Abstract
Introduction: Generalized pustular psoriasis (GPP) is a chronic autoinflammatory disease characterized by fever, erythema, and sterile pustules occurring over large areas of the skin. The immunopathogenesis of GPP remains poorly understood. We aimed to profile serum cytokine levels in patients with GPP and analyze treatment-associated changes in cytokine levels. Methods: Twenty-four patients who were diagnosed with GPP and received drug treatment, together with 28 unaffected controls, were recruited in this study. Levels of 27 cytokines in serum samples from pre- and post-treated patients as well as unaffected controls were assessed using a Bio-Plex multiplex immunoassay. Multivariate analysis of cytokine levels and clinical parameters was conducted. Results: Serum levels of interleukins interleukin (IL)-1β, IL-1Ra, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12 (p70), IL-13, IL-17, granulocyte colony-stimulating factor (G-CSF), interferon-γ (IFN-γ), IFN-γ-induced protein 10 (IP-10), monocyte chemoattractant protein-1, tumor necrosis factor-α, and vascular endothelial growth factor were significantly elevated (p < .05) in patients with GPP compared with controls. Serum levels of IL-6, G-CSF, IL-8, IFN-γ, IL1-Ra, IL-10, IL-13, and IL-1β were correlated with GPP severity (r > 0.4, p < .001). Following treatment, serum levels of IL-6, IL-7, IL-12, IFN-γ, IL-1β, IL-4, IL-8, IL-10, IL-13, G-CSF, and IP-10 were significantly decreased (p < .05) compared with pretreatment levels. Among these cytokines, levels of IL-6, IL-7, IL-8, IL-12, IL-13, and IP-10 were reduced to normal levels. Conclusion: GPP patients exhibited markedly elevated levels of cytokines and chemokines involved in both type 1 and type 2 immune responses, suggesting hypercytokinemia occurs in GPP patients. IL-6 showed the strongest correlation with GPP disease severity and other clinical parameters such as CRP level and fever, thus representing an index for GPP severity. Our data comprehensively documented the cytokine profiles of GPP patients and may provide insights into GPP pathogenesis.
Keywords
Introduction
Psoriasis is a chronic inflammatory disease that affects 0.47% of the population in China. 1 Psoriasis is characterized by abnormal skin with erythron-squamous lesions and can be classified into different types based on clinical features. Generalized pustular psoriasis (GPP) is a rare and potentially life-threatening form of psoriasis and makes up 0.98% of psoriasis cases in China. 1 GPP is characterized by widespread eruption of sterile, subcorneal pustules and epidermal scaling, often coupled with fever, fatigue, and joint or muscle pain. Subtypes of GPP include acute GPP (von Zumbusch psoriasis), circinate and annular pustular psoriasis, pustular psoriasis of pregnancy, and infantile/juvenile pustular psoriasis. 2
Although consensus criteria for diagnosis of GPP have been defined, the pathogenesis of GPP remains poorly understood. GPP is often triggered by environmental factors and immune disorders such as pregnancy, infections, drugs, and hypocalcemia. 3 It has been reported that GPP may be characterized by a distinct pattern of aberrant autoinflammation associated with recessive mutations in IL36RN, a gene encoding interleukin (IL)-36Ra that inhibits the activities of multiple IL-36-associated cytokines (e., IL-36α, IL-36β, and IL-36γ). 4 Loss-of-function mutations in IL36RN lead to downstream activation of nuclear factor (NF)-κB signaling and enhanced secretion of inflammatory cytokines such as IL-1, IL-6, and IL-8. 5 However, IL36RN mutations alone are not sufficient to cause GPP, and a variety of other gene mutations and variants have been implicated in GPP pathogenesis. 6 Recently, we and others have demonstrated that levels of mRNA encoding multiple proinflammatory genes, including IL17 A, TNF, IL1, and IL36, were markedly upregulated in GPP skin biopsies and the peripheral blood mononuclear cells of patients with GPP compared with those of unaffected controls.7,8 These studies suggested that dysregulation of cytokines/chemokines may play a critical role in GPP. However, few systematic evaluations of cytokines/chemokine levels at the protein level in patients with GPP have been conducted.
