Abstract
Aim
To determine whether acute sport-related concussion is associated with functional brain changes in Australian rules footballers.
Methods
Twenty acutely concussed professional Australian footballers were studied with 3 T magnetic resonance imaging and compared to 20 age-matched control subjects. We statistically compared whole-brain local functional magnetic resonance imaging connectivity between acutely concussed footballers and controls using voxel-wise permutation testing.
Results
The acutely concussed football players had significantly decreased local functional magnetic resonance imaging connectivity in the right dorsolateral prefrontal cortex, right inferior parietal lobe, and right anterior insula, compared to controls. No functional brain changes between groups within the default mode network were observed.
Discussion
Acutely concussed footballers had in common decreased functional connectivity within the right lateralized “cognitive control network” of the brain that is involved in executive functions, and the “salience network” involved in switching between tasks. Dysfunction of these brain regions is a plausible explanation for typical clinical features of concussion.
Keywords
Introduction
Sport-related concussion (SRC) refers to a subset of mild traumatic brain injury that is induced by biomechanical forces and typically results in the rapid onset of impairment of neurological function. While most athletes recover uneventfully, SRC may be associated with persistent symptoms or adverse outcomes in some individuals. 1 The acute sequelae of SRC may include an increased risk of musculoskeletal or neurological injury on return to play. 2 The risk factors and evidence for long-term neurodegenerative sequelae are inconclusive; 3 however, some individuals seem to be at greater risk of mood, neurobehavioral and cognitive problems. 4
The clinical diagnosis of acute SRC is based on assessment of a range of domains including: symptoms (e.g. headache, difficulty concentrating, feeling like in a fog, emotional lability, etc.); physical signs (e.g. loss of consciousness, balance disturbance); cognitive impairment (e.g. confusion, slowed reaction times); and neurobehavioral changes (e.g. irritability, not feeling quite right). 1 In the clinical setting, the diagnosis of SRC remains challenging especially given the variable presentation, evolving clinical features, nonspecific symptoms and the absence of a reliable direct biomarker of injury and recovery. 5
The underlying biological basis for both acute and possible long-term problems is not clear. The current consensus is that the acute clinical features of SRC largely reflect a disturbance of brain function rather than a structural or neuropathological injury and, by definition, no abnormality is seen with clinical structural neuroimaging. 6 Meta-analytic findings suggest that cortical regions within three distinct functional brain networks are altered following mild brain trauma. 7 These include (a) the cognitive control network responsible for maintenance of attention and working memory—right frontal cortex and right intraparietal lobule; 8 (b) the salience network responsible for task-switching—right anterior insula and anterior cingulate cortex; 9 and (c) the brain’s core default mode network which is involved in introspective functions—precuneus/posterior cingulate cortex, superior parietal lobules, and ventromedial prefrontal cortex. 10
We hypothesized that voxel-wise functional magnetic resonance imaging (fMRI) would reveal altered local connectivity in these networks, in Australian rules footballers with acute SRC.
Materials and methods
Subjects and ethics
Australian rules football is a sport with frequent and high-velocity head impacts that often results in concussion. 11 All subjects were recruited from professional Australian Football League (AFL) teams within one month of sustaining a concussion, here defined as acute concussion in line with previous reports. 12 Experienced team doctors at each club, who were present at the time of injury, made the diagnosis of concussion in each case according to standardized injury definitions. 12 Inclusion criteria included witnessed impact including the use of side-line video review; 13 the presence of signs and symptoms of SRC; and an abnormal SCAT3 examination. All concussed players recruited into the study had clinical symptoms and/or signs of SRC lasting longer 72 h. AFL team doctors have a consistent approach to the diagnosis and management of SRC due to ongoing research and education programs over the past 20 years.14,15 All players referred in to the study were subsequently reviewed by one of the investigators (MM) at the time of the scan and tested for symptoms and cognitive impairment (including the CogState Sport computerised cognitive screening test battery). In total, 20 professional male AFL footballers were recruited after having suffered a concussion. Twenty age-matched male controls were also recruited; 6/20 healthy controls included nonconcussed footballers from the AFL. All control subjects were age matched and neurologically normal with no history of concussion in the past six months. The 6/20 nonconcussed football controls had a similar past concussion history to the players referred into the study following SRC. All subjects gave informed consent for participation in the study. The study was approved by the University of Melbourne Human Research Ethics Committee (ID: 0830367).
