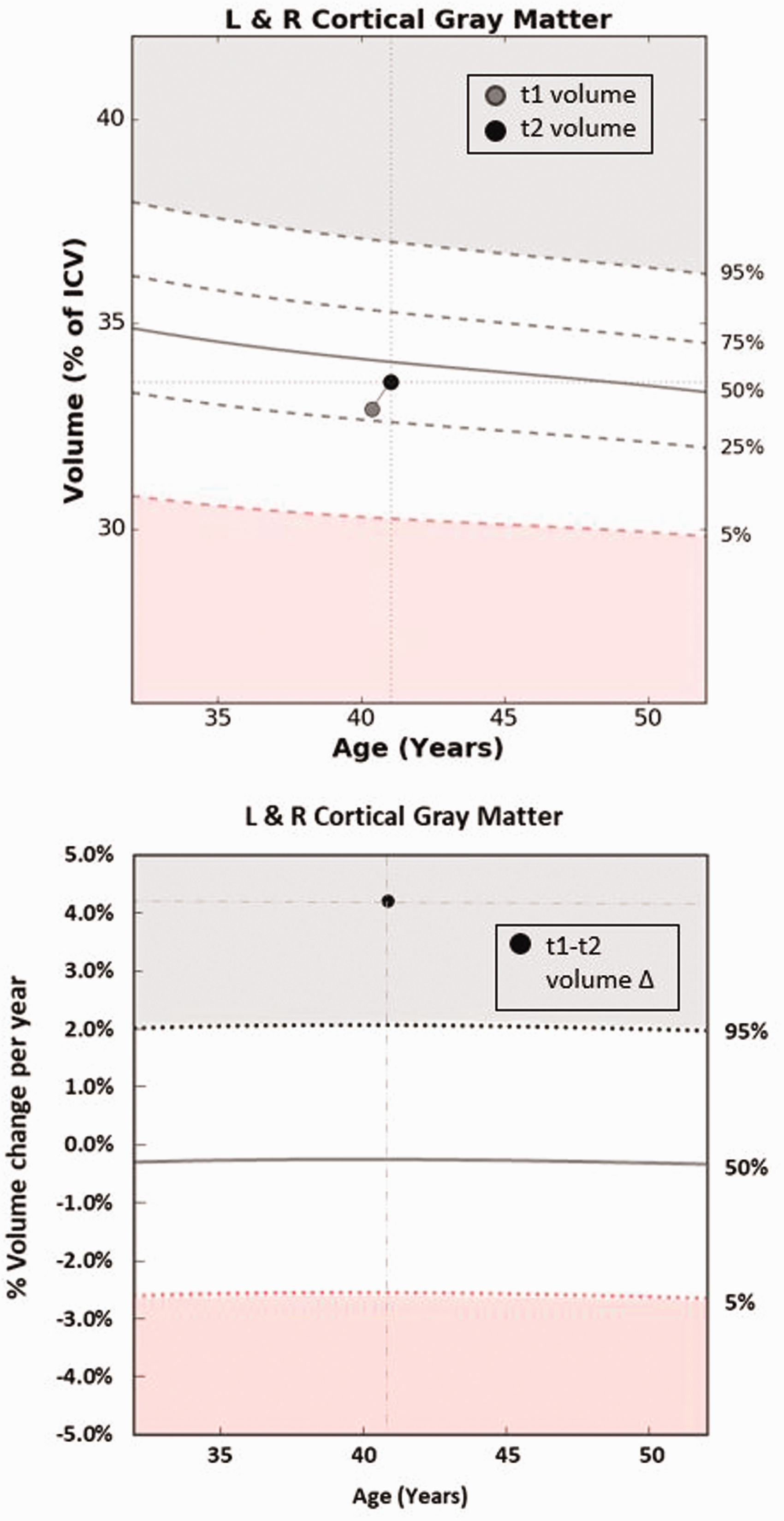
Example of application of t1-t2 brain volume change method in male TBI patient. Top: At t1 (time of first NeuroQuanted MRI, indicated by gray circle), he was 40.4 years old, and his cortical gray matter volume was normal compared to normal men his age, based on the NeuroQuant® normal control data. At t2 (indicated by black circle), he was 41.0 years old, and his volume remained normal but had increased from t1. It was not possible to tell from these two cross-sectional volume measurements if the volume increased at an abnormally fast rate. This top figure is based on the NeuroQuant® report, except for the boxed key, which was added by the authors. Bottom: NeuroGage® analysis showed that his rate of volume increase was 4.2% per year from t1-t2 (solid circle, plotted at the midpoint of the interscan interval, age 40.7). The normal mean % change of brain volume per year (50th normative %tile function in this bottom figure) was determined by differentiating the mean brain volume (50th normative %tile function in the top figure) with respect to time and was found to be −0.25%/year at age 40.7 years. The normal standard deviation of % change of brain volume per year was calculated to be 1.41% per year, based on the NeuroGage® normal control data. The 95th and 5th normative percentiles were calculated as the mean % volume change per year +/− 1.6449 standard deviations respectively. A Z-score was calculated as the patient’s data (4.2%/year) minus the normal mean (−0.25%/year), the quantity divided by the normal SD (1.41%/year), which equalled 3.2, corresponding to the 99.9 normative %tile. His Z score of 3.2 was used in the group analyses.
Statistical analyses
Inspection of distributions of data
Distributions of data were inspected for outliers and distributional characteristics. All the t0-t1, t1-t2, t1 and t2 data were at least approximately normally distributed based on visual inspection of the histograms; therefore, parametric statistics were used.
Testing reliability
Test-retest reliability for NeuroQuant® software versions 2.0 and 2.3 was assessed in the NeuroGage® normal controls using our previously described methods.8,33
The reliability of the volume estimation method was assessed by comparing estimated to measured brain volumes in the normal controls, as described above in the t0-t1 longitudinal analyses section.
Reliabilities were calculated for each brain region using intraclass correlations coefficients (ICCs; 3, 1) based on the terminology of Shrout and Fleiss 44 using SPSS version 25 (Model = 2-Way Mixed; Type = Absolute agreement; Confidence Interval = 95%). To interpret ICC values, the following guidelines were used: ICC < 0.5 indicated poor reliability, 0.5 < ICC < 0.75 indicated moderate reliability, 0.75 < ICC < 0.9 indicated good reliability, and ICC > 0.9 indicated excellent reliability. 45
Comparisons between groups
Two-tailed Z tests were used for all group comparisons, with the NeuroQuant® normal control group expected by design to have a mean of 0 and SD of 1 for each brain region volume.
Results were reported with and without adjustments for multiple comparisons. Such adjustments reduce Type 1 errors (false positive findings) at the expense of increasing Type 2 statistical errors (false negative findings), and for this and other reasons previous authors have argued against using such adjustments. 46 For the current study, Type 2 errors were judged to be at least as important as Type 1 errors. However, other investigators believe that some adjustment is needed to control the risk of Type 1 error. In the current study, we used the following approach: 1) results were reported with Bonferroni adjustments; and 2) also results were reported without adjustments, and calculations regarding the number of positive findings expected by chance alone were provided with each set of unadjusted analyses in order for providing a framework for interpreting the risk of Type 1 error. Accordingly, results associated with significant unadjusted P values but nonsignificant adjusted P values should be interpreted with more caution.
Statistical software
JMP Pro version 14.0.0 was used to perform all statistical analyses except the intraclass correlations, which were performed using SPSS version 25.
