Abstract
Persistent post-concussion symptoms (PPCS) lasting longer than 4 weeks affect 25% of children with mild traumatic brain injury (mTBI) or concussion. Working memory (WM) problems are a common complaint in children with PPCS. Despite normal function on traditional neuropsychological tests, these children exhibit aberrant cortical responses within the dorsolateral prefrontal cortex (dlPFC) and default mode network (DMN) regions – both of which are implicated in WM. Using a prospective, longitudinal cohort study design, we investigated changes in cortical fMRI responses within the dlPFC and DMN during an nback WM task at two timepoints: one and two months post-injury. Across these timepoints, the primary outcome was change in cortical activations (increase in BOLD) and deactivations (decrease in BOLD) of both dlPFC and DMN. Twenty-nine children (mean age 15.49 ± 2.15; 48.3% male) with fMRI scans at both timepoints were included, following data quality control. Student’s t-tests were used to examine cortical activations across time and task difficulty. ANCOVA F-tests examined cortical responses after removal of baseline across time, task difficulty and recovery. Volumes of interest (5 mm sphere) were placed in peak voxel regions of the DMN and dlPFC to compare cortical responses between recovered and unrecovered participants over time (one-way ANOVA). Between one and two months post-injury, we found significant increases in dlPFC activations and significant activations and deactivations in the DMN with increasing task difficulty, alongside improved task performance. Cortical responses of the DMN and bilateral dlPFC displayed increased intensity in recovered participants, together with improved attention and behavioural symptoms. Overall, our findings suggest evidence of neural compensation and ongoing cognitive recovery from pediatric TBI over time between one and two months post injury in children with PPCS. These results highlight the wider and persisting implications of mTBI in children, whose maturing brains are particularly vulnerable to TBI.
Introduction
Pediatric mild traumatic brain injury (mTBI) can have a significant impact on childhood development and cognitive functions. It is an important public health problem, affecting 55.9 million people each year worldwide. 1 mTBI commonly occurs in children, where 20% of children and adolescents less than 16 years of age sustain an mTBI per year.2,3 In the child and adolescent population (aged 0–18 years), falls and contact sport injuries are the most frequent modes of injury. 4 Concussion is a form of mTBI, and the term is sometimes used interchangeably with mTBI. 5 Persistent post-concussion symptoms (PPCS) occur in 50% of children for longer than one month after injury, and in 14% of children for three or more months after injury.6,7 PPCS include physical, cognitive, emotional, and behavioural symptoms such as headaches, fatigue, irritability, insomnia, and concentration and memory difficulties.8,9 In childhood, female adolescents (over the age of 12 years) are particularly at risk of PPCS. 10 Deficits in cognitive functions such as attentional processes, and in particular, working memory (WM) are common in PPCS and may affect a child’s learning capabilities and quality of life.11–18
Working memory is a temporary, capacity-limited memory holding store critical for executive function that matures during puberty, adolescence and early adulthood.15,19 This maturation process is associated with increases in cortical activation in the middle frontal gyrus, especially the dorsolateral prefrontal cortex (dlPFC), a region strongly implicated in WM.20,21 The dlPFC has been extensively researched as a core area for WM in adults.22–27 However, younger children recruit more widespread brain regions during WM tasks,28,29 and their maturation involves a shift from diffuse and posterior activation, to focal and anterior activation. 28
Closely associated with the WM-related activation is the default mode network (DMN). The DMN consists primarily of the posterior cingulate cortex (PCC), retrosplenial cortex and ventromedial prefrontal cortex, and is usually anti-correlated with the WM-related areas.30,31 In healthy individuals, DMN activation is seen during internally-directed attention, such as during planning and introspection, and exhibits a reduced level of activity during the maintenance phase of most attentionally-demanding tasks. 31 However, in adult TBI studies, participants with PPCS have been shown to exhibit abnormal DMN activation during attentionally-demanding tasks. 32 Functional neuroimaging provides insight into both the working memory and default mode networks, in both typically developing children and following mTBI, allowing the pathophysiological and clinical sequelae of injury to be elucidated. 33
Functional neuroimaging studies of WM- and default mode-related areas following mTBI have reported differing results across adult and pediatric populations.34–36 Although different methodologies go some way to explain these differences, they also potentially reflect the vulnerability of an actively maturing region in adolescence so vital for executive function. 37 We have previously reported cortical activation during an nback working memory task in 60 children with PPCS one month following mTBI. 34 Children with PPCS were found to have decreased activation in the dlPFC, and DMN regions (precuneus and posterior cingulate gyri), when compared to 30 children who had recovered following mTBI at the same time point post-injury. 34
The present study aims to examine changes in WM-related cortical activations over time during recovery in children with PPCS. Our study will be the first that longitudinally examines changes in cortical activations during short-term recovery from mTBI in a pediatric population. 12 We hypothesise that WM-related cortical activation in children with PPCS will increase over time in the dlPFC, and decrease concurrently in the DMN. We also hypothesise that changes in cortical activation over time will be related to recovery.
Methods
Study design
The current study was a prospective cohort study which recruited participants from the PLAYGAME trial: a randomised, placebo-controlled, clinical trial of melatonin conducted in children aged 8–18 years with PPCS following mTBI at 4–6 weeks post-injury (NCT01874847) 38 between February 2014 and April 2017 at the Alberta Children’s Hospital, Calgary. In the PLAYGAME trial, participants were randomized to receive placebo (n = 33), 3 mg of melatonin (n = 33); or 10 mg melatonin (n = 33) for 28 days. Concussion and/or mTBI was defined according to the American Academy of Neurology criteria. 39 Exclusion criteria included a loss of consciousness greater than 30 min, or Glasgow Coma Scale (GCS) score of less than 13; mTBI within the previous three months, or failure to recover from a previous mTBI; significant past medical or psychiatric history (attention deficit hyperactivity disorder (ADHD) or mild learning difficulties were not excluded); the use of neuroactive drugs affecting fMRI; an inability to complete questionnaires or evaluations; or a contradiction to MRI. Consent and assent were obtained from the parent and child, respectively. At enrolment, a standardized interview and medical examination were performed by a physician experienced in concussion/mTBI assessment. The study was conducted in accordance with Good Clinical Practice and ethical approval was received by the University of Calgary Health Ethics Research Board (13–0372) and the University of Queensland (2,01,70,01,523).
Participants
Out of 64 eligible children with PPCS and neuroimaging at 4–6 weeks, 29 were included in the current study; where participants were excluded due to single MRI only (n = 22), motion/displacement (n = 11), poor task performance/failed TOMM (A’ score < 0.6; n = 1), or abnormal neuroimaging (n = 1; Figure 1). Participant characteristics are presented in Table 1. The participants in this study completed an nback WM task during fMRI around one month post-injury and again one month later. Out of the 29 participants, 15 participants at the second imaging session had clinically recovered with Post Concussion Symptom Inventory (self-report, PCSI-Y) scores returning to baseline and return to normal activities 40 and 14 participants remained unrecovered at two months post-injury.
