Abstract
COVID-19 continues to strain healthcare systems around the globe. Research has shown a relationship between COVID-19 and an inflammatory response, including neuropathological outcomes. Additionally, studies have shown positive effects of engagement with music on inflammatory responses; music may have potential, as a method, to reduce inflammation triggered by COVID-19. This review compiles exhaustive research from multiple disciplines to account for this possibility. The authors utilized a meta-narrative approach to complete this review. The search was conducted using PubMed, Embase, OneSearch, Primo, Google Scholar, Clinicaltrials.gov, and the bibliographies of relevant articles. In total, 84 articles were included for full-text review, discussion, and analysis. Articles pertaining to music and acoustics encompassed a date range from 1964–2020. Articles referencing COVID-19 spanned the years 2019–2021. This work focused on associations between engagement with music, stress response, blood-brain barrier integrity, inflammation, COVID-19, and neuropathology in preclinical and clinical models. Detailed analysis revealed that engagement with music has the potential to reduce the harmful effects of COVID-19, particularly in the inflammation and blood clotting pathways associated with a range of pathophysiological and neuropathological issues. Further work is warranted to standardize and validate existing methods associating positive effects of engagement with music on the negative effects of COVID-19.
Introduction
Music is a culturally embedded human phenomenon that engages the individual and/or group cognitively, emotionally, and often physically (Clarke et al., 2015). An agreed-on definition of the term “music” across many subdisciplines is “humanly organized sound” (Blacking, 1974). However, this remains an area of debate; research into the origins of music argues for the inclusion of non-human sounds (e.g., bird or whale songs) as part of its definition (Wallin et al., 2001). Scholars in comparative and cross-cultural studies of music argue for clarification of the concept of organization as it applies to the social and cultural contexts in which music is created, practiced, and performed because these differences vary significantly and confound a universal definition (Merriam, 1964). Nonetheless, comparative work has shown significant overlaps and parallels for a universal definition of music from both qualitative (Blacking, 1974), and quantitative perspectives (Huron, 2008).
Regarding music as medicine, a long history exists across cultures of applying music to prevent, heal, and/or cure illness. It is not within the scope of this review to provide an exhaustive account of such history. Rather, for the purposes of this review, we used a meta-narrative approach (Wong et al., 2013) with the understanding that there are entire subdisciplines studying relationships between music and medicine (e.g., medical ethnomusicology; Koen et al., 2008). Longitudinal research in given patient populations has shown marked effects of the application of music to reduce symptoms of depression and anxiety (Aalbers et al., 2017; Ribeiro et al., 2018), high blood pressure (Im-Oun et al., 2018), pain (Johnson & Elkins, 2020), mental illness (Golden et al., 2021), as well as increased positive outcomes of good health (Fancourt & Finn, 2019). Furthermore, music affects a broad range of biological systems (Finn & Fancourt, 2018), including systems involved in psychoneuroendocrinological (Kreutz et al., 2012), neurochemical (Chanda & Levitin, 2013), and psychoneuroimmunological responses to music (Fancourt et al., 2014). Relationships between the immune system and stress pathways are highlighted by Fancourt et al. (2014) as a means to understanding the effects of music upon psychoneuroimmunological response, particularly considering dynamic interactions between multiple biomarkers (e.g., cortisol, interleukins, oxytocin, testosterone, beta-endorphin, immunoglobulins), and physiological and metabolic activities of the body.
Coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome SARS-CoV-2 RNA virus identified in China, proceeded to spread worldwide, inducing a global pandemic. Coronavirus has immunogenic spike proteins in which the immune system creates immunoglobulins and activates an immune response. COVID-19 is primarily targeted via these spike proteins and allows for new potential therapeutic methods. Additionally, cytokines are released in response to viral immune dysregulation to promote homeostasis. Dysregulation of cytokine release results in excess levels of inflammatory cytokines in the immune system, i.e., a cytokine storm. Chemokines are a smaller family of cytokines that mediate immune regulation via leukocyte activation, cell positioning, and provocation of receptors (Majumdar & Murphy, 2021). Cytokines can induce specific chemokine expressions such as C-C motif ligand (CCL) 2, CCL3, CCL4, CCL5, and C-X-C motif ligand (CXCL) 8, which are seen in viral immune responses (Khalil et al., 2021). Furthermore, lung autopsies from patients and in vitro SARS-CoV-2 infections have shown up-regulation of the T cell-specific chemokines CCL2, CCL5, CXCL8, CXCL9, and CXCL10 (Majumdar & Murphy, 2021).
