Abstract
Introduction
Toxic epidermal necrolysis (TEN) is one of the most severe cutaneous adverse reactions with a mortality rate of 30%. Due to a lack of consensus regarding the treatment and management of TEN, therapy is individualized on a case-to-case basis.
Purpose
The scientific literature about Stevens-Johnson Syndrome (SJS) and TEN is summarized and assessed to aid and assist in determining the optimal course of treatment.
Methods
PubMed and Google Scholar, among others, were searched with the keywords: “Toxic Epidermal Necrolysis”, “corticosteroids”, “cyclosporine”, “etanercept”, “intravenous immunoglobulin”, “Stevens-Johnson syndrome” and filtered by year. The research articles generated by the search, and their references, were reviewed.
Results
TEN is a severe dermatological condition that is mainly caused by medicines. World-wide guidelines differ in care plans. As there is no consensus on the management of TEN, this article aims to summarize the efficacy and feasibility of the management aspect of TEN from previous studies. Supportive care is highly accepted, along with early discontinuation of all medicines (hydration & electrolytes). Corticosteroids and cyclosporine have been used in therapy. Intravenous immunoglobulin (IVIG) is currently being administered; however, their efficacy by themselves and in combination remains uncertain.
Conclusion
Current evidence predominantly from retrospective studies suggests no individual treatment has sufficient efficacy and a multi-faceted regimen stands to be favored. Therapeutic regimens from corticosteroids to IVIG are under constant evaluation. The life-threatening nature of TEN warrants further confirmation with more extensive, robust randomized, controlled trials.
Lay Summary
Toxic epidermal necrolysis (TEN) is a serious skin reaction with a 30% chance of mortality. Commonly TEN is caused by medicines and results in a burn like appearance and sensation in patients. Usually administered medicine is cleared effectively by the human body but when the clearance of few metabolites from medicine is disrupted due to few genes, it leads to an ominous response by the body. This response involves several intermediate chemicals that primarily attack skin cells. Treatment guidelines differ globally. Supportive care is highly accepted, along with early discontinuation of all medicine. Currently, a multi-faceted treatment regimen is favored. Treatments like corticosteroids to immunoglobulins are under constant evaluation. Identification of the perfect combination of treatment needs confirmation from robust randomized controlled trials.
Introduction
Toxic epidermal necrolysis (TEN) is a drug-induced potentially fatal condition with mortality ranging from about 15 to 60 %. 1 Given its elusive pathogenesis and contradictory data regarding its therapy, this article attempts to summarize the continuously expanding information regarding various adjunctive therapeutic options for Stevens-Johnson Syndrome (SJS)/TEN.
Methodology
An articles search was carried out using several electronic databases, including PubMed, Google Scholar, MEDLINE, SCOPUS, and Cochrane databases with the appropriate key terms. The articles were searched specifically for the treatment of TEN and SJS. The initial literature search revealed 10,122 articles, which are detailed in Figure 1. All the published articles reported with a description of TEN and its treatment were included in this review.

PRISMA flow chart.
Discussion
Stevens reported the unique presentations of severe purulent conjunctivitis with eruptive fever and stomatitis in 1922, which is now known as the Stevens-Johnson Syndrome (SJS). 2 Later, it was found that erythema multiforme (EM) major and SJS are clinically distinct disorders. 3 In 1956, Lyell used the term ‘toxic epidermal necrolysis (TEN)’ to describe the presentation of a toxic eruption with burn-like sensations and appearances. 4 They can be described as Severe Cutaneous Adverse Reactions (SCARS) characterized by fever and mucocutaneous lesions leading to necrosis and sloughing of the epidermis. Other examples of SCARs include Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS syndrome) and Acute Generalized Exanthematous Pustulosis (AGEP). 5
TEN/SJS can be induced by Allopurinol, Trimethoprim-sulfamethoxazole, and other sulphonamide-antibiotics, aminopenicillins, cephalosporins, quinolones, carbamazepine, phenytoin, phenobarbital and Non-steroidal anti-inflammatory drugs (NSAIDs) of the oxicam-type. 6 In rare cases, it could be caused by an infection by Mycoplasma pneumoniae and Herpes simplex virus.7,8 In fact TEN can be caused by a few malignancies, connective tissue disorders and graft-versus-host disease. The human leukocyte antigen HLA-B*1502 increases susceptibility when induced by carbamazepine. 9
TEN and SJS are two ends of a spectrum, classified by the percentage/extent of the epidermal detachment- SJS (<10% of the body surface [BSA]), TEN (>30% of BSA) and SJS/TEN overlap syndrome (10–30% of BSA).10,11
Clinical presentation
SJS/TEN is an immunological reaction that primarily involves mucocutaneous eruptions. It begins with diffuse erythema, malaise and fever followed by macules development on the face, neck, and torso, which eventually merge to form a larger bulla that is sloughed over the next few days to form erosions and crusts. 5 Often nail and eyebrow loss is involved with keratoconjunctivitis and oral crust and erosions. As the condition progresses, keratinocyte necrosis leads to epidermal detachment and painful blisters.