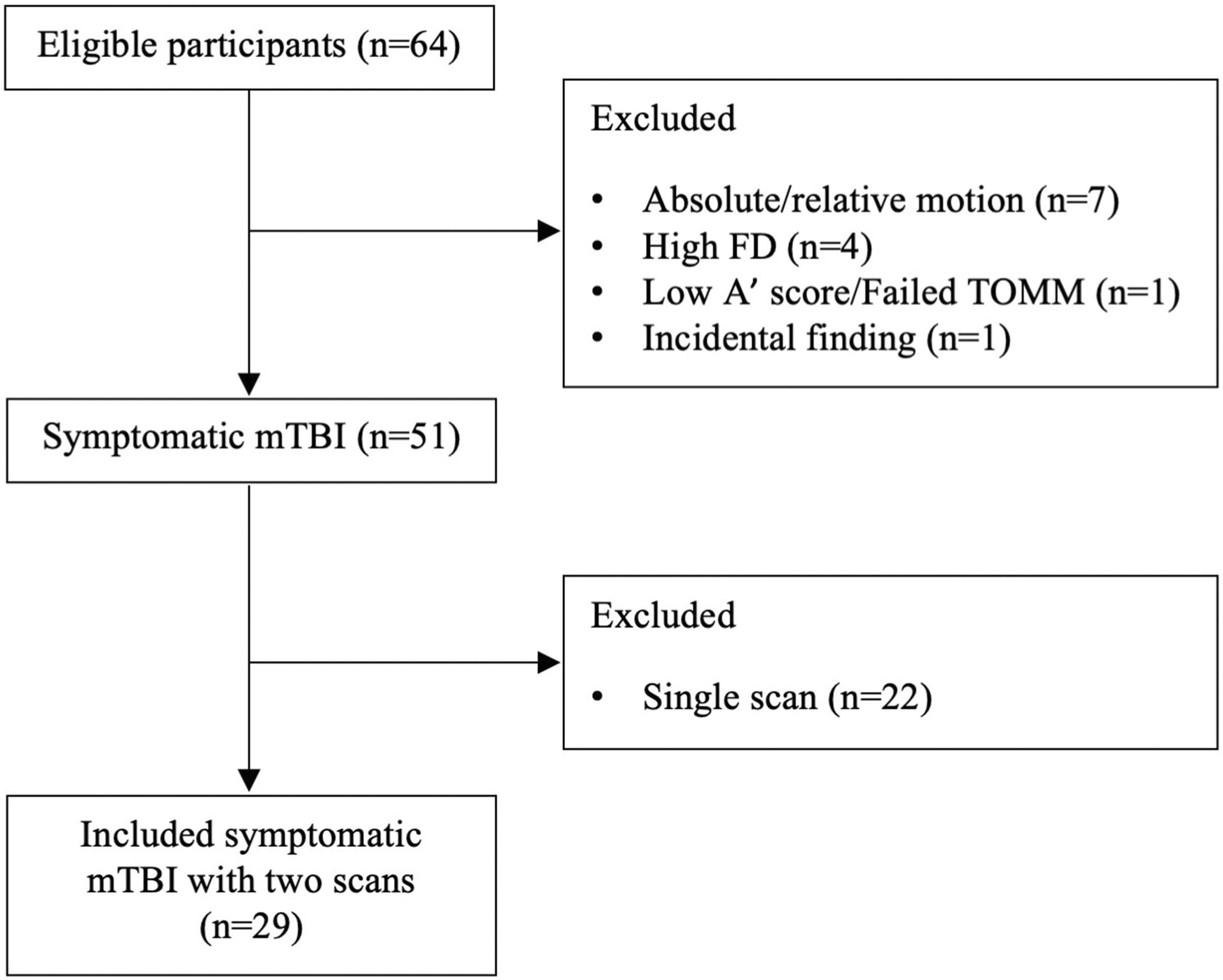
Enrolment into the study. Participant exclusion included single scan, excessive motion, excessive framewise displacement (FD), low A’ score (< 0.6)/failure of TOMM (Test of Memory Malingering), or an incidental finding, and 29 symptomatic participants were included in the final sample. mTBI: mild traumatic brain injury.
Pre-injury characteristics.
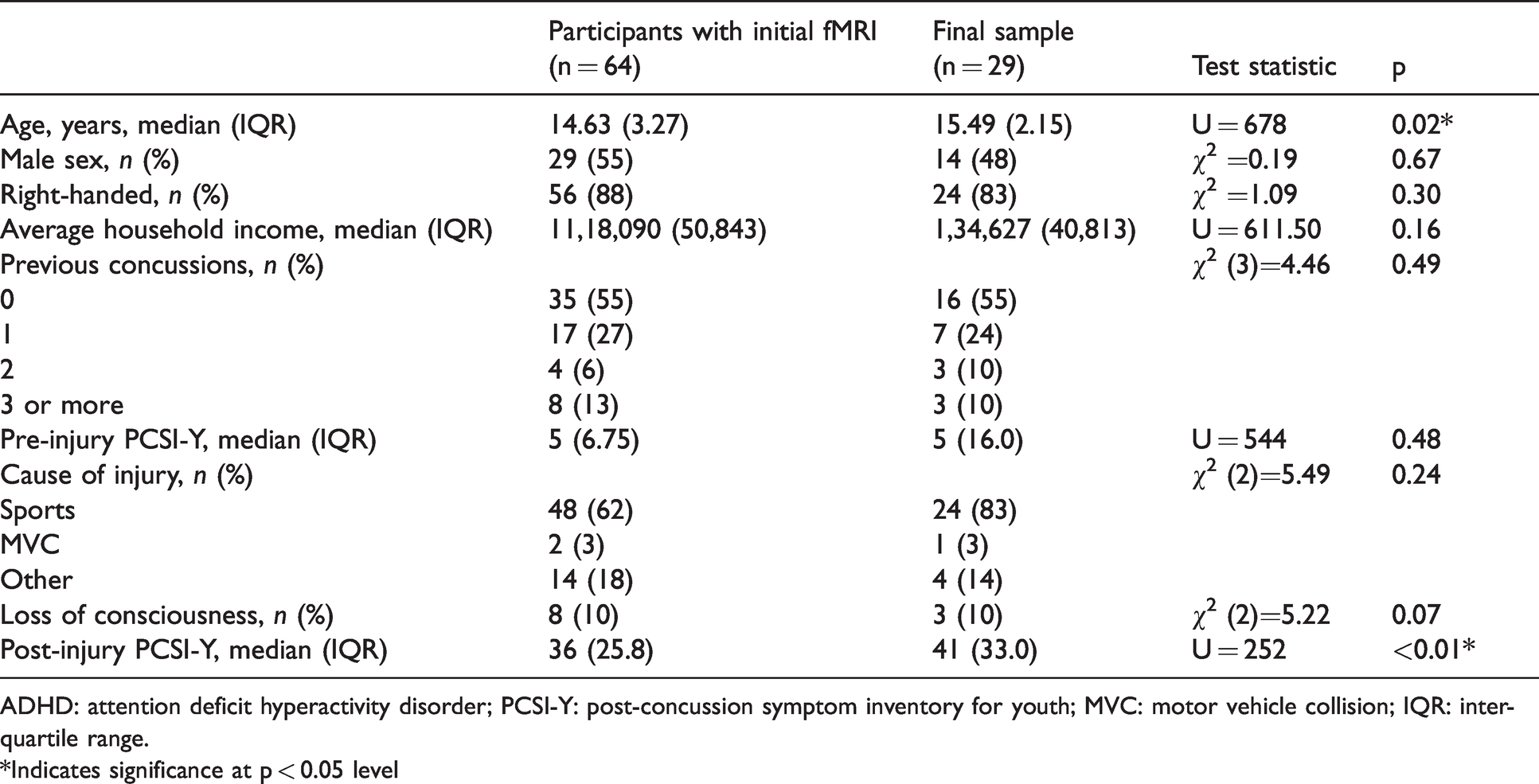
ADHD: attention deficit hyperactivity disorder; PCSI-Y: post-concussion symptom inventory for youth; MVC: motor vehicle collision; IQR: interquartile range.*Indicates significance at p < 0.05 level
Procedure
A standardised clinical assessment was performed by a physician where demographic, medical and injury details were recorded. Cognitive and behavioural measures were completed together with symptom validity testing using the Test of Memory Malingering (TOMM).41,42 Following safety screening, a mock MRI scan for younger children, and training on the nback task with a graduate student or research assistant, MRI was performed at both timepoints.
