Abstract
Pancreatic cancer is one of the most lethal diseases around the world, for hardly detection and poor prognosis. Recent years, functions of the tumor microenvironment and immune cells attract people’s view and there is emerging evidence implicating some immune cells hold the key points in the metabolism, invasion, and metastasis in pancreatic cancer. In this review, we highlight some main immune cells, such as Tumor-associated neutrophils (TANs) and macrophages (TAMs), Pancreatic stellate cells (PSCs), Myeloid-derived suppressor cells (MDSCs), and Regulatory T cells (Tregs). Furthermore, we review current clinical applications and discuss potential values in future.
Keywords
Introduction
Pancreatic cancer (PCa) is a lethal tumor and a major cause of death worldwide. Approximately 45,750 recent PCa deaths have been recently reported in the US alone. 1 PCa is behind only lung cancer and colon cancer in the US in 2018 for cancer-associated deaths. Worldwide, over 400,000 deaths occur annually due to PCa. 2 The five-year survival rates of PCa are one of the poorest (∼5%). 2 On account of the lack of effective early detection methods, patients with early symptoms such as upper abdominal discomfort and loss of appetite are often diagnosed with gastric disease, whilst diagnosed PCa patients frequently exhibit local invasion or metastatic tumors, missing the optimal therapeutic window. 3 The TNF system for PCa states that T1-T3 tumors are “potentially resectable” and often receive surgery. 4 However, as low as 15% of patients are deemed suitable for surgical interventions and most die within 2 years. 5 For advanced and/or metastasis, gemcitabine and erlotinib (EGFR inhibitor) can be efficacious. 6
Amongst PCa subtypes, pancreatic ductal adenocarcinoma (PDAC) is the most common (≥95% of PCa cases). PDAC arises from the epithelial ducts, 7 the reasons for which remain obscure. Pancreatic intraepithelial neoplasias (PanINs), mucinous cystic neoplasms, and intraductal papillary mucinous neoplasms are considered origins of PDAC. The majority are thought to develop from PanINs. 