Abstract
This study conducted a comprehensive analysis of oxidative stress markers in human semen within the context of assisted reproductive technology (ART) and investigated if these markers varied according to male age. 148 semen samples were collected from 139 men presenting at Concept Fertility Centre, Western Australia, from 2009 to 2012. Semen analyses were performed and demographic information including smoking status and abstinence period was collected. Reactive oxygen species production, lipid peroxidation, oxidative DNA damage (8-hydroxy-2′-deoxyguanosine (8-OHdG)), total antioxidant capacity and DNA fragmentation ( terminal deoxynucleotidyl transferase-mediated deoxyuridine diphosphate nick-end labelling) were measured as markers of oxidative stress. Semen parameters and oxidative stress markers were compared against age as a continuous variable, and between males <40 and males ≥40 years of age. Older males aged ≥40 years exhibited higher levels of sperm oxidative DNA damage (8-OHdG) compared to younger males (
Introduction
The effect of male age on fertility is often overlooked, with the majority of age-related fertility research typically focusing on the well-documented effect of female age. However, there is a growing body of evidence indicating that advanced male age can affect fertility, and male reproductive ageing becomes particularly relevant considering the increases in paternal (and maternal) age observed in Australia (Australian Bureau of Statistics, 2012) and other industrialized nations such as the United Kingdom and the Nordic countries (Schmidt et al., 2012). The trend to delay reproduction to a later age is reflected in assisted reproductive technology (ART) demographics, where the average age of both male and female patients has been steadily increasing over the past decade. In Australia and New Zealand, the average age of women undergoing an autologous or recipient ART cycle in 2010 was 36.0 years, while the average age of their partners was 38.3 years with a third of partners aged 40 years or older (Macaldowie et al., 2012). However, despite the scientific and clinical consensus that male age is an increasingly important factor in ART, very little is understood about the mechanisms underlying the relationships between male age and reduced fertility.
While several studies have found that advanced paternal age is linked to a variety of compromised semen parameters including a reduction in seminal volume, percentage of motile sperm and percentage of morphologically normal sperm (Johnson et al., 2015; Kidd et al., 2001; Krausz and Chianese, 2013; Levitas et al., 2007; Oliveira et al., 2014; Stone et al., 2013), the extent to which these impact downstream fertility outcomes in older men remains uncertain (Bellver et al., 2008; Dain et al., 2011; Nijs, 2011). However, these standard semen parameters only provide a limited perspective of reproductive function, and it is likely that there are other underlying biological mechanisms at work. Recent research suggests that age-related decreases in sperm concentration or motility cannot completely account for the relationship between paternal age and reduced pregnancy rate (Koh et al., 2013).
Over the past decade, attention has been drawn to other subcellular measures of sperm quality, most notably DNA damage. Sperm DNA damage is associated with several adverse outcomes including increased miscarriage rates (Zini et al., 2008), abnormal embryonic development (Seli et al., 2004) and compromised chromosomal integrity in the embryo (Aitken and Krausz, 2001). In addition, there is a concern for the health of any resulting offspring with regard to genetic defects and/or disease as a consequence of chromosomal abnormalities (Green, 2010; Kong et al., 2012). Sperm with damaged DNA do not necessarily lose their ability for fertilization (Gandini et al., 2004). This is important to note within an ART context, since procedures such as intra-cytoplasmic sperm injection (ICSI) bypass natural selection mechanisms that may prevent compromised sperm from fertilizing an oocyte in vivo. Sperm DNA damage is also an important factor to consider for an ageing reproductive population, as older male age has been associated with higher levels of DNA damaged sperm (Oliveira et al., 2014; Plastira et al., 2007) and may be a significant contributor to the relationship between male age and reproductive outcome in ART (Humm and Sakkas, 2013).
A major contributor to sperm DNA damage is oxidative stress (Aitken and De Iuliis, 2010). In the brown Norway rat, advanced age has been associated with elevated oxidative stress levels in epididymal spermatozoa (Weir and Robaire, 2007). It therefore follows that increased oxidative stress in the reproductive system of older men may lead to compromised sperm quality, ultimately impacting on fertility outcome. Oxidative stress occurs when highly reactive molecules known as reactive oxygen species (ROS) overwhelm the antioxidant defence capacities of the cell, leading to the damage of various cell components including lipids, proteins and DNA (Halliwell and Gutteridge, 2007). The majority of DNA damage sustained by sperm originates from oxidative stress, as evidenced by high correlations observed between the production of the major oxidative adduct 8-hydroxy-2′-deoxyguanosine (8-OHdG) and DNA fragmentation (Aitken et al., 2010). Oxidative damage in sperm can also occur in the form of membrane lipid peroxidation, where the high levels of polyunsaturated fatty acids are particularly vulnerable to ROS attack, leading to a loss of membrane fluidity and compromised sperm motility (Aitken et al., 1989). It is therefore possible that any age-related increase in oxidative stress in the reproductive system may manifest in decreased fertility outcome by way of compromised sperm quality.