Neurocognitive assessment
The Post-Concussion Symptom Inventory for Youth (PCSI-Y) was used to assess symptoms and classify symptomatic status and recovery. 40 This questionnaire assesses symptoms in four domains: somatic, cognitive, emotional, and sleep. It has been demonstrated to have good internal reliability when measuring PPCS.6,7 Clinically significant symptomatology was defined as a 10 point increase in PCSI as compared with the pre-injury report (assessed at 4 weeks post-injury). 43 The PCSI cognitive domain questions were used to examine subjective cognitive difficulties, including the domain questions, “feeling slowed down”, “mentally foggy”, “difficulty concentrating”, “difficulty remembering”, “feeling dazed” and “becomes confused with directions”. Symptoms were rated from zero to six.
Cognitive ability was assessed using the CNS-Vital Signs computerised cognitive assessment on the day of MRI. 44 This assessment included seven tests: Verbal Memory, Visual Memory, Finger Tapping, Symbol Digit Coding, Stroop test, Shifting Attention test, and the Continuous Performance test (age adjusted, mean [M]= 100, standard deviation [SD] = 15). The CNS-Vital Signs assessment has a good test-retest reliability (r = 0.63–0.82) in the pediatric population, as well as concurrent reliability with traditional neurocognitive testing measures.44–47 Behaviour was measured with the Behaviour Assessment System for Children (2nd Edition, parent report) (BASC-2) which has high reliability and validity in TBI populations.48–50 The Attention subdomain was used as a measure of attention problems. The Behavioural Symptoms Index (BSI) is a composite score of the BASC-2, 51 derived from Hyperactivity, Aggression, Depression, Atypicality, Withdrawal and Attention problems (M = 50, SD = 10). The Behaviour Rating Inventory of Executive Function (BRIEF, parent report) was used to assess executive function and which has very good validity in children with TBI and concussion.52,53 Three summary scores were used: the Global Executive Composite, Behavioural Regulation Index, and the Metacognition Index (M = 50, SD = 10).
fMRI
A letter stimuli-based, visuospatial nback working memory task was designed in Eprime 2.0 (PSTNET Psychology Software Tools Inc., Sharpsburg, PA) with inspiration from Dettwiler and colleagues 35 and was implemented as described by Khetani and colleagues. 34 Each trial consisted of uppercase letters presented in circles around the screen. The task incorporated three levels of working memory load: n = 0, 1, 2. The 0back condition required participants to respond if the presented stimulus matched a pre-defined exemplar, and thus acted as a control condition in terms of the visual and motor processing requirements of the task. The 1back condition required a response when two successive stimuli were identical (in letter and position), and the 2back condition required a response when identical stimuli were separated by a single non-identical image.
During the task, three experimental blocks of each cognitive load were presented, totalling nine blocks (Figure 2). Overall, 60 images were presented; 20% of which were identical to the target (i.e., a correct match). At the beginning of the task, a 20 second introduction was presented, followed by an information and fixation slide before each block. The stimulus and interstimulus interval (ISI) presentation times were 0.5 s and 1.5 s, respectively. Pseudo-randomization of stimuli was undertaken in each block. A practice task was completed by participants outside the scanner, where one block of each cognitive load was completed, with 75% accuracy required to proceed. All participants were able to complete practice training within two attempts. Reaction time and A-Prime (A’), which was calculated as a non-parametric measure using true positives and false positives, were used to measure performance on the nback working memory task. 54 Any participants with an A’ score of below 0.6 on any task condition were excluded from analysis due to potential misunderstanding of the working memory task. 54
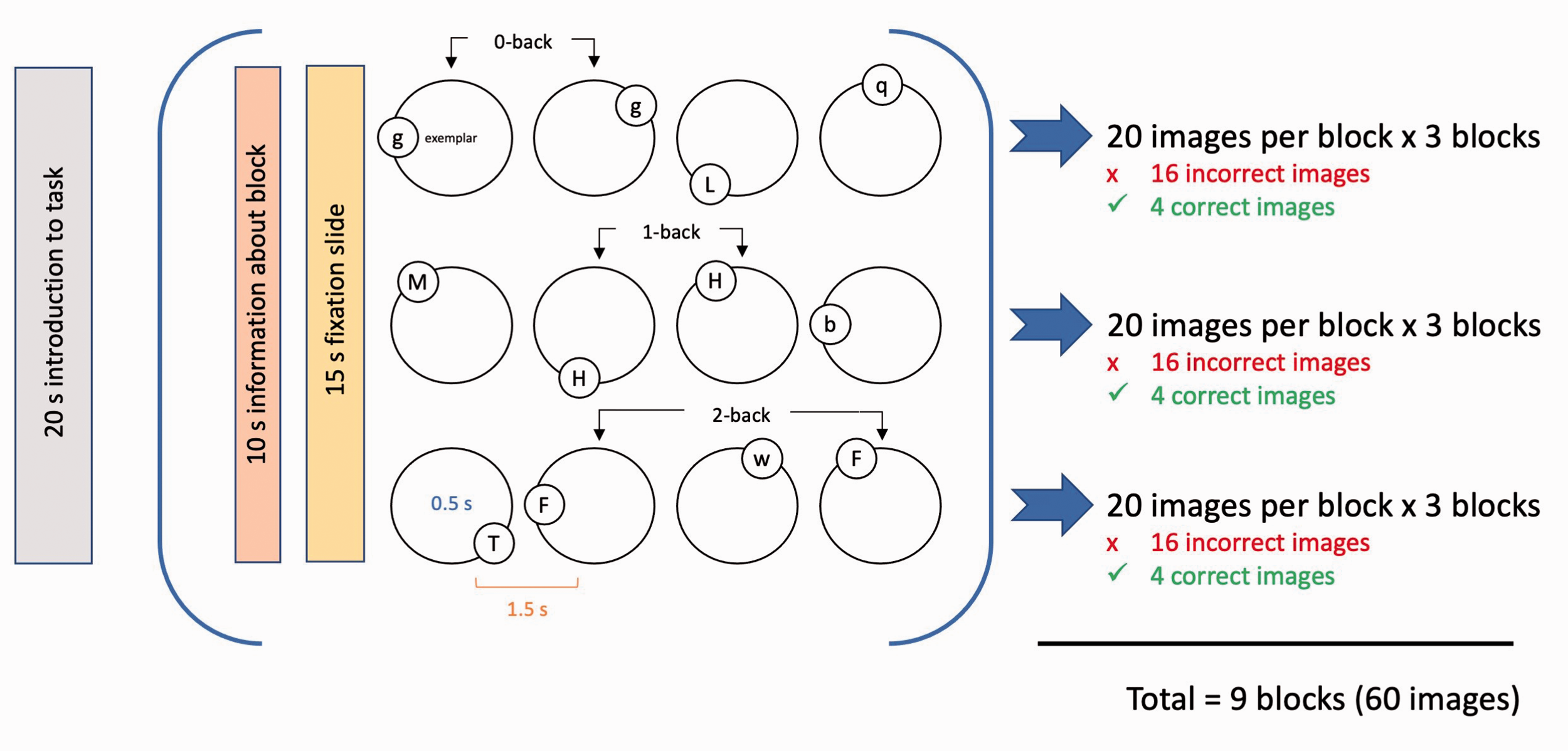
Visual representation of nback task. 33--35
MRI acquisition parameters
Cortical activity corresponding to the nback working memory task was measured by fMRI BOLD signal change. MRI data were acquired at the Alberta Children’s Hospital, using a 3.0 T GE Healthcare Discovery MR 750w Magnetic Resonance scanner with a 32-channel head coil. A Gradient Echo EPI pulse sequence was used to obtain oblique axial plane fMRI images, with the following scanning parameters: repetition time (TR) = 2000 msec, echo time (TE) = 30 msec, flip angle = 90, number of dummy acquisitions = 5, field of view (FOV) = 23 cm, slice thickness = 3.6 mm, number of slices = 317, matrix = 64 x 64, scan time = 10:44 min, interleaved acquisition.