8 An array of genes and signaling pathways have been implicated in the tumorigenesis and progression of PCa, including K‐ras-related proteins, Notch, Hedeghog, Wnt, F‐box proteins, PI3/Akt, and mTOR. Notably, these pathways mediate inflammatory responses.9–15 Inflammation was discovered as a cancer-causing factor in 1863 by Virchow and has since gained intense interest in the progression of PDAC. Inflammation is critical to tissue protection with a loss of tissue homeostasis known to be carcinogenic. 16 Interestingly, organs with chronic inflammation have a heightened risk of carcinogenesis. For example, patients with inflammatory bowel diseases have a higher risk of colorectal cancer, 17 esophageal cancer is more prevalent in patients with Barrett’s disease, 18 and PCa is associated with chronic pancreatitis. 19
Immune cell populations regulate inflammation,
20
and immunotherapy has emerged as a common cancer-targeting strategy. Immuno-regulation can improve the survival rates of PCa patients. In this review, the immune cells regulating carcinogenesis and tumor progression will be discussed, including Tumor-associated neutrophils (TANs) and macrophages (TAMs), Pancreatic stellate cells (PSCs), Myeloid-derived suppressor cells (MDSCs), and Regulatory T cells (T-regs). These cells have been shown be involved in the progress of PDAC; their functions are summarized in Figure 1. We will further discuss the clinical utility of these immune cell populations. The interactions between tumor cells and immune cells.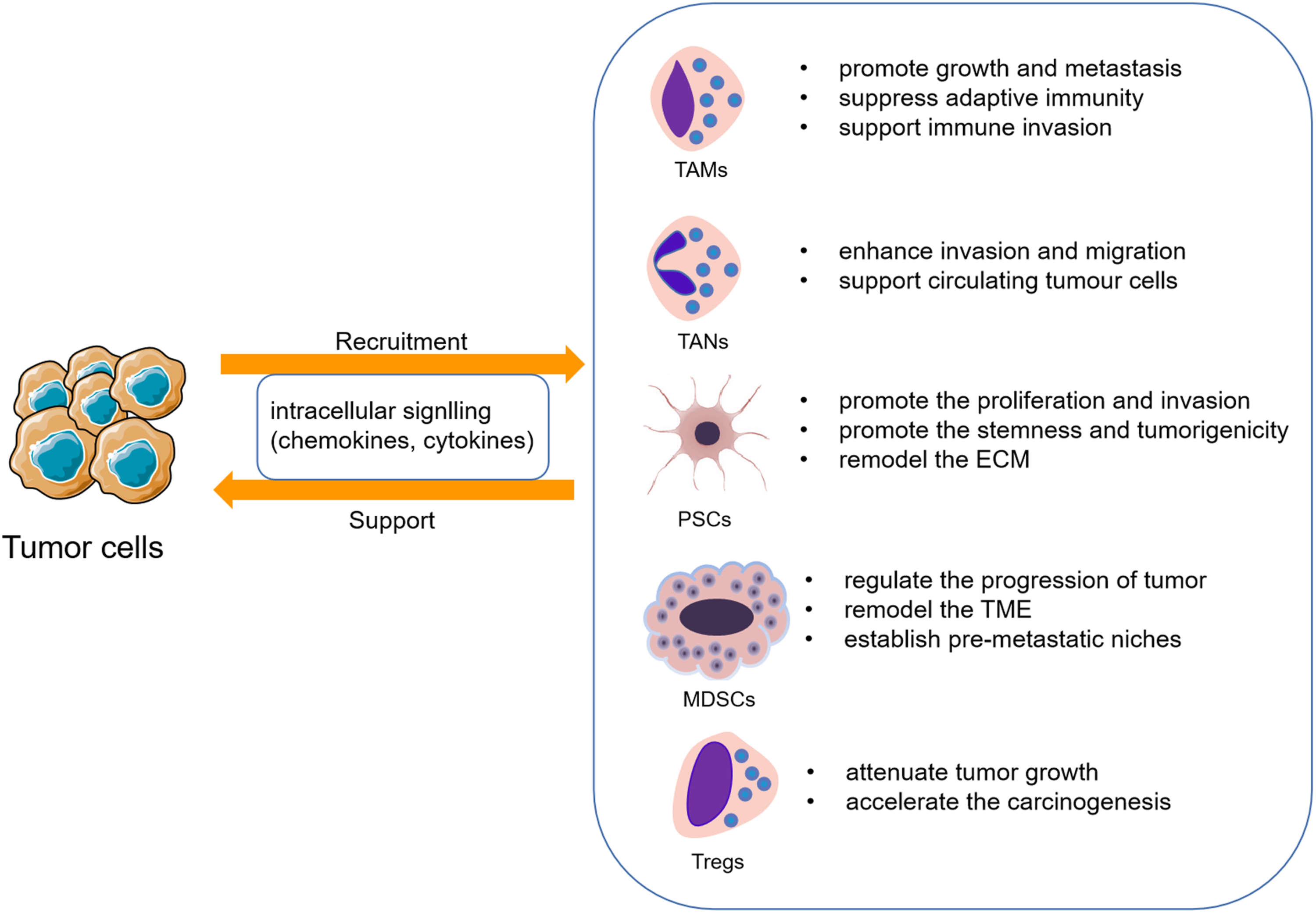
Tumor-associated macrophages
TAMs account for the largest number of invasive monocytes and possess plasticity. A characteristic of PDAC is the abundant invasion of tumor-associated macrophages. In recent years, an array of studies suggest that specific subpopulations of macrophages support tumor progression and macrophages constitute up to 50% of the tumor mass, including tumor infiltration and immune cells in the tumor microenvironment (TME).21,22 Macrophages participate in tumor progression including early carcinogenesis, development, and metastasis. 23 Of note, M1-TAMs are anti-tumoral in cancer-initiating conditions. Upon the establishment of tumors, macrophages become pro-cancerous and switch to the M2 type. 24 Large TAM numbers are frequently associated with poor prognosis. 25
Interaction between TAMs and pancreatic cancer cells