In the current study, we evaluated levels of a large panel of cytokines, chemokines, and growth factors in the periphery of patients with GPP and unaffected controls using a multiplex immunoassay. Levels of these inflammatory factors in pre- and post-treated patients with GPP were examined. By analyzing correlations with clinical parameters and performing multivariate statistical analysis, we identified multiple differentially expressed inflammatory factors and demonstrated their potential implication in GPP pathogenesis. Our study may provide insights into GPP diagnosis and inform development of anti-cytokine therapies.
Methods
Study population
Clinical and laboratory characteristics of patients and unaffected controls enrolled in this study.
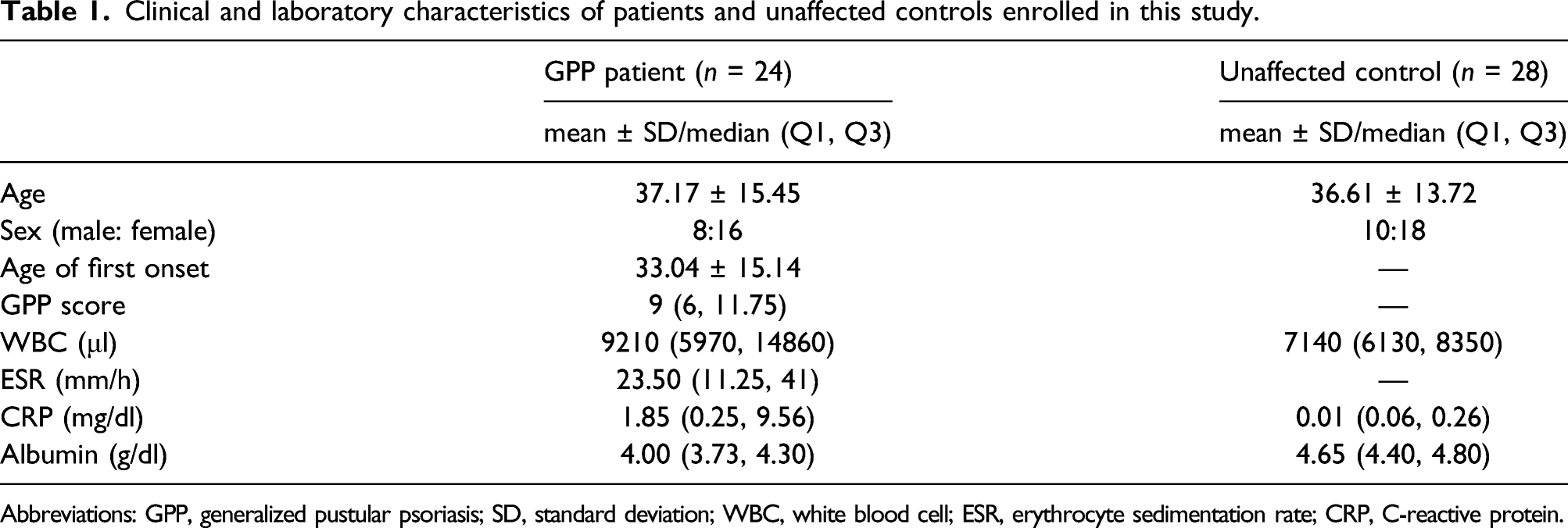
Abbreviations: GPP, generalized pustular psoriasis; SD, standard deviation; WBC, white blood cell; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein.
Volunteers undergoing physical examination were served as controls (10 men and 18 women, mean ± SD age 36.61 ± 13.72 years). Controls had no signs of ongoing inflammation (blood tests and CRP levels were normal), had no history of psoriasis, and were medication-free for at least 3 months prior to physical examination. The study protocol was approved by the Ethics Review Committee of Peking Union Medical College Hospital, Chinese Academy of Sciences (approval number JS-1900–2). All data were treated anonymously.
Diagnostic criteria for GPP
Diagnosis of GPP was made according to established criteria as described previously. 9 All five of the following criteria must have been met: (1) systemic symptoms such as fever and general malaise; (2) multiple, isolated non-infectious pustules in flushed skin all over the body or over a wide area; (3) histopathologically confirmed Kogoj’s spongiform pustules; (4) at least one of the following laboratory results: leukocytosis with left shift, increased ESR, elevated CRP, increased anti-streptolysin O antibody levels, increased IgG or IgA levels, proteinemia, or hypocalcemia; and (5) recurrence of these clinical and histological findings.