Both T1 and T2-weighted images were acquired for registration. The T2-weighted anatomical images were acquired with a T2 FRFSE-XL (fast relaxation fast spin echo) sequence with the following parameters: TR = 6216 msec, TE = 80 msec, echo train length = 26, flip angle = 111, FOV = 23 cm, slice thickness = 3.6 mm, scan time = 0:44 min, interleaved acquisition. The T1-weighted anatomical images were acquired with a FSPGR (fast spoiled gradient echo) BRAVO sequence with the following parameters: TR = 8.2 msec, TE = 3.2 msec, flip angle = 10, FOV = 24 · 24 cm, matrix = 300 · 300, number of slices = 230, slice thickness = 0.8 mm, scan time = 5:32 min.
fMRI processing pipeline
All MRI pre-processing and analysis were completed using the MATLAB SPM software fMRI toolbox (Wellcome Department of Imaging Neuroscience, UCL. Released 2014. Statistical Parametric Mapping 12 for Mac OS, London, UK: The MathWorks, Inc.). Realignment, slice timing correction, coregistration, segmentation, normalisation and smoothing were applied to images (FWHM 8.0 mm). High pass temporal filtering with a 200 s cut-off was used. fMRI images were registered to the extracted T1-weighted image. Images were standardised to the MNI152 2 mm brain template, using non-linear transformation with 12 d.o.f. and 10 mm warp resolution. Motion regressors were modelled within SPM12, and volumes exceeding a framewise displacement threshold of 0.4 mm were excluded. Participants with volumes with greater than 25% excessive displacement and with an absolute displacement > 4.0 mm were excluded from analysis. Flagged volumes in retained participants were censored and motion regressors were included in the first-level general linear model (GLM) analysis. Following scan exclusions, inadequate performance on task (A’ score < 0.6) or a “Fail” on the TOMM, retained participants were incorporated into our first level GLM analysis.
Univariate GLM was used to perform the first-level analysis. The nback task conditions were modelled by three different explanatory variables (EVs), where block onsets were convolved with a double-gamma hemodynamic response function of 38.5 seconds in duration. 55 The following contrasts were run: 1back >0back; 2back >0back; and 2back >1back. Contrast images generated from first-level models were then entered into group-level analyses. To ensure that the task engaged expected WM-related regions, activation maps were generated within each group. Group comparisons were undertaken using second-level analyses, involving t-tests and ANCOVA F-tests. De-meaned regressors for age, sex, and task were included in the model since these factors may influence working memory related activation and deactivation.56–58
Statistical analyses
Descriptive statistics were performed using the IBM SPSS Statistics software (IBM Corporation Version 26 for Mac OS, Armonk, NY). The Shapiro-Wilk test was used to test normality of the data. Mann-Whitney U-test was used to assess non-parametric differences between groups (recovered vs unrecovered and included vs. excluded participants). Change over time was assessed using paired Student’s t-test (BASC, CNSVS, BRIEF) and Wilcoxon signed-rank test (A’ scores, PCSI) using the Bonferroni multiple comparison correction.
Cortical activation and deactivation responses were evaluated using MATLAB SPM 12 software. Group level two-sample t-tests were used to derive significant differences in cortical responses across the three nback tasks with age and gender as covariates. To control for any change in task performance over time, all analyses included the A’ score as a covariate. A difference analysis was also undertaken, where 0back (control condition) performance was subtracted from 1back and 2back task performance. ANCOVA F-tests with age and gender as covariates were then performed on the 1back – 0back and 2back – 0back conditions to compare change in cortical activations and deactivations over time and between groups (recovered; unrecovered). To compare groups (recovered; unrecovered), a volume of interest (VOI; 5 mm sphere) analysis was conducted. For each participant, VOIs were extracted at the peak of the group average response. These VOIs were placed in statistically significant regions of the dlPFC and DMN, to ascertain localized changes in BOLD signal intensity within these areas in each individual. One-way ANOVAs were used to compare changes in BOLD values between groups in activations and deactivations, independently, in the PCC and dlPFC, with a Benjamini-Hochberg correction. As the sample of participants in this study had been part of a larger clinical trial which assessed the effects of placebo, 3 mg or 10 mg melatonin on clinical symptoms following TBI, we performed a repeated-measures 2x3 level ANOVA (treatment group, time) to determine if there was any influence of treatment group on cortical activations across the three nback conditions in our sample of 29 participants.
All statistical contrasts were first identified via a cluster-level threshold at p < 0.001 uncorrected, with multiple comparisons correction accounted for via family-wise error (FWE) pFWE < 0.05. All fMRI-related analyses were corrected for multiple comparisons using the in-built SPM Random Field Theory Method. 59 Post-hoc analyses of region of interest values between recovered and unrecovered participants were corrected for multiple comparisons at a significance threshold of pFDR (false discovery rate) = 0.05 due to a minimal number of post-hoc comparisons and more liberal correction approach than a FWE approach used for fMRI contrasts. Cortical activations and deactivations within the dlPFC were measured via the Stanford University Functional Region of Interest (ROI) with voxel radius of 5 mm in all analyses. 60
Results
Overall, 29 participants were eligible for the study (mean age 15.49 (range 8.9 – 17.8; IQR 2.15) years, 48.3% male). Both parent (P) and youth (Y)-reported PCSI scores significantly decreased over time between one and two months post-injury (PCSI-Y: median = 22, IQR = 24 and 7.5 (13) at one and two months, respectively; PCSI-P: 41 (35), 15 (24.8) at one and two months, respectively; both p < 0.001). Although the 29 participants were similar in most demographic and clinical details, they were more likely to have higher initial post-injury PCSI scores and were older than participants with single imaging (Table 1). Almost half of participants had experienced at least one previous concussion. Most mTBIs were sports-related (n = 24, 82.8%). Participants were scanned first at 4 – 6 weeks post-injury (M = 37.6 days, SD = 6.2), and again at 8 – 10 weeks post-injury (M = 68.5 days, SD = 6.9). This will be hereafter referred to as one and two months post-injury.
Cognition
Performance on neurocognitive measures and working memory task performance is reported in Table 2. Overall, mean neurocognitive function was well within the normal range. Neurocognition scores significantly increased over time (t = 2.8, 95% CI = 2.8, 7.8, p < 0.01). Visual Memory scores decreased over time (t = -2.7, 95% CI = -13.4, 1.8, p < 0.05). However, neither the neurocognition nor visual memory changes survived post-hoc Bonferroni correction. Performance on the 2back task significantly increased at two months post-injury (Z = -3.8, p < 0.001), which remained significant after a post-hoc Bonferroni correction.
Neuropsychological outcomes at one- and two-months post-injury in children with repeated neuroimaging.