Furthermore, acute respiratory disease syndrome (ARDS), a symptom of COVID-19, is caused by an excessive release of chemokines. Chemokines function in the immune response to a coronavirus by leading immune cells to the viral infection. Yet, an excessive release of chemokines can cause hyperinflammation. This reaction can be linked to ARDS, potentially highlighting a link between chemokines and COVID-19 (Khalil et al., 2021). It may be that engagement with music could systematically address effects of COVID-19 upon the immune system due to music's effects upon psychoneuroendocrinological (Kreutz et al., 2012), neurochemical (Chanda & Levitin, 2013), and psychoneuroimmunological functions (Fancourt et al., 2014).
This review analyzes numerous empirical studies associating engagement with music and inflammatory biomarkers related to COVID-19, with a focus on neurological/neuropathological effects. Although its focus is on the effects of music, effects of sound-based studies (e.g., white vs. no noise at a particular decibel level; white noise vs. drum sound) investigating anatomical and physiological outcomes in animal models are included to complement the music-based clinical interventions. The introduction discusses music, COVID-19 and the chemokine/cytokine storm. The body of the article expands upon these topics and incorporates/ evaluates stress response, blood-brain barrier (BBB) integrity, and the pro-inflammatory and anti-inflammatory biomarkers and hormones associated with engagement in and responses to music. It concludes with a discussion of the relationships between music, COVID-19, inflammation, biomarkers, and neuropathology.
Methods
Application of the meta-narrative review method (Wong et al., 2013) brought awareness to the complementary ways in which music interventions address inflammatory responses associated with COVID-19. Study selection was completed through hand searching (Hopewell et al., 2007). Independent researchers conducted a comprehensive search and analysis of experimental and observational studies, and reviews using PubMed, Embase, OneSearch, Primo, Google Scholar, and Clinicaltrials.gov. Search strategies and inclusion/exclusion criteria were iteratively developed. A flow diagram indicating stages of inclusion/exclusion with reasons was not included as we did not conduct a formal meta-narrative review.
Search terms and phrases began with a foundation of “COVID-19 and inflammation” and “Music and Inflammation.” After several reiterations of this foundation, for example, “SARS-CoV-2 and inflammation,” the scope of our analysis expanded, and searches specifying biomarkers, neuropathology, and blood clotting were included. The search terms used were as follows: “COVID-19” and “psychological effects”; “COVID-19” and “neurological effects”; “neurology” and “COVID-19”; “inflammation” and “neurology” and “COVID-19”; “interleukin levels” and “COVID-19”; “interleukin 6 levels” and “COVID-19”; “interleukin 10 levels” and “COVID-19”; “cytokines” and “COVID-19”; “music” and “neurology”; “music” and “neurology” and “inflammation”; “music therapy” and “inflammation”; “music therapy” and “neurology”; “music therapy” and “pain”; “music therapy” and “psychological effects”; “music therapy” and “stress response”; “music therapy” and “stress response” and “neurology”; “music” and “cytokines”.
Four independent reviewers screened article abstracts and titles and conducted full-text analysis from potentially relevant research articles. All searches were conducted between August 2020 and December 2021. Selection and appraisal of articles required an iterative process of relevance judgements specific to the topic areas of interest. Each researcher reviewed articles independently. Discussion among reviewers determined the inclusion or exclusion of articles based on study design, potential methodological bias, competing interests, and overall focus upon topic areas of interest to this review, namely ways in which music has been used to address inflammatory biomarkers shown to associate with symptoms of COVID-19. Eighty-four articles, published between 1964 and 2021, were included for full-text review and discussion. Thirty-five articles were selected for data extraction and display in one of two tables: Table 1 includes 16 clinical and two preclinical studies using music or noise to affect inflammatory biomarker concentrations; Table 2 includes 16 clinical and one preclinical study focused on biomarker concentration changes due to effects of COVID-19.
Inflammatory biomarker concentration changes due to exposure to music: literature review.

ACC = anterior cingulate cortex, ACTH = adrenocorticotropic hormone, β-EP = β-endorphin, CO2 = carbon dioxide, CORT = cortisol, d = day, DHEA = dehydroepiandrosterone, F = female, fMRI = functional magnetic resonance imaging, GH = growth hormone, hr = hours, IgA = immunoglobulin A, IL = interleukin, M = male, min = minute, mo = month, M/F = male to female ratio, n = number of participants, NA = noradrenalin, NK = natural killer, POMS = Profile of Mood States, SAS = Self-Rating Anxiety Scale, sec = second, TNF = tumor necrosis factor alpha; VAS = visual analogue scale, wk = week, yr = years.
Biomarker concentration changes due to COVID-19: literature review.