Results
Reliability
ICCs comparing NeuroQuant® 2.0 to 2.3 software versions ranged from 0.95 to 1.00, showing excellent reliability for all brain regions (online supplementary file “ICCs for NQ 2.0 v 2.3” https://www.dropbox.com/sh/n96hsfrgu4lg0iv/AAC-rgKKrMdibBxSfT7KE4cGa?dl = 0). Therefore, the volume results from either software version were considered to be interchangeable for the purposes of the current study.
ICCs comparing measured brain volumes to estimated brain volumes showed that reliabilities ranged from good to excellent: WBP (.92), GM (.85), CWM (.77), and SCN+IFT (.77) (see figures in online supplementary file “Reliability of vol estim method” https://www.dropbox.com/sh/n96hsfrgu4lg0iv/AAC-rgKKrMdibBxSfT7KE4cGa?dl = 0).
Comparing patients to normal controls for t0-t1 volume change
The groups of patients and normal controls were compared with respect to estimated change of brain volume from time 0 (t0) to time 1 (t1) (Table 1). The patients had more regions of abnormal t0-t1 volume change (4 of 4 regions with P < .05) than would have been expected by chance alone (5% abnormally small + 5% abnormally large = 10% abnormal; 10%x4 regions = 0.4 regions). Cerebral white matter atrophied abnormally fast, and the other 3 regions enlarged abnormally fast. 2 of the 4 regions survived Bonferroni corrections.
Comparisons of brain volume changes from time 0 (t0) to time 1 (t1) between patients and NeuroQuant® normal controls (converted to Z scores), where t0 was the time just before injury, and t1 was the time of the first NeuroQuanted MRI.
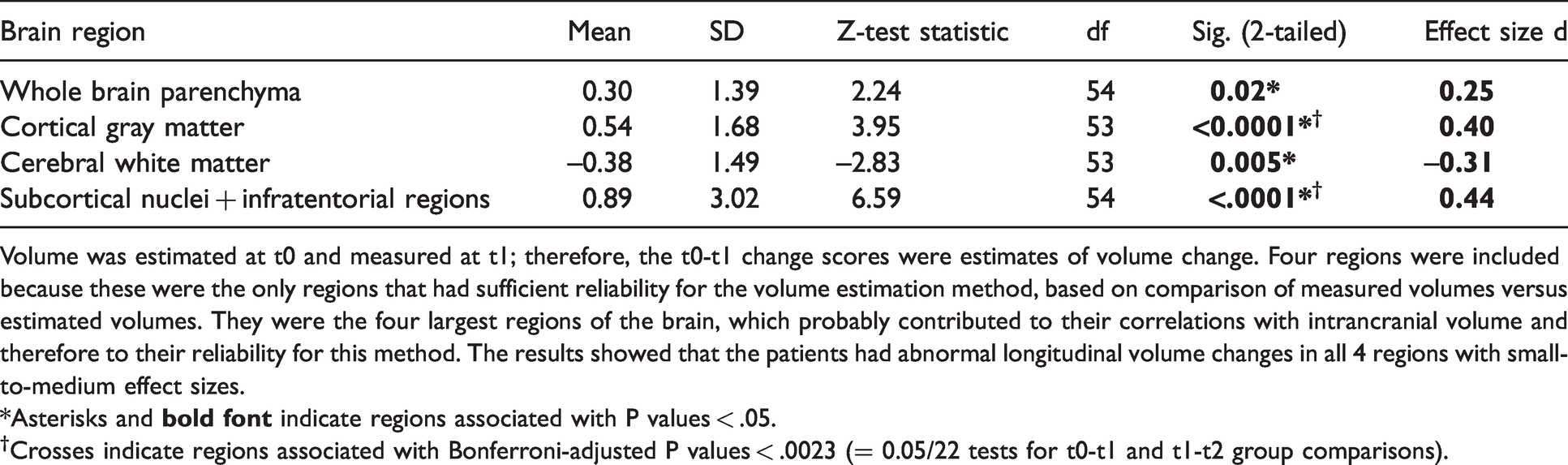
Volume was estimated at t0 and measured at t1; therefore, the t0-t1 change scores were estimates of volume change. Four regions were included because these were the only regions that had sufficient reliability for the volume estimation method, based on comparison of measured volumes versus estimated volumes. They were the four largest regions of the brain, which probably contributed to their correlations with intrancranial volume and therefore to their reliability for this method. The results showed that the patients had abnormal longitudinal volume changes in all 4 regions with small-to-medium effect sizes.
*Asterisks and
†Crosses indicate regions associated with Bonferroni-adjusted P values < .0023 (= 0.05/22 tests for t0-t1 and t1-t2 group comparisons).
Comparing patients to normal controls for t1-t2 volume change
The groups of patients and normal controls were compared with respect to change of brain volume from t1 to t2 (Table 2). The patients had more regions of abnormal t1-t2 volume change (9 of 18 regions with P < .05) than would have been expected by chance alone (5% abnormally small + 5% abnormally large = 10% abnormal; 10%x18 regions = 1.8 regions). All abnormal volume changes involved longitudinal parenchymal enlargement (or similarly, ventricular diminution). 6 of 9 regions survived Bonferroni correction.
Comparisons of brain volume changes from time 1 (t1) to time 2 (t2) between patients and NeuroQuant® normal controls (converted to Z scores), where t1 was the time of the first NeuroQuanted MRI, and t2 was the time of the second NeuroQuanted MRI.
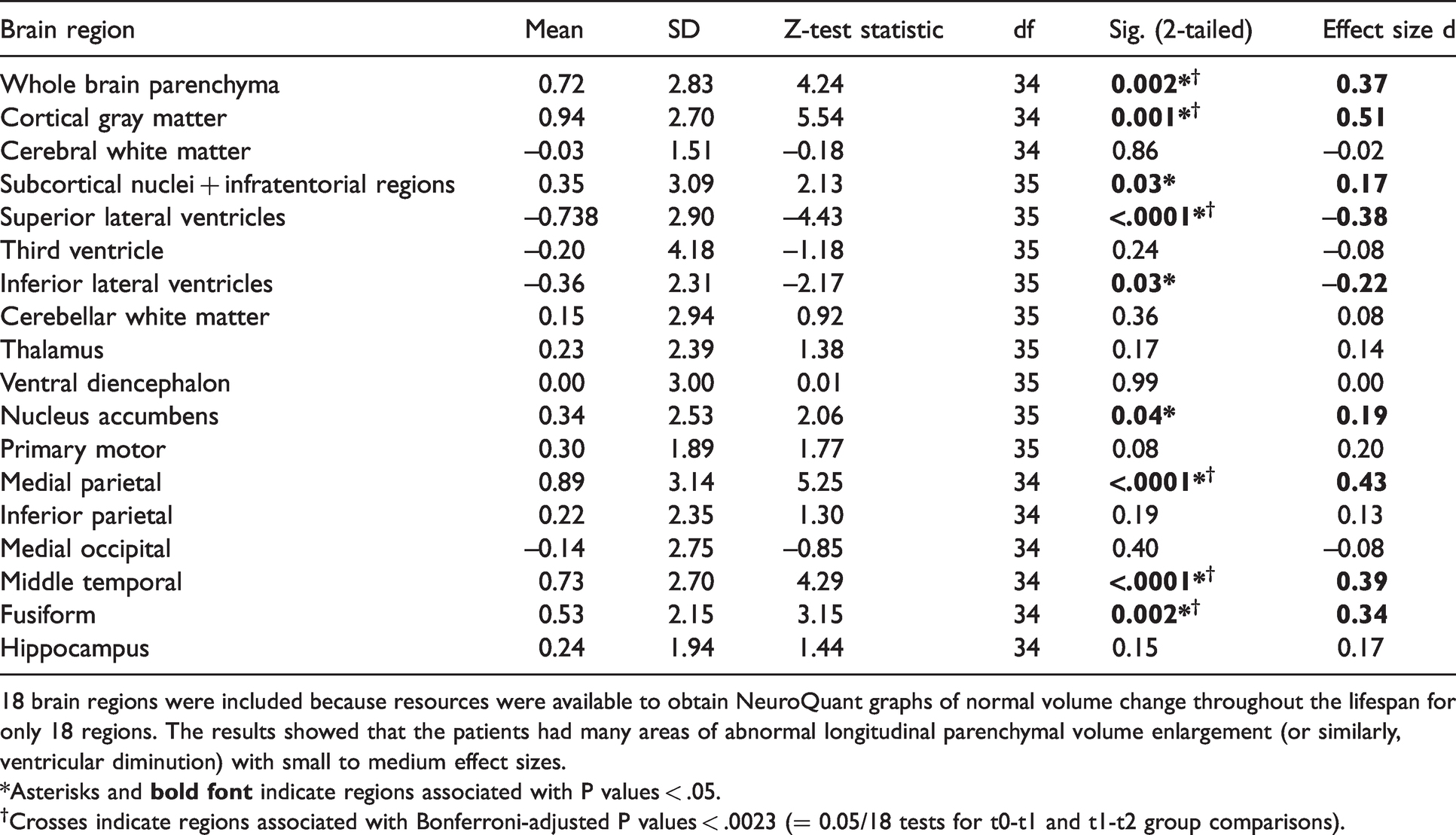
18 brain regions were included because resources were available to obtain NeuroQuant graphs of normal volume change throughout the lifespan for only 18 regions. The results showed that the patients had many areas of abnormal longitudinal parenchymal volume enlargement (or similarly, ventricular diminution) with small to medium effect sizes.
