Abstract
To investigate the effects and potential mechanisms of chicoric acid (CA) on LPS-induced inflammatory response in A549 cells. 0–800 μM CA was added to A549 cells to determine the toxicity of CA using MTT assay. Then, 2 μg/mL LPS and 50 μM CA were simultaneously added to A549 cells to investigate the effects of CA. In order to investigate the effects of miR-130a-3p and IGF-1 on LPS-induced A549 cells, cells were infected with inhibitor of miR-130a-3p and si RNA IGF-1. The levels of inflammatory cytokines such as IL-1β, IL-6, and TNF-α were measured by real-time RT-PCR and enzyme-linked immunosorbent (ELISA) assay. The IGF-1 pathway and NF-κB expression were measured using immunoblot assay. Moreover, a luciferase activity assay was used to indicate the binding site of miR-130a-3p on the 3′UTR of IGF-1. 0–50 μM CA had no toxicity on A549 cells. Thus, we chose 50 μM CA for the following study. CA attenuated the inflammatory response by LPS through decreasing IL-1β, IL-6, and TNF-α levels and increasing IGF-1/IGF-1R expression. Inhibition of miR-130a-3p reduced the inflammatory response and restored IGF-1/IGF-1R pathway induced by LPS. Furthermore, luciferase activity results indicated that miR-130a-3p directly targeted IGF-1 to regulate inflammatory response. CA alleviates LPS-induced inflammatory response through miR-130a-3p/IGF-1pathway in A549 cells.
Introduction
Lung inflammation is usually caused by lesions or exposure to toxins, pollutants, irritants, and allergens and is clinically observed as immune infiltration. 1 The dysregulation of lung epithelial cells is a key contributor to lung inflammation. 2 Airway epithelial cells identify pathogens through toll-like receptors and stimulate NF-κΒ pathway to promote IL-6, IL-1β, and TNF-α secretion. 1 Insulin growth factor-1 (IGF-1) is a multifunctional hormone with high molecular structure similarity to insulin. 3 IGF-1 binds its receptor, IGF-1R, activating intracellular signaling. It has been indicated that IGF-1/IGF-1R pathway is related to pulmonary development and lung inflammation. 4 IGF-1 inhibits lung inflammation and remodeling induced by anaphylactogen. 5 Hypoxia-induced reduction in IGF-1 is responsible for chronic pulmonary inflammation in postnatal rats. 6 Thus, the exploration of drugs targeting IGF-1 may be a potential way to alleviate lung inflammatory diseases.
Chicoric acid (CA) is a type of hydroxycinnamic acid that belongs to the phenylpropanol family and is mainly found in the roots of many plants such as chicory, purple coneflower, and basil. 7 It has been found in BV-2 cells and mice brain that CA can improve the inflammatory response stimulated by lipopolysaccharides (LPS) through inhibition of NF-κB pathway.8, 9 CA has also been reported to decline IL-1β, IL-6, and TNF-α mRNA expression. 10 In LPS-induced acute lung injury mice, CA decreased immune cell infiltration and the production of inflammatory cytokines. 11 Nevertheless, the potential mechanisms of CA in lung inflammatory response remain unknown.
MicroRNAs are non-coding RNAs with a length of 18–23 nucleotides. By binding to specific sites of target mRNA, they can post-transcriptional down-regulate and inhibit the expression of target genes. 12 Accumulating evidence suggests that microRNAs are involved with lung diseases, among which miR-146a, miR-24, miR-155, and miR-127 have been proved to alleviate pulmonary inflammation.13–16 As a member of microRNAs family, miR-130a is involved in many diseases, such as cancers, 17 spinal cord injury, 18 and kidney diseases. 19 The miR-130a-3p has been shown to mediate inflammatory responses in LPS-induced BV2 cells via IGF-1/IGF-1R pathway. 18 However, the function of miR-130a-3p in pulmonary inflammatory is still needed to be clarified.
In the present study, the inflammatory response was stimulated by LPS in human lung A549 epithelial cells. We explored whether CA could attenuate LPS-induced inflammation through miR-130a-3p/IGF-1 pathway in A549 cells.
Materials and methods
Cell cultures
Human lung A549 epithelial cell line was obtained from the China Center for Type Culture Collection (Wuhan, China). A549 was cultured by high-glucose DMEM with 10% FBS (5% CO2, 37°C). To determine the effect of CA, 2 μg/mL LPS (Sigma, USA) and 50 μM CA (Sigma, USA) were simultaneously added to the cells. After incubated for 24 h, the cells and culture medium were collected for further study.
MTT assay
A549 cells were plated at 1 × 106/mL in a 96-well plate and cultured for 24 h. Then, 0–800 μM CA was added to the cells. After 24 h incubation, 15 μL MTT was added to the cells and incubated for 6 h. 100 μL DMSO was added to the cells. The optical density was measured at 570 nm through a microplate reader.
Real-time RT-PCR
Primer sequences for PCR.

Cytokine measurement
The levels of IL-1β, IL-6, and TNF-α in culture supernatant were evaluated by ELISA kit obtained from Boster (Wuhan, China), in line with the manufacturer’s instructions.
Western blotting
The total proteins were obtained using protein lysis buffer (DingGuo, China). The quantitation of proteins was measured using BCA kit (DingGuo, China). Proteins were isolated using 10–15% SDS–PAGE gels and then transferred onto a PVDF membrane. The antibodies for IGF-1, IGF-1R, p- NF-κB, NF-κB, and β-actin were used for detection. The bands images were analyzed by Image J software.