Note: CCL2, C-C motif ligand 2; CCL3, C-C motif ligand 3, CSF = cerebrospinal fluid, CRP = C-reactive protein, CNS = central nervous system, d = days, F = female, hr = hour, ICU = intensive care unit, IFNγ, interferon, IL = interleukin, M = male, M/F = male to female ratio, MLR = monocyte/lymphocyte ratio, n = number of participants, NfL = neurofilament light chain, NLR = neutrophil/lymphocyte ratio, PTSD = post-traumatic stress disorder; SII = systemic immune-inflammation index, TNF = tumor necrosis factor alpha, wk = weeks, yr = year.
For a more complete understanding of the topics discussed, no articles were barred from analysis because of their publication date. No quality assessment or assessment of bias template was used. This study was limited to articles originally published or translated into English. Selection criteria included articles with research methods that focused on the effect of music-based therapeutics on human physiology and inflammation, as well as COVID-19 and central nervous system (CNS) pathophysiology. Analysis of music and acoustic parameters was not limited by delivery methods, style, or genre.
Results
General Overview of Music, Inflammation, Stress, and Biomarkers
A book chapter of the neurochemical functions of music (Koshimori, 2019) indicates that music impacts multiple neurotransmitters (e.g., dopamine, serotonin, norepinephrine, and acetylcholine) and neuropeptides (e.g., beta-endorphin, oxytocin, and arginine vasopressin). A recent systematic review provided evidence that music interventions have effects on both physiological stress-induced biomarkers and self-reported psychological stress responses (Wong et al., 2021). Additionally, experimental evidence has established that stress directly induces physiological responses, including alterations to inflammatory signaling cascades, the HPA axis, and both the peripheral and central renin-angiotensin system (Welcome & Mastorakis, 2020). Physiological responses are similarly influenced in viral states (Alifano et al., 2020; Silverman et al., 2005). Therefore, this review evaluated the qualitative and quantitative effects of music and acoustics on pathophysiology, inflammatory biomarkers, and immune and stress responses. It should be noted that noise/music can become a stressor depending on exposure/administration methods. For example, Shukla and a team of researchers found that mice experienced statistically significant increases in pro-inflammatory cytokines (TNF, IL-1α, IFNγ, and IL-6) and a statistically significant decreases in anti-inflammatory cytokines (IL-4 and IL-10) after being exposed to 100dB of music for 2 hrs a day for several days (Shukla et al., 2020).
Pro-inflammatory Biomarkers
IL-6 is a cytokine that plays a major role in signaling an inflammatory response. In acute infections, this allows the body to increase blood circulation in afflicted areas, recruit cells to eliminate a pathogen, and differentiate needed cells for a particular threat. However, the continued creation of IL-6 is known to promote several chronic health conditions. For example, patients with systemic lupus erythematosus have higher levels of IL-6 than healthy controls (p = 0.009) (Linker-Israeli et al., 1991).
Currently, researchers are exploring the potential of music-based therapeutics to treat adverse effects of inflammatory conditions. An experiment by Koelsch et al. (2016) showed that exposure to uplifting music allowed 143 participants to better respond to physical stressors, primarily by lowering the release of pro-inflammatory cytokines such as IL-6 (p = 0.001). However, the relevance of the content and intensity of the music used in such interventions remains unknown. For example, researchers found that rats, which are cognitively impacted by music in an entirely different manner than human models, experienced a similar decline in IL-6 levels when periodically exposed to drumming that mimicked the rhythm of the human heartbeat (Kim et al., 2015). Overall, evidence suggests that music-based interventions are capable of decreasing IL-6 levels in organisms. As shown by Okada and colleagues, music-based therapeutic intervention decreased the levels of IL-6 in older adult patients who suffered from autoimmune diseases and patients who suffered from heart failure (Okada et al., 2009). Notably, the impact was seen in patients with pre-existing conditions, including dementia. These results and similar findings from other experiments indicate that music-based interventions can possibly be utilized to decrease the adverse pro-inflammatory effects of IL-6 in patients with COVID-19.
Interferon γ (IFNγ) is a type II interferon produced during antiviral innate immune responses (Lee & Ashkar, 2018). IFNγ acts in response to inflammatory signaling cascades to recruit various molecules and enhance the inflammatory response (Zhang, 2007). Ongoing research has been conducted to establish a cause and effect relationship between various Type I and Type II interferons with the hope of better regulating inflammatory responses in humans. Type II interferons are released from natural killer cells. Wachi and colleagues investigated expression rates in response to recreational music-making activities. In this experiment, IFNγ mRNA expression decreased in response to music participation. This decrease in expression is consistent with findings supporting the anti-inflammatory properties of acoustic therapy in human models (Wachi et al., 2007).
Tumor Necrosis Factor (TNF) is a cytokine that acts on the vascular endothelium and endothelial leukocytes to promote pro-inflammatory responses in the human body. In conjunction with chemokine release, the local effects of TNF on endothelial cells result in inflammatory responses (Bradley, 2008). Music-based interventions can reduce the adverse effects of TNF in inflammatory diseases by reducing its concentration. Kim and colleagues regularly exposed 15 mice to the sound of a Buk drum; these mice consequently experienced a statistically significant decrease in TNF expression (p < 0.05) (Kim et al., 2015). These findings indicate that music-based interventions can be utilized as an affordable, noninvasive way to manage excessive TNF production.