SJS/TEN patients are vulnerable to cutaneous infections owing to compromised membrane integrity. 12 Aside from the cutaneous presentation, it has ophthalmological and genitourinary involvement and is known to compromise several organs. Glomerulonephritis, hepatitis, urethritis, phimosis, vaginal synechiae, pneumonia, pulmonary edema, and hypoxemia accompany skin sloughing is seen in up to 90% of cases. 13
In severe cases, the epithelium slides off in entire sheets (Nikolsky sign). Skin biopsy is crucial to diagnose SJS/TEN with certainty. 14 The characteristic pathologic finding is full-thickness epidermal necrosis. 15
Prognosis of SJS/TEN
The Score of Toxic Epidermal Necrolysis (SCORTEN) criteria was introduced in 2000 to measure the severity of illness for TEN. A score (0–7) is determined by the presence or absence of several risk factors that demonstrated excellent accuracy between expected and actual mortality rates. 16 The score is used to generate the expected mortality rate for the patient. 17
Although the scale allows close predictions of actual mortality rates, it displays time-associated deterioration in calibration, leading to overestimating mortality risk. 18 It has been reported to overestimate SJS/TEN-related in-hospital mortality in various populations; therefore, a minor modification based on the population being considered may increase the predictive accuracy of the original SCORTEN. 19
Originally, SCORTEN was supposed to be calculated within 24 h of admission; however, it was found that its predictive value was most accurate when calculated on the 3rd day of hospitalization. Therefore, it is now suggested that the SCORTEN should be calculated on day 1 and day 3 of hospitalization.
20
Serial analysis using SCORTEN was determined to be better than analysis performed only on day 1.
19
Scoring system of toxic epidermal necrolysis based on different variables- SCORTEN criteria.

Probability of mortality based on SCORTEN criteria.

A new prognostic scale called the ABCD-10 score (Age, Bicarbonate, Cancer, Dialysis, 10% body surface area) has been recently proposed as an alternative to the SCORTEN. In contrast to SCORTEN, ABCD-10 incorporates prior dialysis and weights up the impact of cancer. Even though ABCD-10 accurately predicted in-hospital mortality, 21 SCORTEN was a better predictor of inpatient mortality in a North American cohort of patients. 22 SCORTEN should therefore be used for mortality prognostication in epidermal necrolysis instead of ABCD-10. 18 Even though SCORTEN behaved as a reliable predictor of mortality in patients with TEN, outperforming the newer ABCD-10, acute renal failure (ARF) was an early event associated with a poor prognosis. Therefore, ARF could represent a prognostic marker to be considered in the future. 23 (Table 3).