In PCa, macrophages assist tumor proliferation, invasion, and metastasis. 26 As previously discussed, cancer-related inflammation is a hallmark of cancer. TAMs are involved in tumor progression through the secretion of pro-inflammatory cytokines or chemokines. M2-polarized macrophages suppress adaptive immunity via cytokines such as IL-10 and IL-12 and chemokines such as CCL13 and CCL18. 27 In addition, M2-polarized macrophages enhance Th-2 responses through enhanced cytokine secretion; thus, inhibiting the anti-tumor immune responses mediated by T cells.28,29 PDAC tumors create a hypoxic TME and promote angiogenesis creating a hypoxic and nutrient-deprived environment. During tumor progression, TAMs secrete HIF-1α, a transcription factor for many angiogenesis responsive genes. 24 Upon distant tumor cell migration, TAMs release CCL2 that recruits inflammatory monocytes, converting them into metastasis macrophages. 30 Of note, malignant cells are cleared by the immune system; however, PCa evades the immune response through the creation of an immunosuppressive niche, a major reason for its poor prognosis. The secretion of IL-10 by the TAMs contributes to immune evasion. TAMs that express CD120a+ and CD120b+ promote T cell apoptosis and anti-tumor responses, enhancing the ability of PDAC to undergo immune evasion. 31 Chang et al. have reported that ezrin in PDAC-derived small extracellular vesicles can modulate the polarization of macrophages to the M2 phenotype and promote PDAC metastasis, while knockdown of ezrin promoted the M1 phenotype. 32 Another report found that exosomal K-Ras protein from tumor cells could be taken up by macrophages resulting in a switch to an M2-TAM phenotype via STAT3-dependent fatty acid oxidation to promote the tumor progression. 33 Zhang et al. found that TAMs could protect cancer cells from complement-dependent cytotoxicity (CDC) by up-regulating CD59 via the IL-6R/STAT3 signaling pathway. 34 On the other hand, tumor cells release chemokines that recruit TAMs.
The clinical application of TAMs in the treatment of pancreatic cancer
Relevant clinical application of TAMs in PCs treatment.

Tumor-associated neutrophils