MRI acquisition
All imaging was performed on a 3 T Siemens Skyra MRI system (Siemens, Erlangen, Germany). Data acquisition included a resting-state fMRI sequence to assess functional connectivity and a T1-weighted anatomical image for quality control and spatial normalization purposes. 10 min of resting-state fMRI data (200 volumes) were acquired with 44 interleaved 3 mm slices, repetition time = 3000 ms, echo time = 30 ms, voxel size = 3 × 3 × 3 mmand acquisition matrix of 72 × 72. T1 weighted images were acquired using an MPRAGE sequence, with repetition time = 1900 ms, echo time = 900 ms, voxel size 0.9 × 0.9 × 0.9 mm3, and acquisition matrix of 256 × 256 × 192.
fMRI pre-processing
The acquired resting-state fMRI data were preprocessed using an in-house pipeline based on scripts from the Statistical Parametric Mapping 12 toolbox (http://www.fil.ion.ucl.ac.uk/spm/) and the Data Processing & Analysis for Brain Imaging toolbox. 16 Functional images were slice-time corrected and realigned before co-registering to the subject’s T1 weighted image. The T1 weighted images for each subject were segmented into grey matter, white matter, and cerebrospinal fluid components, and spatially normalized using a diffeomorphic registration algorithm to create an average group template in Montreal Neurological Institute coordinate space. 17 Functional images were transformed to the same space by application of a concatenation of the subject’s coregistration and group spatial normalization transformations. Average white matter and cerebrospinal fluid signal and 24 motion parameters 18 were regressed from the functional data. The data were further filtered to exclude frequencies outside the range of 0.01 to 0.08 Hz. To mitigate potential adverse effect of excessive head movement we excluded from analysis any fMRI time-points with head-movement exceeding 0.50 mm. 19
fMRI local connectivity analysis
Local functional connectivity was assessed using regional homogeneity, 20 a metric widely used to study whole-brain local network function, 21 also in mild traumatic brain injury.22,23 Regional homogeneity is a voxel-wise measure that quantifies the Kendall’s coefficient of concordance between an fMRI time-series of a single voxel and its neighbors within a 3 × 3×3 voxel cube: that is, the concordance between a voxel and its 26 nearest neighbors (i.e. synchronicity within 0.729 cm3 of cortex). After regional homogeneity calculation, the voxel-wise spatial maps were intensity-normalized into z-scores 24 and smoothed using an 8-mm full-width-at-half-maximum Gaussian kernel.
Voxel-wise statistical analysis
We used a mass univariate two-sample t-test to determine voxel-wise fMRI local connectivity differences between groups using FSL’s randomise (a nonparametric permutation inference tool). 25 We used 5000 permutations in this study, which means that we generated a statistical null distribution by randomly re-allocating all subjects 5000 times. This whole-brain and voxel-wise permutation approach also corrects for multiple comparisons at a family-wise error rate of p < 0.05.
Results
Standard structural MRI (including T1 and T2 weighted images) was assessed by experienced reviewers for each individual subject, and no abnormalities were detected in any of the acutely concussed or control subjects. In-scanner head motion was similar between the two groups (Table 1).
Overview of subjects included in this study.
