Abstract
Background:
Renal failure in sarcoidosis is rare and data on its long-term outcome are scarce.
Aim:
To investigate the pattern of renal involvement in sarcoidosis, its clinical course and response to treatment in the long-term.
Methods:
A single-center retrospective study with review of renal biopsies and medical charts was performed.
Results:
Between January 2005 and December 2016, seven patients with sarcoidosis underwent a kidney biopsy. This is equivalent to a frequency of 1.6% in a total of 434 biopsies from native kidney performed in our institution. All patients presented with renal failure. Five patients had granulomatous interstitial nephritis (GIN) and one patient each interstitial nephritis without granuloma and nephrocalcinosis. Three patients had concomitant glomerular disease: IgA nephropathy (n = 2), membranous and focal proliferative glomerulonephritis (n = 1). Most patients (n = 5) presented with hypercalcemia. All patients initially received oral prednisolone 1 mg/kg/day (n = 3) or 0.5 mg/kg/day (n = 4), respectively, with subsequent tapering or suspension. One patient was started on azathioprine after 18 months to spare steroids. After a mean follow-up of 59 months mean estimated glomerular filtration rate (eGFR) had improved from 19 ± 7 at presentation to 49 ± 16 mL/min. No patient required dialysis. All patients started on prednisolone 1 mg/kg/day developed transient diabetes mellitus while patients on 0.5 mg/kg/day did not. Renal function improvement did not differ between both treatment groups.
Conclusion:
GIN was the most common diagnosis in sarcoidosis patients with renal failure. Initial hypercalcemia was observed in the majority. Early steroid treatment lead to sustained renal function improvement.
Introduction
Sarcoidosis is a multisystem inflammatory disease of unknown etiology characterized by non-caseating granulomas with epitheloid cells and multinucleated giant cells. Sarcoidosis may affect any organ—leading to organ dysfunction. It most frequently affects the lungs, lymph nodes, eyes and skin. 1
Renal involvement is rare. Renal failure occurs in 0.7%–4.3% of sarcoidosis patients2–4 and may lead to end-stage renal failure requiring permanent dialysis in 4%–10% of patients with renal involvement.5,6 Renal injury is frequently due to disturbances in calcium metabolism, leading to nephrolithiasis and/or nephrocalcinosis. 7 Hypercalciuria is observed in about 30% and hypercalcemia in about 10% of patients with sarcoidosis in general. 8 However, hypercalcemia is more common in patients with renal involvement ranging from 24% 9 –37%. 6 Granulomatous interstitial nephritis (GIN) is another cause of renal impairment in sarcoidosis and is present in 7%–27% of all patients in autopsy studies. 10 In series from native kidney biopsies from patients with sarcoidosis granulomatous interstitial nephritis is observed in 30%–79%, while in non-granulomatous interstitial nephritis it is observed in 14%–44% of all cases.5,6,9,11 Glomerular disease is less common and found in 4%–26% of native kidney biopsies in sarcoidosis.5,6,9,11 The spectrum of glomerular diseases associated with sarcoidosis includes membranous nephropathy, IgA nephropathy (IgAN), minimal change disease and proliferative lupus nephritis with membranous nephropathy and IgAN being the most frequent. 12 Sarcoidosis may recur in patients after renal transplantation 13 and rarely affect the transplanted kidney in the form of GIN. 6
Corticosteroids are the standard of care in renal sarcoidosis. 14 In patients who do not respond to steroids or develop steroid toxicity other immunosuppressive agents such as azathioprine, mycophenolate mofetil or TNFα inhibitors are used. 14
Only few studies have investigated the pattern of renal involvement in sarcoidosis, its clinical course and response to treatment in the long term. Here, we present the histological pattern and clinical course of renal sarcoidosis in our institution and provide data on long-term outcome.
Patients and methods
In the present retrospective study we reviewed all 434 native kidney biopsies performed in our institution (Kliniken Maria Hilf, Mönchengladbach, Germany), a large urban tertiary care hospital, between 1 January 2005 and 31 December 2016. Patients with sarcoidosis according to the statement of the American Thoracic Society, the European Respiratory Society and the World Association of Sarcoidosis 15 in whom any alternative diagnosis had been excluded were identified from medical records. Initial analysis concerned 9 patients; 2 of them did not meet the inclusion criteria because of the absence of sufficient criteria to diagnose sarcoidosis (i.e. isolated renal granulomatosis, n = 2). All patients presented with renal failure. Taking the epidemiology of the disease into account we considered that a sample size of 7 sarcoidosis patients with renal failure could be reasonable for such a rare clinical condition.
For all cases the demographic and clinical information at time of biopsy were recorded and included: age at biopsy, sex, relevant past medical history, extrarenal involvement, serum creatinine (mg/dL), creatinine clearance, 24-h proteinuria, urinalysis, blood cell count, serum calcium (mval/L), C-reactive protein, and serum angiotensin-converting enzyme (ACE). Glomerular filtration rate (GFR) was estimated according to the Modification of Diet in Renal Disease Study (MDRD) calculation. 16 Moreover, intact parathormone (iPTH), serum 25OH-vitamin D3 (25OHD3), serum 1,25-dihydroxy-vitamin D3 (1,25DihydroxyD3) and soluble Interleukin II-receptor (sIL2R) levels were recorded, when available. Sarcoidosis therapy, including steroids and/or immunosuppressive agents, was documented for all patients.
