Abstract
Objective
To report the observed effect of erenumab in mild posttraumatic headache migraine phenotype with and without aura.
Background
There is no clinical algorithm of pharmacotherapy for migraine following posttraumatic headache. Most migraine preventatives that are typically used are either ineffective or not tolerated.
Methods
Seven patients who met the clinical criteria for migraine with or without aura following posttraumatic headache who failed or were intolerant of conventional migraine preventatives were treated with erenumab 140 mg subcutaneously. Most had no history of migraine. In those patients with a history of migraine, the posttraumatic headache migraine headaches were different than the past migraine experience. Descriptive headache intensity or disability using the Head Impact Test-6 and monthly headache days were recorded before and after treatment. All patients were debilitated on presentation and demonstrated no signs of spontaneous resolution.
Results
Patients responded with a 95% (SD 1.22, p < .001) reduction in headache days. All Head Impact Test-6 scores went from disabling to non-disabling without adverse effects. Most required only one dose of erenumab with no migraine recurrence. Onset of efficacy often became apparent within days to four weeks. Extended follow-up six months after treatment revealed no relapses.
Conclusions
Erenumab is effective in the treatment of posttraumatic headache with migraine phenotype in this small cohort. Large-scale studies are urgently required for this highly prevalent, disabling, condition which has no effective established treatment.
Introduction
The prevalence of posttraumatic headache (PTH) is approximately three million of which 20% are adolescence. 1 Most patients who present with PTH will manifest migraine phenotype which has all the characteristics of migraine with and without aura.2,3 All patients represented in this observational study fell under the International Classification of Headache Disorders-vs 3 (ICHD3) classification as acute headache attributed to mild traumatic injury to the head 5.1.2. 4 The PTH phenotype was migraine: typical features include throbbing pounding headache, photophobia, nausea, vomiting, visual aura, brainstem aura, and inability to engage socially, in school, or work. Most of these patients are treated with preventatives used in non-traumatic migraine patients, but the response is usually inadequate.
Erenumab is a CRRP-receptor monoclonal antibody which is Food and Drug Administration (FDA)approved for the preventative treatment of migraine. It has an excellent safety profile, impressive efficacy data, can be effective as quickly as one week, and has virtually no adverse effects except for very low incidence of injection site reactions and constipation. Erenumab was the first of the three current FDA approved CGRP monoclonal antibodies. It is the only antibody targeting the CGRP receptor. Of the three FDA approved CGRP monoclonal antibodies, erenumab has the longest (3 yr) open label extension data. 5 Six patients were treated with erenumab with clinical migraine phenotype following PTH.
Methods
All patients met the criteria for migraine with or without aura as delineated in ICHD3. The etiology of their migraines was head trauma which was defined as PTH secondary to mild traumatic injury to the head. All patients had normal exams and head imaging. Three patients had a remote history of migraine six months or longer with symptoms that differed from the PTH-induced migraine phenotype and their history of migraine. General informed consent was obtained from all patients. Erenumab is FDA approved for the preventative treatment of migraine with and without aura. An AHS publication recently recommended using CGRP mAbs in the pediatric population where other preventives have failed or not tolerated. 6 All patients failed prior treatments and were disabled from migraine with intense high-frequency headache, and inability to work, attend school, or engage in social circles. Ineffective preventative treatments given for at least four weeks alone or in combination which were discontinued with erenumab administration included daily topiramate, amitriptyline, gabapentin, doxepin, venlaxafine, memantine, ibuprofen, naproxen, and ondesterone. Abortives such as non-steroidal anti-inflammatory drugs (NSAIDS) triptans, or over the counter non prescription (OTC) analgesics were allowed but were never taken daily and their need tapered off quickly with the rapid response to erenumab. The patients were relatively healthy and confounding variables such as non-morbid obesity and concurrent minor illnesses were not significant to be considered in the outcome analysis. No patient had medication overuse headache syndrome, pregnancy, secondary gain motivation, or psychiatric disorders. Baseline monthly headache days (MHDs) and Head Impact Test-6 (HIT-6) were recorded and restated two months after receiving erenumab 140 mg. Only one patient (patient 5) required a second dose, and the MHD and HIT-6 were recorded two months after the second dose. Doses were administered in the office by a certified medical assistant. Data were assessed retrospectively.
Results
Patients experienced a 95% (SD 1.22, p < .001) reduction of MHDs in two months, with headache remission often commencing within a week of treatment. Prior to treatment with erenumab, all six patients had HIT-6 scores above 60 indicating severe disability. Two months posttreatment, all HIT-6 scores fell below 49 which is no disability. Patients were very satisfied with the reduction of headache, photophobia, nausea, and dizziness. All patients returned to normal activities of daily living, work, and school activities. Following discharge from treatment, patients required either no abortives or rare use of OTC analgesics. Those patients with a history of migraine had no difference in response to erenumab. There were no adverse effects reported. There were no relapses. Findings are summarized in Table 1.