Anti-inflammatory Biomarkers
IL-10 is an anti-inflammatory cytokine stimulated by a pathogenic host immune response. As a means to regulate the immune and subsequent inflammatory response, IL-10 plays a critical role in infection pathogenesis, hemodynamics, and tissue homeostasis (Iyer & Cheng, 2012). Wachi and his team found decreases in IL-10 mRNA expression in 40 healthy males (mean age = 38.4; SD = 8.4) due to participation in recreational music-making activity over 6 months (Wachi et al., 2007). Thus, evidence supports the theory that music, auditory, and acoustic interventions have significant physiological effects on biomarker titers and mRNA expression levels, and as such, these therapies may be useful in regulating inflammatory pathways.
Immunoglobulin A (IgA) plays a role in immune homeostasis and stands out as a prevalent antibody isotype after secondary exposure to an antigen. In conjunction with multiple receptors, signaling cascades, and dendritic cells, IgA has been confirmed to play a significant role in T-cell and anti-inflammatory regulation in viral and non-viral physiological responses (Mkaddem et al., 2014). Knight and Rickard found that listening to a prepared music selection, i.e., Pachelbel's Canon in D major, in conjunction with a stressor actively increased salivary IgA levels (Knight & Rickard, 2001). Interestingly, Hirokawa and Ohira demonstrated in different experimental conditions, where participants served as their own controls, that salivary IgA levels were not significantly altered between test conditions when listening to various music categories (Hirokawa & Ohira, 2003).
Hormones
Cortisol actively works as an anti-inflammatory hormone and has long been linked to stress- and illness-induced inflammatory pathways. Adrenocorticotropic hormone (ACTH) is released by the anterior pituitary gland and targets the adrenal gland to stimulate the production of cortisol; it is closely linked to the production and eventual effects of cortisol.
Both acute and chronic stress states have been shown to increase pro-inflammatory biomarkers and decrease anti-inflammatory biomarkers. This trend has been upheld experimentally in cases involving cortisol. Separate teams led by Koelsch and Thoma established that in stress-induced states, ACTH and both serum and salivary cortisol levels increased significantly (p < 0.05) (Koelsch et al., 2016; Thoma et al., 2013).
In clinical and non-clinical settings, music and acoustic therapies have been experimentally shown to reduce serum and salivary cortisol levels. For example, Le Roux and colleagues demonstrated a decrease in serum cortisol and cortisol to DHEA ratios when patients with respiratory conditions were involved in music-based interventions (le Roux et al., 2007). Khan and his team of researchers further established that critical care and postoperative patients experienced decreased cortisol titers in response to music intervention (Khan et al., 2018). There is reasonable evidence to indicate that music can influence underlying inflammatory pathophysiology, which serves as a precursor to ACTH and cortisol release, evidenced by a decrease in titer levels during acoustic activity.
Epinephrine and norepinephrine are hormones that are discharged in stress-induced immune responses. Levels of norepinephrine increased with exposure to uplifting music in one experiment testing the effects of various types of music on immune responses (Hirokawa & Ohira, 2003). Therefore, we can conclude that norepinephrine reacts to stress stimuli, and music type plays a role in trafficking norepinephrine in response to stimuli. Furthermore, another study gathered data demonstrating that epinephrine and norepinephrine concentrations differed between experimental groups participating in and those withheld from music exposure conditions during stressor events (Chlan et al., 2007). These experimental outcomes support the idea that epinephrine and norepinephrine play a role in the immune response system and can be influenced by music exposure and mood or stress states.
COVID-19 and Relationships Between Stress, Inflammation, Biomarkers, and Neuropathology
Blood-Brain Barrier, Stress Response, and COVID-19 as a Stressor
The BBB comprises cerebral microvascular endothelial cells and is surrounded by astrocytes, pericytes, perivascular microglia, oligodendrocytes, neurons, and the extracellular matrix. The BBB regulates the movement of molecules such as water, ions, nutrients, and metabolites from the blood to the histo-hematic barrier, and furthermore, the CNS (Welcome & Mastorakis, 2020).
Stress is a response to a threatening stimulus and has uncomfortable and/or harmful effects. Moderate stress levels can enhance performance, but exceeding these levels potentially prompts CNS disorders such as post-traumatic stress disorder, anxiety, depression, etc. The hypothalamic-pituitary-adrenal (HPA) axis is the primary mediator of stress as it regulates hormone production and distribution in response to stress stimuli (Welcome & Mastorakis, 2020).