Native kidney biopsies were processed for light microscopy, immunohistochemistry, and electron microscopy. All renal biopsies were viewed by a renal pathologist.
We evaluated the outcome and treatment for each patient. Patients were followed until their last visit in the outpatient clinics or in hospital between November 2016 and February 2017 or until death. Serum creatinine, mean estimated glomerular filtration rate (eGFR), and blood cell count were recorded at last follow-up. Moreover, serum calcium levels, urinalysis, and C-reactive protein levels were documented, when available. The need for dialysis was specified.
A descriptive analysis was used to study the distribution of continuous variables (i.e. means, medians, standard deviations, ranges) and the frequencies of categorical variables (numbers and percentages). A paired t-test was used to compare laboratory data at renal sarcoidosis presentation and last follow-up. A value of p < 0.05 was considered significant.
The study was approved by the ethics committee of the medical faculty at RWTH University of Aachen (EK 294/17).
Results
During the observation period, 7 patients (6 male/ 1 female; mean age 65 ± 10 years) with sarcoidosis underwent a kidney biopsy. This is equivalent to a frequency of 1.6% in a total of 434 biopsies from native kidney performed in our institution. In five patients sarcoidosis was newly diagnosed, two had prior known sarcoidosis. Six of the seven patients had lung involvement and one patient each gastrointestinal, bone marrow and neurologic involvement at the time of biopsy. In two patients cardiac involvement was diagnosed during subsequent follow-up (Table 1).
Patient characteristics, renal pathology and treatment.
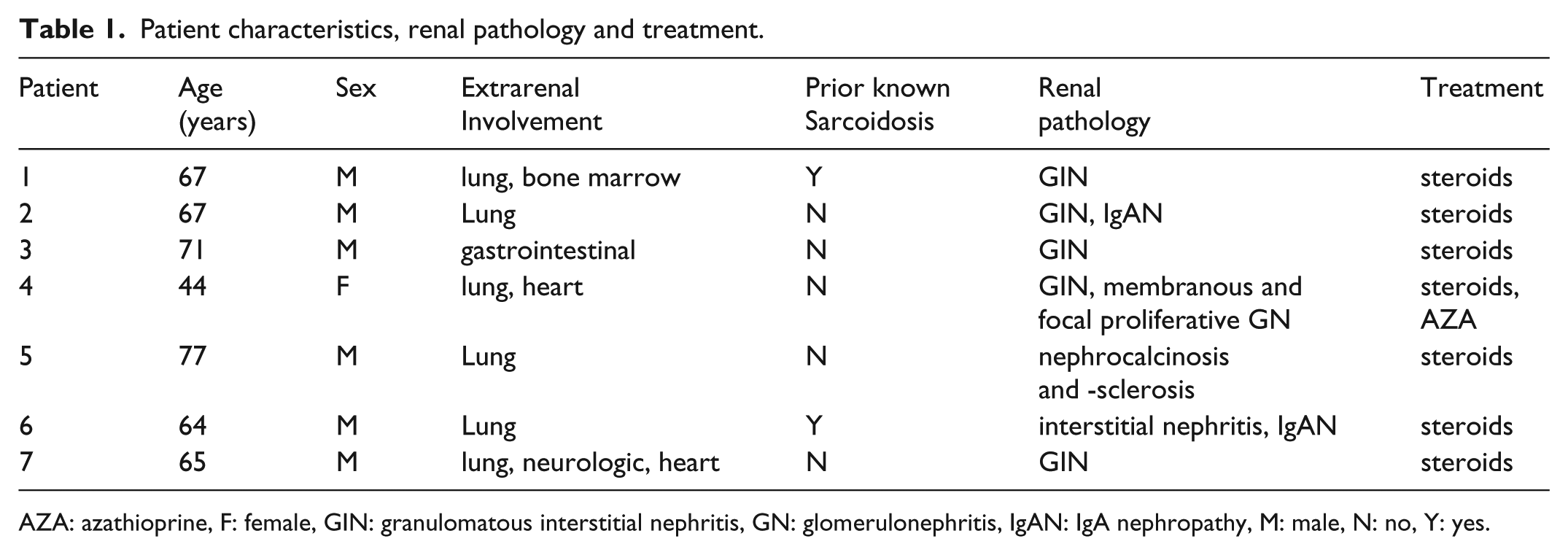
AZA: azathioprine, F: female, GIN: granulomatous interstitial nephritis, GN: glomerulonephritis, IgAN: IgA nephropathy, M: male, N: no, Y: yes.