There are two basic pathways in which age may affect fertility via oxidative stress. One possibility is that older men have increased levels of ROS and/or decreased antioxidant capacities in semen (Cocuzza et al., 2008). Alternatively, ROS and antioxidant levels are not necessarily altered with age, but the vulnerability of the sperm cell to oxidative damage increases. A reduced capacity to repair sperm DNA strand breaks in older men compared to younger men has been noted (El-Domyati et al., 2009).
Although many oxidative stress parameters have been previously studied in semen, research that considers multiple measures of oxidative stress within a single subset of samples is lacking, particularly in relation to male age. The aim of the present study was to conduct a comprehensive analysis of oxidative stress markers in human semen within the context of ART and to investigate if these markers varied according to male age. Based on epidemiological studies reporting decreases in male fertility with age occurring around the age of 40 years (De La Rochebrochard et al., 2006; Sartorius and Nieschlag, 2010), we hypothesized that men aged ≥40 years would have increased levels of seminal oxidative stress markers (increased ROS generation and decreased antioxidant capacity), and that their sperm would exhibit elevated levels of oxidative damage to the sperm membrane and DNA.
Materials and methods
Study population
The study population consisted of 139 men presenting at Concept Fertility Centre, Western Australia, between 2009 and 2012. A total of 148 semen samples were analysed from this cohort, with no more than two samples collected from each man. The population included patients presenting with male factor infertility, as well as men with no known fertility problems who were submitting a semen sample (e.g. partners of female patients). Semen was collected by masturbation into sterile containers. Only leftover portions of samples following clinical analysis were used in this study and no aspect of patient treatment was compromised as a result. The only inclusion criterion was the presence of sperm in the ejaculate (i.e. no azoospermic samples) and that there was at least 0.5 ml of neat semen left over after clinical use. All men provided informed consent, and the project was approved by The University of Western Australia Ethics Committee (RA/4/1/2506) and the King Edward Memorial Hospital for Women Ethics Committee (1470/EW) prior to commencement of the study.
Semen analysis
Standard semen analyses were performed by laboratory staff at Concept Fertility Centre according to World Health Organization (WHO) guidelines (World Health Organization, 2010). Parameters assessed included sperm concentration, sperm morphology (on initial presentation of subject only) and sperm motility. Motile sperm concentration following PureSperm (NidaCon Laboratories AB, Sweden) density gradient centrifugation (DGC) was also assessed.
Lifestyle characteristics
At the time of sample production, participants completed a questionnaire regarding various lifestyle characteristics. The questions comprised: the presence of febrile illness during the preceding 3 months (‘illness’), medications consumed in the preceding 3 months (‘medications’), antioxidant supplementation during the preceding 3 months (‘antioxidants’) and cigarette smoking during the preceding 3 months (‘cigarettes’). As per standard protocol at the clinic, a sexual abstinence period of 2–3 days was recommended to all participants prior to sample collection; however, men were not excluded from analyses if they did not adhere to these guidelines. The most common antioxidant supplementation reported was Menevit (Bayer Healthcare) with 6 of the 18 men (33%) reporting antioxidant supplementation taking this. No medications known to impact on sperm function were reported. The variation in lifestyle variables including abstinence period was investigated in relation to age, oxidative stress and sperm quality (detailed in section ‘Results’).
Fertility status
WHO standards were used to classify men as either being fertile or subfertile according to their sperm sample parameters. As per WHO guidelines, fertile men were those with normozoospermic samples where sperm concentration, progressive motility and morphology met WHO reference values (World Health Organization, 2010). A total of 140 samples had sufficient data (i.e. data available on sperm concentration, motility and morphology) to classify them as either fertile or subfertile; 113 samples were classified as fertile and 27 were classified as subfertile.
Measures of sperm ROS generation and oxidative damage
Sample preparation
From each neat semen sample, 0.5 ml was separated using a discontinuous gradient (80% and 40% PureSperm in human tubal fluid (HTF); NidaCon laboratories AB, Sweden) and the remainder was centrifuged at 350 ×
The isolated high-density sperm pellet from the discontinuous gradient was washed with 2 ml of in-house HTF medium and centrifuged at 400 ×
ROS generation
Intracellular ROS generation was measured using dihydroethidium (DHE) based on protocols set out by De Iuliis et al. (2006). Briefly, 50 µl aliquots of fresh sperm preparations that had been stripped of leucocytes were labelled with DHE (Sigma-Aldrich, Australia) and Sytox green (SyG; Molecular Probes, USA) to give final concentrations of 4 µM and 0.05 µM, respectively, in a final volume of 400 µl. Cells were incubated in the dark for 15 min at 37°C before DHE and SyG fluorescence was measured using a FACScalibur flow cytometer (Becton Dickinson, USA). Argon laser excitation at 488 nm was coupled with emission measurements using 530/30 band pass (green) and 585/42 band pass (red) filters for FL1 and FL2, respectively. Sperm-specific events were gated according to forward- and side-scatter properties. Within the sperm population, non-viable SyG-positive cells were gated out, and the mean DHE fluorescence value of the remaining cells used as an indicator of ROS generation. All samples were assayed in duplicate.