Blood collection and serum preparation
Blood samples (5–10 mL) were obtained from both patients and unaffected controls following written informed consent. Blood was collected in endotoxin-free silicone-coated tubes without additives. Blood samples were allowed to sit at room temperature for 1 h then centrifuged at 3000 r/min for 10 min at 4°C. Serum was then collected and stored at −80°C in sterile screw-cap tubes and thawed immediately prior to analysis.
Multiplex immunoassay
Serum levels of IL-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-12 (p70), IL-13, IL-15, IL-17, eotaxin, macrophage chemoattractant protein (MCP)-1, macrophage inflammatory protein (MIP)-1α, MIP-1β, interferon-gamma (IFN-γ); IFN-γ-induced protein 10 (IP-10), granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), fibroblast growth factor (FGF)-basic, platelet-derived growth factor (PDGF)-bb, regulated on activation, normal T cell expressed and secreted (RANTES), tumor necrosis factor (TNF)-α, and vascular endothelial growth factor (VEGF) were measured using Bio-Plex Pro Human Cytokine 27-plex Assay kit (Cat# M500KCAF0Y, Bio-Rad, Hercules, CA, USA) and a Bio-Plex 200 System according to the manufacturers' instructions. Cytokine standards were included on each plate and all standards and samples were run in duplicate. Data were analyzed using Bio-Plex Manager 6.1. Cytokine concentrations (pg/mL) were calculated in keeping with the mean fluorescence intensity values of standards that were run simultaneously.
Statistical analyses
Statistical analyses were performed using SPSS version 22 (SPSS Inc., Chicago, IL, USA). Mann–Whitney U-tests were used for nonparametric comparisons between groups. Normally distributed continuous data were presented as means ± standard errors of the means (SEMs) or means ± SDs. For non-normally distributed continuous variables, data were presented as medians (interquartile ranges). Correlations were assessed using Spearman’s test. Two-sided values of p less than 0.05 were considered statistically significant.
Results
Comparison of serum cytokine levels in patients with GPP and unaffected controls
To comprehensively assess potential disturbances in serum cytokines associated with GPP, we examined levels of serum cytokines in 24 patients with GPP and pustular flare as well as 28 unaffected controls. Taking advantage of the xMAP multiplex technology, levels of 27 cytokines were simultaneously measured (Supplementary Table 1). As shown in Figure 1, compared with levels in unaffected individuals, levels of type 1 cytokines including IL-1β (p < .001), IL-1RA (p < .001), IL-7 (p < .001), IL-12 (p < .001), IFN-γ (p < .001), and TNF-α (p < .01) were significantly elevated in sera from patients with GPP (Figure 1(a)). Among type 2 cytokines, levels of IL-4 (p < .001), IL-5 (p < .001), IL-6 (p < .001), and IL-10 (p < .001) were significantly elevated in patients with GPP (Figure 1(a)) compared with unaffected controls. Levels of the type 3 cytokine IL-17 (p < .001) were also elevated in patients with GPP. Moreover, levels of the growth factors G-CSF (p < .001) and VEGF (p < .001) (Figure 1(b)) as well as those of the chemokines IL-8 (p = .006), IP-10 (p = .002), and MCP-1 (p = .001) were significantly elevated in patients with GPP compared with unaffected controls. Levels of the chemokine eotaxin (p = .03) (Figure 1(c)) were significantly decreased in the sera of patients with GPP compared with unaffected controls. Comparison of serum cytokine levels measured using xMAP multiplex technology between GPP patients (n = 21) and unaffected controls (n = 28). (A) Levels of cytokines. (B) Levels of growth factors. (C) Levels of chemokines. Data represent means ± SEMs. *p < .05, **p < .01, *** p < .001, n. s., not significant by Mann–Whitney U-test.