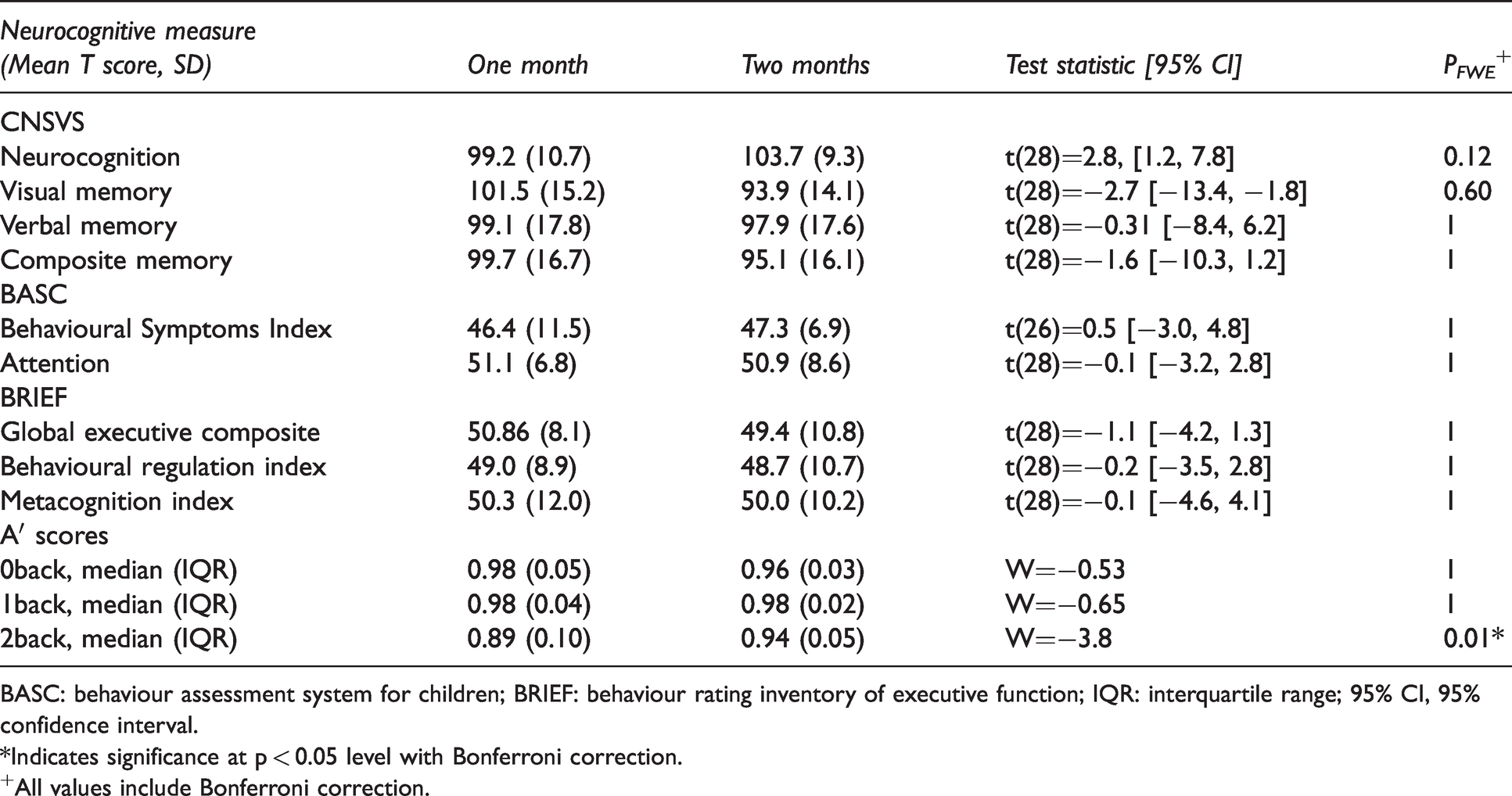
BASC: behaviour assessment system for children; BRIEF: behaviour rating inventory of executive function; IQR: interquartile range; 95% CI, 95% confidence interval.
*Indicates significance at p < 0.05 level with Bonferroni correction.
+All values include Bonferroni correction.
Working memory BOLD response
After controlling for task performance, age, and gender, there were significant differences in WM task-related cortical activation over time (pFWE < 0.05; Figure 3). All changes over time occurred while children were actively recovering, demonstrated by decreased PCSI scores over time. MNI coordinates and associated Brodmann Areas for all significant activations are presented in Table 3. During the 0back task (Figure 3(a)), increased activation at one month compared to two months post-injury was seen in the dlPFC bilaterally (voxel coordinates in mm [x, y, z]: 30, 17, 47; Z = 20.4 and -33, 14, 47; Z = 19.6) as well as in the posterior cingulate cortex (PCC; -9, -67, 47; Z = 21.8; Figure 3(a)). No significant differences in activity were seen at two months compared to one-month post-injury. During the 1back, increased right dlPFC activation (33, 14, 47; Z = 16.7) as well as some PCC activation (-9, -40, 50; Z = 14.7) was seen at one month compared to two months post-injury (Figure 3(b)); whereas less dlPFC activation and significantly increased PCC deactivation was seen over time (Figure 3(c)). During the 2back, no significantly increased activation was seen at one month compared to two months post-injury. At two months post-injury (Figure 3(d)), increased deactivation was seen over time in the right dlPFC (33, 11, 50; Z = 12.0) and PCC (9, -43, 50; Z = 14.8). There was no effect of melatonin on cortical response over time compared to placebo. Task performance (A’), age, and gender did not significantly influence cortical responses.
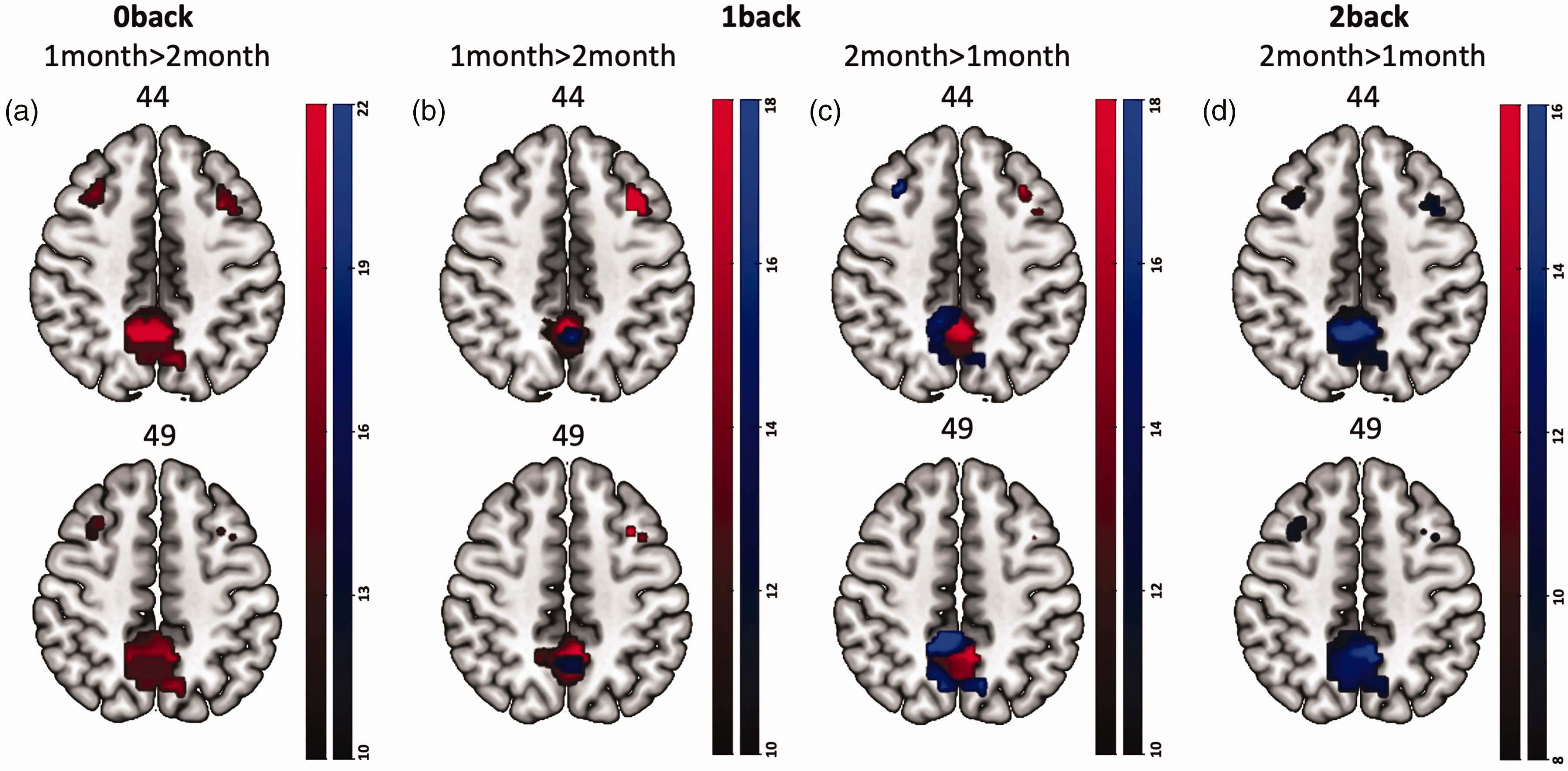
Significant working memory (WM) task-related changes in neural activation of the dlPFC and DMN over time between one and two months post-injury, by WM task load (2back > 1back > 0back; n = 29). Overall, increased deactivation of the DMN occurred over time and with increased task load. Red represents increased activation over time (i.e., significantly increased at two months); blue represents increased deactivation over time. (a) 0back WM condition cortical activation at one month post-injury; (b) 1back WM task cortical activation at one month post-injury; (c) 1back WM task cortical activation at two months post-injury; (d) 2back WM task cortical activation at two months post-injury. Activations represent statistically significant regions thresholded at KE = 20, with cluster level correction (pFWE < 0.05). Slice numbers shown above axial slices. Stanford University Functional Imaging in Neuropsychiatric Disorders Lab dlPFC and DMN masks used to create region of interest in all contrasts. Colour bars represent Z-scores for each contrast. Neurological convention displayed (left side of brain shown on left). WM: working memory; dlPFC: dorsolateral prefrontal cortex; DMN: default mode network; FWE: family-wise error.