All patients presented with renal failure. Mean serum creatinine level at presentation was 3.6 ± 1.2 mg/dL, eGFR was 19 ± 7 mL/min and mean creatinine clearance 26 ± 16 mL/min (Table 2). Hypercalcemia was present in 5 of 7 patients at presentation (mean 5.9 ± 0.4 mval/L, range 5.4–6.3 mval/L). In patients with hypercalcemia iPTH levels were lower than the normal range in 3 of 5 patients (iPTH < 15 pg/mL) and normal in 2. The serum 25OHD3 level was low in all patients (25OHD3 < 30 ng/mL) while the 1,25DihydroxyD3 level was normal in 4 of 5 patients with hypercalcemia and low in one (Table 3). Most patients presented with mild anemia (mean Hb 12.1 ± 1.6 g/dL) and moderately elevated CRP levels (mean CRP 1.7 ± 1.7 mg/dL, normal range 0.0–0.3 mg/dL). Levels of ACE were elevated in four, normal in one, und low in two patients (mean 25 ± 20 mU/mL, range 0.1–50.6; normal range 8–28 mU/mL). Soluble Interleukin II-receptor (sIL2R) was determined in three patients at presentation and was elevated in all (mean 9759 ± 4780, range 6006 –15140; normal range, < 710 U/mL).
Renal function at renal sarcoidosis presentation and at the end of follow-up.
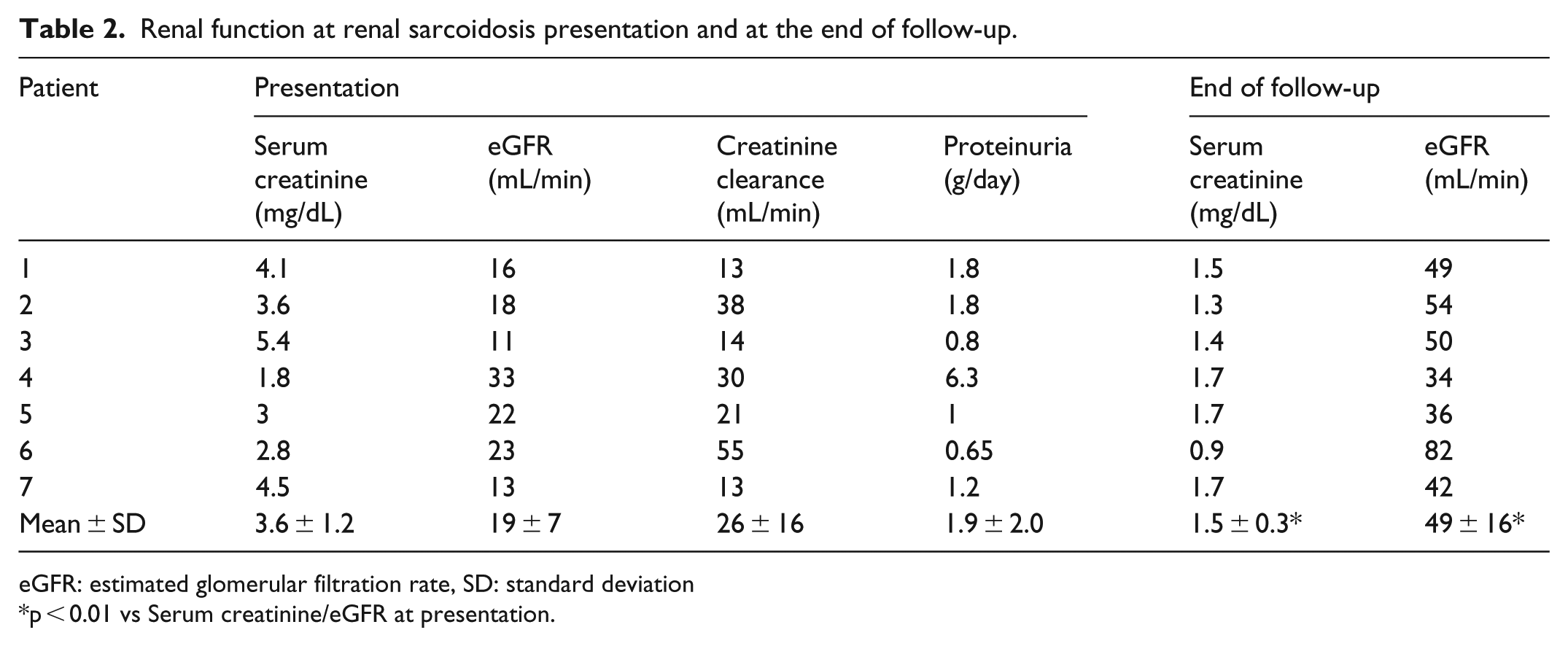
eGFR: estimated glomerular filtration rate, SD: standard deviation
p < 0.01 vs Serum creatinine/eGFR at presentation.
Serum calcium, vitamin D and iPTH at renal sarcoidosis presentation; duration of follow-up and serum calcium at the end of follow-up.
