Abstract
Keywords
Introduction
Major depressive disorder (MDD) is a mood disorder that affects about 10 to 20% of the population across the world at any stage of their life. 1 It increased the mortality rate by inducing suicidal tendencies and declines the overall health which contributes to the greater risk of disease burden. 2 Several studies showed that people with mood disorders or psychosis, like schizophrenia, 3 obsessive-compulsive disorder (OCD), 4 depressive disorder,5,6 and bipolar disorder have a strong association with acute infection or autoimmune diseases caused by inflammatory mediators, like cytokines, supported by genome-wide association and epidemiological studies.6–9 Apart the association of inflammatory conditions and mental disorder, there are several potential neuropathological pathways10,11 such as reduced hippocampal neurogenesis, 12 synaptic plasticity, 13 glutamatergic excitotoxicity, 14 mutual neuroimmune exchanges 15 and alterations in microglial function 16 may involve in the neurobiology of depressive disorder. Previous studies reported an increased number of granulocytes and monocytes due to a low-grade inflammatory condition, 17 increased levels of acute-phase, 18 altered inflammatory cytokine, and chemokine levels19,20 in MDD than healthy controls (HCs). Other studies showed that cytokines cross the blood-brain barrier, and some of them can transport signals to the central nervous system (CNS).21,22 Some meta-analyses determined an altered level of peripheral c-reactive protein (CRP), IL-6, IL-12, TNFα, and IL-4 in acute depression than HCs.6,11,18,19,23 A meta-analysis of clinical trials on anti-inflammatory drugs showed some antidepressant effects.23,24 Therefore, circulatory inflammatory cytokines are promising candidate markers for depression risks. 25
Inflammatory cytokines play a vital role in brain function and an abnormal level of these markers produces detrimental effects on the brain. 26 A previous study showed that elevated levels of different inflammatory and proinflammatory cytokine markers affect the different clinical stages of MDD and depressive episodes. 27 Most meta-analyses reported the role of circulatory cytokine compounds among chronic MDD patients with antidepressant therapy.6,19 Some recent meta-analyses confirmed that the acute phase of depression is an inflammatory state where the alterations of pro-and anti-inflammatory cytokines occurred. CRP is a protein and proinflammatory marker, which levels were increased during depression, 28 and associated with a high degree of symptoms in MDD. MCP-1 plays an important role in the modulation of other cytokines via stimulating the expression of interleukin (IL)-4 in the TH-2 system, 18 which is found to be changed in MDD. An altered level of serum IFN-γ is linked with the drug-naïve MDD 29 and used as an early risk assessment tool for depression. TNF-α, a glycoprotein hormone, plays a pivotal role in the etiology of depression. 30 A meta-analysis reported the changes in circulatory cytokine levels during the first-episode psychosis 31 and depressive disorder. 32 This may directly contribute to the progress of depressive symptoms. 33 A meta-analysis of 24 studies showed that circulatory TNF-α and IL-6 levels increased in MDD than HCs. A high level of heterogeneity among these studies was reported. 19 Another meta-analysis showed that the circulatory levels of IL-6 were increased in MDD than HCs and no such alterations were observed for TNF-α and IL-1β. 18 Another meta-analysis of 82 studies with a high degree of heterogeneity reported the upregulated levels of IL-6, TNF-α, IL-10, IL-13, IL-18, IL-12, and downregulated levels of interferon-gamma (INF-γ) in peripheral blood of MDD patients than HCs. 32 Therefore, the levels of peripheral inflammatory cytokines might serve as potential risk assessment markers in MDD. However, it is unknown to what extent antidepressant therapy is associated with the alterations of peripheral cytokines in depression. A concrete understanding of the causal mechanism of MDD is a prerequisite in developing treatment strategies and preventive plans related to inflammatory cytokines. In addition, a large number of new studies are required to reduce the sources of heterogeneity in evaluating the involvement of inflammatory cytokines in patients with depression than HCs.