Tight junction and adherens junction proteins at the BBB are downregulated due to stress, which causes hyperpermeability or a “leakage” in the BBB. When the permeability of the BBB is altered, cerebral neuroinflammation, neurodegeneration, as well as behavioral and other cognitive disorders that are seen in stress-related conditions can occur. The influx of pro-inflammatory factors, such as interleukin (IL)-23 and TNF, from the peripheral circulation to the BBB aids in the mediation of the compromised permeability. A mouse model of chronic social defeat stress illustrated this phenomenon. The intrusion of IL-6 from peripheral circulation caused BBB hyperpermeability through a decreased expression of tight junction proteins, which resulted in depression-like symptoms (Menard et al., 2017). This study displays that an increase in pro-inflammatory cytokines occurs in conjunction with stress-induced behaviors and this relationship demonstrates that treatment with TNF or hydrogen peroxide can potentially alter BBB permeability (Lum et al., 2003; Welcome & Mastorakis, 2020). These studies support the theory that excessive stress (and its accompanying physiological changes) stemming from COVID-19 infection may perpetuate many of the same stressed induced outcomes.
Varied Neurological Involvement of COVID-19
According to a variety of literature, neurological manifestations are common for patients with COVID-19 (Chen et al., 2020). SARS-CoV-2 enters the CNS, specifically astrocytes, macrophages, and microglia, and induces a pro-inflammatory reaction. Inflammatory mediator levels are elevated, including IL-1β, IL-2, IL-4, IL-6, IL-10, IFNγ, and TNF. Additionally, antibodies and immunoglobulin G (IgG), immunoglobulin M (IgM), and IgA levels increase as the virus progresses.
Mohammadi and his team found that cytokine storm created by SARS-CoV-2 increased the progression of neurodegeneration and potentially neurodegenerative disease development. SARS-CoV-2 induces continuous systemic inflammation, which may prompt an increase in microglial activation. They also postulated that these activated microglia secrete increased levels of pro-inflammatory cytokines, such as IL-1β, IL-6, IL-12, and TNF, which could aggravate neurodegeneration and lead to Alzheimer disease.
Patients with Parkinson’s disease show increased pro-inflammatory cytokines, such as IL-1β, IL-6, TNF, and IFNγ, in the cerebrospinal fluid (CSF). Thus, SARS-CoV-2 could aggravate neurodegeneration progression in patients with Parkinson’s disease resulting from an increased number of cytokines distributed into the CSF in patients with COVID-19. Additionally, microglial activation occurs because of increased cytokine release, which can compound the activation that patients with Parkinson’s disease commonly experience. Furthermore, a cytokine storm influenced by SARS-CoV-2 infection in the CSF dysregulates dopamine activity and may influence Parkinson’s disease progression.
Moreover, cytokine storms of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF, increase the risk of ischemic and hemorrhagic stroke. This has been demonstrated in experimental models that display anti-cytokine intake, resulting in decreased stroke recurrence (Mohammadi et al., 2020). Research has proposed that excessive levels of IL-6 obstruct T-cell differentiation into regulatory cells and, therefore, increase inflammation and demyelination in mice (Jung et al., 2019). This continuous inflammation and induction of myelin destruction may lead to delayed multiple sclerosis progression.
In the acute stages of Guillain–Barré Syndrome (GBS), immune responses toward pro-inflammatory cytokines occur and levels of IL-1β and IL-6 in the CSF increase. In addition, the increased pro-inflammatory cytokine development as a result of SARS-CoV-2 may contribute to neurodegeneration and thus GBS progression. Also, the damaged integrity of the BBB promotes the permeation of lymphocytes and could lead to GBS (Mohammadi et al., 2020).
Pro-inflammatory Biomarkers
Several studies have described the abundance and role of IL-6 in influencing COVID-19 symptoms. For example, K18-hACE2 mice had a 500-fold increase in IL-6 mRNA production in lung and brain cells when infected with a strain of COVID-19 (Kumari et al., 2021). In addition, in clinical trials with patients infected with COVID-19, patients had a statistically significant increase in IL-6 production when experiencing a severe case of COVID-19 (p < 0.05) (Henry et al., 2020). Similarly, a research team led by Han found statistically significant evidence that elevated IL-6 levels indicated severe symptoms (e.g., organ dysfunction, death) (Han et al., 2020). These studies in humans and mice suggest that increased levels of IL-6 impact immunological dysregulation and contribute to symptoms. In a clinical setting, a study by Lavillegrand and colleagues found similar results where COVID-19 patients who had higher levels of IL-6 were more likely to have a severe case of COVID-19 (Lavillegrand et al., 2021). These two studies demonstrate the pivotal role of an elevated IL-6 concentration in producing immune dysregulation and escalating symptoms in COVID-19 patients.