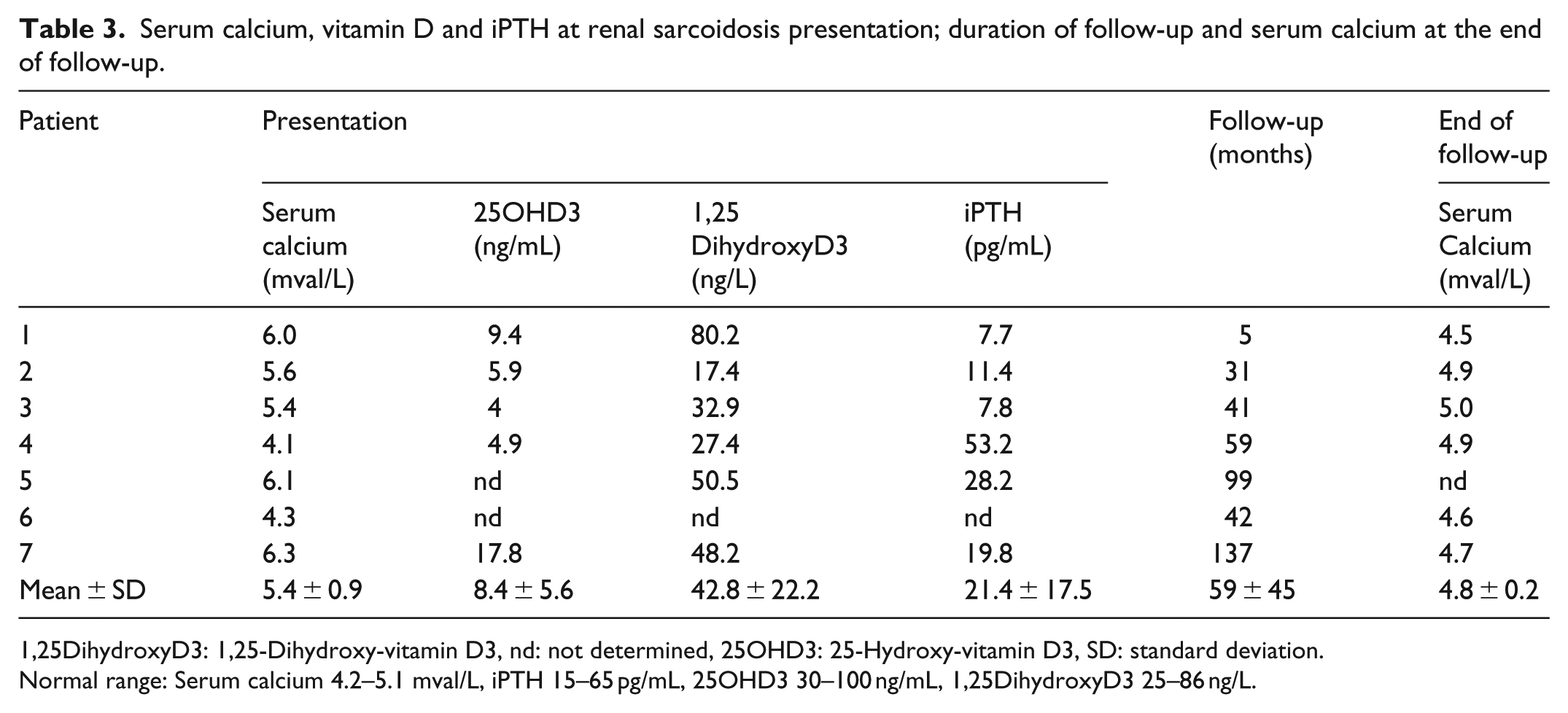
1,25DihydroxyD3: 1,25-Dihydroxy-vitamin D3, nd: not determined, 25OHD3: 25-Hydroxy-vitamin D3, SD: standard deviation.
Normal range: Serum calcium 4.2–5.1 mval/L, iPTH 15–65 pg/mL, 25OHD3 30–100 ng/mL, 1,25DihydroxyD3 25–86 ng/L.
Proteinuria was usually moderate (mean 1.9 ± 2.0 g/day, range 0.65–6.3 g/day; Table 2). The only patient with nephrotic-range proteinuria (6.3 g/day) had concomitant membranous and focal proliferative glomerulonephritis. On urinalysis none of the patients had leukocyturia. Mild microhematuria was present in 6 and moderate microhematuria in one patient with concomitant IgA nephropathy.
An informative percutaneous renal biopsy containing more than 6 glomeruli was performed in all patients (mean 10 ± 2 glomeruli). Five patients presented with non caseating granulomatous interstitial nephritis (GIN; Figure 1), one patient with interstitial nephritis without granuloma, and one patient with nephrocalcinosis (Figure 2; Table 1). Interstitial inflammatory cell infiltration was observed in all patients. In the patient with nephrocalcinosis this was restricted to foreign body reaction in areas of interstitial calcification. Interstitial fibrosis and tubular atrophy were present in all cases to varying degrees. Giant cells were associated with GIN in 3 of 5 patients (Figure 1). Tubulointerstitial microcalcifications were observed in six patients, including one with overt nephrocalcinosis. In the two patients without hypercalcemia microcalcifications were also observed on biopsy. Three patients had additional glomerular disease: two patients IgAN and one patient membranous and focal proliferative glomerulonephritis (Figure 3).