In this meta-analysis, we aimed to systematically review a large number of studies for the first time to investigate the association of inflammatory cytokines, like CRP, TNF-α, INF-γ, and MCP-1, in drug-naïve MDD patients and healthy controls (HCs) patients with MDD.
Method
The present study evaluated the studies that compared CRP, TNF-α, INF-γ, and MCP-1 levels between MDD patients and HCs through an observational study design. We conducted this study according to the PRISMA guidelines. 34 Two independent researchers (AAM and MUA) performed the literature search, screening after reading the titles or abstracts, final inclusion decision after full-text review, data acquisition, and methodological quality evaluation of studies. They resolved the discrepancies between them through conversation during the process. If any confusion evolved, they consulted the issues with a third researcher (MRI).
Search strategy
Database search strategy (from inception to 14 February 2021).

Inclusion and exclusion criteria
The present study included the original articles with the following criteria: (1) observational studies having HCs; (2) studies used DSM or ICD for the diagnosis of MDD patients; (3) measured peripheral cytokines in the participants aged 18 years or above; (4) assessment of peripheral CRP, TNF-α, INF-γ, and MCP-1 levels; (5) drug-naïve first-episode MDD patients.
Data extraction and methodological quality assessment
For each target inflammatory cytokine, we retrieved means and variance estimations (standard deviation or standard error of the mean) of MDD patients and HCs. Additionally, we extracted the information of (1) first author, (2) publication year, (3) sample size (number of patients and controls), (4) proportion of female gender, (5) age and BMI, (6) duration of illness (months), (7) severity of depression, (8) treatment, and (9) main findings of the studies. We recorded the above information on a standardized data collection form. The methodological quality of all studies was assessed following the Newcastle Ottawa Scale (NOS). 