Lipid peroxidation
Lipid peroxidation was measured using 4,4-difluoro-5-(4-phenyl-1,3-butadienyl)-4-bora-3a,4a-diaza-s-indacene-3-undecanoic acid (BODIPY) based on the protocols set out by Guthrie and Welch (2007) and Aitken et al. (2007). In short, 50 µl aliquots of fresh sperm preparations that had been stripped of leucocytes were labelled with BODIPY (Invitrogen Life Technologies) and propidium iodide (PI; Sigma-Aldrich) to give final concentrations of 2 µM and 9.6 µM, respectively, in a final volume of 400 µl. Cells were incubated in the dark for 30 min at 37°C before BODIPY and PI fluorescence was measured using a FACScalibur flow cytometer (Becton Dickinson) as described above. Within the sperm population, non-viable PI-positive cells were gated out, and the mean BODIPY fluorescence value of the remaining cells was used as an indicator of lipid peroxidation. All samples were assayed in duplicate.
Oxidative DNA damage – 8-OHdG production
Oxidative DNA damage was measured using a fluorescein isothiocyanate (FITC)-labelled protein which binds to the 8-oxoguanine moiety of 8-oxoguanosine (OxyDNA Test; Argutus Medical, Ireland). The assay was carried out on fixed cells following the manufacturer’s instructions and protocols described by De Iuliis et al. (2009) without modification and determined by flow cytometry. Within the sperm population, 8-OHdG-positive cells were gated based on FITC fluorescence. The percentage of 8-OHdG positive cells was reported as an indicator of oxidative DNA damage.
Flow cytometry
All flow cytometric analyses of DHE, BODIPY and 8-OHdG were performed with a FACSCalibur flow cytometer (Becton Dickinson) with two laser lines at 488 nm and 635 nm, using CellQuest Pro software (Becton Dickinson). Data was then extracted using FlowJo Version 7.6 (multiplatform). Samples were run for 2 min each in duplicate, or until 10,000 live cells had been gated.
Rainbow fluorescent particles (BD Biosciences, Australia) were used to calibrate instrument settings before each run. Unstained samples and positive controls were included in each assay. Sperm collected from the low-density portion of the PureSperm gradient was used as the DHE positive control. For the BODIPY positive control, cells were treated with 80 µM of ferrous sulphate to promote the lipid peroxidation chain reaction (Aitken et al., 2007). The positive control for the 8-OHdG assay was made by treating cells with 2 mM of hydrogen peroxide and 2 mM of dithiothreitol prior to fixation. Elevated responses were observed in the positive controls for all the above assays (independent
DNA fragmentation – Terminal deoxynucleotidyl transferase-mediated deoxyuridine diphosphate nick-end labelling assay
To assess DNA fragmentation, the ApopTag Plus In Situ Apoptosis Detection Kit (S7101, Chemicon International, Australia) was used to carry out the terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine diphosphate nick-end labelling (TUNEL) assay. Briefly, 20 µl of neat semen was smeared over silanized glass microscope slides (Superfrost Plus, LabServ, Canada) and air dried. Slides were then washed in PBS and permeabilized in 0.1% Triton X-100 (Sigma-Aldrich, Australia). DNA strand breaks were detected using the kit according to the manufacturer’s instructions and as detailed by Taylor et al. (2009). The percentage of sperm with DNA fragmentation was calculated by counting 400 sperm per sample under 1000× microscopy using an oil immersive lens. A negative control slide was assayed with each run using reaction buffer instead of working strength TdT enzyme.
Measures of antioxidant defences in the seminal plasma
Total antioxidant capacity
The total antioxidant capacity (TAC) of the seminal plasma was measured using an antioxidant assay kit (Cayman Chemical Company, USA) according to the manufacturer’s instructions and protocols set out by Mahfouz et al. (2009). The assay is based on antioxidants in the sample inhibiting the oxidation of 2,20-azino-di-[3-ethylbenzthiazoline sulphonate] (ABTS) to ABTS+ by metmyoglobin. Samples were diluted in the ratio 1:10 using assay buffer (Cayman Chemical Company) prior to assaying. Trolox (Cayman Chemical Company) standards and reagents were prepared according to the manufacturer’s instructions. Standards were assayed in triplicate, while all samples were assayed in duplicate. ABTS+ production was measured by reading absorbance at 405 nm on a plate reader (Multiskan RC; Labsystems, Finland). Results were reported as micromolar Trolox equivalents.
Statistics
All statistical analyses were performed using SPSS version 15.0. All semen parameters (except percentage of motile sperm), abstinence duration, daily number of cigarettes smoked and all oxidative stress measures (ROS generation, lipid peroxidation, 8-OHdG production, DNA fragmentation and TAC) were not normally distributed. With the exception of the DNA fragmentation TUNEL data, these were log-transformed to improve the normality of the sample prior to statistical analyses. For clarity, untransformed values are presented in tables and figures in the section ‘Results’. Pearson correlations were used to relate oxidative stress markers to male age as a continuous variable, while independent student’s
Data for all variables was not always available for all samples due to a lack of information provided by patients on their questionnaire, restrictions of sample volume or patient clinic records being incomplete. For the DHE assay, four cases (all from one run) were excluded from statistical analyses due to controls running significantly higher during that run. The actual sample size for each analysis is indicated in the relevant tables.