Neutrophils regulate acute inflammation. 42 Neutrophils account for ∼60% of all leukocytes in the circulation and are recruited to the sites of infection or inflammation, interacting with an array of immune cells. 43 In traditional immunology, neutrophils act in host defenses, immunomodulation, and tissue injury, and patients with neutrophil deficiencies (such as neutropenia) are more susceptible to microbial and fungal infections. 44 When the inflammatory response occurs, neutrophils are recruited from the peripheral blood to the sites of inflammation. Neutrophils then produce ROS, granular proteins, and cytokines to kill microorganisms and clear infections. 45 Of note, the neutrophils that originate from myeloid precursors, the primary responsive cell types of the innate immune response, 42 are involved in tumor progression, malignant transformation, anti-tumoral immunity, and angiogenesis, 43 with some studies suggesting that TANs and peripheral neutrophils and granulocytic MDSCs in the bone, spleen, and blood regulate tumor progression. 46
Interaction between Tumor-associated neutrophils and pancreatic cancer cells
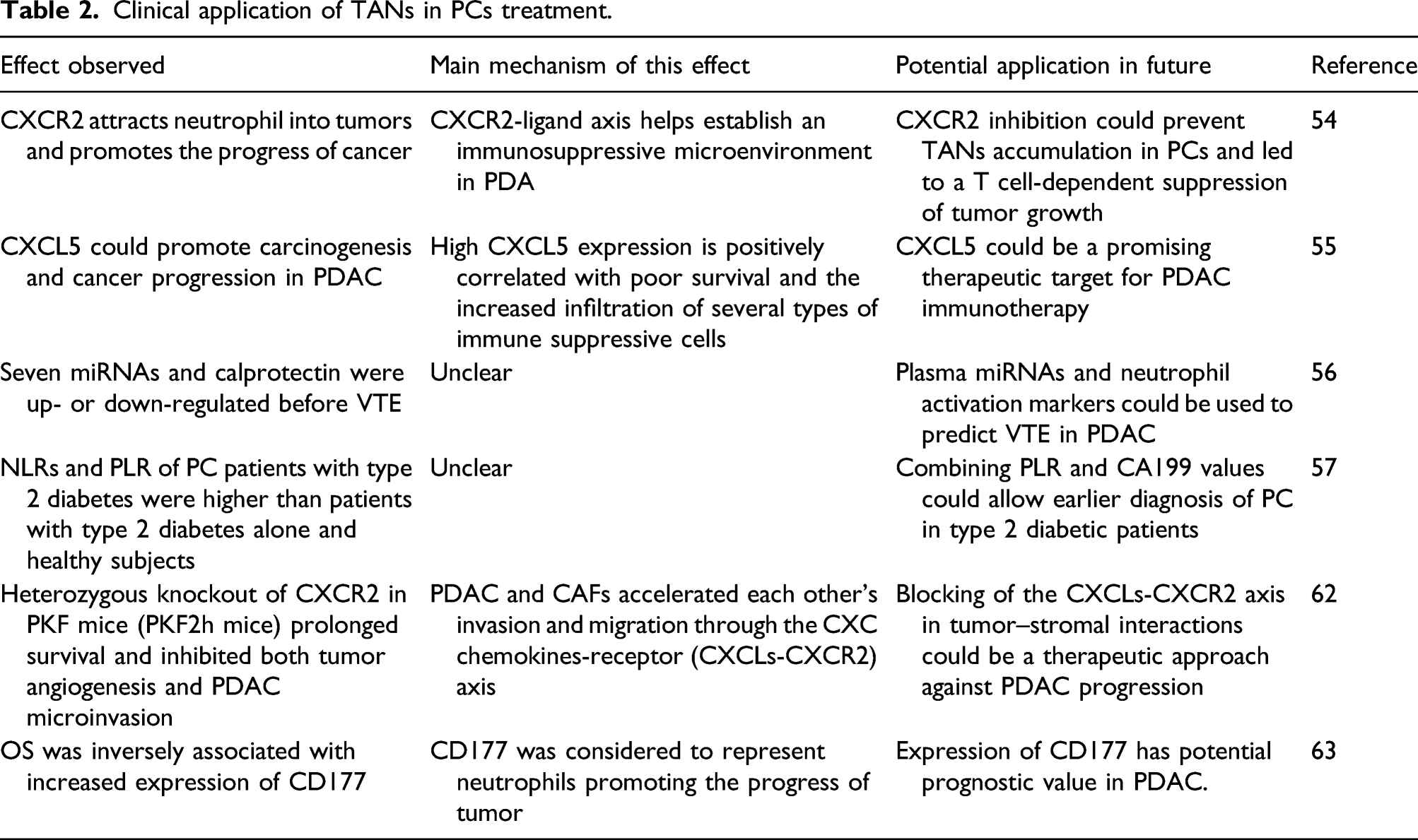
Similar to the TAMs, TANs promote oncogenic (referred to as N2 TANs) and anti-tumor (or N1) phenotypes,46,47 that differ according to the degree of activation rather than polarization. 48 In PCa, N1-like and N2-like neutrophils act in the early stages of tumor growth. The proliferation and maturation of neutrophils are dependent on G-CSF and GM-CSF, 49 which in PCa cells recruit neutrophils to the tumor site. 50 The presence of increased levels of TANs is often associated with poor clinical outcomes. 51 TANs produce proteases such as PR3, elastase, cathepsin G, and MMP-8 and -9 to enhance PCa cell invasion and migration. 52 Another report has found that TANs can support circulating tumor cells (CTCs), the precursors of metastasis, in PCa, promoting their survival and extravasation; it is possible that vimentin-positive CTCs may be biomarkers for the diagnosis and treatment of patients with PCa. 53 Chao et al. have found that CXCR2 inhibition by genetic ablation prevented neutrophil accumulation in pancreatic tumors and led to a T cell-dependent suppression of tumor growth. 54
Clinical applications relevant to Tumor-associated neutrophils in pancreatic cancer
Clinical application of TANs in PCs treatment.