35 NOS score of more than five stars indicates the better methodological quality of a study.
Statistical analysis
We conducted all necessary statistical analyses using Microsoft Excel (version 2016) and Review Manager 5.4 (RevMan 5.4). We measured the heterogeneity and inconsistency of all combined results applying the Cochran’s chi-square Q-test and I2-index. The presence of significant heterogeneity or inconsistency was considered where
Results
Retrieved studies
Initially, we retrieved 9,506 studies (PubMed: 8162, ProQuest: 1,226, Scopus: 50, Web of Science: 49, and PsycINFO: 19), and 962 were excluded as duplicates. Then, we considered 8544 journal articles for screening. We excluded 8,371 citations after viewing titles and abstracts. After assessing the full text of the remaining 173 articles, again we excluded 150 (97 excluded after full-text reviewed due to irrelevant results, 47 no control group, and six MDD not assessed based on DSM-5 or ICD-10) articles based on the inclusion criteria. Finally, we included 23 case-control studies (CRP: 6, INF-ɤ: 5, MCP-1: 4, and TNF-α: 8) were included for final systematic review.29,41–62 PRISMA flowchart for the study selection process has been presented in Figure 1. PRISMA flow diagram shows literature search and study selection process.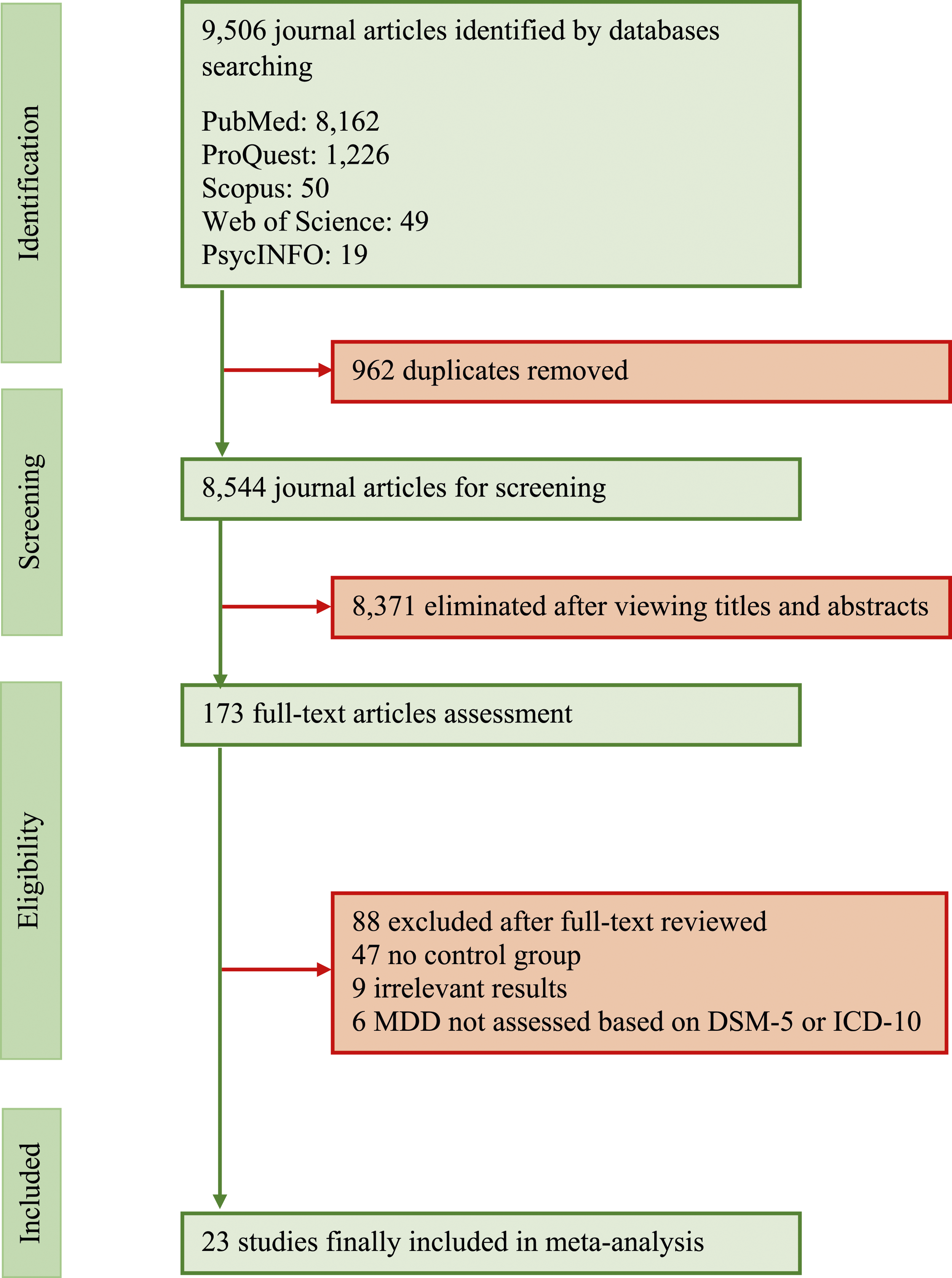
Characteristics and quality of included studies
Description of the study and participants.

Note: MDD: major depressive disorder; M/F: male/female; BMI: body mass index.
Summary inflammatory marker findings in MDD patients compare to controls.

Note: MDD: major depressive disorder;
Quality assessment of the included studies based on NOS.