*Asterisks and
†Crosses indicate regions associated with Bonferroni-adjusted P values < .0023 (= 0.05/18 tests for t0-t1 and t1-t2 group comparisons).
Graphs of volume change (t0-t1 and t1-t2) versus time after injury are shown in Figures 3 to 5.
Comparing patients to normal controls for t2 volume
The groups of patients and NeuroQuant® normal controls were compared with respect to cross-sectional brain volume at t2, and the results are shown in Table 3. (Recently we reported a similar analysis for t1 data 19 ). The patients had more regions of abnormal t2 volume (25 of 39 regions with P < .05) than would have been expected by chance alone (5% abnormally small + 5% abnormally large = 10% abnormal; 10% × 39 regions = 3.9 regions). 22 of the regions were abnormally large, and 3 regions were abnormally small. Sixteen of the 25 regions survived Bonferroni correction.
Comparisons of cross-sectional brain volumes at time 2 (t2) between patients and NeuroQuant® normal controls (converted to Z scores), where t2 was the time of the second NeuroQuanted MRI.
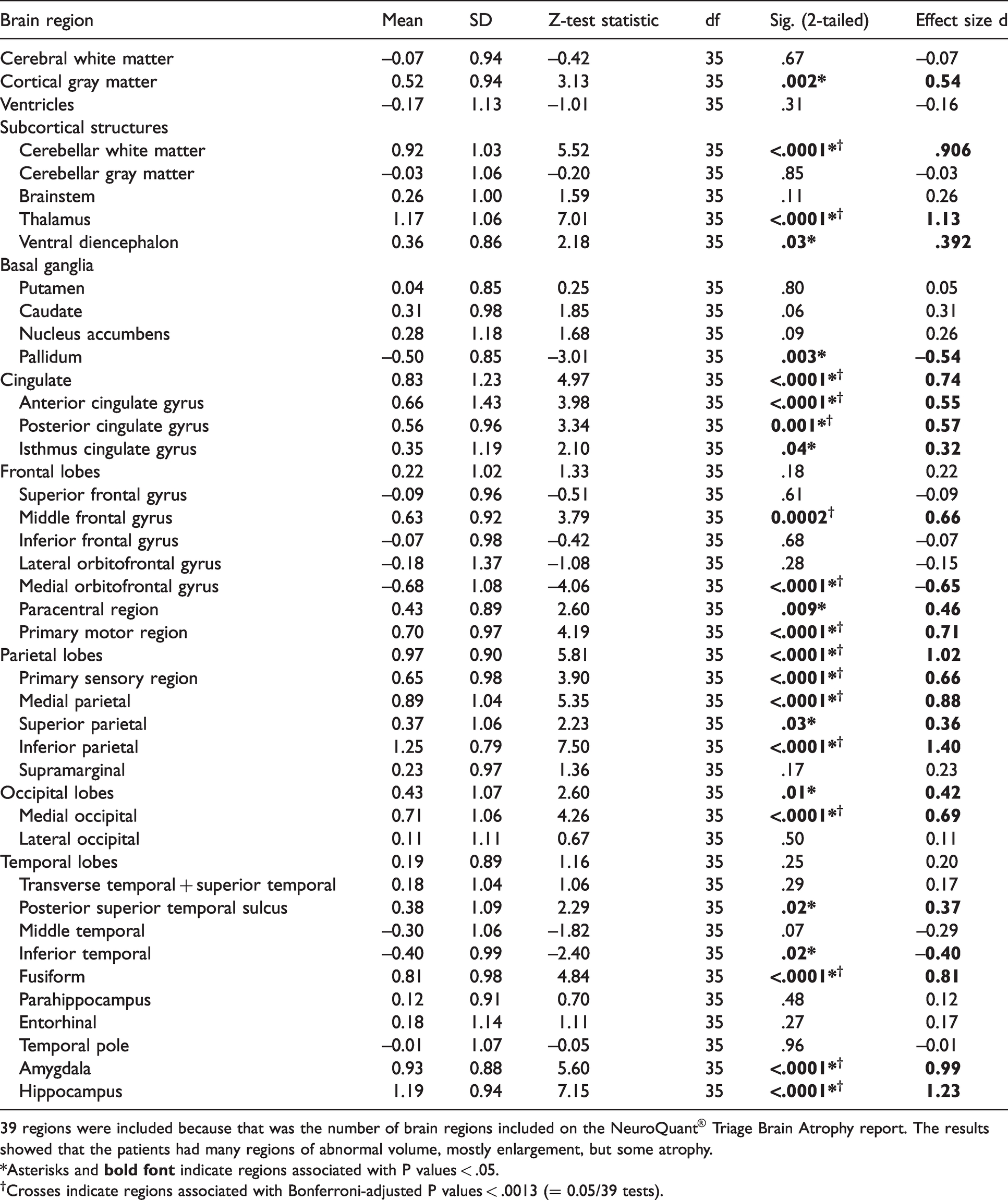
39 regions were included because that was the number of brain regions included on the NeuroQuant® Triage Brain Atrophy report. The results showed that the patients had many regions of abnormal volume, mostly enlargement, but some atrophy.
*Asterisks and
†Crosses indicate regions associated with Bonferroni-adjusted P values < .0013 (= 0.05/39 tests).
Discussion
Main findings
The main findings of this study were that patients with chronic mild or moderate TBI had abnormally fast longitudinal enlargement of multiple brain regions, including cerebral cortical gray matter, subcortical nuclei + infratentorial regions (SCN+IFT), and several cortical gyral regions.