Pancreatic stellate cells
Pancreatic stellate cells are located in endocrine and exocrine PCa tissues. 64 In 1982, PSCs were identified in mouse pancreatic ducts and have abundant cytoplasmic lipid droplets and vitamin A levels. 65 PSCs were also shown to be present in healthy pancreatic samples of rats and humans. 66 Recently, the PSCs in islets have been termed fibrogenic islet stellate cells, 67 which have fewer lipid droplets than typical PSCs and are more easily activated by stimulators, to reduce proliferation and migration. 68 In recent years, PSCs have been shown to regulate extracellular matrix (ECM) turnover through the regulation of matrix-degrading enzymes, 69 and the ECM causing the fibrosis is primarily formed by PSCs. PSCs can be divided according to two distinct biological phenotypes, namely, quiescent and activated PSCs. Quiescent PSCs express high levels of glial fibrillary acidic protein and desmin and when activated to myofibroblasts, express high levels of α-SMA, and display enhanced collagen I-III, fibronectin, and ECM components, thus promoting pancreatic fibrosis. 70
The interaction between Pancreatic stellate cells and pancreatic cancer
Jiang J observed that nerve growth factor (NGF) from PSCs could promote the proliferation and invasion of cancer cells by activating the PI3K/AKT/GSK signaling pathway. 71 It has also been found that TGF-β1 secreted by PSCs negatively regulates L1CAM expression through canonical TGF-β-Smad2/3 signaling, promoting the stemness and tumorigenicity of pancreatic cancer cells. 72 Steins et al. found that the stroma from high-grade PDAC contained both reduced collagen and activated PSCs, identified colony-stimulating factor 1 (CSF-1) as the ligand that deactivated PSCs, and suggested that inhibition of its receptor CSF1R may be a useful therapeutic target. 73 Kuntze observed that stimulating Piezo1 activated PSCs by inducing Ca influx, leading to increased cellular motility and ECM traction, suggesting that environmental pH might influence the mechanical output of PSCs and favor the early local invasion of PDAC cells. 74 Mounting evidence suggests that cancer cells influence PSCs via mitogenic and fibrogenic factors to enhance their activation, proliferation, migration, and ability to remodel the ECM. 75 During tumor inflammation, TGF-β1, PDGF, IL-1β, IL-6, and TNF-α are released into the PCa TME to stimulate qPSCs.76,77 PSC activation leads to elevated production and secretion of these molecules. 78 Proteins secreted from the activated PSCs are also associated with cancer progression and epithelial-to-mesenchymal transition (EMT), known to occur during embryonic development, tissue regeneration, organ fibrosis, and tumor formation. 79 EMT occurs during the initial stages of pancreatic tissue fibrosis, and activated PSCs enhance the expression of EMT-related genes. 80 Other studies found that PSCs can inhibit apoptosis, increase tumor invasion, and promote metastasis. 81
The clinical application of Pancreatic stellate cells in the treatment of Pancreatic cancer
Clinical application of PSCs in PCs treatment.