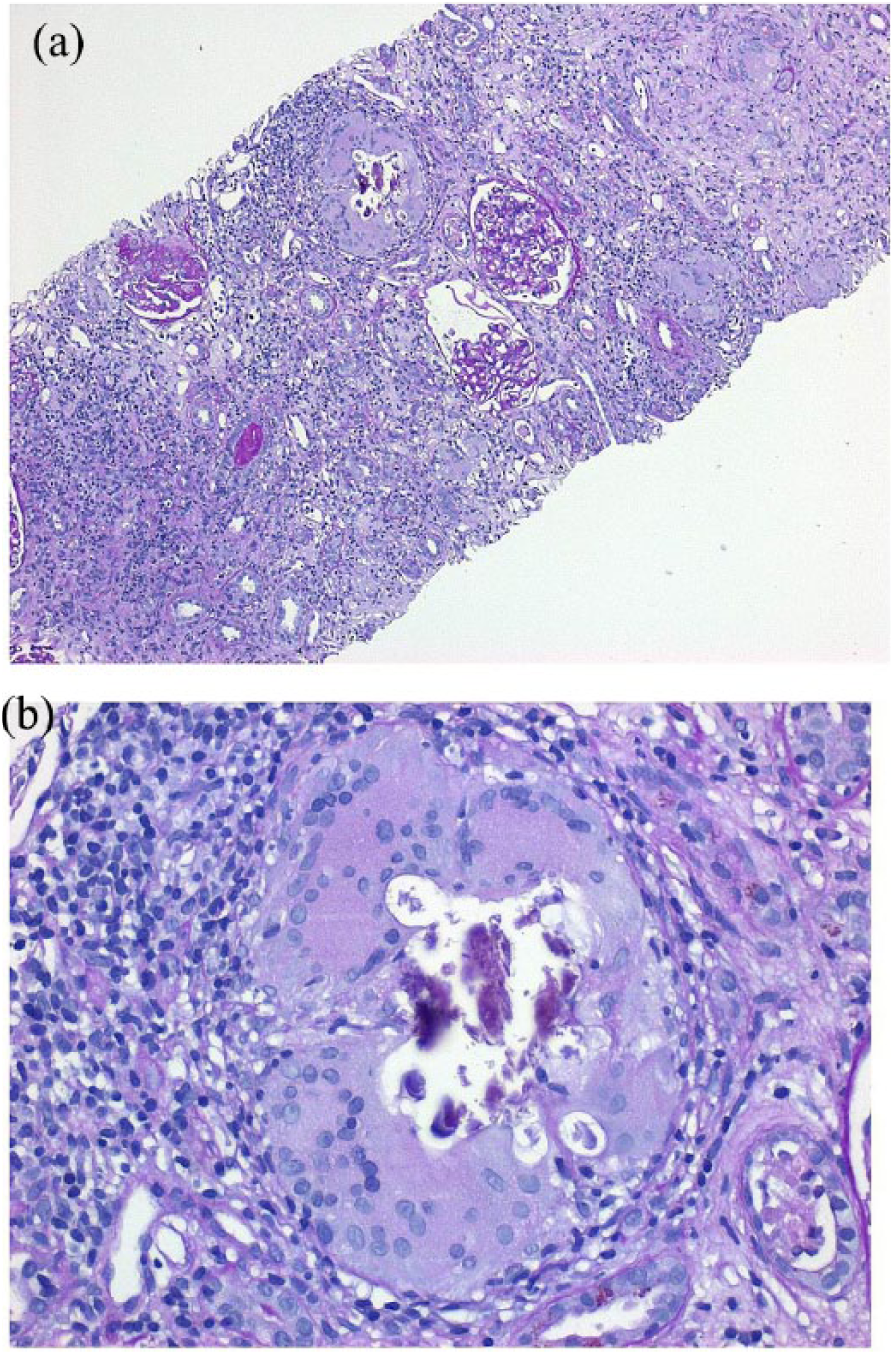
Granulomatous interstitial nephritis with epithelioid cells, a giant cell (b), tubular atrophy and interstitial inflammation. PAS stain (a) 100× and (b) 400×.
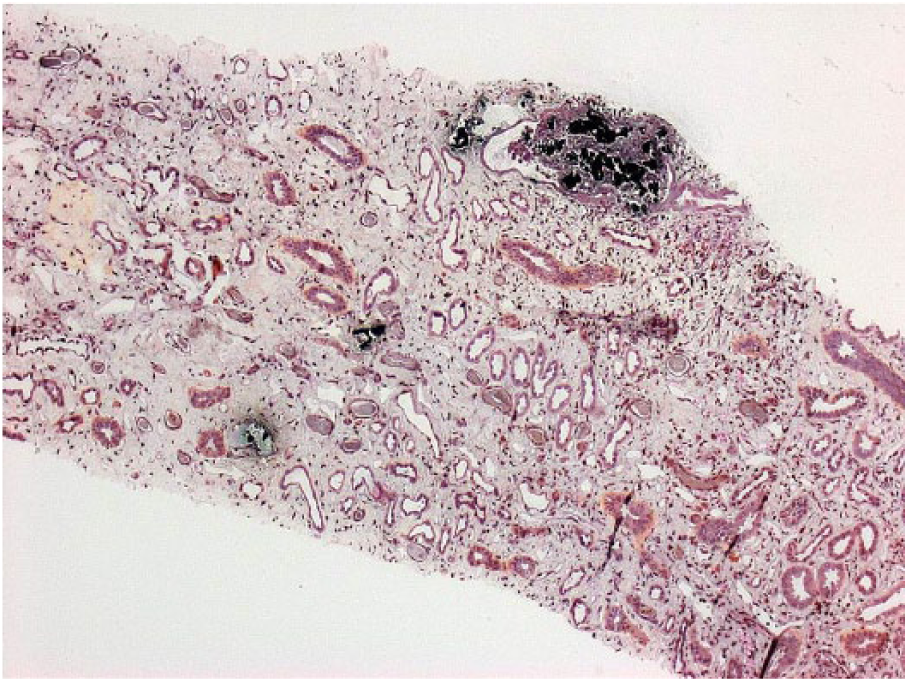
Tubular and interstitial calcifications as well as tubular atrophy and fibrosis. Von Kossa stain: 100×.