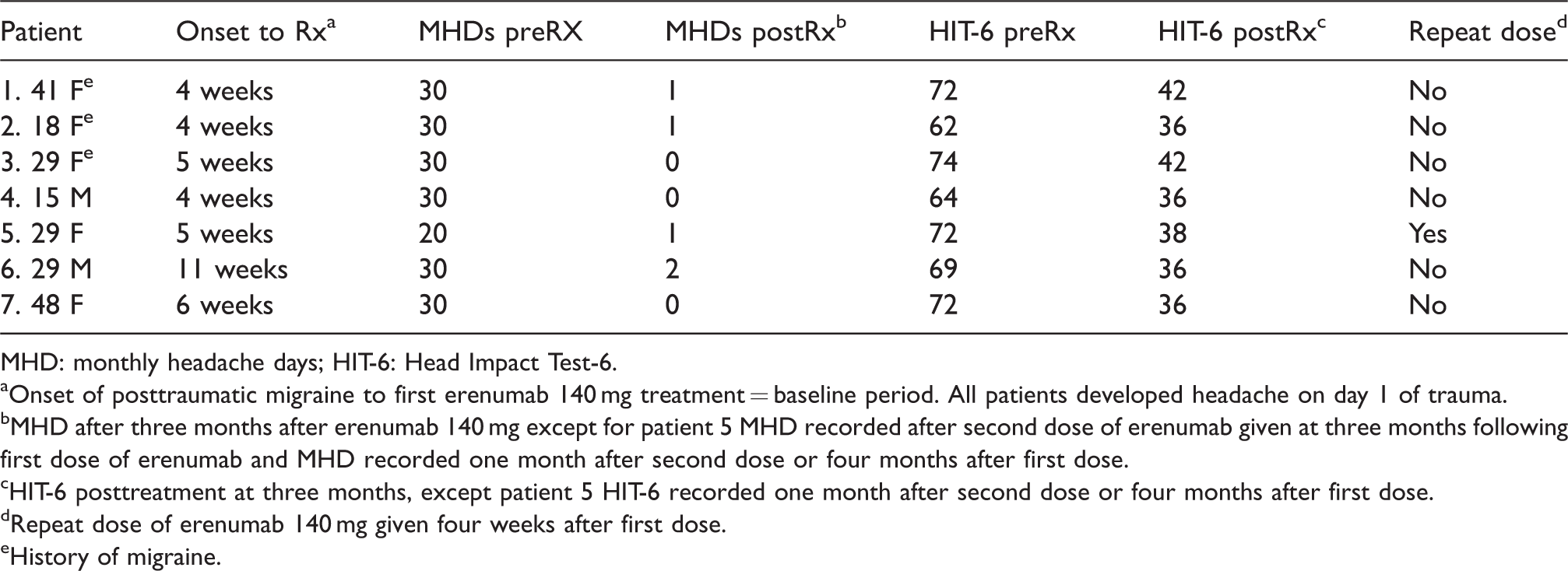
MHD: monthly headache days; HIT-6: Head Impact Test-6.
aOnset of posttraumatic migraine to first erenumab 140 mg treatment = baseline period. All patients developed headache on day 1 of trauma.
bMHD after three months after erenumab 140 mg except for patient 5 MHD recorded after second dose of erenumab given at three months following first dose of erenumab and MHD recorded one month after second dose or four months after first dose.
cHIT-6 posttreatment at three months, except patient 5 HIT-6 recorded one month after second dose or four months after first dose.
dRepeat dose of erenumab 140 mg given four weeks after first dose.
eHistory of migraine.
Discussion
These seven patients were severely symptomatic from migraine that followed mild posttraumatic head injury. Each patient went from severe disability to no headache disability and 95% reduction of MHDs with the reduction often commencing within a few weeks of receiving erenumab 140 mg. There were no adverse effects.
The real impact of erenumab on these patients was striking. Patient 6 was a 29 y/o laborer who fell off a truck, hit head, and experienced daily migraine with no history of migraine. For 11 weeks he could not return to work and was unresponsive to vestibular therapy, topiramate, amitriptyline, gabapentin, and non-daily triptan and NSAID abortives. Three weeks after one dose of erenumab, he went back to work with rare headaches. Patient 4 was a 15 y/o male student wrestler who was thrown to the floor during wrestling with head trauma, had daily migraines and could not attend school due to severe pounding headaches and photophobia worsened with academic engagement. Within five days of erenumab, he was asymptomatic and back to his academic classes.
There are no FDA approved treatments for PTH with migraine phenotype. Studies have shown that conventional treatment of PTH migraine phenotype results in 26% relief and 74% no relief.7,8 Differences in regional volumes, cortical thickness, surface area, and curvature were noted comparing PTH migraine phenotype to migraine patients using functional magnetic resonance imaging. 9 However, CGRP receptors are ubiquitous in the cortex of the entire brain. 10 The trigeminal vascular access of CGRP-R mAbs via the meninges may explain the robust response of these patients to erenumab. Perhaps there is amplification of the trigeminovascular system in PTH migraine phenotype which would account for the failure of conventional migraine preventatives which do not directly antagonize CGRP receptors which is the mechanism of action of erenumab. The presence of pre- and postsynaptic receptors can explain the efficacy of CGRP-mAbs. In a rat model of concussion, headache and pain-related behaviors were mediated through a CGRP-dependent mechanism. Concussed rats’ tactile pain and hypersensitivity were inhibited by anti-CGRP antibody treatment. 11
Conclusions
This is a small observational study on the effect of erenumab on patients with PTH-migraine phenotype with no control group. A placebo effect cannot be excluded, and the original studies that led to FDA approval of erenumab for episodic and chronic migraine demonstrated efficacy with a placebo effect of approximately 30%. None of these patients had psychiatric or secondary gain issues, or currently active preexistent migraine. Many had failed previous migraine preventatives. The efficacy and safety of erenumab 140 mg in the virtual elimination of migraine was extremely impressive with no adverse effects. Concussion with PTH migraine phenotype is a public health problem. This pilot observation study should lead to larger studies to validate these findings and determine if erenumab, which is safe and effective, should be in the forefront for the treatment of PTH with migraine phenotype.
Footnotes
Author’s contribution
JAC: Had full access to all the data in the study, integrity of the data and the accuracy of the data analysis; study concept and design; analysis; interpretation of data; drafting of the manuscript; critical revision of the manuscript for important intellectual content; statistical analysis; technical or material support; study supervision. JAC is the speaker for Supernus and Amgen.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