Myeloid-derived suppressor cells
In the 1990s, extramedullary hematopoiesis (EMH) and neutrophilia were identified during tumor progression, promoting tumor vascularization and immune-evasion. This phenomenon was associated with increased serum hematopoietic, abnormal myeloid cell differentiation and colony-stimulating capacity. 87 Abnormal myeloid cells were first characterized as natural suppressors (NS) that inhibit CTL activity. 88 NSs lack membrane markers for mature T cells, B cells, and NK cells, as well as macrophages 89 and were termed MDSCs with potent immune-regulatory activity. 90 MDSCs exist as granulocytic or poly-morphonuclear MDSCs (PMN-MDSCs) that are similar to neutrophils and monocytic MDSCs (M-MDSCs) that are similar to monocytes. 91 Recent studies have identified another population of MDSCs that induce colony-forming activity and other myeloid precursors, named early-stage MDSCs. 92 In general, MSDCs regulate the progression and metastasis of tumors through immunosuppression, TME remodeling, the establishment of a pre-metastatic niche, angiogenesis, cancer cell stemness, and EMT.93-95
The interaction between Myeloid-derived suppressor cells and pancreatic cancer
There is emerging evidence that PDAC cells can release pro-inflammatory metabolites, such as granulocyte-macrophage colony-stimulating factor (GM-CSF), to favor the expansion and accumulation of MDSCs.50,96 In addition, Trovato et al. found that high levels of MDSCs were associated with poor patient prognosis and that STAT3 was crucial for monocyte re-programming. 97 In other research, higher MDSC levels were found in PC compared to healthy controls and premalignancy. 98 ETS homologous factor (EHF) is a tumor suppressor in PDAC. Liu et al. observed that tumoral EHF deficiency could cause MDSC accumulation while EHF overexpression might improve the PDAC immunotherapy checkpoint. 70 Obesity is often considered as a risk factor related to the poor prognosis of cancer patients, including PDAC. Turbitt et al. found that adiposity induces MDSC accumulation which might favor an immunosuppressive environment that promotes tumor growth. 99
The relevant clinical application of Myeloid-derived suppressor cells in pancreatic cancer
Clinical application of MDSCs in PCs treatment.

Regulatory T cells
Treg cells are the main mediators of immune homeostasis and are responsible for the regulation of diverse immune responses including autoimmunity, allergies, microbial infections, and cancers. 104 However, their inhibitory activity often antagonizes protective immunity and this may favor disease progression. 105 PDAC is characterized by an immunosuppressive microenvironment which favors the growth of cancer and resistance to therapy. Treg cells undoubtedly play an essential role in this.