Independent prognostic factors for in-hospital mortality in patients With SJS/TEN and ABCD-10 SJS/TEN mortality prediction model score. 21
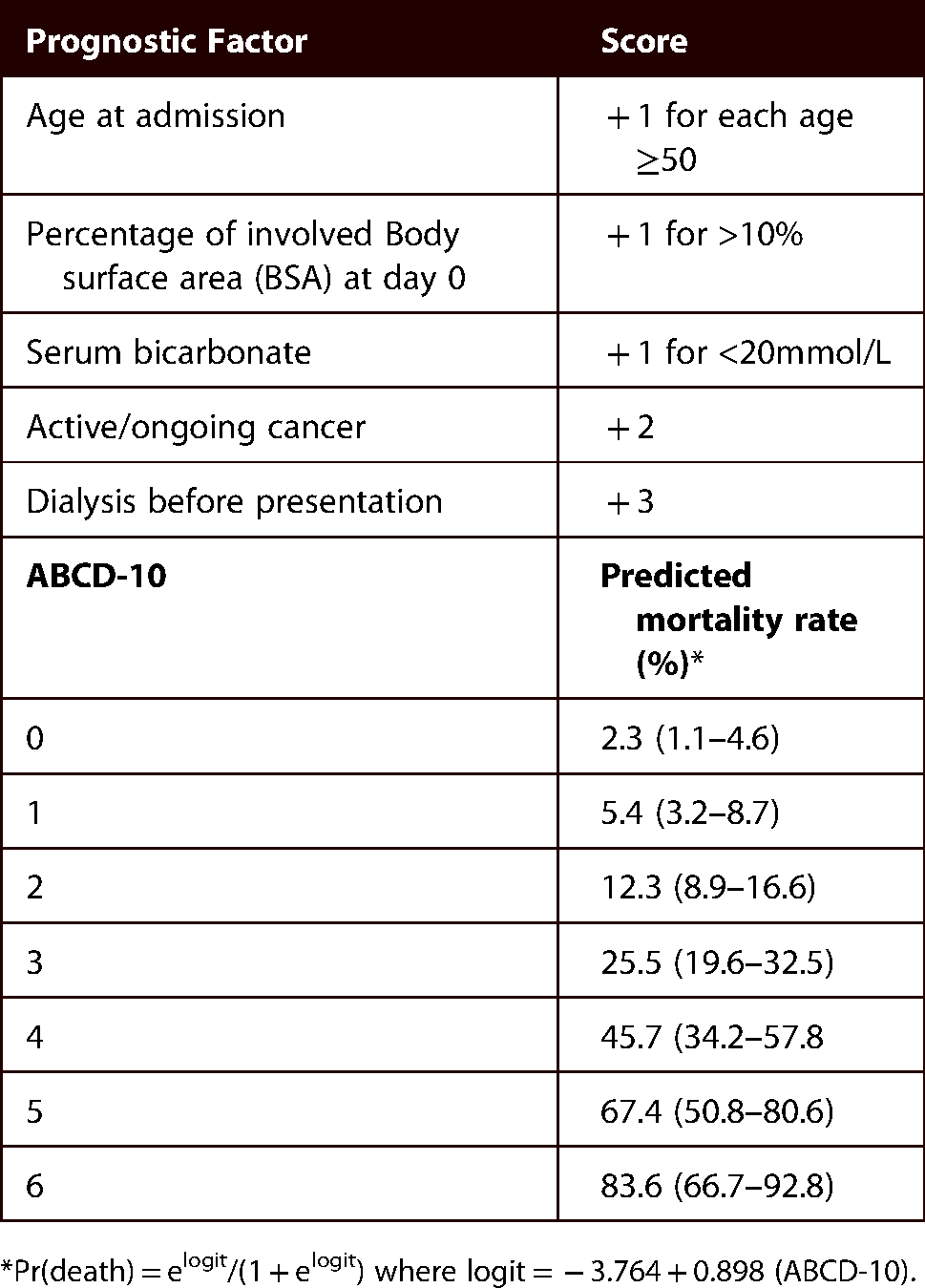
*Pr(death) = elogit/(1 + elogit) where logit = − 3.764 + 0.898 (ABCD-10).
Pathophysiology
SJS/TEN is an immune-mediated delayed-type hypersensitivity reaction due to ineffective clearance of specific drug metabolites. 24 All the nuances of these complex systemic responses are currently unclear. However, it is postulated that drug antigens trigger a cytotoxic response in keratinocytes.5,25 A genetic component and several other chemokines like Granulysin, IL-15, IL-13, Fas ligand have been associated with keratinocyte cell death. 26 IL-33 and TGF-beta1 are potential biomarkers for assessing disease severity in SJS/TEN. 27 Interleukin-15 upregulated granulysin production and was associated with severity and mortality in SJS/TEN. 28
Drug metabolism, immune- function and T-cell clonotypes are known to play a role in the presentation and development of SJS/TEN. The drug exposure generally precedes the onset of symptoms by 1 to 3 weeks. Some high-risk drug classes include anti-infective sulphonamides, anti-epileptic drugs, anti-gout agents, antibiotics, anti-psychotic non-steroidal anti-inflammatory drugs (NSAIDs) of the oxicam type, allopurinol, nevirapine and chlormezanone.
HLA molecules and genetic variants of cytochrome P450 2C have been associated with SJS/TEN in various ethnicities and populations.

Some herbal remedies and new biologicals have also been investigated as potential risk factors.29–31
Since soluble ligands were key suspects owing to the small number of immune infiltrates. Soluble ligand theory based on the interaction of an apoptotic cell-surface receptor Fas and a soluble form of its ligand was implicated in blister formation. Fas-Fas ligand (FasL) interactions were implicated in keratinocyte apoptosis due to its elevated levels in peripheral blood mononuclear cells (PBMCs) from SJS/TEN patients.