Interferon γ (IFNγ), an additional example of a common pro-inflammatory cytokine, was shown by Xiong and colleagues to be elevated in symptomatic children infected with COVID-19 relative to asymptomatic children infected with COVID-19 (p < 0.05). Notably, the data from this study also stated that symptomatic children had higher concentrations of other pro-inflammatory cytokines, for example: IL-6 and TNF (Xiong et al., 2020). This supports the claim that the simultaneous increase in several pro-inflammatory cytokines is a major element of the immune dysregulation and resulting symptoms in COVID-19 patients. Additionally, a separate study focused on COVID-19's impact on the central nervous system displayed that in organisms with COVID-19, TNF experiences a 750-fold increase in concentration in the brain and throughout the rest of the body (Kumari et al., 2021).
Anti-inflammatory Biomarkers
IL-10 is an anti-inflammatory cytokine involved in pathogenic immune responses (Iyer & Cheng, 2012). IL-10 production is increased in response to inflammation, injuries, and stress-induced immune responses (Curtin et al., 2009; Slavish & Szabo, 2019).
In a clinical study conducted in Wuhan, China, Han and colleagues found higher serum levels of cytokines in patients with COVID-19 than in control patients. Elevated levels of cytokines, including IL-10, are part of the inflammatory immune responses in the lungs (Han et al., 2020). Additionally, researchers analyzed cytokine levels in the CSF and serum of patients with COVID-19. The CSF of patients with inflammatory neurological conditions had increased levels of IL-10, whereas non-inflammatory neurological disorders did not have the same marked increase in IL-10 (Gomes et al., 2021). These studies suggest that IL-10 is one of many biomarkers of COVID-19-related inflammation and should be considered a predictor of disease severity.
Furthermore, IL-4 can exhibit anti-inflammatory behaviors via the up-regulation of cytokine inhibitors (Renu et al., 2020). T-helper type 2 (Th2) is associated with inflammation pathways in regards to the production of humoral immunological responses, like increases in antibodies (Renu et al., 2020). Specifically, IL-4 cell promotion is symbiotic of Th2 cells; the interleukin blocks Th1 pathways to promote Th2 pathways and thus generation of cells (Renu et al., 2020). In COVID-19 patients Th2 cell levels were high, an indication of an increased IL-4 presence (Renu et al., 2020). The presence of SARS-CoV-2 spike proteins increases the activation of Th2 cells and has the potential to change immunological responses to the protein as well as causing COVID-19 sepsis, overall promoting degeneration of the immune system (Renu et al., 2020) (Figure 1).
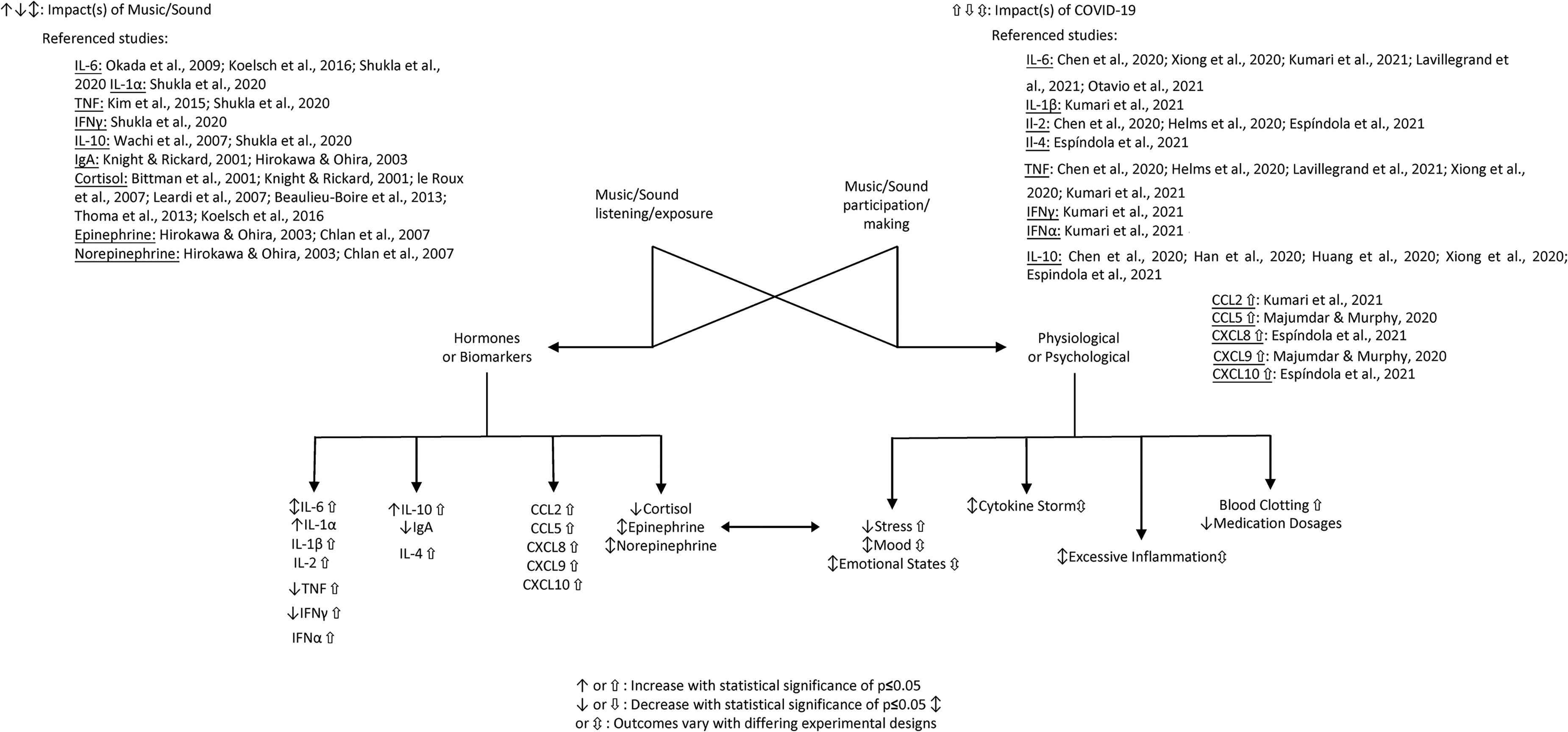
Music/sound has differing experimental definitions with parameters varying in specific music types/styles (e.g., multiple classical music selections, self-assessed favorite music, techno music, white noise), noise/decibel levels, and administration methods (headphones, stereo audio, live instrument exposure/manipulation). The reviewed studies primarily defined music administration through experimental methodologies based on listening/exposure to provided music sources or through hands-on manipulation or participation in the creation or playing of music. These methodological differences largely evaluated similar outcomes (biomarker/physiological alterations) and, as such, are included together. An analysis of COVID-19 literature established that the biomarkers altered upon COVID-19 infection are like those altered via music involvement; thus, the effects of each are compared and illustrated. Alterations depicted are based on evaluation of experimental research/articles included in more depth in Table 1 and Table 2.