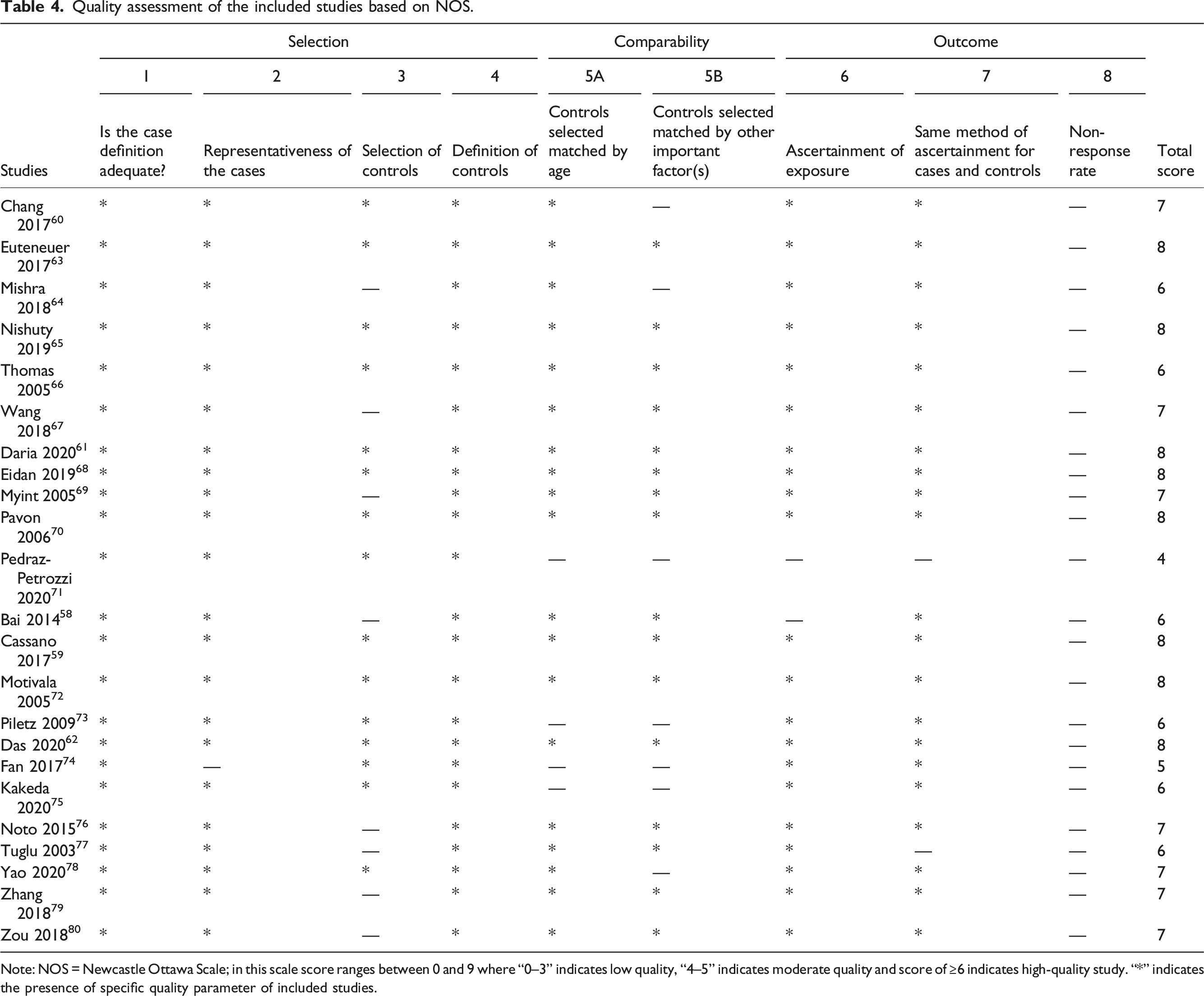
Note: NOS = Newcastle Ottawa Scale; in this scale score ranges between 0 and 9 where “0–3” indicates low quality, “4–5” indicates moderate quality and score of ≥6 indicates high-quality study. “*” indicates the presence of specific quality parameter of included studies.