Multivariate analysis of serum cytokine levels
Because various serum cytokines/chemokines were substantially dysregulated in patients with GPP, we next investigated whether levels of a subset of cytokines could be used to discriminate between patients with GPP and unaffected controls. We applied principal component analysis (PCA) to serum cytokine data from patients and unaffected controls. PCA is an unsupervised dimension reduction technique that allows identification of combinations of variables that best describe the extent to which samples are alike; the results are then visualized as a plot. Using this analysis, we found that unaffected controls clustered together and apart from GPP patients by principal component (PC)-1, which accounted for 40.2% of the observed variability, while GPP samples were more widely dispersed (Figure 2(a)). This analysis demonstrated the different characteristics of cytokine expression patterns in patients with GPP and healthy donors. Next, we performed a hierarchical cluster analysis of cytokine levels in patients with GPP and unaffected controls. By calculating all pairwise distances among individuals, the clustering analysis clearly divided our study cohort into two major groups: cluster A and B. Cluster A consisted primarily of unaffected controls, while cluster B consisted primarily of patients with GPP (Figure 2(b)). To evaluate the diagnostic potential of cytokine levels, we performed a linear discriminant analysis. This method is used to identify a group of variables that separate two or more data classes. We found that a group of cytokines including IL-17, IL-4, IL-5, IL-12, IFN-γ, IL-6, IL-10, and G-CSF was able to discriminate between patients with GPP and controls (p < 0.0001) (Figure 2(c)). Thus, these cytokines represent potential indexes for GPP diagnosis. Associations between serum cytokine levels and GPP. (A) Principal component analysis. The projection of 21 patients with GPP and 28 unaffected controls was described by 27 serum cytokine variables for the first two principal components. (B) Hierarchical cluster analysis of serum cytokine levels in patients with GPP and unaffected controls. Results are displayed as a heat map and dendrogram. Each column represents the cytokine profile of an individual patient. Each row represents a different cytokine. (C) Correlation analysis of serum cytokine levels with GPP occurrence by linear discriminant analysis. *p < .05, **p < .01, ***p < .001, ****p < .0001.
Correlation analysis of serum cytokine levels and disease severity
Evaluation of disease severity by GPP scores.
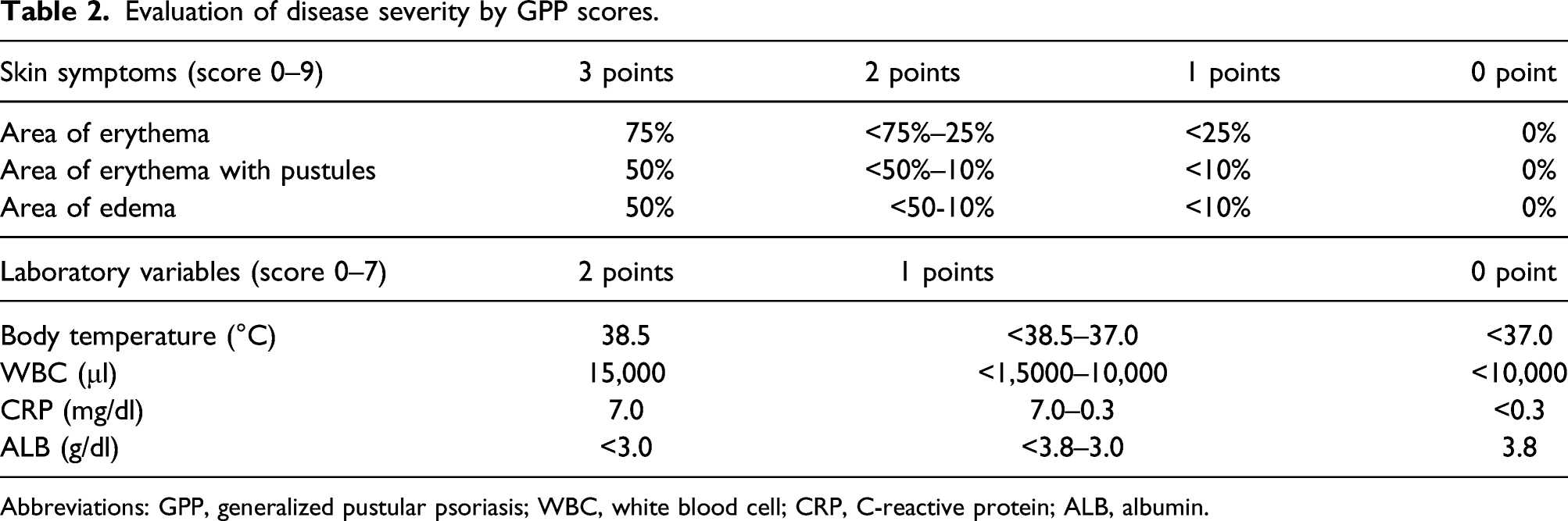
Abbreviations: GPP, generalized pustular psoriasis; WBC, white blood cell; CRP, C-reactive protein; ALB, albumin.