Note: All coordinates displayed are ±5 mm for simplicity.
BOLD: blood oxygen level-dependent; PCC: posterior cingulate cortex; LDLPFC: left dorsolateral prefrontal cortex; RDLPFC: right dorsolateral prefrontal cortex.
After removing the effect of the control condition (0back), changes in cortical activity were noted over time (see Figure 4). During the 1back task, increased deactivation was seen over time in the left dlPFC (-45, 14, 38; Z = 13.7) and the dorsal part of the PCC (9, -67, 47; Z = 16.1), whereas increased activation was seen over time in the ventral part of the PCC (3, -40, 50; Z = 13.8) (pFWE < 0.05; Figure 4(a)). During the 2back task, activation was seen mostly in the left dlPFC (-33, 23, 44; Z = 14.9), and deactivation mostly in the right dlPFC (38, 25, 35; Z = 19.8), although both activation and deactivation was seen bilaterally (Figure 4(b)). In the PCC, ventral activations and dorsal PCC deactivations were seen during the 2back task, in similar regions to that seen during the 1back. Overall, all cortical responses increased in intensity as task difficulty increased.
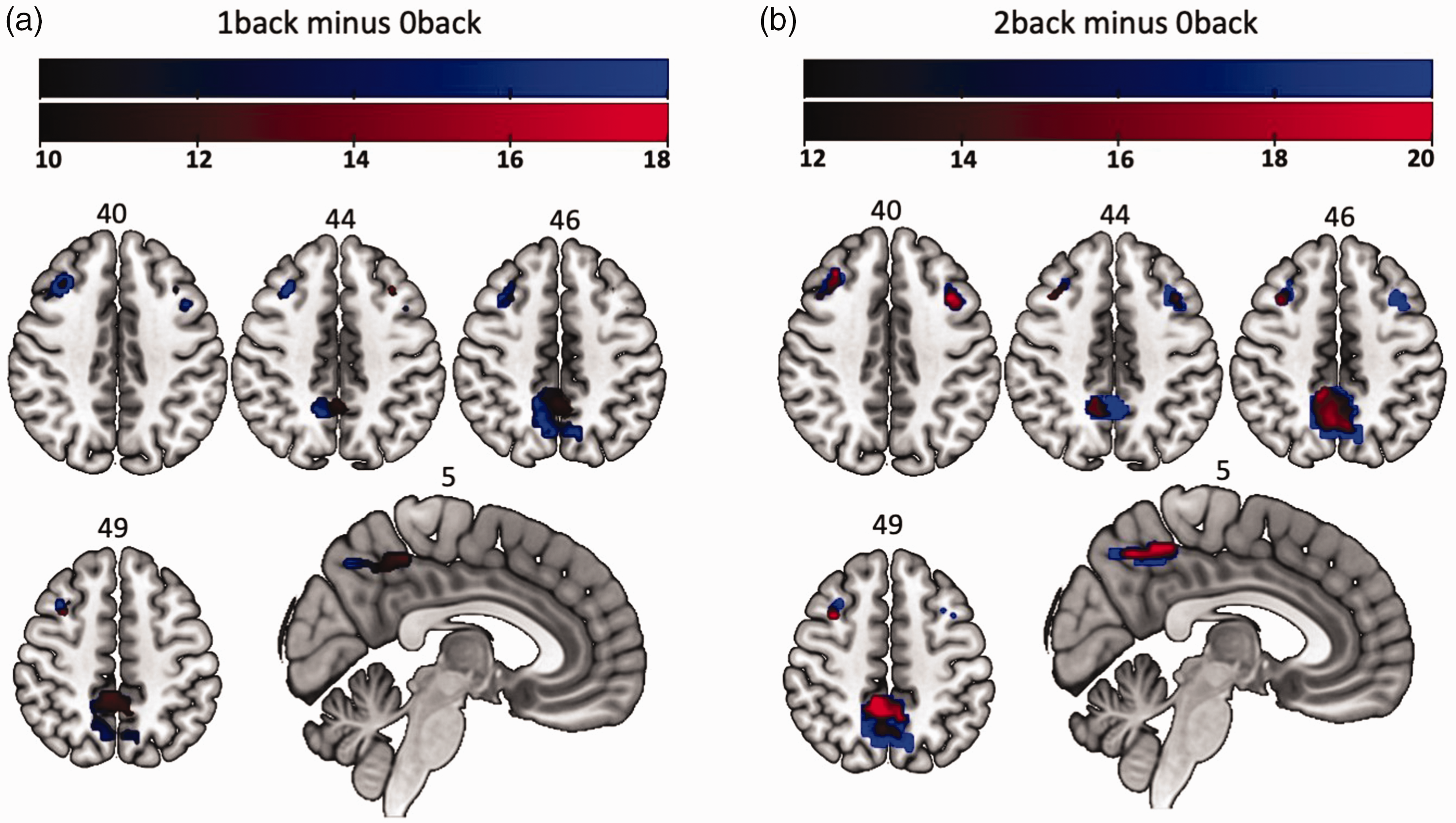
Effect of removing baseline (0back) on WM task-related cortical changes over time in 1back and 2back tasks. (a, b) Two months vs. one month WM task-related activations and deactivations over time (n = 29). Within the PCC, ventral activations and dorsal deactivations were noted. Overall, increased activation of the bilateral dlPFC, and increased deactivation of the DMN was seen over time with increasing task difficulty. Red represents increased activation over time (i.e., significantly increased at two months); blue represents increased deactivation over time. Activations represent statistically significant regions thresholded at KE = 20, with cluster level correction (pFWE < 0.05). Stanford University Functional Imaging in Neuropsychiatric Disorders Lab dlPFC and DMN masks used to create region of interest. Colour bars represent Z-scores for each contrast. Slice numbers are shown above axial slices. Neurological convention displayed (left side of brain shown on left).WM: working memory; dlPFC: dorsolateral prefrontal cortex; DMN: default mode network; LDLPFC: left dorsolateral prefrontal cortex; FWE: family-wise error
Effect of recovery status
Of the 29 symptomatic participants, 15 had clinically recovered by two months post-injury. The recovered and unrecovered participants were similar in handedness (χ2 (1) = 0.05, p = 0.83), average household income (U = 79, p = 0.61), age (U = 60, p = 0.15), pre-injury symptom scores (PSCI-Y; U = 120, p = 0.15), and task performance (measured by A’ score) in all three levels of task difficulty and across both timepoints (all p > 0.05). Children in the unrecovered group were more likely to be female (χ2 = 4.32, p = 0.04), and more likely to have had previous injuries (χ2 = 9.2, p = 0.03). Attention t score (U = 59.5, p < 0.05) and Behavioural Symptoms Index score (U = 52.5, p = 0.04) were significantly lower in recovered compared to unrecovered participants at two months post-injury. In measuring change over time, during the 1back, recovered participants displayed significantly higher intensity of activations in the ventral PCC, deactivations of the dorsal PCC, both activations and deactivations in the right dlPFC (RDLPFC), and activations in the left dlPFC (LDLPFC) compared to their unrecovered counterparts (pFDR < 0.05; Figure 5(a) and (b)). There were three outliers in the unrecovered group (Δ BOLD intensity = 10.87, 10.91, 10.91). These data points were checked and verified to be valid. Although these outliers were included in the final analysis, it is important to note that censoring these values still yielded a trend-level effect (puncorrected = 0.052). During the 2back, recovered participants had greater BOLD signal intensity of activations and deactivations of the LDLPFC.