Meta-analysis for inflammatory markers in MDD patients and control subjects
We compared peripheral inflammatory cytokine levels between the MDD patients and HCs using forest plot graphs. Six studies compared the circulatory CRP levels of MDD patients and HCs.43,47,49,53,57,59 These studies involved 362 MDD patients and 318 control subjects. The CRP levels in all studies were converted to mg/L. The results showed no significant differences of circulatory CRP levels between MDD patients and HCs (SMD with a random-effects model: 0.18, 95% CI: −0.85–1.23, z = 0.35, Forest plot showing effect sizes for mean differences in C-reactive protein levels in MDD patients compared with healthy controls. All the C-reactive protein levels were converted to mg/L.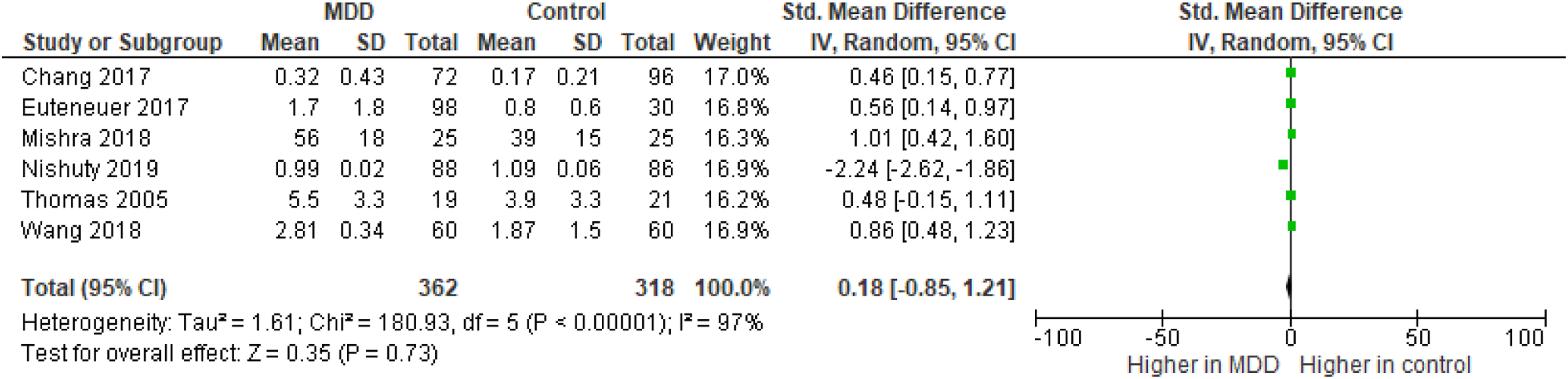
Five studies compared the peripheral INF-ɤ levels between MDD patients and HCs.29,43,47,52,55 These studies involved 274 MDD patients and 294 control subjects. The INF-ɤ levels in all studies were converted to pg/mL. The results showed no significant difference of circulatory INF-ɤ levels between MDD patients and HCs (SMD with a random-effects model: −0.05, 95% CI: −2.72–2.62, z = 0.03, Forest plot showing effect sizes for mean differences in interferon-gamma levels in MDD patients compared with healthy controls. All the interferon-gamma levels were converted to pg/mL.
Four studies compared the peripheral MCP-1 levels between MDD patients and. Volunteers.41,42,51,52 These studies involved 271 MDD patients and 279 control subjects. The MCP-1 levels in all studies were converted to pg/mL. The results showed no significant variations of peripheral MCP-1 levels between MDD patients and HCs (SMD with a random-effects model: 0.70, 95% CI: −0.09–1.49, z = 1.73, Forest plot showing effect sizes for mean differences in monocyte chemoattractant protein-1 levels in MDD patients compared with healthy controls. All the monocyte chemoattractant protein-1 levels were converted to pg/mL.
Finally, eight studies compared the peripheral TNF-α levels between MDD patients and HCs.62,74–80 These studies involved 459 MDD patients and 451 control subjects. The TNF-α levels in all studies were converted to pg/dL. The results showed that there were significantly higher levels of circulatory TNF-α in MDD patients than HCs (SMD with a random-effects model: 1.04, 95% CI: 0.69–1.39, z = 5.84, Forest plot showing effect sizes for mean differences in tumor necrosis factor-alpha levels in MDD patients compared with healthy controls. All the tumor necrosis factor-alpha levels were converted to pg/dL.
Discussion
An earlier meta-analysis of seven studies reported an increased CRP levels in MDD patients, 18 strongly associated with a higher risk of suicidal behaviors among MDD patients than HCs. 68 However, an association of peripheral INF-γ, MCP-1, and TNF-α levels in depression was unreported in previous meta-analysis. Another recent meta-analysis found an elevated levels of CRP and TNF-α in depression but undetermined the level of MCP-1 levels compared to HCs and the level of peripheral INF-γ in major depression. 63 In addition, a high degree of heterogeneity was noticed in previous study.