32
A group demonstrated TNF-α and IFN-γ from T-cells that triggered upregulation of FasL-positive keratinocytes and increased iNOS production causing Fas-FasL and Caspase-8-induced apoptosis.
33
However, they were later found to down-regulate cytotoxic lymphocytes.
34
Perforin-positive lymphocytes were found in dermal lesions and blisters of SJS/TEN patients.
35
Elevated levels of perforin, Granzyme B, TNF-alpha and FasL were associated with the presentation of SJS/TEN.
36
Inhibiting perforin and granzyme B resulted in decreased keratinocyte death.
37
Granzyme B positive T-lymphocytes in lesions were found to be primed towards specific drug epitopes. In the absence of anti-Fas antibody inhibition, perforin-Granzyme B was concluded to be the causative agent of cytotoxicity.
38
Granulysin concentration in blister exudates was associated with SJS/TEN severity. Cytotoxic killer T-cells release granulysin, and natural killer cells were highly expressed in blister fluid samples. Since granulysin injection-induced SJS/TEN-like dermal conditions in a murine model, it was confirmed as a crucial modulator for keratinocyte necroptosis.
39
CD1 + and CD14 + cells have also been suspected of contributing towards keratinocyte death in SJS/TEN via TRAIL (TNF-related apoptosis-inducing ligand), also known as Apo-2L and TNF-like weak inducer of apoptosis (TWEAK). However, many factors contributing to TEN/SJS in virus-induced and autoimmune aetiologies are unknown. 40
Management
SJS/TEN compromises the mucosal membrane and dermal integrity, accompanied by multi-organ complications. Due to its high mortality and morbidity, early management at intensive care units would play a critical role in improving prognosis. While the UK guidelines recommend withdrawal of the causative drug,41–43 on the other hand, the Japanese guidelines recommend systemic corticosteroid therapy. 44 Conservative management and supportive care have been preferable approaches to treat SJS/TEN.
Supportive care should be set up to monitor respiratory and renal function, nutrition,
45
pain management,
46
and to prevent infections.
47
Despite the similarity of burn appearances, SJS/TEN patients have lower fluid requirements, higher oxygen consumption and double the metabolic rates of patients with similarly sized burns.48–51 SJS/TEN patient care requires a maintenance of room temperature between 30–32°C.
52
Anti-shear wound care where loose skin is used as a dressing instead of surgical debridement has been debated in several groups; a recent retrospective chart review study has shown that anti-shear approach induces an end-result comparable to those predicted for surgical debridement. Anti-shear approach also has benefits of averting painful dressing changes, avoiding sedation as well as anesthesia.
53
Contemporary silver-impregnated nylon sheet dressings are also an attractive option for TEN which must be changed every three days. A recent retrospective analysis has shown that these novel dressings reduce the dressing change frequency, associated pain, and infection. These dressings can be customized according to patient's body surface area involved. The silver sheets are saturated with sterile water and must be rewet every four hours.
54
The patient's clinical status, comorbidities, local availability, and cost should be considered if adjuvant systemic therapy is being entertained.
Cutaneous infections are the most prevalent in SJS/TEN patients administered systemic corticosteroids; special care should be taken regarding dermal care and topical wound treatment. Corticosteroid therapy has been known to predispose to systemic infections and decrease the overall survival rate.55–60 However, other studies have found a beneficial effect of corticosteroid therapy in SJS/TEN. However, these studies did not show statistically significant results.61,62 A study has demonstrated that short-term use of corticosteroids increases survival without causing infections in patients.
63
IVIGs have been highly debated since several case reports worldwide indicated contradictory patient benefits and mortality compared to supportive care in SJS/TEN. Recently researchers scrutinized treatment results of multiple therapies in TEN. This meta-analysis demonstrated that IVIG is efficacious when combined with corticosteroids. Research also reported encouraging outcomes for calcineurin inhibitor, plasmapheresis and etanercept.
64
More extensive randomized controlled trials are required to get a more precise verdict regarding IVIG therapy in SJS/TEN.65–73
Plasmapheresis removes causative agents like drug metabolites and cytotoxic chemokines from the blood of SJs/TEN patients. It must be carried out regularly and is safe due to minimal adverse effects. It can be used when supportive therapy or systemic corticosteroids are not a viable option.
74
Even though it decreases serum cytokine levels, its efficacy in terms of patient survival remains controversial.75–78
From the few studies carried out using TNF-α inhibitors like etanercept and infliximab for SJS/TEN treatment, it can be surmized that it is a promising option.79–83 However, additional studies are required to gather confirmatory data regarding the efficacy of TNF-α inhibitors.