Abnormal longitudinal brain volume changes
Summary of longitudinal findings
More specifically, the brain volume estimation method was used to estimate volume at t0 (just before injury) and compare it to volume measured at t1 (after injury). Based on this method, cortical gray matter and SCN+IFT enlarged abnormally fast. The latter two findings constituted a partial replication of our previous findings, which were based on a subsample of the current patients.12,16 Longitudinal enlargement of SCN+IFT from t0-t1, found in the current study, correlated anatomically with the previously reported t1 cross-sectional enlargement of several of the subcomponents of SCN+IFT, including cerebellar white matter, thalamus, ventral diencephalon, caudate nucleus, amygdala and hippocampus. 19
Cerebral white matter appeared to atrophy during the t0-t1 interval, with a small to medium effect size (d = -0.31). However, the associated statistical test of inference (with P = .005) did not survive Bonferroni adjustment (adjusted alpha = .0023), raising a question about whether it was due to a Type 1 statistical error. Was there evidence to suggest that it was a valid finding? Figure 4 showed that, if there was atrophy, it occurred mostly within the first year after injury. A post hoc exploratory analysis showed that cerebral white matter atrophied in the subset of patients scanned within 1 year after injury (N = 21 patients, mean Z score = -0.87, SD = 2.25, Z-test statistic = -3.97, df = 20, P < .0001, effect size d = -.53). In our previous study, which was based on a subset of the current sample of patients, we presented evidence that there was cross-sectional atrophy of cerebral white mattter, especially within the first year after injury 19 (p. 14). More generally, there have been previous reports of longitudinal cerebral white matter atrophy in patients with chronic mild TBI 9 and chronic moderate or severe TBI (for reviews, see1,2) Overall, in patients with chronic mild or moderate TBI, it seemed likely that there was longitudinal cerebral white matter atrophy within the first year of injury (but not after the first year), and that finding should be considered preliminary.
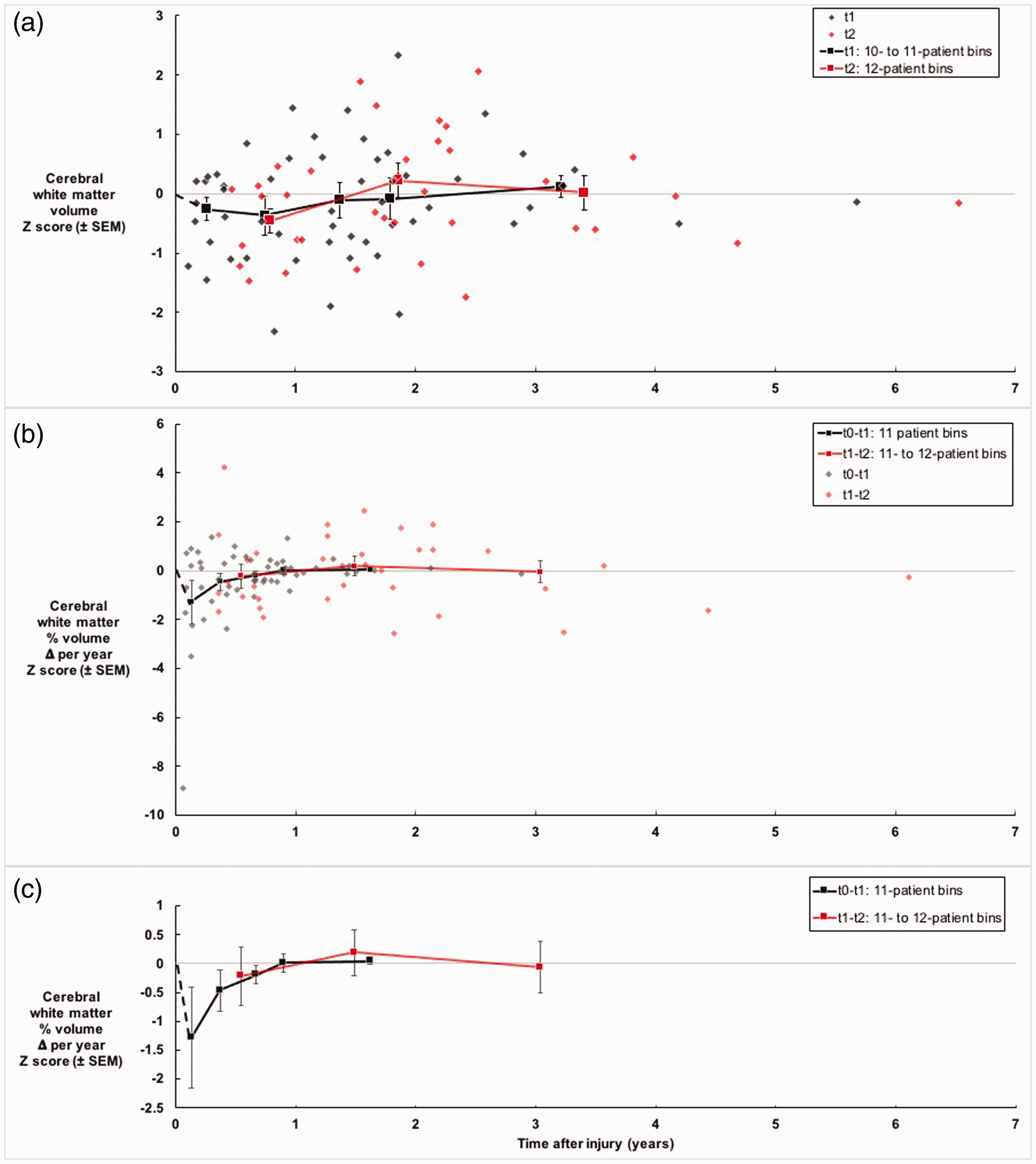
Cerebral white matter volume (measured at t1 and t2) and volume change (measured from t0-t1 and t1-t2) versus time after injury. (a) Volumes (t1 and t2) reached small-to-medium effect sizes (Z scores around -0.4) within 10 months after injury and returned to normal over the next 2 years. (b) Volume changes (t0-t1 [estimated] and t1-t2 [measured]) reached large effect sizes (Z scores around -1.2) within a couple months after injury and returned to normal over the next two years, indicating that volume was atrophying through the first year but had stopped atrophying by the end of the first year after injury. (c) Zoom-in on the trendlines from section (b).
Traditional longitudinal measurements (t1-t2 change), based on measuring—not estimating--t1 and t2 volumes, showed abnormal enlargement of whole brain parenchyma, cortical gray matter, and several cortical gray matter subregions, including the medial parietal cortex, middle temporal gyrus and fusiform gyrus. Similarly, there was progressive diminution of the superior lateral ventricles, consistent with longitudinal enlargement of adjacent parenchymal regions.
Examination of the relationships between longitudinal volume changes and time after injury (Figures 1 to 3) showed different patterns for the 3 major subdivisions of the brain as defined in this study: 1) for cortical gray matter, volume enlarged rapidly within the first few months after injury and continued to enlarge until approximately 3 years after injury; 2) for cerebral white matter, volume decreased rapidly within the first few months after injury then tended to plateau over the next couple years; and 3) for SCN+IFT, volume enlarged rapidly within the first few months after injury but began decreasing around 2 or 3 years after injury.