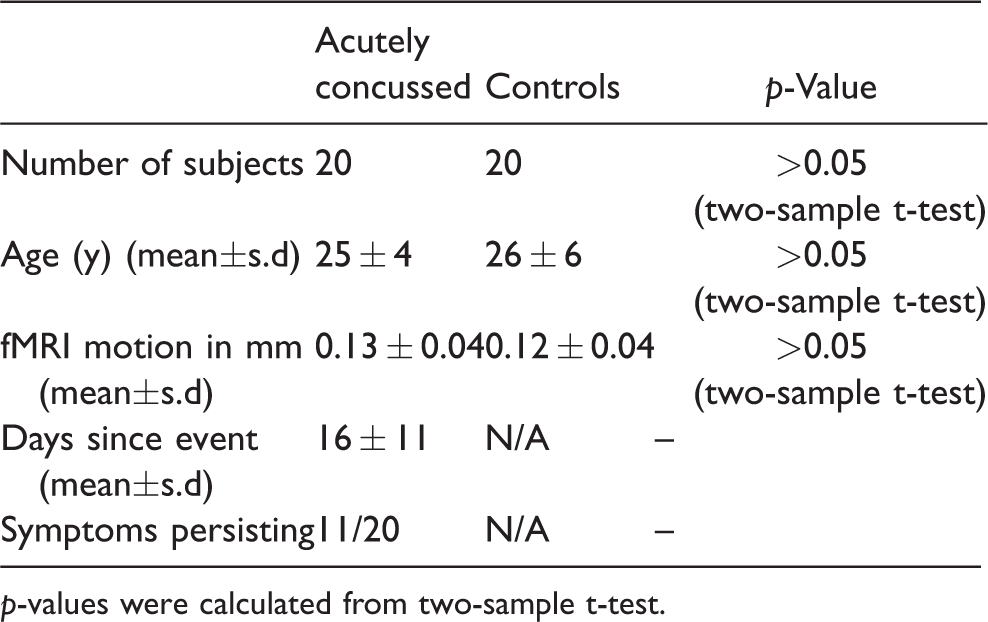
p-values were calculated from two-sample t-test.
Local connectivity is decreased in concussed footballers
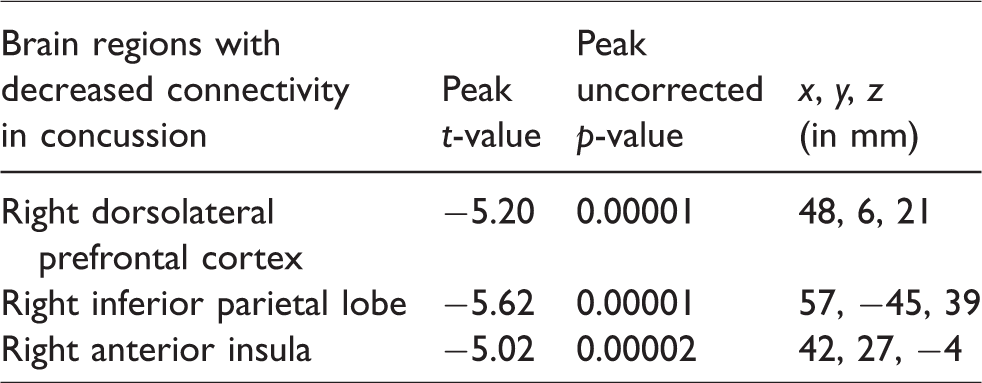
Whole-brain and voxel-wise regional homogeneity showed fMRI local connectivity differences between groups in the right hemisphere. As seen in Figure 1 and Table 2, acutely concussed subjects displayed significantly decreased local connectivity in the right dorsolateral prefrontal cortex extending into right inferior frontal cortex, right inferior parietal lobe, and the right anterior insula, compared to controls (family-wise error p < 0.05, using threshold-free cluster enhancement with 5000 permutations). See also Supplementary Figure 1 for an unthresholded voxel-wise statistical fMRI local connectivity map.
Summary statistics: threshold-free cluster enhancement (family-wise error rate p < 0.05; 5000 permutations).