Results
Age ranged from 24 years to 58 years with a mean of 36.4 ± 0.53 years. Forty-six subjects (30.9%) were aged ≥40 years, which reflects male age at the clinic (approximately 1/3 aged ≥40 years) and is consistent with reports that one-third of ART recipients’ partners are aged ≥40 years in Australia and New Zealand (Macaldowie et al., 2012).
Table 1 describes the overall semen quality of the cohort. Men abstained for 4.91 days (standard deviation = 3.56) prior to sample collection (range = 1–28 days). The frequency of reported lifestyle factors that may impact on semen parameters is presented in Table 2.
Mean, SD and ranges of sperm motile concentration, sperm motility, sperm morphology, total sperm count following DGC and live sperm count following DGC.
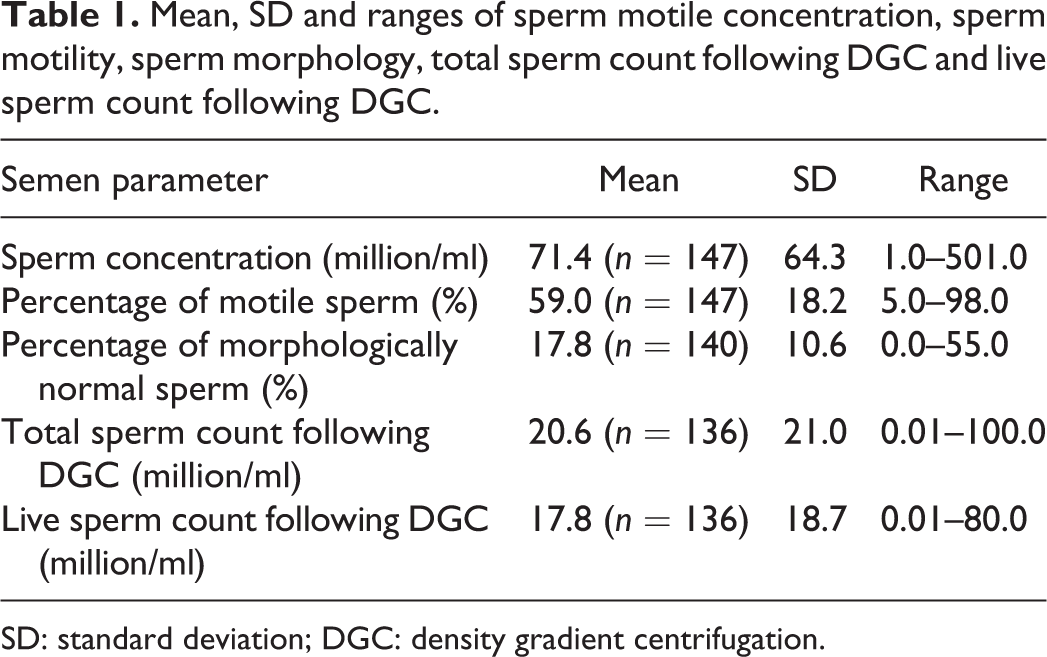
SD: standard deviation; DGC: density gradient centrifugation.
Reported lifestyle characteristics of males aged <40 years and males aged ≥40 years.
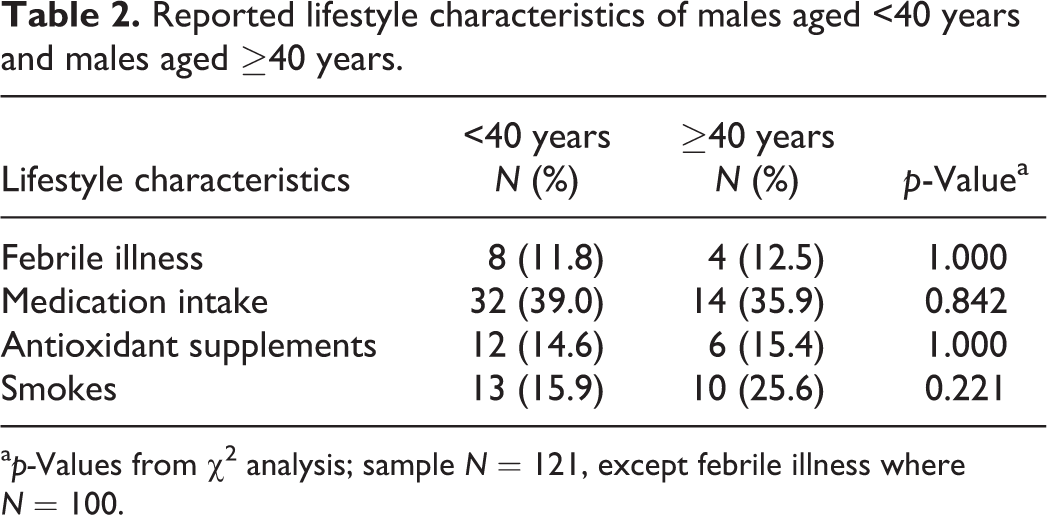
a
Older men aged ≥40 years had similar abstinence lengths (5.5 ± 0.9 days,
Correlations between semen parameters and oxidative stress markers
Increased oxidative stress was associated with poorer semen parameters (Table 3). However, TAC was not significantly associated with any semen parameter or any other oxidative stress marker with the exception of an inverse relationship with oxidative DNA damage as measured by percentage of 8-OHdG positive cells.