Discussion
The evidence presented in this review indicates the potential for the use of music-based therapeutic interventions as a treatment for symptoms of COVID-19, specifically disease-mediated alterations in cytokine concentrations. However, no formal study has tested this prediction. Therefore, we cannot comment on the extent to which music-based interventions can be used to treat COVID-19 patients. Several studies show increases in numerous pro-inflammatory biomarkers, such as IL-6 and cortisol, when an individual suffers from an inflammatory disease such as COVID-19. This literature review also found inverse relationships; some studies documented music-mediated decreases in many of the same biomarkers that COVID-19 increased, such as IL-6. This is a significant finding due to the pivotal role of these cytokines and hormones in causing the potentially dangerous “cytokine storm” in patients with COVID-19 (Hojyo et al., 2020).
Inclusion of experiments from different global regions and populations leads us to understand that the anti-inflammatory effects of music/sound therapy may not be culturally specific or caused by exposure to a particular genre of music. The majority of symptomatic COVID-19 cases are considered to primarily affect respiration. However, in addition to commonly expressed lung infiltrates and symptoms, including cough, breathing difficulties, and respiratory failure, there is growing evidence that COVID-19 plays a role in both cardiovascular and cerebrovascular diseases, as well as hemorrhage incidence. Separate teams led by Chen and Ranucci conducted studies that demonstrated that patients with COVID-19 infection experience an increase in clot strength and factors contributing to clot severity, such as increased platelet, fibrinogen, ferritin, and D-dimer levels (Chen et al., 2020; Ranucci et al., 2020). Similarly, Varatharaj and colleagues found that in cases of COVID-19 infection, there were statistically significant increases in cerebrovascular events and, more specifically, increased occurrences of ischemic stroke and intracerebral hemorrhage (Varatharaj et al., 2020). These studies suggest that in addition to the typical expression of symptoms, COVID-19 influences blood, affecting cerebral processes and structure.
New research has indicated that the extracellular traps created through NETosis may negatively affect patients with COVID-19. It is now suggested that when neutrophils undergo NETosis at an abnormally high rate to counter a COVID-19 infection, there is an increased risk of thrombosis occurring in capillaries in the lungs. (May, 2021). The neutrophils that undergo NETosis are activated by several different signals: notably, this includes pro-inflammatory cytokines like TNF (Gupta et al., 2010). As shown in this paper, music-based interventions tend to lower the levels of the pro-inflammatory cytokines that are needed for excessive neutrophil activation. Therefore, exposure to therapeutic music may lower the chance that a patient undergoes thrombosis during a COVID-19 infection. The validity of this statement needs to be experimentally tested.