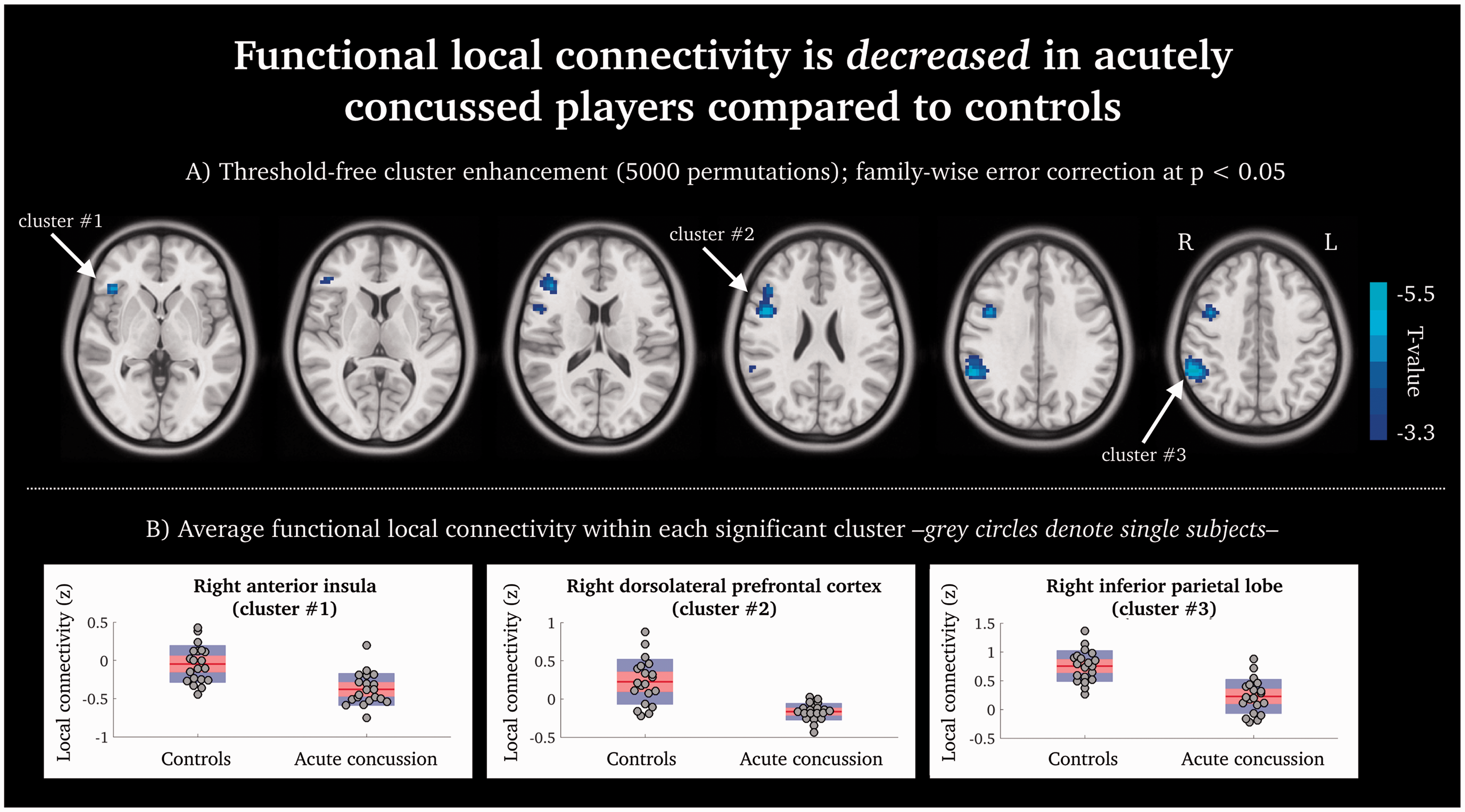
Functional local connectivity is decreased in acutely concussed players compared to controls. (a) Statistically significant brain regions at family-wise error rate p < 0.05; 5000 permutations using threshold-free cluster enhancement. Cool-scaled color bar denotes the magnitude of voxel t-values. (b) Individual-level z-normalized fMRI local connectivity values where each grey circle denotes an individual subject; red line is the group mean; red shaded area is the 95th percentile of the mean value; and blue shaded area is one standard deviation from the mean.