The interaction between Treg cells and PC cells
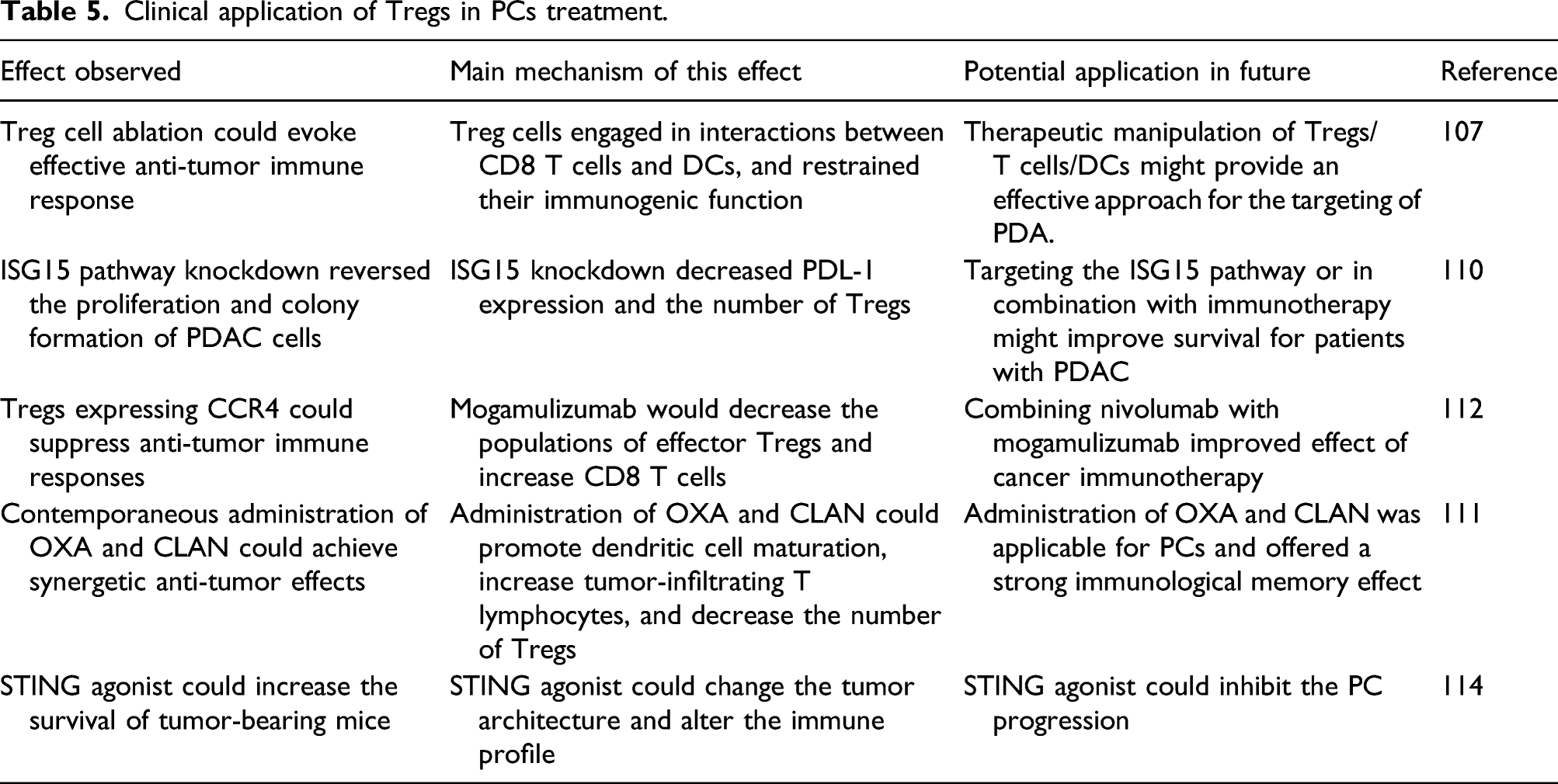
In the context of PDAC, the level of Treg cells is considered a prognostic factor as PC cells can recruit the Tregs at an early stage with the Treg cell count increasing along with the development of cancer. 106 On the other hand, Treg cells depletion can cause attenuation of tumor growth. 107 However, other research reports that the deletion of Treg cells may accelerate PDAC carcinogenesis through increased recruitment of myeloid cells, leading to further immune suppression. 108 Hypoxia has been shown to be an important factor in promoting cancer growth. In PDAC, a hypoxic microenvironment contributes to the recruitment of Tregs and depletion of cytotoxic T cells.
The relevant clinical application of Treg cells in PDAC
Clinical application of Tregs in PCs treatment.
Conclusion
In conclusion, PCa remains a threat to human health due to difficulties in diagnosis, poor therapeutic effects, and poor prognosis. With improvements in living standards, its incidence is increasing on a yearly basis, and the 5-year survival rates remain less than 5%. In recent years, the treatment of PCa has emphasized the importance of early diagnosis and metastasis control, in which immune regulation plays a key role. This review summarizes recent findings on the immune regulation of PCa and proposes new clinical applications. To date, the immune regulation of PCa is still insufficient, and further clinical assessments are needed. However, immune mechanisms have the unique advantage of assisting early diagnosis, clinical treatment, and predicting prognosis in PCa.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (81672402), Provincial Natural Science Foundation of Jiangsu, China (BK20171305), Jiangsu Key R & D Program Social Development Project, China (BE2018689), Research Programs of Jiangsu Provincial Commission of Health and Family Planning, China (H201434), “Six one Project” Research Projects of High-level Medical Personnel of Jiangsu Province (LGY2016054), “Six Talents Peak” High-level Talent Selection and Training Project of Jiangsu Province (2014-WSW-038), Science and Technology Support Program Project of Zhenjiang, China (SH2014024), and Qing Lan Project Funding of Jiangsu Colleges and Universities.