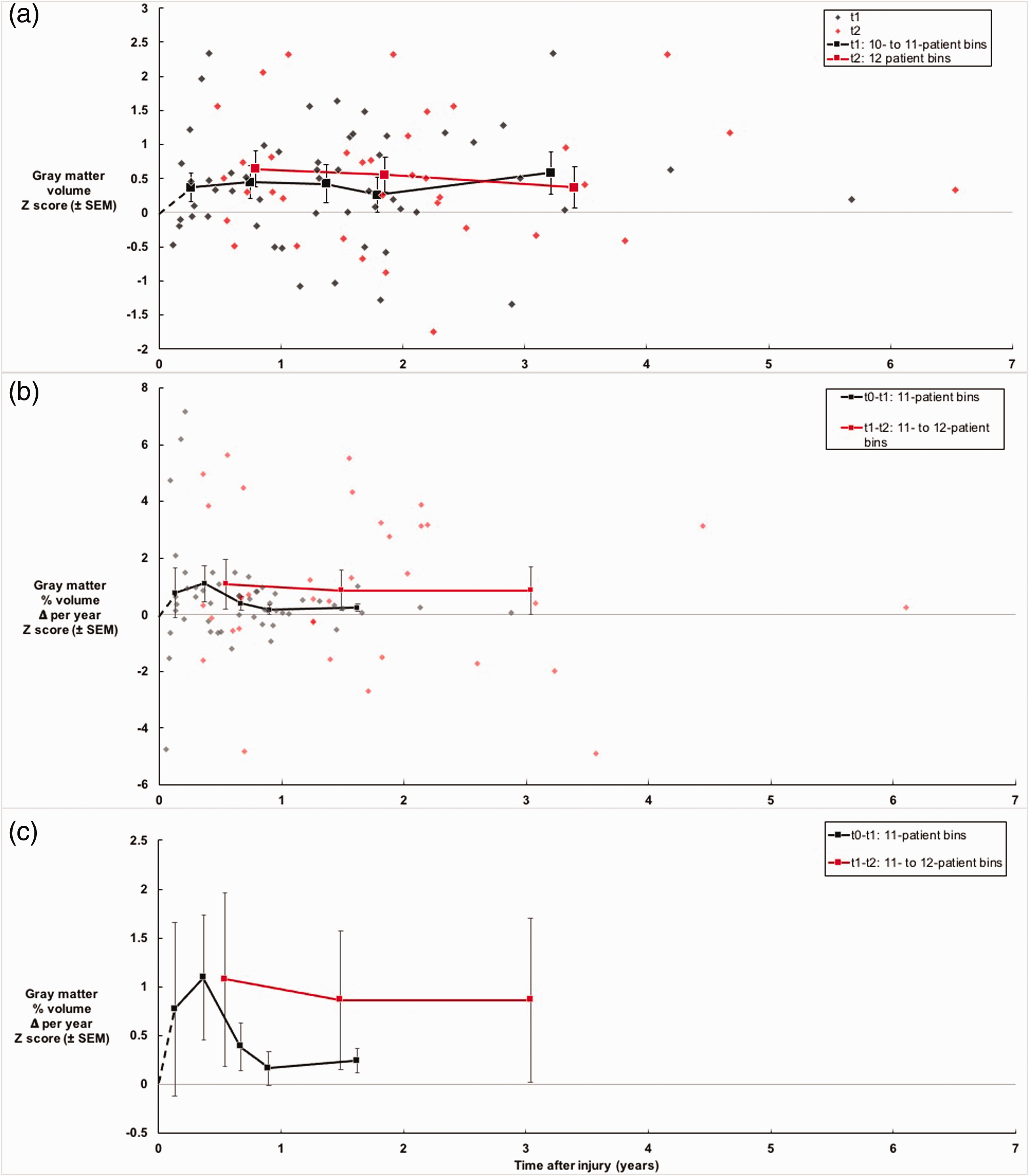
Cortical gray matter volume (measured at t1 and t2) and volume change (measured from t0-t1 and t1-t2) versus time after injury. The following comments about sections (a) through (c) apply to Figures 3 through 5. For sections (a) and (b), patient data are shown as individual data points and averaged over bins to examine trends over time. Section (c) is a zoom-in on the trendlines from section (b), achieved by omitting the individual data points. Because the patients were screened to exclude those who had pre-injury disorders affecting brain volume, it was likely that they had normal brain volume or volume change (Z score = 0) before injury (at t0). Therefore, for each graph, a theoretical line was drawn from the point (x = 0, volume Z score =0) to the first data point based on the average of measured data, and the line was dashed to indicate that it was a theoretical trendline. (a) Volumes (t1 and t2) reached medium effect sizes (Z scores around 0.5) within a few months after injury and plateaued for over 2 years. (b) Volume changes (t0-t1 [estimated] and t1-t2 [measured]) reached large effect sizes (Z scores around 1) within a couple months after injury and decreased partially for the next two years (Z scores averaging around 0.6), indicating that volume was enlarging at a rapid pace through the first year but at a more moderate pace during the second and third years after injury. (c) Zoom-in on the trendlines from section (b).
Literature review
Although there have been many reports of longitudinal brain atrophy in patients with moderate to severe TBI (for review, see 1–3,21), the pattern of volume abnormalities in mild to moderate TBI is less clear. Table 4 shows a brief review of 9 previous longitudinal studies that were similar to the current study; that is, they involved adult patients with mild or moderate TBI, measured brain volume or cortical thickness, and involved mechanisms of injury such as motor vehicle accidents and falls, not military or sports injuries.
Review of previous studies of MRI brain volume in patients with chronic mild or moderate TBI.

Only studies similar to the current study were reviewed. See the Discussion section for discussion. Key: mTBI-mild TBI; VBR-ventricle-to-brain ratio.
The finding of abnormally fast enlargement of whole brain parenchymal volume has not been reported previously and stands in contrast to reports of longitudinal atrophy by our group and others.6–8,12 However, the study by Hofman et al. had no control group (so whole brain volume decreased significantly, but it was not clear if the rate of decrease was abnormally fast); and the study by MacKenzie et al. had only 7 patients and 4 normal controls. It was not clear why our current study differed from our previous studies with respect to this finding, but possibilities include improved methods of our current study as follows: 1) much larger sample sizes; 2) a later, improved version of NeuroQuant® software (software version 2 versus software version 1); 3) use of age-matched normal control data in the current study but not in the earlier ones; 4) increased levels of treatment and rehabilitation in our patients over the years possibly led to greater brain volume enlargement. 47
The finding of abnormally fast increase of total cerebral cortical gray matter volume from t0-t1 (with an effect size of 0.4) was consistent with our previous finding, 12 which also found an effect size of 0.4; but the effect size in the previous study was not associated with a significant result for the associated test of statistical inference, which in retrospect probably was due to lower power than in the current study. The findings of enlargement of cortical gray matter for t0-t1 and t1-t2 in the current study generally was consistent with previous findings of longitudinal thickening of multiple cortical gray matter regions. 15 However, two other studies found longitudinal atrophy (decreased volume or cortical thickness) of cortical gray matter regions.9,13The reasons for the different findings were not clear but could have been due to multiple differences in methods.
The finding of abnormally fast cerebral white matter atrophy was a partial replication of our previous studies, which was based on a smaller subset of the current sample of patients.8,12,16 Atrophy of white matter in cingulate regions was reported by Zhou et al. 9 No other studies of patients similar to ours reported cerebral white matter atrophy, but most of the other studies measured whole brain parenchymal volume or cortical gray matter volume (or thickness) and not cerebral white matter.