Exploratory analyses were also undertaken by computing Pearson correlation coefficients between fMRI local connectivity (average scores within the right dorsolateral prefrontal cortex, right inferior parietal lobe, and right anterior insula) and time since concussive event, rate of recovery, and neurocognitive test performance, across all concussed footballers. No significant correlations were found after correcting for multiple comparisons, although our study is likely underpowered for neurocognitive and correlational analysis.
Discussion
SRC and its impact on the pathophysiological functioning of the brain has received widespread interest in recent years. 26 However, we still do not fully understand physiological brain changes that occur after a concussion, and conventional clinical neuroimaging is usually normal. Therefore, we utilized an advanced MRI method to help quantify functional MRI changes in acutely concussed Australian rules footballers. We observed differences between acutely concussed Australian rules footballers and controls in brain regions comprising the right lateralized cognitive control network and the salience network (Figure 1), concordant with previous meta-analytic findings in people with mild traumatic brain injury. 7 These are brain networks known to be lateralized to the right hemisphere.8,9 Contrary to previous reports, we did not observe any functional brain changes within the default mode network.
Functional brain changes in acutely concussed footballers
We found decreased intrinsic fMRI connectivity within right fronto-parietal regions in acutely concussed footballers. These regions are involved in a cognitive control network that is important for attention and goal directed behaviors. 27 We also observed decreased intrinsic fMRI connectivity within the anterior insula, which is a part of the brain's salience network important for task-switching. 9 The cognitive control network is also switched ‘on’ and ‘off’ by the right anterior insula, given its role in regulating brain states. 28 These functional deficits are likely to be a part of wider networks affected by the head impact. Dysfunction of these networks may explain some of the most common cognitive symptoms seen in acute SRC.
Intuitively, a “global” impact to the head might be expected to lead to diffuse and widespread brain changes. Data from finite element analysis modeling of unhelmeted SRC in Australian rules football and rugby show regional differences in tissue deformations in the brain. 29 There are examples in the traumatic brain injury literature that “global” head impacts are associated with selective brain alterations as determined by neuroimaging. Sharp et al. 30 have outlined several accounts of selective damage to the anterior insula and anterior cingulate cortex (i.e. the right lateralized salience network) and the brain’s default mode network. We observed that the group level effects of acute SRC are limited to brain structures in the right hemisphere and observed no changes within the brain’s core default mode network. We postulate that this is due to the relatively mild head injury these football players experience, compared to other traumatic brain injury cohorts. 31 For example, Bonnelle et al. 32 demonstrated that the structural deterioration of the default mode network is a predictor of persistent cognitive symptoms in moderate/severe traumatic brain injury.
Limitations and future directions
One limitation of our study is the varying time since injury across concussed footballers. This is an inevitable consequence of recruitment of players from professional sport, where the benefits of having a homogeneous group (diagnosed and managed by experienced team doctors) must be balanced against the timing of assessment/imaging. Nevertheless, we observed that all twenty concussed footballers had similar functional connectivity scores (Figure 1(b)), suggesting that dysfunction of fronto-parietal and insular structures persist throughout the first month after the head impact. This is consistent with findings by Maier et al. 23 who show greater fMRI local connectivity abnormalities at one month, compared to one week, following SRC in collegiate athletes. Longitudinal research is needed to further understand the evolution and recovery of brain changes in Australian rules footballers. Such studies will better define the time course of changes following concussion.