Our present systematic review and meta-analysis of studies assess the level of CRP, INF-γ, MCP-1, and TNF-α in the peripheral fluid of drug-naïve MDD patients and healthy controls (HCs). We found an increased level of TNF-α in MDD patients than HCs but not the other inflammatory markers, like CRP, INF-γ, and MCP-1. Even after Bonferroni adjustment, peripheral TNF-α levels remained higher in MDD patients compared to HCs, and did not notice any consistent association between TNF-α and MDD. Study design, age, sex, the severity of depression, comorbidities, and cytokine assay method may contribute to the heterogeneity among the studies. However, it is not practically possible to consider all the potential confounding factors in a meta-analysis.
C-reactive protein
The abnormalities of inflammatory cytokines in depression getting increased attention in recent years. 74 Among the cytokines, CRP is one of the key inflammatory markers in depression. An increased level of CRP enhances the permeability of blood-brain barriers that might allow direct or indirect effects of this cytokine on CNS, 75 indicating a strong association of CRP with a high degree of symptoms of MDD, supported by a previous study. 28 In addition, other studies showed that CRP influences the activities of microglia and astrocytes, and enhanced glial cell proliferation. 64 The above mechanisms confirmed the CRP association in damaging neurons that might cause mental illness. 72 However, the findings from the present meta-analysis did not support the association between circulatory CRP levels and the pathophysiology of depression.
According to the sensitivity analysis, we observed significant heterogeneity among the studies that compared peripheral CRP levels between MDD patients and HCs. The presence of this heterogeneity may be due to the dissimilar methods followed by the studies. Also, we noticed that highly-sensitive and standard CRP estimations were inconsistent among the studies. 69 A meta-analysis reported no significant alteration in circulatory CRP levels between antidepressant-treated MDD patients and HCs. However, they observed a significant heterogeneity among the studies. Moreover, meta-regression analysis failed to find an association between sample sizes, age, sex, and BMI, with increased CRP levels in depression. In some cases, this might happen as false-negative results due to few included studies with low statistical power. 65 Also, the researchers observed that psychiatric medicines and antidepressant drugs might elevate peripheral CRP levels. 76 Thus, a change in inflammatory responses might increase circulatory CRP levels in depression.
Interferon-gamma
IFN-γ, produced from Th1 lymphocytes is another proinflammatory cytokine that triggers inflammation via promoting cytotoxic activity and natural killer (NK) cell activation. 70 This mechanism is directly related to the progress of depressive symptoms. 71 An altered level of serum IFN-γ is observed in the drug-naïve MDD 29 and considered as an early risk assessment tool for depression. However, we did not find any evidence to support the involvement of IFN-γ in the development of depression according to the present meta-analysis. Some earlier meta-analyses with small samples size support the current findings related to IFN-γ levels in depression.11,19,30 Therefore, we recommend further studies with high sensitivity to establish the actual relationship between circulatory levels of IFN-γ and MDD.
Monocyte chemoattractant protein-1
MCP-1 plays an important role in the regulation of other cytokines via potentiating the expression of interleukin (IL)-4 in the TH-2 system. 18 An increased level of chemokine MCP-1 provokes the depression66,73 through potentiating chemotaxis and macrophage activation. 77 In the CNS, the role of MCP-1 is only beginning to be discovered. 67 However, some other studies have put a question mark on its role in psychiatric disorders. 78 In this present meta-analysis, we did not find a significant alteration in peripheral MCP-1 levels between depressed and non-depressed individuals. Also, a previous meta-analysis observed elevated MCP-1 levels in depression compared to non-depressed individuals; however, the result was not significant after considering heterogeneity publication bias.79,80 Therefore, further high-quality meta-analyses are recommended to establish the association of peripheral MCP-1 levels with major depression.