The finding of abnormally fast increase of SCN+IFT also was a partial replication of our previous studies.8,12 The finding generally was inconsistent with the previously reported findings of longitudinal atrophy of the caudate, putamen and amygdala, 17 although SCN+IFT includes many regions other than just the ones reported by Zagorchev et al.
In summary, our literature review of longitudinal studies of MRI brain volume in patients similar to ours (including our previous studies) found that few studies existed, and the results generally were mixed. The most agreement was found for cerebral white matter atrophy, which was found by our 3 studies and 1 other study. There were mixed findings for whole brain parenchyma, cortical gray matter, and SCN+IFT, with some studies showing atrophy and others showing enlargement. The reasons for the different findings were unclear but may have been due to multiple differences in methods, including brain volumetry software, patient samples (including nature and severity of injury), and time after injury. However, the NeuroQuant® database used in the current study may have constituted a better normal control group than those of other studies because it was based on a much higher number of controls (over 4,000 throughout the lifespan) and because it was the only one FDA-cleared for clinical use. Overall, it seemed likely that longitudinal atrophy of cerebral white matter, enlargement of cortical gray matter, and enlargement of SCN+IFT was a characteristic of at least some patients with chronic mild or moderate TBI.
Implications of longitudinal versus cross-sectional design
Since the current findings partially replicated and extended our previous findings, why are they important? Our previous studies were based mostly on cross-sectional data. The current study was based mainly on longitudinal (t1-t2) data, thus providing a new basis for important theoretical implications, as discussed below in this section. Because atrophy after TBI has been reported commonly, we will focus our discussion on the newer findings of widespread enlargement.
As a first step toward understanding
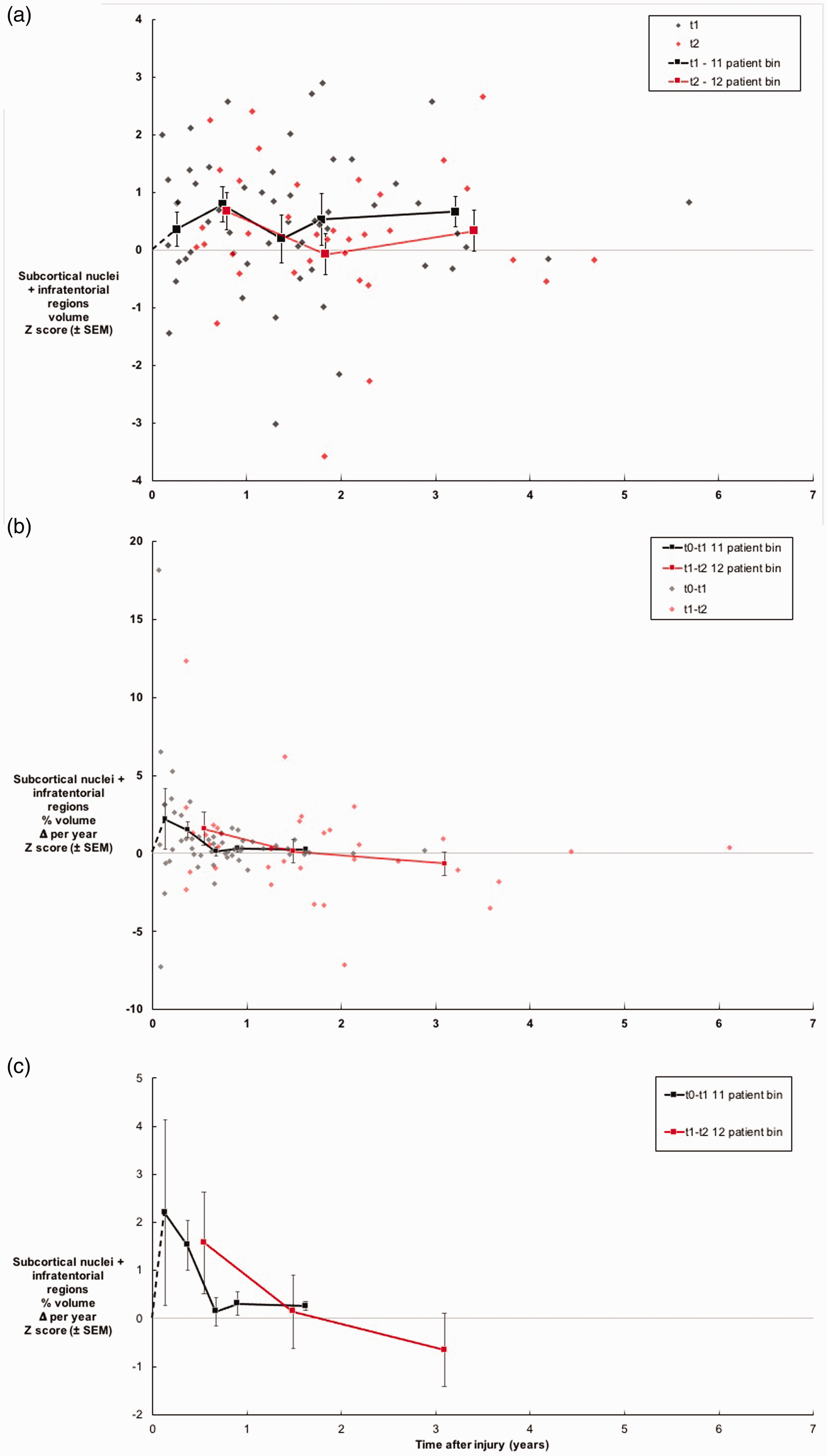
Subcortical nuclei + infratentorial regions (SCN+IFT) volume (measured at t1 and t2) and volume change (measured from t0-t1 and t1-t2) versus time after injury. (a) Volumes (t1 and t2) reached large effect sizes (Z scores around 0.8) within 10 months after injury and decreased partially over the next 2 years. (b) Volume changes (t0-t1 [estimated] and t1-t2 [measured]) reached very large effect sizes (Z scores around 2.2) within a couple months after injury, and then steadily decreased until 3 years after injury, indicating that volume was enlarging through the first year, changing little during the second year, but atrophying around 3 years after injury. (c) Zoom-in on the trendlines from section (b).
Abnormal cross sectional brain volume
At t2, the patients (N = 36) had many regions that were abnormally large and a few that were abnormally small. The pattern of abnormal volumes was very similar to that observed in our previous study which examined t1 volumes using a group of patients that was mostly overlapping but larger (N = 50) than in the current study because more data were available at t1 than t2. 19 Thus, although the current study’s t1-t2 analyses showed multiple significant increases of volume, the changes were not large enough to have a major impact on the pattern of cross-sectional volume abnormalities at t1 compared to t2.
Theoretical importance of abnormal enlargement
What causes the abnormal brain volume enlargement in patients with chronic mild or moderate TBI? Two leading possibilities include compensatory hypertrophy and chronic neuroinflammation.
Compensatory hypertrophy
Regarding possible compensatory hypertrophy, there have been many studies showing that when brain regions perform more tasks, they enlarge.47–60
There are a few possible causes or subcategories of the hypothesized compensatory hypertrophy that are discussed below, including: 1) injured brain regions becomes more active and enlarge; 2) uninjured brain regions connected to the injured brain regions become more active and enlarge; and 3) the injured regions atrophied in the post-injury acute phase and later enlarged as recovery occurred.