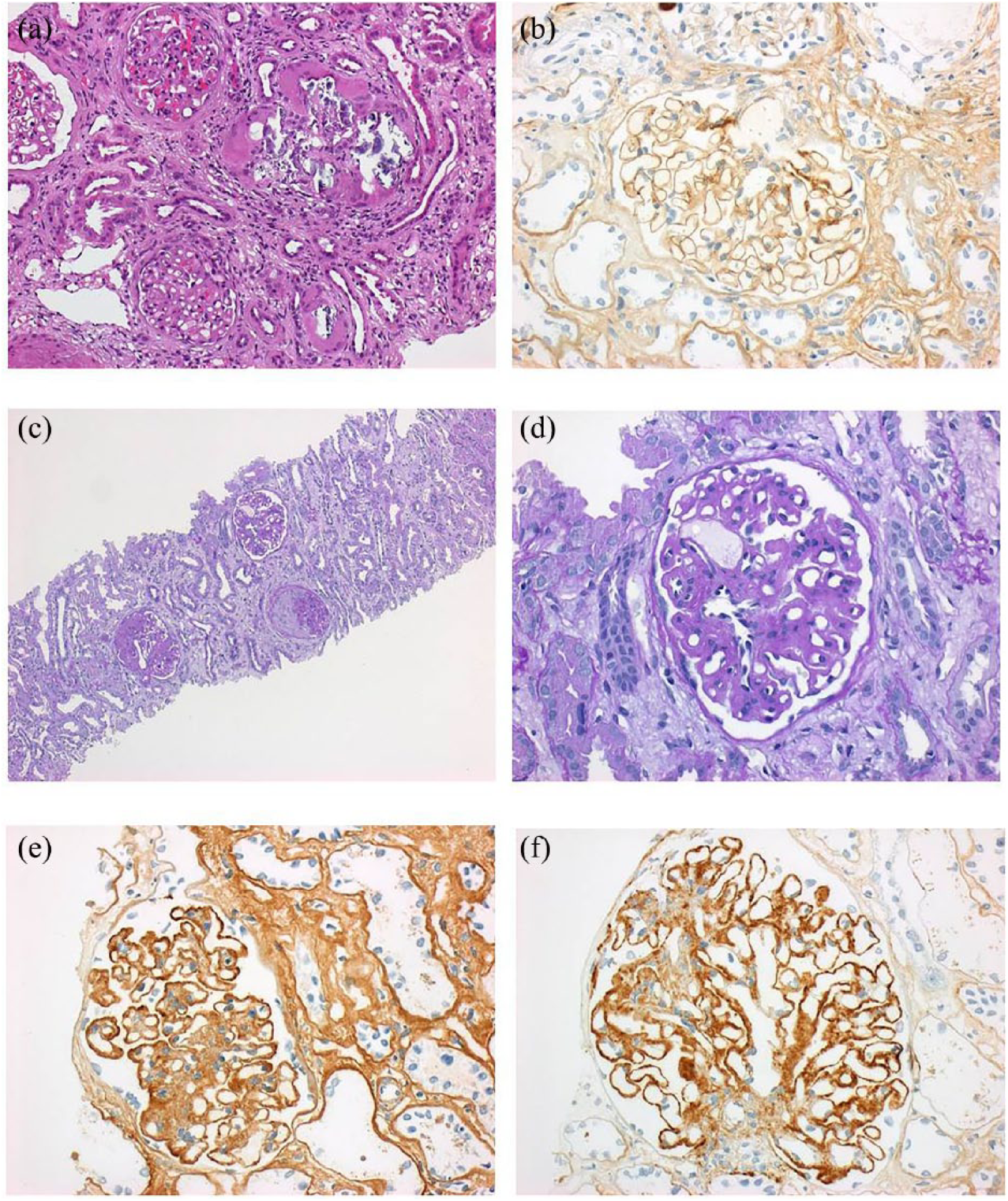
(a) IgA-GN and renal involvement in sarcoidosis: Interstitial calcification with giant cell (a), mesangial granular IgA deposits (b) 400×. (c), (d) Focal crescentic membranous glomerulonephritis (PAS, 100× and 400×). (e), (f) Granular IgG- and C3c-deposition along the glomerular basement membrane (400×).