Associations between serum cytokine levels and GPP scores. (A) Heat map displaying Spearman correlations between GPP scores and levels of 27 serum cytokines. (B–I) Spearman correlation between serum cytokine levels, GPP scores, and CRP levels in GPP patients. R, correlation coefficient.
Comparison of cytokine levels in patients with GPP pre- and post-treatment
We next examined changes in cytokine levels in patients with GPP in response to drug treatment. Sera were collected from 12 patients with pustules at three consecutive time points: at presentation with pustular psoriasis (pretreatment), at the time of complete disappearance of pustules (stage 1), and at 4 weeks post-stage 1 (stage 2). Therapeutic choices for each patient were in accordance with disease severity and morphology. The drugs used to treat each patient are listed in Supplementary Table 3. We found that levels of IL-6, IL-7, IL-12, and IFN-γ were substantially decreased at both stage 1 and stage 2, while levels of IL-1β, IL-4, IL-8, IL-10, IL-13, G-CSF, and IP-10 were substantially decreased at stage 2 only (Figure 4(a)). Thus, 4 weeks of continuous treatment post-pustule disappearance contributed to remission of hypercytokinemia in patients with GPP. No substantial differences were observed in levels of IL-2, IL-9, IL-15, FGF-basic, GM-CSF, MIP-1α, MIP-1β, RANTES, and PDGF-bb in association with treatment (Figure 4(a) and Figures 1(a) to (c)). Levels of several cytokines markedly increased in patients with GPP, including IL-5, MCP-1, IL-17, TNF-α, and VEGF, were not substantially affected by treatment (Figure 4(a) and Figures 1(a) to (c)). We further asked to what extent levels of these cytokines decreased at stage 2. In patients with GPP, levels of IL-6, IL-7, IL-8, IL-12, IL-13, and IP-10 had returned to normal levels by stage 2 (p > .05), while levels of IL-1β, IL-4, IL-10, IL1-Ra, IFN-γ, and G-CSF were still significantly higher compared with unaffected controls (p < .05). Considering that GPP is a systemic inflammatory disease with a high relapse rate, we postulate that insufficient normalization of peripheral cytokines may contribute to GPP recurrence. Analysis of serum cytokine levels in patients with GPP pre- and post-treatment. (A) Serum cytokine levels in patients with GPP before treatment and after treatment for different lengths of time. *p < .05, **p < .01 (comparison between pretreatment and stage 1); #p < .05 (comparison between pretreatment and stage 2); all p values from Mann–Whitney U-test. (B) Comparison of serum cytokine levels between unaffected controls and patients (either pretreatment or at stage 2).
Discussion
GPP is a severe autoinflammatory disease associated with systemic inflammation, high fever, and general malaise. Autoinflammatory disorders usually occur following innate immune system dysfunction and are associated with cytokine and chemokine alterations in the periphery. 10 Our understanding of the role of serum cytokine/chemokine levels in GPP is very limited,11,12 likely because few analyses have been performed using samples from patients with GPP, a sporadic disease. In the current study, we performed quantitative analyses of serum cytokine/chemokine levels in 24 patients with GPP using xMAP multiplex technology. These analyses revealed that multiple proinflammatory cytokines/chemokines (IL-1, IL-1β, IL-1Ra, IL-4, IL-1Ra, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12, IL-13, IL-17, TNF-α, IP-10, G-CSF, INF-γ, and MCP-1) were markedly elevated in patients with GPP compared with unaffected controls. Our data suggest that a “cytokine storm” or hypercytokinemia occurs in patients with GPP, which may have fatal consequences.