Correlations between semen parameters and oxidative stress markers.a
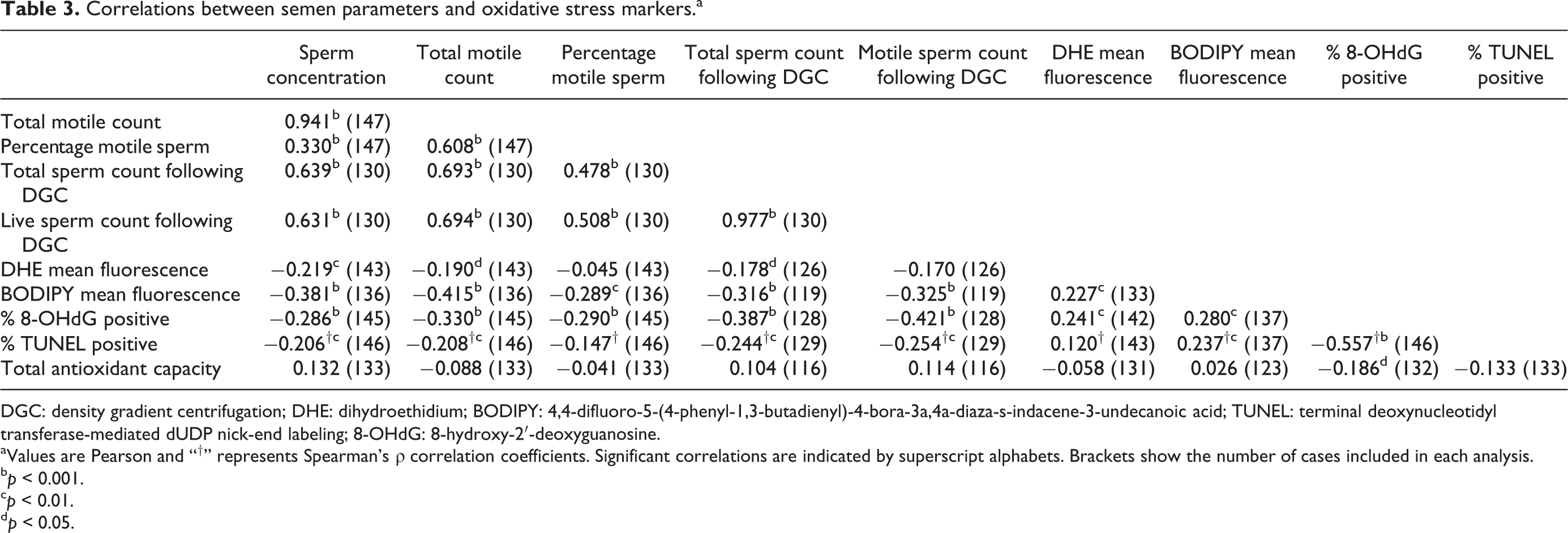
DGC: density gradient centrifugation; DHE: dihydroethidium; BODIPY: 4,4-difluoro-5-(4-phenyl-1,3-butadienyl)-4-bora-3a,4a-diaza-s-indacene-3-undecanoic acid; TUNEL: terminal deoxynucleotidyl transferase-mediated dUDP nick-end labeling; 8-OHdG: 8-hydroxy-2′-deoxyguanosine.
aValues are Pearson and “
b
c
d
Effect of age on semen parameters
Males aged ≥40 years had poorer total and live sperm concentrations following DGC compared to males aged <40 years (Table 4). However, neat semen parameters of sperm concentration, total motile count and percentage of motile sperm did not differ between older and younger males.
Comparisons of semen parameters between males aged <40 years and males aged ≥40 years.a

DGC: density gradient centrifugation.
aValues are means and SEM. Samples sizes are indicated by round brackets.