Patients were started on steroids 2 days after kidney biopsy was performed (median = 2, range –5–18 days). All patients initially received oral prednisolone at doses of 1 mg/kg per day (n = 3) or 0.5 mg/kg per day (n = 4) with subsequent tapering or suspension. One patient with GIN and concomitant membranous and focal proliferative GN as well as lung and heart involvement received azathioprine after 18 months to spare steroids (Table 1). At the end of follow-up 3 patients were on oral prednisolone at doses between 2.5 and 10 mg per day, one patient received azathioprine without steroids, and 3 patients received no immunosuppression. The median duration of steroid treatment was 37 months (range 1–55 months). Three patients developed transient diabetes mellitus: one was diet controlled, one treated with insulin, and one with oral antidiabetics. All patients who developed diabetes had been started on high dose oral prednisolone, that is 1 mg/kg per day. However, upon steroid tapering blood glucose normalized and antidiabetic treatment could be stopped in all.
After a mean follow-up of 59 months (median 42; range 5–137 months), the mean eGFR was 49 ± 16 mL/min (range 34–82 mL/min; Table 2 and Figure 4), the mean serum creatinine 1,5 ± 0,3 mg/dL. Serum calcium levels were normal in all patients (mean 4.8 ± 0.2 mval/L, n = 6, Table 3). Mean hemoglobin level had increased to 13.7 ± 1.9 mg/dL and CRP decreased to 0.5 ± 0.5 mg/dL. Proteinuria was assessed in five patients and was trace in four, negative in one. One patient died 42 months after renal biopsy of an unknown cause. Four weeks before death he had a normal serum creatinine of 0.9 mg/dL. Renal function improvement did not differ between both treatment groups: median eGFR improved by 28 mL/min from initial presentation to last follow-up in patients started on prednisolone 1 mg/kg/day as compared to 36 mL/min in patients started on prednisolone 0.5 mg/kg/day. No patient required dialysis at presentation or during follow-up.
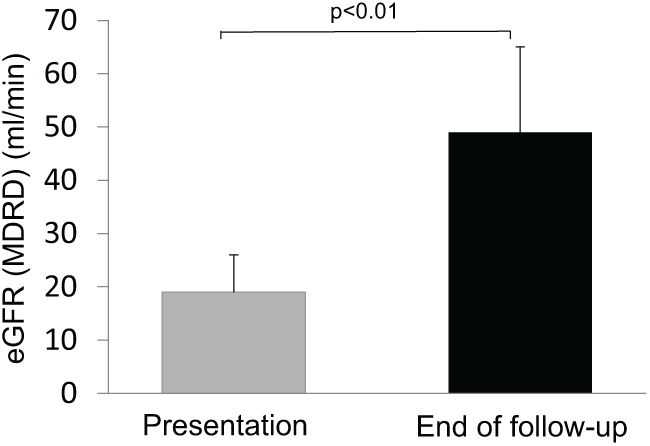
Mean eGFR at presentation and at last follow-up.
Discussion
Renal failure in sarcoidosis is uncommon, occurring in 0.7%–4.3% of sarcoidosis patients.2–4 Only few studies provide data on response to treatment and long-term outcome in these patients.5,6,9,11 In the current study patients were followed for a median of 3.5 years (median 42, mean 59 months) while median follow-up was 2 to 3 years in most studies5,6,11, and 7 years in one. 9
GIN was the most frequent form of renal involvement in sarcoidosis in our series. This corresponds to findings in most studies where 37%–79% of sarcoidosis patients undergoing a renal biopsy revealed GIN.5,6,9 In contrast, Löffler et al. 11 found non-granulomatous interstitial nephritis to be the most common histological entity accounting for 44% of their cases followed by GIN with 30%. Interstitial nephritis without granuloma was the second most frequent form of renal involvement in most studies accounting for 14%–21% of cases.5,6,9 Moreover, in 10%–11% of sarcoidosis patients presenting with renal disease, nephrocalcinosis is observed.9,11 Interstitial calcifications are, however, also found in patients with granulomatous and non-granulomatous interstitial nephritis,5,6,9 probably reflecting the high frequency of hypercalciuria and hypercalcemia in these patients. In our series, the majority of patients exhibited initial hypercalcemia and tubulointerstitial microcalcifications were also observed in patients initially not presenting with hypercalcemia. Bagnasco et al. 6 described hypercalcemia in 37% of patients with biopsy-proven GIN, while Mahevas et al. 5 observed hypercalcemia in 34% of patients with granulomatous and non-granulomatous interstitial nephritis. In contrast, in sarcoidosis without renal involvement, hypercalcemia is apparently less frequent and observed in about 4% of cases. 17 In the presence of hypercalcemia, iPTH and 25OHD3 levels were low or within the normal range in renal sarcoidosis patients in the current study as also observed by others. 5 Surprisingly, 1,25DihydroxyD3 levels were normal in most of the patients in our series. Hypercalcemia in these patients therefore is unlikely due to an increased systemic conversion of 25OHD3 to 1,25DihydroxyD3 by the enzyme 1-α hydroxylase produced by an activated macrophage system. In that case, one would expect elevated 1,25DihydroxyD3 levels. One possible explanation for this observation is the production of parathormone-related peptide (PTHrP) by sarcoid granulomas as previously described.18,19 Unfortunately, PTHrP levels were not available in the present study. Alternatively, hypercalcemia may have been caused by an increased conversion of 25OHD3 to 1,25DihydroxyD3 on the tissue level by activated local macrophages in sarcoid granulomas without leading to increased systemic 1,25DihydroxyD3 levels. 20 Molecular studies have shown a discrepancy between high cellular vitamin D levels and low serum vitamin D levels in sarcoidosis patients. 21