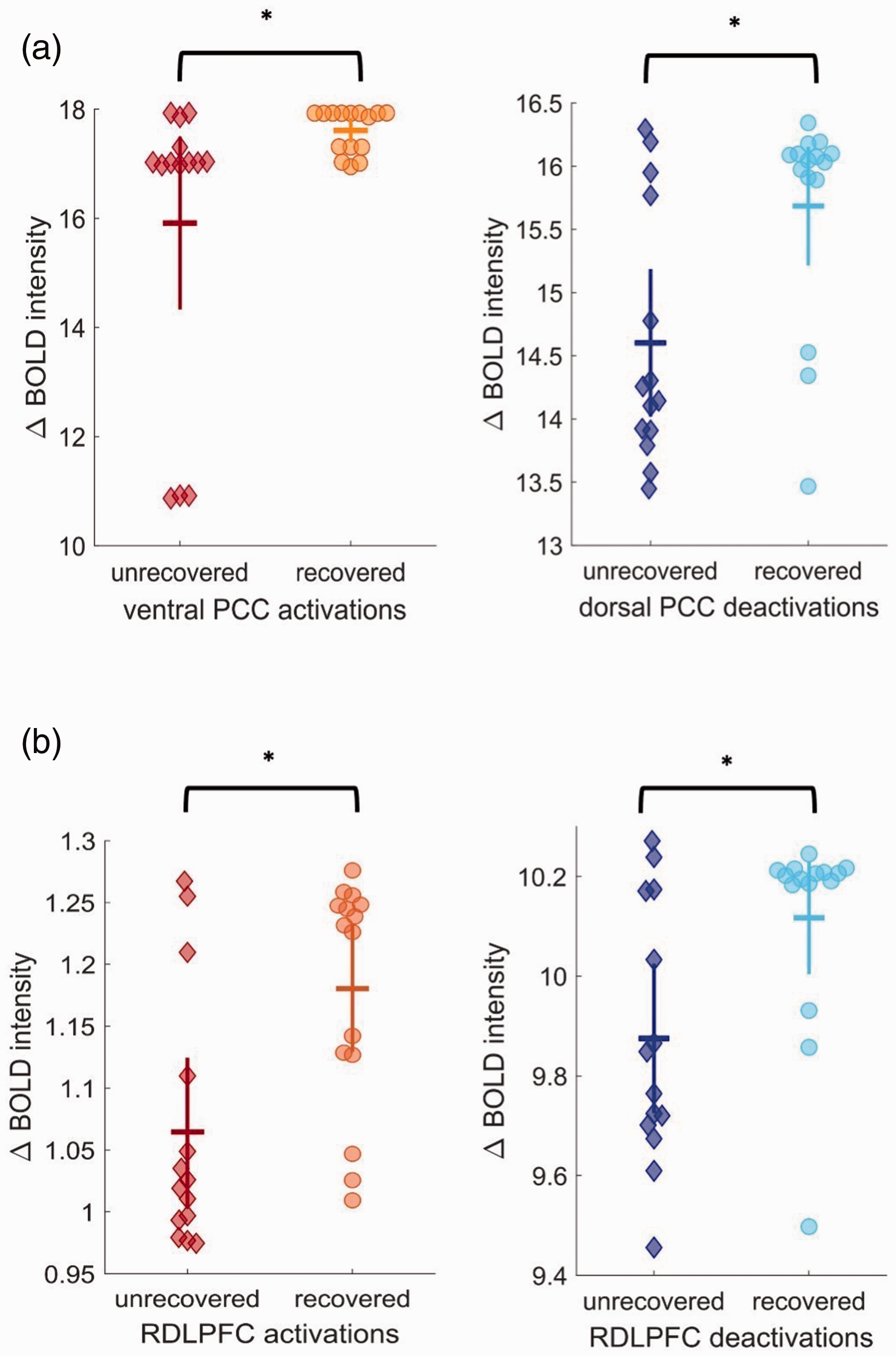
Significant changes in (a) posterior cingulate cortex (PCC) and (b) right dlPFC (RDLPFC) peak BOLD activation and deactivation signal intensity between unrecovered and recovered participants over time during the 1back task (n = 29). Recovered participants displayed greater intensity of activations in the ventral PCC, deactivations of the dorsal PCC, and both activations and deactivations in the RDLPFC compared to unrecovered participants. Recovered participants also showed greater intensity of activations in the left dlPFC (LDLPFC) during the 1back, and both activations and deactivations of the LDLPFC during the 2back (not shown here). Red and dark blue diamonds represent BOLD activations and deactivations in unrecovered participants, respectively; orange and light blue circles represent BOLD activations and deactivations in recovered participants, respectively. BOLD: blood oxygen level-dependent; dlPFC: dorsolateral prefrontal cortex; Δ: change * indicates significance at pFDR < 0.05.
Discussion
This study is the first to examine changes in WM-related cortical responses within the dlPFC and DMN longitudinally in children with PPCS during recovery. Over time, and with increased task difficulty, we found significant increases in dlPFC activation, as well as increases in activation and deactivation of distinct posterior DMN areas (PCC) during a working memory task. The intensity of WM-related cortical activations and deactivations within these regions was higher in participants who had clinically recovered by two months post injury during the 1back task. These results provide a valuable contribution to the scarce literature in the field of pediatric TBI, highlighting the implications of injury during a time of maturation and development.
We have previously reported increased deactivation of the DMN and hypoactivation of the dlPFC during a WM task in children with PPCS at one month post-injury relative to children with mTBI and good recovery, and so postulated that DMN interference may contribute to some of the cognitive symptoms so common in PPCS. 34 In our current study, we hypothesised that over time there would be increased dlPFC activation, and decreased DMN interference. Our hypotheses were partially upheld in that there was increased dlPFC activation over time, which was most prominent in the 2back task. There was also greater deactivation in the PCC (posterior part of the DMN). However, we also found evidence of continuing DMN interference at 2 months post injury (increased PCC activation, and some areas of dlPFC deactivation).