b
cIndependent samples
Effect of age on oxidative stress parameters
There were no differences in DHE fluorescence, BODIPY fluorescence, DNA fragmentation or TAC between older and younger men (Figure 1). Older men had significantly higher levels of 8-OHdG positive cells compared to younger men (Figure 1(c)). When age and fertility status (fertile/subfertile) were included together in a two-way ANOVA, subfertility was positively associated with the percentage of 8-OHdG positive cells (
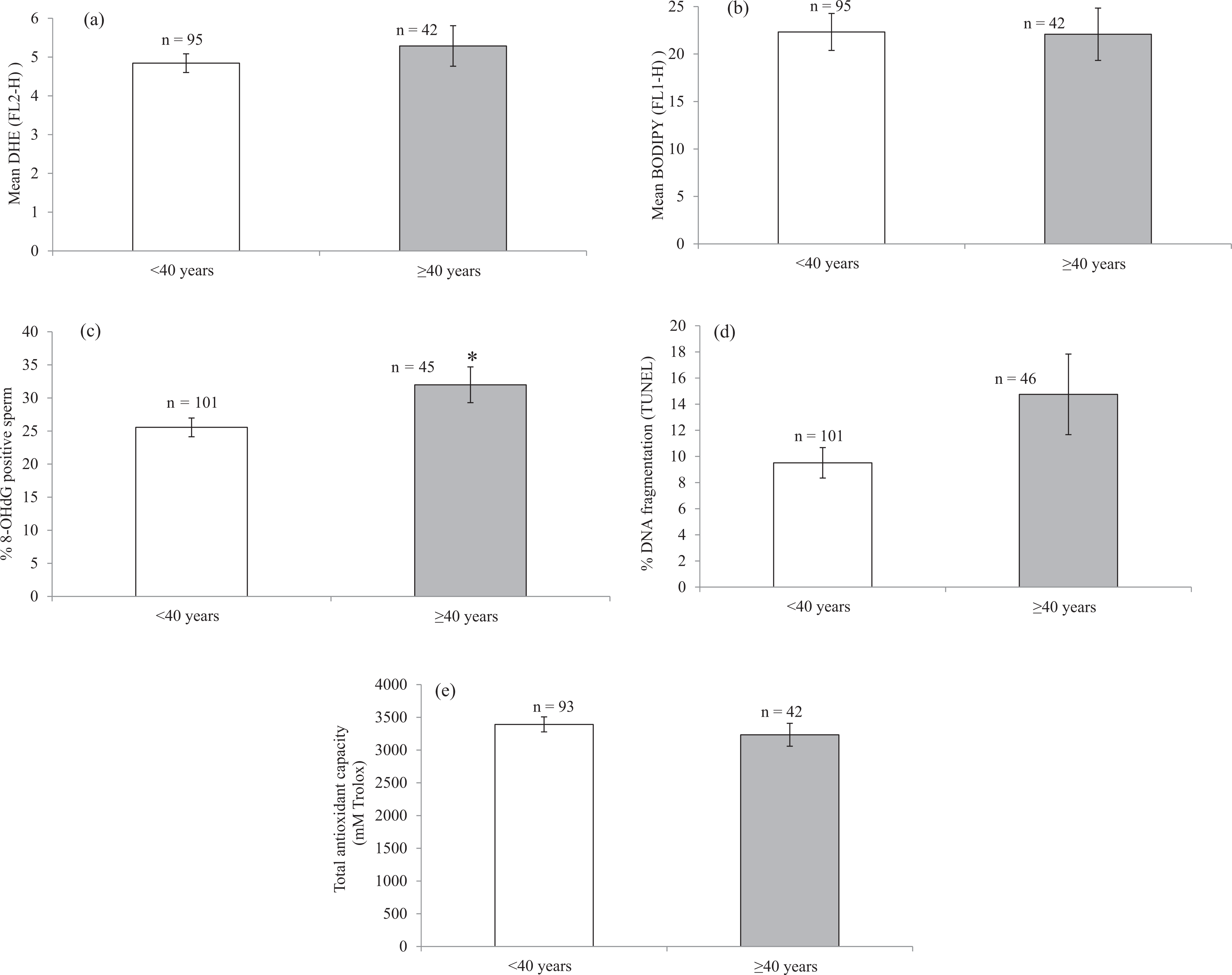
Comparisons of oxidative stress markers between males aged <40 years and males aged ≥40 years. Plots show (a) mean DHE fluorescence, (b) mean BODIPY fluorescence, (c) percentage of 8-OHdG positive sperm, (d) percentage of DNA fragmentation TUNEL-positive sperm and (e) total antioxidant capacities.
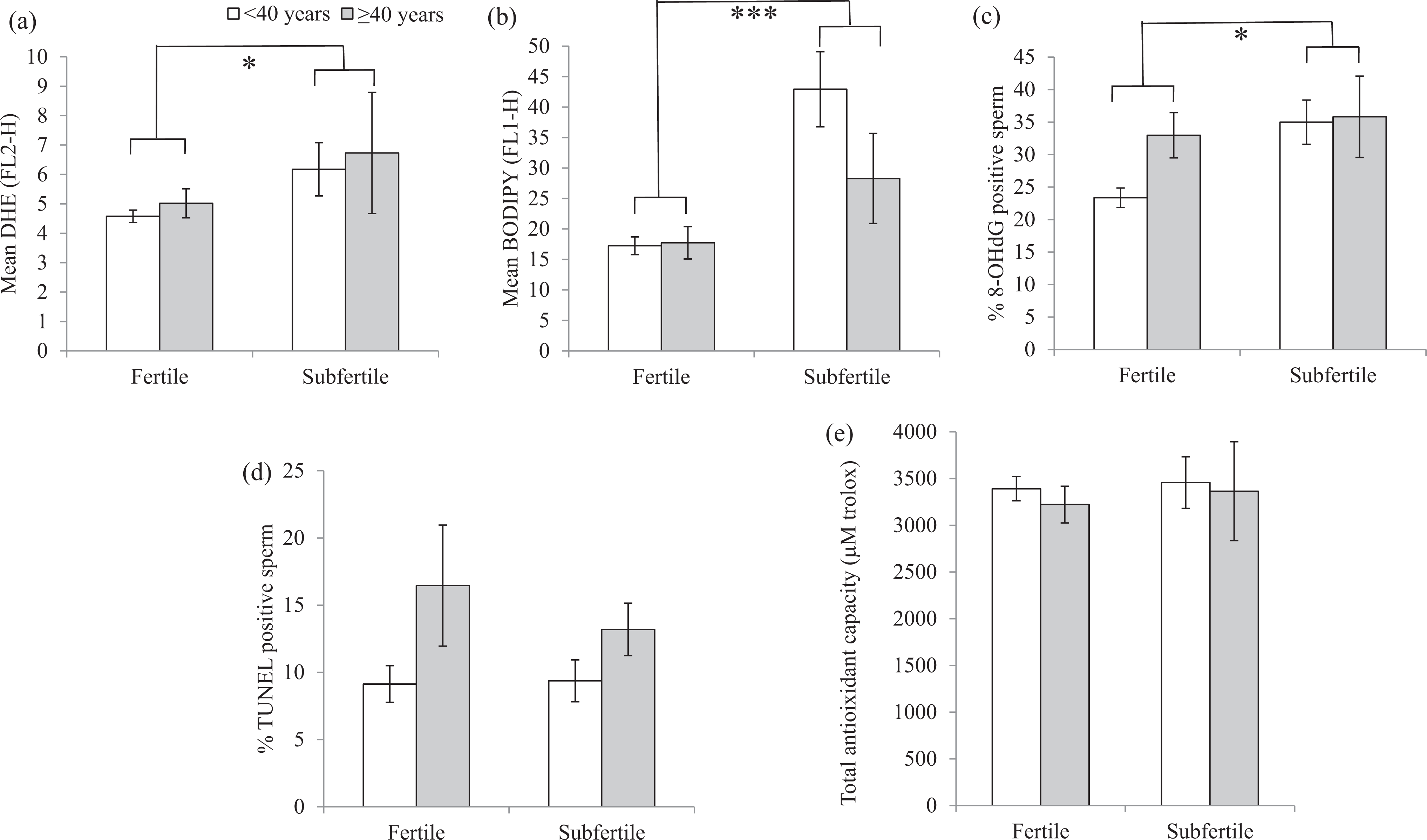
Oxidative stress markers (DHE fluorescence, BODIPY fluorescence, % 8-OHdG positive sperm, % TUNEL positive sperm and total antioxidant capacity) between men aged <40 years and ≥40 years according to fertility status. There are no significant interactions between age and fertility status.
Discussion
The main findings of this study were that older males aged ≥40 years exhibited poorer live sperm count after DGC and higher levels of sperm oxidative DNA damage compared to younger males. However, ROS production, lipid peroxidation, general DNA fragmentation and TAC levels did not significantly vary with age. Independent of male age, subfertile samples generally had elevated levels of oxidative damage.
Oxidative damage to sperm DNA arises via increased exposure of the cell to oxidative stress, compromised capacity of the cell to cope with oxidative stress or a combination of the above two pathways. On first glance, the results from the present study suggest that older men do not have increased levels of oxidative stress. Instead, it appears that their sperm have a greater vulnerability to ROS attack compared to younger men. Defective remodelling mechanisms during spermiogenesis result in poorly packaged sperm chromatin that contains base adducts including 8-OHdG which is formed as a consequence of oxidative stress (Barratt et al., 2010). It is therefore possible that older males are more prone to errors during spermiogenesis, leading to reduced quality of the sperm DNA, rendering it more susceptible to oxidative attack.