Tumor necrosis factor-alpha
TNF-α, a glycoprotein hormone, plays a pivotal role in the etiology of depression. 30 Inflammatory cytokine TNF-α has several roles in host defense mechanisms. 70 However, some human studies did not find a direct anti-inflammatory effect of antidepressants among MDD patients. 27 Therefore, the role of inflammatory cytokines in the pathophysiology of depression is not clear. Few meta-analyses did not find any significant relation between circulatory levels of TNF-α and depressive disorder.24,61 In this meta-analysis, we found circulatory levels of TNF-α significantly increased in depression compared to HCs before and after the Bonferroni adjustment. We can explain this finding by several neuro-psycho-inflammatory mechanisms. T-lymphocytes, macrophages, and natural killer cells help to produce TNF-α. The inflammatory response system (IRS) can activate the hypothalamic-pituitary-adrenal (HPA) axis. Therefore, the elevated level of TNF-α destroys the serotonin (5-HT) neurotransmitter.80,81 Also, increased TNF-α accelerates dopamine metabolism to induce depressive symptoms. 82 Therefore, we recommend this elevated circulatory TNF-α as a sensitive marker to predict depression risk compared to CRP, IFN-γ, and MCP-1.
Strengths and limitations
The present study has some positive aspects. The main positive aspect of this study is the systematic method to analyze possible confounding factors. We assessed the effect of probable disease-related factors (e.g., drug-naïve depression, first episode of depression), demographic profile (e.g., age, sex, and BMI), and statistical errors (e.g., data and study quality). Another strength is the multivariate meta-analytic approach to eliminate the impact of diversity in this study. Also, this is the first meta-analysis that reported circulatory CRP, TNF-α, INF-γ, and MCP-1 in drug-naïve MDD patients. Besides, we only included case-control studies using DSM or ICD for the assessment of patients in this meta-analysis. However, the present study has several limitations. Only four inflammatory cytokines (CRP, TNF-α, INF-γ, and MCP-1) were investigated in this study. The lack of repeated measurements for inflammatory cytokines is a limitation in this meta-analysis. We only are concentrating on circulatory inflammatory cytokines that might not reflect the inflammatory process in the CNS. Also, we performed this meta-analysis considering study-level data. Therefore, we recommend future meta-analyses of patient-level data with repeated measurement of cytokines considering the inflammatory process at CNS to clarify the actual association between depression and inflammatory cytokine.
Conclusion
The present study showed an upregulated level of peripheral TNF-α but not CRP, INF-γ, and MCP-1 involve in depression, indicating a strong association of circulatory TNF-α levels with the inflammatory state of depression in MDD. Finally, the present study results are not consistent with the idea that the depressive state “causes” elevated levels of inflammatory cytokines. Therefore, we suggest further large-scale meta-analyses to confirm the current findings.
Supplemental Material
Supplemental Material - Evaluation of inflammatory cytokines in drug-naïve major depressive disorder: A systematic review and meta-analysis
Supplemental Material for Evaluation of inflammatory cytokines in drug-naïve major depressive disorder: A systematic review and meta-analysis by Md Rabiul Islam, Md Sohan, Sohel Daria, Abdullah Al Masud, Maizbha Uddin Ahmed, Arpita Roy and Mohammad Shahriar in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgments
All the authors are thankful to Mohammad Fahim Kadir for his help in data identification.
Author contributions
Conception and design of study: Md. Rabiul Islam, Md. Sohan, and Sohel Daria. Acquisition of data: Abdullah Al Masud and Maizbha Uddin Ahmed and Arpita Roy; analysis and interpretation of data: Md. Rabiul Islam, Abdullah Al Masud and Maizbha Uddin Ahmed, and Arpita Roy. Drafting the manuscript: Md. Sohan and Sohel Daria. Revising the manuscript critically for important intellectual content: Md. Rabiul Islam and Mohammad Shahriar. Finally, all authors approved the version of the manuscript to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Informed consent
Informed consent for patient information to be published in this article was not obtained because the present study only reported and analyzed the published articles from publicly available sources.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