Psoriasis is considered as a Th1/Th17 cell-mediated immune disease. Cytokines produced by these types T cells, including IFN-γ, IL-1 (α, β), IL-36 (α, β, γ), IL-22, TNF-α, IL-23, and IL-17, have been implicated in keratinocyte hyperproliferation and immune cell infiltration. 13 In patients with GPP, we also observed substantial elevation of cytokines produced by Th1 or Th17 cells, including IFN-γ, IL-1β, TNF-α, and IL-17. Notably, Th2 cytokines, including IL-4, IL-5, IL-6, IL-7, IL-8, and IL-10, were also markedly elevated in patients with GPP. This finding differs from prior studies of patients with psoriasis, which found that levels of serum Th2 cytokines, including IL-4 and IL-10, were unaffected. 14 - 17 This discrepancy may arise because GPP is associated with an immune regulation network that is overlapping but distinct from that of psoriasis vulgaris, as revealed by a recent gene profiling study. 7 Moreover, a serum cytokine analysis in six patients with GPP indicated that levels of IL-4 and IL-10 were considerably associated with disease severity. 11 Because IL-4 and IL-10 negatively control excessive Th1 cytokine production, we postulate that a negative feedback circuit may exist in patients with GPP, and that the balance between Th1 and Th2 T cells may be critical for the outcome of GPP treatment. Interestingly, when we examined cytokine/chemokine levels at different time points post-GPP treatment, we found that levels of Th1 and Th2 cytokines decreased in a sequential pattern: levels of the Th1 cytokines IFN-γ and IL-12 decreased at the time of complete disappearance of pustules, whereas levels of the Th2 cytokines IL-4 and IL-10 decreased several weeks later. We speculate that the delayed decrease of IL-4 and IL-10 levels may help to stop excessive inflammation in patients with GPP.
Using multivariate statistical analysis, we found that multiple factors were considerably associated with GPP disease status. First, levels of IL-17, IL-4, IL-5, IL-12, IFN-γ, IL-6, and IL-10 were significantly correlated with GPP occurrence (p < .001). Second, levels of IL-6, G-CSF, IL-8, IFN-γ, IL1-Ra, IL-10, IL-13, and IL-1β were significantly correlated with GPP scores (r > 0.4, p < .001). Third, levels of IL-7, IL-12, IL-13, IP-10, and IL-6 returned to normal following treatment (p > .05). These data may be valuable for diagnosis for GPP as well as for evaluation of therapeutic efficacy. Of note, IL-6 showed the strongest correlation with disease severity (r = 0.683, p < .0001) and was the sole factor associated with fever in patients. In addition, serum levels of IL-6 decreased rapidly to normal levels following treatment. Release of IL-6 from macrophages promotes neutrophil differentiation and activation, which may contribute to formation of pustules. 18 Thus, IL-6 could play an important role in GPP pathogenesis. Moreover, anti-IL-6 treatments have been applied to multiple autoimmune and inflammatory diseases, including rheumatoid arthritis, 19 systemic sclerosis, 20 Crohn’s disease, 21 and systemic lupus erythematosus. 22 Therefore, it would be interesting to investigate whether dysregulation of cytokines/chemokines, especially IL-6, could be potential targets for anti-cytokine therapies in patients with GPP or other types of psoriasis. Collectively, our data comprehensively depicted cytokine expression profiles in patients with GPP patients prior to and following treatment. These analyses provide new insights into GPP pathogenesis and diagnosis. However, our results were based on a limited number of samples as GPP is an extremely rare form of psoriasis. Additional studies with larger sample sizes will be needed to corroborate these results.
Conclusions
Patients with GPP showed significantly elevated levels of cytokines and chemokines involved in type 1 and type 2 immune responses. Levels of some type 1 and type 2 cytokines decreased following treatment, albeit with different kinetics. IL-6 showed the strongest correlation with GPP disease severity and other clinical parameters such as CRP level and fever. IL-6 also decreased rapidly in response to drug treatment and normalized to levels similar to those of unaffected controls. Thus, IL-6 may represent an index of GPP severity. Further studies of larger cohorts should be conducted to verify these initial findings.
Supplemental Material
sj-pdf-1-eji-10.1177_20587392221076450 – Supplemental Material for Profiling and multivariate analysis of serum cytokines in patients with generalized pustular psoriasis
Supplemental Material, sj-pdf-1-eji-10.1177_20587392221076450 for Profiling and multivariate analysis of serum cytokines in patients with generalized pustular psoriasis by Lingyan Wang, Jing Pan and Hongzhong Jin in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the National Natural Science Foundation of China (82073450), the National Key Research and Development Program of China (2016YFC0901500), and CAMS Innovation Fund for Medical Sciences (CIFMS) No.2021-I2M-1–059. We thank Dr. Zhuo Zhou for critical reading of the manuscript. We thank Liwen Bianji (Edanz) (![]() ) for editing the English text of a draft of this manuscript.
) for editing the English text of a draft of this manuscript.
Ethics approval
The Ethics Review Committee of Peking Union Medical College Hospital, Chinese Academy of Sciences reviewed and approved this study (ethical approval number JS-1900–2).
Informed consent
Written informed consent was obtained from all participants prior to enrollment in the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