Calcineurin inhibitors like cyclosporine inhibit cell-mediated cytotoxicity. They deter FasL, NFkb and TNF-α production, thus hampering keratinocyte apoptosis. Cyclosporine shows promise as a therapy in SJS/TEN and has been reported to decrease mortality rates.84–87 Compared to the above adjuvant therapy options, SJS/TEN patients treated with cyclosporine present fewer side effects.
88
However, the clinical efficacy and therapeutic benefits of cyclosporine must be validated on larger sample size.
Benefits rendered from systemic corticosteroids, IVIG, cyclosporine, TNF-α inhibitors and plasmapheresis are still under constant scrutiny as studies have not provided satisfactory evidence for systemic treatment in SJS/TEN.62,89 Since every stage of the disease accounts for different systemic behavior, treatment must be tailored for individual patients. In the acute phase, immunosuppressive might be helpful; however, it might hinder epithelialization and predispose the patient to infection when skin detachment develops.
However, many studies including IVIG and corticosteroid combination therapy also show statistically insignificant survivability in SJS/TEN compared to IVIG or corticosteroid treatment alone, warranting a need for more data and clinical studies.61,90–92 When a patient presents with severe symptoms and is refractory to corticosteroid therapy, a combination of IVIG or plasmapheresis (PP) therapy is added to systemic corticosteroid therapy. Combined PP and IVIG therapy have shown contradictory results in a few studies.93,94 Therefore, more studies are needed to ascertain further the viability of these therapeutic approaches as standardized care.
It is proven that there is an absence of agreement on the most potent and efficacious treatment. 64 Pragmatic and practical approach considering cost, availability of medicines, and accessibility of clinical expertise would be best in deciding final treatment protocol. 64
Role of randomized control trials in life threatening situations like TEN is debatable as randomization to a less effective control arm could reduce the survival. This paves way for consideration of contemporary systemic reviews or metanalysis in management of TEN. A systemic review and network meta-analysis assessing the systemic immunomodulating therapies for SJS/TEN overlap and TEN has demonstrated that corticosteroid and intravenous immunoglobulin combination therapy reduce mortality risk. This study also labels cyclosporine and etanercept as promising therapy. 95
Another multi-variate meta-regression and network meta-analysis has confirmed that cyclosporine and immunoglobulins plus corticosteroids are linked with reduced mortality than forecasted by SCORTEN. However, reduction in SCORTEN based standardized mortality ratio (SMR) was not attained by any treatment. 96
A latest retrospective pan-European, multicenter, cohort study demonstrated that systemic glucocorticoids and intravenous globulins were associated with reduced chances of infection while cyclosporine was associated with increased frequency body surface area detachment in acute phase. 97 In contrast, a landmark network meta-analysis which compared the efficacy of treatment interventions in decreasing mortality of TEN patients has concluded that cyclosporine can decrease the mortality in TEN patients. While other findings of this study were consistent with similar studies showing encouraging results with amalgamation of corticosteroids plus intravenous immunoglobulin and etanercept. 98
Optimal facility to treat TEN
Ideal facility to manage TEN must be a tertiary or higher care hospital with burns unit as they have the skilled professionals for managing wounds, dressing, optimal fluid management and pain relief. 99
Conclusion
SJS/TEN is a stimulus (predominantly drug) -induced hypersensitivity reaction of the skin with exceptionally high mortality and relatively unknown pathogenesis. These patients mostly reach healthcare facility in emergency. The condition is difficult to diagnose in the early stages, and no predictive biomarkers have been validated for routine management yet. The preliminary treatment still resorts to withdrawing offending drugs immediately and establishing supportive care. Emergency room clinical practitioners must be familiar with SJS/TEN. Several adjuvant therapies have shown promise, and combination therapy with corticosteroids and immunoglobulins is the step forward. TNF-α inhibitors and Calcineurin inhibitors have shown initial promising results. Local availability, clinical expertise and cost factors of adjuvant agents should be considered first after initial supportive care. However, more large-scale studies are required to prescribe and derive internationally accepted treatment flow charts as that will lead to benchmark therapeutic option with a certain degree of confidence.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
How to cite this article
Piyu Parth Naik. A contemporary snippet on clinical presentation and management of toxic epidermal necrolysis. Scars, Burns & Healing, Volume 8, 2022. DOI: 10.1177/20595131221122381