Glomerular diseases are less frequent than tubulointerstitial lesions in sarcoidosis and observed in 4%–26% of kidney biopsies.5,6,9,11 The spectrum of glomerular lesions found in the context of sarcoidosis is heterogenous with membranous nephropathy and IgAN being the most common. 12 The existence of a direct molecular link between sarcoidosis and glomerular damage remains unclear. However, cytokines produced by sarcoid granulomas, circulating immune complexes, and hypergammaglobulinemia may contribute to glomerular injury in sarcoidosis. On the other hand, the glomerular lesions observed, such as IgAN, may merely coincide with sarcoidosis. It has been suggested that glomerular lesions in sarcoidosis are less steroid-sensitive than interstitial lesions. 12
In the present study, serum ACE levels were elevated in only 4 of 7 patients (57%). In other studies, 18% and 55% of patients, respectively, exhibited elevated ACE levels at renal sarcoidosis presentation.5,9 Serum ACE, therefore, seems to lack specificity for both diagnosis and assessment of disease activity. Another possible marker of disease activity, sIL2R, was determined in three patients in our series at presentation and was markedly increased in all. However, in renal function impairment sIL2R may increase due to impaired clearance which may lead to an overestimation of disease activity. 22 Data on the course of sIL2R levels in renal sarcoidosis are currently not available and the role of sIL2R as a biomarker in renal sarcoidosis and renal function impairment therefore remains to be elucidated.
Corticosteroid treatment is the standard of care in renal sarcoidosis. 14 However, no standardized protocol regarding the dose and duration of therapy exists. In our series, all patients were started early at initial presentation on oral prednisolone at doses of 0.5–1 mg/kg per day with subsequent tapering—a dose recommended by most authors.5,9 Patients responded well to steroid treatment in the present study with sustained improvement in eGFR at last follow-up. However, only one of seven patients recovered normal renal function. On average, patients were kept on steroids for 18 months in most studies.5,6 In contrast, Rajakariar et al. 9 recommended maintaining patients on low-dose steroids indefinitely to prevent relapses. Initial intravenous pulse methylprednisolone may be beneficial but data are scarce. 5 Generally, duration and dose of steroid therapy need to be balanced against potential adverse events such as the development of diabetes or osteoporosis. In our series, all patients started on prednisolone 1 mg/kg per day developed transient diabetes mellitus, while none of the patients who started on prednisolone 0.5 mg/kg became diabetic. The use of steroid-sparing agents such as azathioprine is an option in patients who develop adverse events or relapse on maintenance steroids.5,9
There are several limitations of our study. First, the number of patients was small and was acquired from a single institution. Therefore, large prospective studies need to be conducted in the future to further investigate calcium metabolism, markers of disease activity, and treatment strategies in renal sarcoidosis. The second limitation is the retrospective design of the study and information was only available from medical records and kidney biopsies.
Conclusion
In our study, GIN was the most common form of renal involvement in sarcoidosis patients with renal failure. Hypercalcemia at presentation was frequent in these patients. The mechanism of hypercalcemia with low 25OHD3 and normal 1,25DihydroxyD3 levels in renal sarcoidosis, however, is not clear and remains to be elucidated. Early administration of oral steroids with subsequent tapering led to long-term renal function improvement in sarcoidosis patients with renal involvement. Our series indicates that prednisolone 0.5 mg/kg may be sufficient to induce remission. Prospective studies on steroid induction therapies with and without pulse methylprednisolone, as well as steroid-sparing agents for maintenance therapy are, however, desirable.
Supplemental Material
Corrigendum_NPC_834928_1 – Supplemental material for Renal involvement in sarcoidosis: Histologic findings and clinical course
Supplemental material, Corrigendum_NPC_834928_1 for Renal involvement in sarcoidosis: Histologic findings and clinical course by Shirin Naderi, Kerstin Amann and Ulf Janssen in Nephrology @ Point of Care
Footnotes
Author contributions
S.N. contributed to the study conception, data collection, data analysis, data interpretation, and manuscript drafting and revision. K.A. contributed to the manuscript drafting and revision. U.J. contributed to the study conception, data interpretation, intellectual content, and manuscript revision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the ethics committee of the medical faculty at RWTH University of Aachen. Ethics approval number: EK 294/17.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Due to the retrospective nature of this study, no patient consent was required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