Subhypothesis 1: the injured brain regions become more active and enlarge. This is supported by studies of patients with brain disorders showing that increased brain activity leads to brain enlargement.47,55,59,60
Subhypothesis 2: uninjured brain regions account for the enlargement. This is indirectly supported by studies of people without brain disorders showing that increased brain activity leads to brain enlargement.48–54,56–58
Subhypothesis 3: reversal of previous atrophy. Under this scenario, the observed longitudinal enlargement would be consistent with brain regions changing from abnormally small to normal cross-sectional volume. However, this hypothesis would not explain the abnormal cross-sectional enlargement reported in this and other studies12,13,15,19 and therefore seems unlikely to be true.
Chronic neuroinflammation
An alternative explanation for the cause of the brain enlargement is chronic neuroinflammation. Previous investigators have found that TBI causes both acute and chronic neuroinflammation,61–68 which can persist for years after injury. 63 So it would be reasonable to hypothesize that chronic neuroinflammation and edema result in abnormally large brain volume.
Empirical evidence for this idea comes from a study of a group of patients with mild TBI, which found that abnormal enlargement of the anterior and posterior cingulate regions was associated with posttraumatic headache and a serum marker of neuroinflammation. 69 In a post hoc attempt to explore this possibility, we attempted to partially replicate their findings.
Each of our patients was assessed for posttraumatic headaches using the approach recommended by Zasler et al., 70 pp. 932–953, including the most common types of posttraumatic headaches--tension-type, neuropathic (also called neuralgia) and migraines, 70 p. 949. T-tests were performed within between subgroups of patients based on headache type to test the hypotheses that each headache type was associated with volume abnormalities in the anterior or posterior cingulate gyrus.
Results showed that patients with neuropathic headaches had larger volume of the posterior cingulate cortex (P = .01), with a medium-to-large effect size, although the P value did not survive Bonferroni adjustment (adjusted alpha = .05/6 tests = .008) (Table 5). Given the a priori finding of an association, the Bonferroni adjustment may have been too conservative. 46 Overall, this finding of an association between the neuropathic headaches and abnormal enlargement of the posterior cingulate cortex provided moderately good support for the previous similar finding by Niu et al. 69
Comparisons of cingulate brain volumes (Z scores) at time 1 (t1) between patient subgroups based on type of posttraumatic headaches.
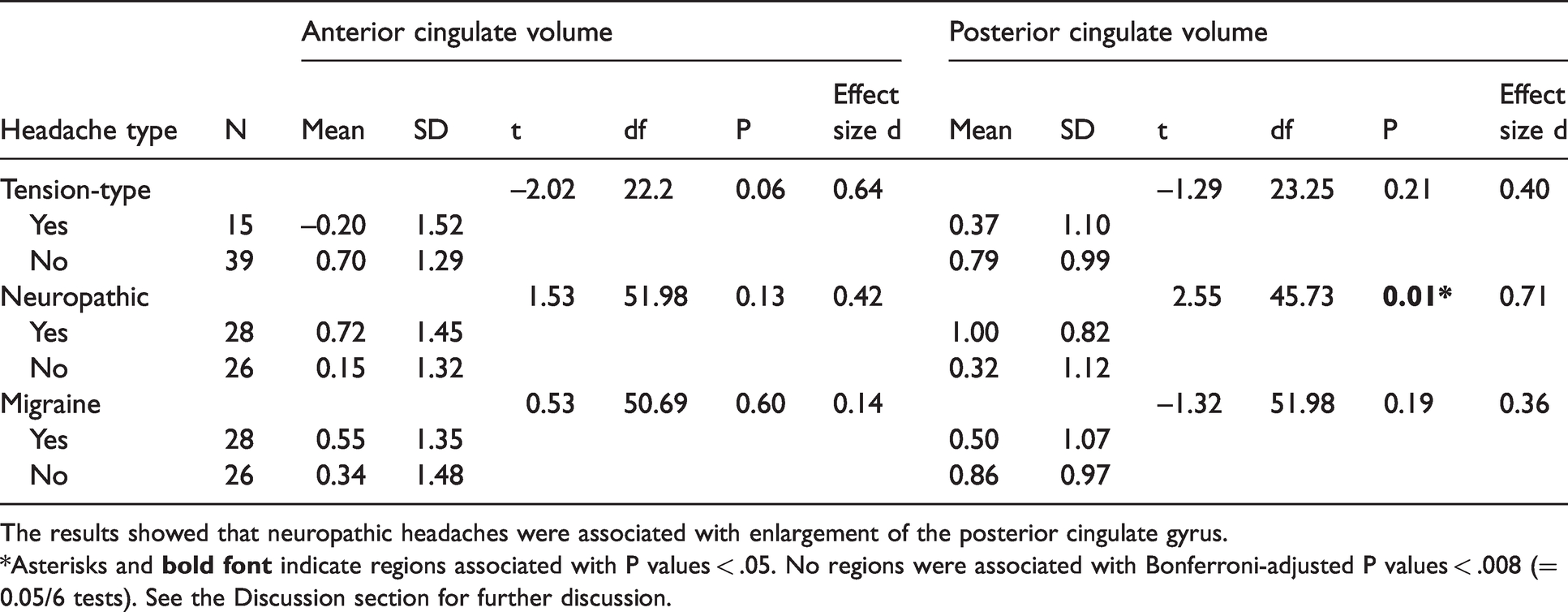
The results showed that neuropathic headaches were associated with enlargement of the posterior cingulate gyrus.
*Asterisks and
As those authors pointed out, there is a higher prevalence of chronic posttraumatic headache in mild TBI than in moderate or severe TBI, which is surprising because most symptoms are less severe in mild TBI. Similarly, abnormal enlargement appears to occur more often in mild TBI, which is surprising because most volume abnormalities have been reported to occur less often in mild TBI. Given these similarities and our replication of Niu and colleagues’ finding of an association between posttraumatic headaches and cingulate enlargement, it is interesting to hypothesize that these phenomena share a pathophysiology that occurs more often in mild TBI than in severe TBI.
Niu et al. provided a theoretical framework for our hypothesis based in part on their finding that posttraumatic headaches were associated with increased serum levels of CCL2, a chemokine that increases rapidly after TBI, causing inflammation. Accordingly, Niu et al. hypothesized that TBI leads to inflammation, causing gray matter hyperreactivity, volume increase and altered pain perception.
Limitations
The current study examined patients with mild or moderate TBI who had symptoms that persisted for months to years after the injury and sought treatment at a TBI specialty clinic. Therefore, these results may not apply to other TBI patients, for example, patients with severe TBI, or patients with mild TBI who have complete resolution of symptoms within hours to days. Also, given the naturalistic outpatient setting of the current study, the patients participated in rehabilitation, and most of them took neuropsychiatric medications for their symptoms. It was unknown whether those factors affected brain volume.
Limitations of the current study included that it was not possible to definitively evaluate the effects of education on the findings because education data was not available for the NeuroQuant® normal controls. However, given that the mean years of education for the patients (14.3; SD = 3.0) was similar to the mean years of education for the U.S.A. population in 2012 (13.3 years), it seemed unlikely that there would have been large effects of education on brain volume. It would be helpful for future studies to include measurements of levels of education.
Many of the patients in the current study had posttraumatic stress disorder (PTSD). A recent review found that PTSD is associated with atrophy of gray and white matter in prefrontal cortex and cingulate cortex; atrophy of the corpus callosum and hippocampus; and abnormal enlargement of the amygdala. 71 The findings of atrophy of the cerebral white matter overlaps with the same finding in the current study and many other studies of TBI. The finding of abnormal enlargement of the amygdala also overlaps with the same finding in the current study. However, the finding of atrophy of cortical gray matter regions and the hippocampus, although overlapping with studies of moderate and severe TBI, stand in contrast to the current findings of abnormal enlargement of those structures. Thus, there were some findings from the current study which may have been due to TBI or PTSD, but other findings appeared to be relatively specific to chronic mild or moderate TBI. Further research on patients with and without TBI, and with and without PTSD, will be needed to further clarify these issues.