Another limitation of this study is that only 6 of 20 control subjects were nonconcussed professional footballers, despite making considerable effort to recruit such subjects. However, permutation testing creates a null distribution where all the 40 subjects from our two groups (group 1 = 20 acutely concussed footballers; group 2 = 20 matched healthy controls) are randomly allocated into new group assignments, a process that is repeated 5000 times. Given that the six nonconcussed footballer controls were ideal controls for this procedure, and are included in this permutation testing, it is unlikely that we could observe any significant differences in functional connectivity between our groups if nonconcussed footballers had the same brain patterns as concussed footballers.
It is also important to note that our results reflect brain changes that concussed footballers have “in common” as a group. Individual football players may also display additional areas of fMRI local connectivity changes, with our group findings reflecting a subset of areas that are common to the diagnosis of concussion. In future, it will be important to further characterize functional brain changes within individuals, understanding those that are biomarkers of concussion, and which features predict ongoing or chronic post-concussion symptoms. In this study, we focused on local fMRI connectivity changes in acute concussion. In future work, it may be beneficial to examine more spatially distant or global fMRI connectivity. For example, given our present results, one might hypothesize long-range changes between brain regions located in the right sided salience and central executive network.
In conclusion, we observed functional brain changes in the right fronto-parietal and anterior insular cortex in acutely concussed Australian rules footballers compared to a group of matched controls. Dysfunction of these brain areas may provide a plausible explanation for some of the typical clinical features of concussion.
Supplemental Material
Supplemental material for Functional brain effects of acute concussion in Australian rules football players
Supplemental Material for Functional brain effects of acute concussion in Australian rules football players by Graeme D Jackson, Michael Makdissi, Mangor Pedersen, Donna M Parker, Evan K Curwood, Shawna Farquharson, Alan Connelly, David F Abbott and Paul McCrory in Journal of Concussion
Footnotes
Acknowledgements
The authors wish to thank the AFL Doctors Association, AFL Players Association, Dr. Patrick Clifton (AFL Operations and Innovations Manager) and Dr Peter Harcourt (AFL Medical Director) for their support of the project. They also thank all the recruited subjects who gave their valuable time to participate in this study. The Florey Institute of Neuroscience and Mental Health acknowledges the strong support from the Victorian Government and in particular the funding from the Operational Infrastructure Support Grant. We also acknowledge the facilities, and the scientific and technical assistance of the National Imaging Facility (NIF), a National Collaborative Research Infrastructure Strategy (NCRIS) capability, at the Florey node and The Victorian Biomedical Imaging Capability (VBIC).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Paul McCrory is a co-investigator, collaborator, or consultant on grants relating to mild TBI funded by several governmental organizations. He is co-chair of the International Concussion in Sport Group. He has been reimbursed by the government, professional scientific bodies, and sporting bodies for travel costs related to presenting research on mild TBI and sport-related concussion at meetings, scientific conferences, and symposiums. He received consultancy fees in 2010 from Axon Sports (US) for the development of educational material (which was not renewed) and has received research funding since 2001 from CogState Inc. He does not hold any individual shares in any company related to concussion or brain injury assessment or technology. He did not receive any form of financial support directly related to this article.
Dr Michael Makdissi is a consultant Sport and Exercise Medicine physician at Olympic Park Sports Medicine Centre and team physician for the Hawthorn football club (AFL). He is a member of the International Concussion in Sport Group. He receives research funding from the AFL and nonfinancial research support from CogState Pty Ltd. He has attended meetings organized by the International Olympic Committee (IOC), National Football League (USA), National Rugby League (Australia), and FIFA (Switzerland); however, he has not received any payment, research funding, or other monies from these groups other than for travel costs. He is an honorary member of concussion working/advisory groups for AFL, Australian Rugby Union, and World Rugby.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the National Health and Medical Research Council (NHMRC) of Australia, grant number #1127007, #1060312 (GJ Practitioner Fellowship) and #1026383 (PM Practitioner Fellowship). DA and SF are supported by fellowship funding from NIF. Funding for the MRI scans was provided by the AFL.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