A reduced capacity to cope with oxidative stress may also arise from defective DNA repair mechanisms. Studies on the brown Norway rat and mice have reported an age-related increase in 8-OHdG levels in sperm, as well as a decreased germ cell capacity to repair DNA damage in older animals (Ozkosem et al., 2015; Paul et al., 2011). Human males possess some ability to repair oxidative DNA damage via 8-oxoguanine-DNA glycosylase-1 (OGG1) excision of 8-OHdG, although this mechanism has been shown to be limited compared to the more efficient rat system (Olsen et al., 2003). It is possible that a compromised OGG1 repair system in older males is responsible for the higher 8-OHdG levels observed in this study. Future studies investigating the efficacy of the OGG1 repair system in older men are therefore warranted.
Another explanation for the findings of this study may be that older males have increased seminal oxidative stress levels leading to increased oxidative DNA damage, but these differences in oxidative stress levels between older and younger men have not been observed. This may be due, in this study, to the sample processing steps undertaken prior to ROS analyses. ROS measurements were made on sperm taken from the high-density portion of the PureSperm gradient to reflect sperm used for assisted reproduction. However, ROS-generating sperm are more likely to be in the low-density portion of the gradient as these cells retain their cytoplasmic droplet around the midpiece where the ROS-producing mitochondria is located (Koppers et al., 2008). Mitochondrial ROS generation by sperm is hence associated with an increase in lipid peroxidation and a subsequent reduction in sperm motility (Dandekar et al., 2002). This ROS-induced decrease in motility leads to these cells being excluded during ART processing (Aitken et al., 2013).