To measure brain volume, this study used NeuroQuant software instead of better-known programs like FreeSurfer or other programs commonly used in university research settings. A PubMed search showed that there have been many more peer-reviewed published articles using FreeSurfer (1946 articles) than NeuroQuant® (91 articles). Why use software that has many less publications to support it?
Although NeuroQuant has fewer peer-reviewed articles to support its use, 91 articles and FDA clearance provide a good basis for its reliability and validity, as discussed in the NeuroQuant® software was used for cross-sectional brain volume analysis section.
Furthermore, most or all clinical researchers agree that university-based research findings should provide benefits or applications for patients in real-world clinical settings. However, there is a large gap between the research on advanced MRI brain imaging techniques and their clinical availability.72,73 In clinical practice, to order a brain volume analysis of a patient, the clinician must choose a software tool that has not only a sound research basis but also commercial availability. In 2007, NeuroQuant® achieved both goals, and now it is relatively easy for a clincian to order a brain volume analysis on a patient.
In contrast, FreeSurfer and similar programs are not available for use in typical clinical settings. This is due in part to the fact that FreeSurfer and most university research-based programs are not FDA-cleared and do not have large, well-characterized and well-controlled normal control datasets that allow reliable and valid analyses on individual subjects.
NeuroQuant® generally has been found to have good to excellent reliability with FreeSurfer by our group,33,34 and others.30,35,40,74
Given the multiple studies showing good inter-method reliability and other types of reliability for NeuroQuant®,8,75,76 we believe that it is likely that the results of the current study would show good reliability with results from studies using programs like FreeSurfer. Nevertheless, it will be important to test this idea directly.
Finally, we will discuss the limitations of the brain volume estimation method, a relatively novel method. The t0-t1 longitudinal analyses were based on estimation, not measurement, of brain volume at t0 (time of injury). Actual measurement of volume at t0 before accidental injury would have been highly preferable. However, this approach would be difficult and expensive because many healthy people would need to be scanned for every one who subsequently had a TBI. For this reason, a study of this type has never been done, and the volume estimation method was developed as a partial solution to this problem.
How reliable is the volume estimation method? The reliability of estimated versus measured volumes was good for the 4 large brain regions used in the current study. Not surprisingly, test-retest reliabilities for estimated-versus-measured volumes were lower than for measured-versus-measured volumes, which had excellent reliability.
Furthermore, due to the lower reliabilities for estimated-versus-measured volumes, the variances for t0-t1 change data were markedly larger than for t1-t2 change data. An example of this can be seen in Figures 1 and 2, where the normal range for t0-t1 cortical gray matter volume change at 45 years of age was -0.1% ± 7.6% per year (Figure 1), whereas the corresponding range for t1-t2 change was -0.2% ± 2.3% per year (Figure 2). But despite the greater variances for t0-t1 data, the current study still found statistically significant t0-t1 volume changes for all 4 brain regions.
Is estimation of volume change from t0-t1 more informative than cross-sectional measurement at t1? Cross-sectional volumes at t1 adjust for intracranial volume, so in some sense they are similar to t0 volume estimation, which uses measurement of intracranial volume as a core feature. But cross-sectional volumes do not take advantage of the longitudinal design, which often is more sensitive for detecting brain volume abnormalities. 21 For example, previously we reported that a subset (N = 50) of the current sample of patients (N = 55) did not have statistically significant cross-sectional atrophy of cerebral white matter (effect size = −2.3; P = .13) (although other evidence showed that there likely was atrophy, especially within the first 10 months of injury). 19 In contrast, in the current study, the longitudinal t0-t1 analyses found statistically significant cerebral white matter atrophy (effect size = −3.1; P = .005); the effect size was 35% larger ( = [0.8/2.3]×100%), due mainly to the increased power of the longitudinal design. Thus, the t0-t1 analysis, in comparison with the t1 cross-sectional analysis, provided clearer statistical evidence for the finding of cerebral white matter atrophy.
Similarly, the t0-t1 estimates of volume change complement the t1-t2 measured data because, unlike the t1-t2 data, they cover the acute and subacute stages after injury.
In summary, compared to actually measuring volume at t0, estimating volume at t0 is an inferior method. But compared to not having a measurement or even estimate of volume change during the critically important t0-t1 interval, estimating t0-t1 volume change is a useful approach based on its reliability and validity for distinguishing patients from normal controls. Therefore, the best approach may be to combine t0-t1 estimates with traditional cross-sectional and longitudinal measurements.
Conclusions
In contrast to previous studies that found longitudinal brain volume decrease in patients with chronic mild or moderate TBI, the current study found more longitudinal brain volume enlargement than atrophy. However, there was atrophy in some regions. An almost universal limitation in this area is the lack of brain volume measurement before injury. Given this limitation, one of the most interesting aspects of the current finding of post-injury longitudinal increase of volume is that it adds indirect evidence that the previous findings of cross-sectional enlargement12,13,15,16,19 were due to post-injury enlargement and not due to pre-injury abnormalities. This conclusion is based on the idea that, because the current study found that longitudinal enlargement occurred after injury (with t1 and t2 both measured after injury), then it was more likely that longitudinal enlargement also occurred earlier in the post-injury course (between t0 and t1).
The finding that chronic mild or moderate TBI is characterized by cross-sectional and longitudinal brain volume enlargement stands in contrast to many previous studies of patients with moderate to severe TBI, which have consistently found brain atrophy. In turn, the contrasting findings suggest that mild TBI is not simply a mild form of severe TBI but is fundamentally different with respect to brain volume changes and pathophysiology, raising hope--for example--that treatments that did not work for severe TBI might work for mild to moderate TBI. Other ideas for future research include measuring volume change closer to the time of injury, and examination of correlations between volume changes and changes in pathophysiological measures (including measures of neuroinflammation), neuropsychological performance or clinical symptoms.
Footnotes
Acknowledgements
The following information is required to be included in publications that used data from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database. Data used in the preparation of this article were obtained from the ADNI database (adni.loni.ucla.edu). The ADNI was launched in 2003 by the National Institute on Aging (NIA), the National Institute of Biomedical Imaging and Bioengineering (NIBIB), the Food and Drug Administration (FDA), private pharmaceutical companies and non-profit organizations, as a $60 million, 5-year public-private partnership. The primary goal of ADNI has been to test whether serial magnetic resonance imaging (MRI), positron emission tomography (PET), other biological markers, and clinical and neuropsychological assessment can be combined to measure the progression of mild cognitive impairment (MCI) and early Alzheimer's disease (AD). The Principal Investigator of this initiative is Michael W. Weiner, M.D., VA Medical Center and University of California - San Francisco. ADNI is the result of efforts of many co-investigators from a broad range of academic institutions and private corporations, and subjects have been recruited from over 50 sites across the U.S. and Canada. MRI data collection and sharing for the ADNI normal control participants was funded by the Alzheimer's Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12–2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.;Cogstate;Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.;NeuroRx Research; Neurotrack Technologies;Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics.The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
Declaration of conflicting interests
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ross is CEO of NeuroGage LLC, a company which produces Neurogage® software. In 2017, he was a paid consultant for CorTechs Labs, Inc., which produces NeuroQuant® software. None of the other authors had competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