Mild TBI in childhood is sustained during a period of considerable developmental change for WM- and DMN-related areas. In typically developing populations, large-scale maturational changes occur in the regions responsible for healthy WM and DMN functions during teenage years. Maturation of WM-related areas is marked by increasingly stronger recruitment of the dlPFC and a change from activation of diffuse and posterior brain regions to focal and anterior regions. 28 Similarly, network connectivity of the DMN increases as children mature,61–64 and becomes progressively less activated (or increasingly deactivated) during sustained attention tasks. 65 These ongoing changes must be considered when interpreting the results of our study.
The dlPFC has an important role in modulating WM task performance, 66 where increased dlPFC activation has been reported in both adult and adolescent TBI populations during WM tasks. Similarly to our study, Dettwiler et al. 35 studied dlPFC activation during a WM task in 15 adults with clinical recovery from mTBI at 2 weeks, 1 month and 2 months post-injury. Compared to healthy controls, subacute increases in cortical activations were found in bilateral dlPFC regions over time. Previously, we reported dlPFC hypoactivation in children with PPCS at one month post-injury when compared to children who had recovered from their mTBI. 34 In our current study, we found evidence of increased activation of the dlPFC during a WM task over time, especially during the 2back task. In order to assess how changes in cortical activations were influenced by symptom improvement, we compared those children with good and poor recovery. Here, we found that overall net increases in signal intensity of cortical activations and deactivations in the dlPFC over time were linked with recovery. Changes in cortical activity within the dlPFC may be associated with WM-related compensatory mechanisms,67,68 and may demonstrate the broader nature of task engagement networks normally present in children. 28
Suppression of the default mode network is recognized as a correlate of task engagement, where the DMN exhibits linear deactivation as task difficulty increases. 69 In working memory research, the DMN is traditionally regarded as anticorrelated with WM-related areas. Here, increased prefrontal activation is associated with decreased activation in the DMN (i.e. the external focus of attention during WM processing corresponds with decreased internal engagement of DMN attention processes). 31 However, anticorrelation of the executive control network and DMN has been shown to occur following adult TBI. A recent fMRI study in adult chronic TBIs (mild to severe) at approximately 8.5 months post-injury and 5 weeks later found decreased anticorrelation between executive control and posterior default mode networks 32 which was suggestive of possible neural compensation mechanisms occurring in these brain networks during recovery. In our study, we also noted decreased anticorrelation between DMN and executive control networks. This finding provides evidence of similar compensatory mechanisms present in pediatric mTBI to those seen in adult mTBI. In addition, our findings extend observations made within these networks from resting-state data in the same cohort,70,71 where changes in DMN activity between one and two months post-injury may suggest the ongoing presence of neural compensation and cognitive recovery following mTBI.
Neural compensation and recovery seems to be closely tied to posterior regions of the DMN, where regions such as the PCC have been noted to be implicated in mild to moderate TBI. 72 The PCC has been suggested as a highly heterogeneous network hub and shows variable levels of cortical activity depending on arousal state73–75 and age. 76 During maturation, increased anticorrelation of the DMN and task-positive regions has been observed, where between the ages of 10 to 13 years, participants exhibited increasing segregation between the PCC and executive areas. 76 The PCC is affected in mTBI, where functional connectivity studies have shown abnormally high PCC connectivity to be associated with more efficient response speeds, which may reflect a novel mechanism for cognitive recovery after TBI. 31 In healthy populations, the PCC shows a complex functional organisation, where multiple functionally-distinct networks have been identified within the region. 77 A previous study by Sharp et al. found that as cognitive demands increased, the ventral aspect of the PCC displayed less integration with the rest of the DMN and less anticorrelation with the executive network (including dlPFC); whereas the dorsal aspect of the PCC exhibited better integration within the DMN and increased anticorrelation with the executive network. 77 In a later study, the same group found that PCC deactivation during an attentionally-demanding task was isolated to signals arising from the dorsal PCC. 78 We noted a similar anticorrelation pattern in our study, where distinct increases in ventral PCC activation and dorsal PCC deactivation occurred with increasing task difficulty over time, alongside improved task performance. These results were further supported by changes noted in the PCC of recovered, compared to unrecovered participants over time. It is important to note that direct testing for correlation between ventral PCC activity and cognitive performance, as well as ventral PCC activity and PCSI scores could not be directly tested due to insufficient data. Thus, these relationships should be treated as inferences rather than direct correlations. Nonetheless, we observed that the intensity of posterior DMN activations and deactivations significantly increased in recovered participants, compared to unrecovered participants. Notably, the changes in ventral PCC activation may predominantly reflect the contribution of three outliers (shown in Figure 5); however, a trend-level effect was still noted if these outliers were censored. Thus, decreased power in our study may have influenced these data points. These recovery-specific changes also reflected a dorsal and ventral PCC distribution. Overall, the differences in ventral and dorsal PCC activity are likely to suggest evidence of DMN normalization in our participants over time. 77
Although this is one of few longitudinal task fMRI studies following mTBI and the only longitudinal study in children with PPCS, it has several limitations. The most prominent limitation is the lack of a control group in this study, particularly at the second study timepoint, which affects interpretations of changes in cortical responses over time during WM processing. Comparisons of healthy and mTBI participants with early recovery could provide more information on the natural history of cortical activations seen over time. However, our results suggest that it is reasonable to assume that the cortical activations seen over time related to hyperactivation in mTBI recovery. In addition, participant refusal to undergo repeat imaging significantly decreased power. Moreover, during this study, we used a continuous nback WM task, which has a good test-retest reliability. 79 However, the nback was structured so that participants only pressed a button when the stimuli matched, and not when there was a mismatch. Although this was implemented to make the task less complicated and easier to understand for the children, as well to reduce motion artifacts, it may have decreased the sensitivity of task performance. Finally, the exclusion of participants with serious illnesses or neurological conditions may limit the generalisability of our findings; however, it reduced the likelihood of confounding. 80
It is important to note that most available literature relating to cortical activation during recovery from mTBI encompasses adult populations, and thus it is unclear whether the discussed findings can be generalised to a child population. Despite the potential differences in WM and DMN circuitry between adults and children, there is a paucity of longitudinal fMRI studies conducted on WM in pediatric mTBI. In order to accurately understand deficits in WM following mTBI in children, further longitudinal pediatric studies during recovery from mTBI are crucial. In the future, functional connectivity studies measured during executive functioning task paradigms should be conducted to investigate the longitudinal relationship between dlPFC and DMN networks both in children with mTBI and matched control groups. These task-related connectivity findings have the potential to extend upon key observations of whole-brain resting-state networks in childhood mTBI, and provide further insight into the dynamic reconfiguration of brain networks and how they relate to cognitive performance outcomes resting-states. 81
Conclusions
We examined the changes in cortical responses within working memory- and default mode-related areas over time in children with persistent post-concussion symptoms during recovery from mTBI. Over time and with increasing task difficulty, increases in bilateral dlPFC activation and both activation and deactivation in distinct areas of the PCC were noted, the intensity of which were increased in recovered participants. Our findings extend results from our previous study, thus suggesting neural compensation and ongoing recovery of cognitive functions over time between one and two months post injury. On the whole, this study has provided valuable insights into WM functions during longitudinal recovery from pediatric mTBI. Our findings contribute to understanding changes in activity of the dlPFC and DMN during increased working memory loads in children with PPCS and highlight the wider implications of mTBI in children, whose maturing brains are particularly vulnerable to TBI.
Footnotes
Acknowledgements
We extend our gratitude to all participants who were involved in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Canadian Institutes of Health Research (Grant 293375), the Alberta Children’s Hospital Research Institute, and the University of Calgary.