It is commonly reported that exposure or participation in music-based therapies influence the cardiovascular system and, subsequently, blood flow. Independent studies led by Knight, Lai, and Wang concluded that participation in such activities markedly altered blood flow, decreasing heart rates and lowering systolic and diastolic blood pressures (Knight & Rickard, 2001; Lai et al., 2013; Wang et al., 2015). Experimentation in this realm commonly suggests a relationship between music and physiological function. These results indicate that in cases of COVID-19 and similar disease processes, music could play a role in managing cerebrovascular disease and hemorrhage by influencing blood flow.
A recent report on biomarker and therapeutic discovery (May, 2021) associates long-term COVID-19 symptoms with pulmonary, cardiovascular, and autonomic systems. In highlighting the patient complaints specific to autonomic dysfunction, the author suggests that particular to cases involving dysautonomia (a group of conditions associated with the problematic function of the autonomic nervous system), antibodies produced in response to SARS-CoV2 infection may attack the autonomic nervous system and affect the ability of normal nerve function to facilitate regular blood flow to the brain.
Because of limited knowledge on how the stress response impacts the immune system, there continues to be ambiguity regarding the mechanism by which music reduces stress and improves immunological outcomes. Research in integrative health interventions (e.g., yoga) more broadly is providing complementary evidence to the potential positive effects of music interventions upon manipulation of stress to decrease inflammation (Bushell et al., 2020). A review by Bushell et al. (2020) found that yoga reduced the levels of pro-inflammatory cytokines (Bushell et al., 2020). Whether these findings translate to a promising method to mitigate the severity of COVID-19 infection would require a separate study. However, it is noteworthy that a handful of studies have shown that yoga positively affects patients who live with highly inflammatory diseases such as systemic lupus erythematosus (Middleton et al., 2018). Furthermore, due to increasing emphasis on the role of an “eicosanoid storm” in developing immune dysregulation in COVID-19 patients (Hammock et al., 2020), future research on how music-based intervention regulates biomarkers involved in eicosanoid regulated pathways should also be investigated. Finally, the impact of music listening on the concentration of IL-1 in humans needs to be further investigated. At the time of writing this article, there is an insufficient number of articles that have published data on IL-1 level changes due to music to make any meaningful conclusions on its impact.
In conducting this literature review, we encountered limitations finding relevant articles because of the novelty of our topic. For example, at the time of this writing, no studies have directly researched the impact of music/noise on patients with COVID-19. Additionally, because SARS-CoV-2 is a relatively new disease, there are limitations in data collection concerning sample size and demographics (e.g., age, gender, pre-existing medical condition), a lack of variation in research techniques and methodologies, a lack of standardized experimental precautions (including double-blind arrangements), and an ever-evolving understanding of the COVID-19 viral process. Additionally, limitations arose standardizing information pertaining to COVID-19 with regard to the numerous COVID-19 variants and their subsequent differences.
A lack of standardization concerning music articles and experimentation, specifically regarding music type, decibel levels, forms of administration, and demographics, made findings complicated to quantify, compare, and evaluate. Additionally, compiling data from both human and animal model studies posed limitations to the qualification, quantification, and comparison of relevant results. An incomplete understanding of the impact music/sound has on animal models posed difficulties in concisely comparing seemingly relevant data sets between different animal and human models. Finally, all articles were limited to those available to the University of Florida, online, and in English.
The consensus among available research is that music has measurable and statistically significant impacts on the human body's inflammatory and physiological regulatory processes. Many of the pathways in inflammatory specific viral processes, specifically COVID-19, overlap with the areas altered by music exposure and participation; therefore, further research and experimentation are required to establish if there is a direct effect between music exposure/therapy and viral inflammation, especially in cases of cerebrovascular impact.
Footnotes
Action Editor
Eckart Altenmüller, Institut für Musikphysiologie und Musikermedizin, Hochschule für Musik, Theater und Medien Hannover.
Peer Review
Two anonymous reviewers.
Author Contributorship
Conceptualization, S.D. and A.C.; investigation, J.S., J.C. and A.P.; resources, S.D.; writing, J.S., J.C., A.P., and A.C.; revising and editing, S.D., J.S., J.C., A.P., and A.C.; funding acquisition, S.D.; All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This research did not require ethics committee or IRB approval. This research did not involve the use of personal data, fieldwork, or experiments involving human or animal participants, or work with children, vulnerable individuals, or clinical populations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health, Department of Defense, (grant number NS095166 , NS103036, NS110008, NS116076, AZ180127).