In contrast to lipid peroxidation, damage to the DNA occurs relatively downstream in the process of oxidative damage (Aitken et al., 2013). An oxidative attack to sperm DNA induces the generation of high levels of the 8-OHdG enzyme (Valavanidis et al., 2009). It is possible that in vivo, high levels of ROS not only attack the DNA of the ROS-generating sperm themselves but may also diffuse to neighbouring cells and cause oxidative DNA damage there. These neighbouring sperm may not possess the same defects in chromatin remodelling during spermiogenesis and can otherwise be of relatively good quality themselves with no retention of excess cytoplasm and no excessive ROS production. These cells are therefore isolated and selected during discontinuous density gradient selection. Nevertheless, they sustain oxidative DNA damage from the high levels of ROS produced by adjacent defective sperm. As such, the present study may only be picking up the increased oxidative DNA damage in older men but not any age-related increases in ROS production or lipid peroxidation.
In this study, older men had decreased sperm concentrations and motile sperm counts after DGC. Neat semen parameters were also lower, however did not reach statistical significance possibly due to greater variation. The age-related decrease in total and motile sperm counts following DGC may reflect higher levels of ROS-generating sperm in the neat ejaculate of older men; this would result in a greater number of cells being discarded following DGC, reducing post-DGC sperm counts as discussed earlier.
Within an ART context, age-related low sperm concentrations and motile counts can be bypassed using technologies such as ICSI, unlike age-related increases in oxidative DNA damage which persist and remain undetected during routine ART procedures. Notably, 8-OHdG levels in the present study were highly correlated with increased DNA fragmentation indicating that a significant proportion of DNA fragmentation in sperm is linked to oxidative stress. This is in agreement with several previous studies (Sakkas and Alvarez, 2010; Santiso et al., 2010) and highlights the significance of oxidative stress in the aetiology of DNA damage and subsequent fertility outcome. However, DNA fragmentation levels were similar between younger and older men suggesting that other factors may be contributing to DNA fragmentation in younger men including pollution, radiation and dietary deficiencies (Hadwan et al. 2014; Morris, 2002; Rubes et al., 2007).
Analyses of seminal plasma TAC revealed no age-related variations in antioxidant defences between younger and older men. This indicates that the elevated oxidative DNA damage in older men is not due to compromised antioxidant capacity of the seminal plasma. TAC was also not significantly associated with any semen parameter or oxidative stress measure other than 8-OHdG. It is worth noting that in vivo TAC levels of the seminal fluid may not predict oxidative stress in sperm during ART because of isolation of sperm from the seminal plasma during ART processing. Our observed association between antioxidant supplementation and TAC is encouraging, although it is not possible to ascertain if dietary antioxidant intake was the direct cause of high TAC in the seminal fluid. It is possible that men who reported antioxidant intake were more conscious of their health in general, leading to a variety of possible factors such as better diet that would contribute to improved TAC.
Decreased sperm concentration and motility in the neat sample as well as lower total and live sperm counts following DGC were associated with high levels of oxidative DNA damage, suggesting that compromised overall sperm quality is generally accompanied by oxidative stress. Furthermore, samples classified as subfertile possessed significantly higher levels of oxidative stress, including raised DHE, BODIPY and 8-OHdG levels. This is in agreement with the previous studies reporting increased ROS production (Saleh et al., 2003), lipid peroxidation (Nakamura et al., 2002) and 8-OHdG levels (Kodama et al., 1997; Nakamura et al., 2002) in subfertile men relative to fertile controls. The effect of age seen in our study may have been mediated by fertility status since the effect of age on 8-OHdG production was not significant when fertility status was included in the statistical model. This suggests that older men have poorer quality sperm in general (Johnson et al., 2015), which relates to the higher levels of oxidative stress; however, causal direction cannot be confirmed.
It is important to consider the potential limitations of this study. The samples and subjects used in this study were from an ART cohort, rather than the general community. The findings are therefore most relevant and applicable within the context of fertility treatment. Samples were only included if they had sufficient volume as samples with very low volumes could not be aliquoted for the various measures. This may have led to underrepresentation of samples from subfertile men. Consequently, any observed adverse effects of male age and semen quality are conservative. Finally, TAC was used rather than individual measures of specific antioxidants. It is possible that some antioxidants provide better protection against oxidative stress in older men than others.
In conclusion, to the authors’ knowledge, this is the first study to demonstrate an age-related elevated level of oxidative DNA damage in human spermatozoa. We propose that this elevated oxidative DNA damage may contribute to the decreased pregnancy rate previously observed in older sperm donors undergoing donor insemination (Koh et al., 2013). This is supported by the findings of the previous research which indicates that 8-OHdG production is predictive of clinical pregnancy rates during intrauterine insemination (Thomson et al., 2011). The fact that an age-related increase in 8-OHdG production continues to be observed in samples that have been processed by DGC indicates that oxidative stress is indeed relevant to ART, as sperm processing techniques are intended to isolate sperm with the best fertility potential for insemination. These findings indicate that the sperm with elevated levels of 8-OHdG in older men are not removed during sperm processing. This is likely to adversely impact on treatment outcomes such as pregnancy success rates and also carries the risk of increasing the chance of chromosomal abnormalities in any resultant offspring.
Footnotes
Acknowledgement
The authors acknowledge the facilities, and the scientific and technical assistance of the Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy, Characterisation & Analysis, The University of Western Australia, a facility funded by the University, State and Commonwealth Government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