Inhibition of miR-130a-3p
The miR-130a-3p inhibitor and negative control (NC) obtained from IBS Bio (Shanghai, China) were used to detect the effect of inhibition of miR-130a-3p. Briefly, miR-130a-3p inhibitor or NC oligos were transfected into cells with Lipofectamine 2000 in opti-mem (Invitrogen) and incubated for 24 h. Then, 2 μg/mL LPS was added to cells and incubated for 24 h. After that, the cells and culture medium were collected for further study. The cells transfected with NC oligos were taken as control group.
siRNA transfection
A549 cells were infected with IGF-1 siRNA (IBS Bio, Shanghai, China) using siRNA Transfection Reagent (IBS Bio, Shanghai, China). After 24 h incubation, cells were treated with 2 μg/mL LPS or combined with miR-130a-3p inhibitor for 24 h. Then, the cells and culture medium were collected for further evaluation. The cells without transfection were taken as control group.
Dual-luciferase reporter assay
Cells were cotransfected with IGF-1-3′UTR wild (3′UTR-WT), IGF-1-3′UTR mutant (3′UTR-MU), or IGF-1-3′UTR negative control (3′UTR-NC) with miR-130a-3p-mimics or miR-NC using Lipofectamine 2000 (Invitrogen) for 36 h. Then, the cells were collected, and the luciferase activity was measured based on the instructions.
Statistical analysis
Data were shown as mean ± SD. Independent samples-t test was used to detect differences between two groups, and one-way ANOVA with LSD post hoc test was used for multiple groups. GraphPad Prism 9 (GraphPad Prism, USA) was used for analyses. A difference with p < .05 was indicated as statically significant.
Results
CA partially inhibited the inflammatory response induced by LPS in A549 cells
To investigate the role of CA, we first performed an MTT assay to evaluate the toxicity of CA in A549 cells. The result showed that 0–50 μM CA had no toxicity on A549 cells, thus we chose 50 μM CA for the following study (Figure 1(a)). Next, A549 cells were treated with LPS to establish a cell model of pulmonary inflammation. LPS significantly increased mRNA expression of IL-1β, IL-6, and TNF-α compared with control group (Figures 1(b) to (d)). Consistent with that of mRNA expression, LPS increased inflammatory cytokines levels in the culture medium compared to control group (Figures 1(e) to (g)). Moreover, CA treatment partially inhibited the increase of inflammatory cytokines stimulated by LPS (Figures 1(b) to (g)). CA inhibited the inflammatory response induced by LPS. To evaluate CA toxicity in cells, A549 cells were treated with 0–800 μM CA for 24 h and measured the cell viability with MTT assay 
CA partially inhibited the up-regulation of miR-130a-3p and down-regulation of IGF-1 pathway induced by LPS
As shown in Figure 2(a), the miR-130a-3p expression was significantly increased after LPS treatment, while CA treatment partially repressed the up-regulation of miR-130a-3p. In addition, we measured IGF-1 pathway and NF-κB activation. In A549 cells induced by LPS, IGF-1 and IGF-1R protein expression was down-regulated, and NF-κB phosphorylation was up-regulated, which were partially reversed after CA treatment (Figures 2(b) to (e)). CA inhibited the up-regulation of miR-130a-3p and down-regulation of IGF-1 pathway stimulated by LPS. The miR-130a-3p expression was measured by real-time RT-PCR 
Inhibition of miR-130a-3p diminished the inflammation stimulated by LPS
In order to study the effect of miR-130a-3p, the miR-130a-3p inhibitor was transfected into A549 cells. The miR-130a-3p inhibitor repressed the elevation of miR-130a-3p induced by LPS (Figure 3(a)). Inhibition of miR-130a-3p declined the levels of inflammatory cytokines stimulated by LPS (Figure 3(b)–(g)). Besides, by inhibiting miR-130a-3p, the IGF-1 and IGF-1R expression was strengthened, and NF-κB phosphorylation was declined (Figures 3(h)–(k)). Suppression of miR-130a-3p reduced LPS stimulated inflammatory response. In order to explore the effect of miR-130a-3p in LPS-induced A549 cells, A549 cells were treated with miR-130a-3p inhibitor. Real-time RT-PCR was used to evaluate the mRNA levels of miR-130a-3p, IL-1β, IL-6, and TNF-α 
MiR-130a-3p directly targeted IGF-1
Bioinformatics analysis indicated that IGF-1 might be a target gene for miR-130a-3p (Figure 4(a)). We conducted a dual-luciferase reporter assay to determine whether miR-130a-3p bind to IGF-1. The results demonstrated that miR-130a-3p mimics remarkably decreased the luciferase activity of the IGF-1-WT, indicating that miR-130a-3p might directly bind IGF-1 to regulate inflammatory response (Figure 4(b)). IGF-1 was the targeted gene for miR-130a-3p. Bioinformatics analysis indicated that IGF-1 gene was a potential target for miR-130a-3p 
IGF-1 silencing enhanced the inflammatory response induced by LPS
To verify the efficacy of IGF-1 on inflammatory response, the miR-130a-3p inhibitor and siRNA IGF-1were cotransfected into LPS-induced A549 cells. The mRNA expression of IGF-1 was markedly silenced compared to control group (Figure 5(a)). After silencing IGF-1, the inflammatory cytokine levels were enhanced compared to control group, which were repressed following miR-130a-3p inhibitor treatment (Figures 5(b)–(g)). Additionally, IGF-1 silencing facilitated NF-κB expression, and inhibition of miR-130a-3p suppressed NF-κB expression (Figures 5(h)–(k)). IGF-1 silencing enhanced the inflammatory response induced by LPS. To verify the efficacy of IGF-1 on inflammatory response, siRNA IGF-1was transfected into LPS-induced A549 cells. The mRNA expression of IGF-1 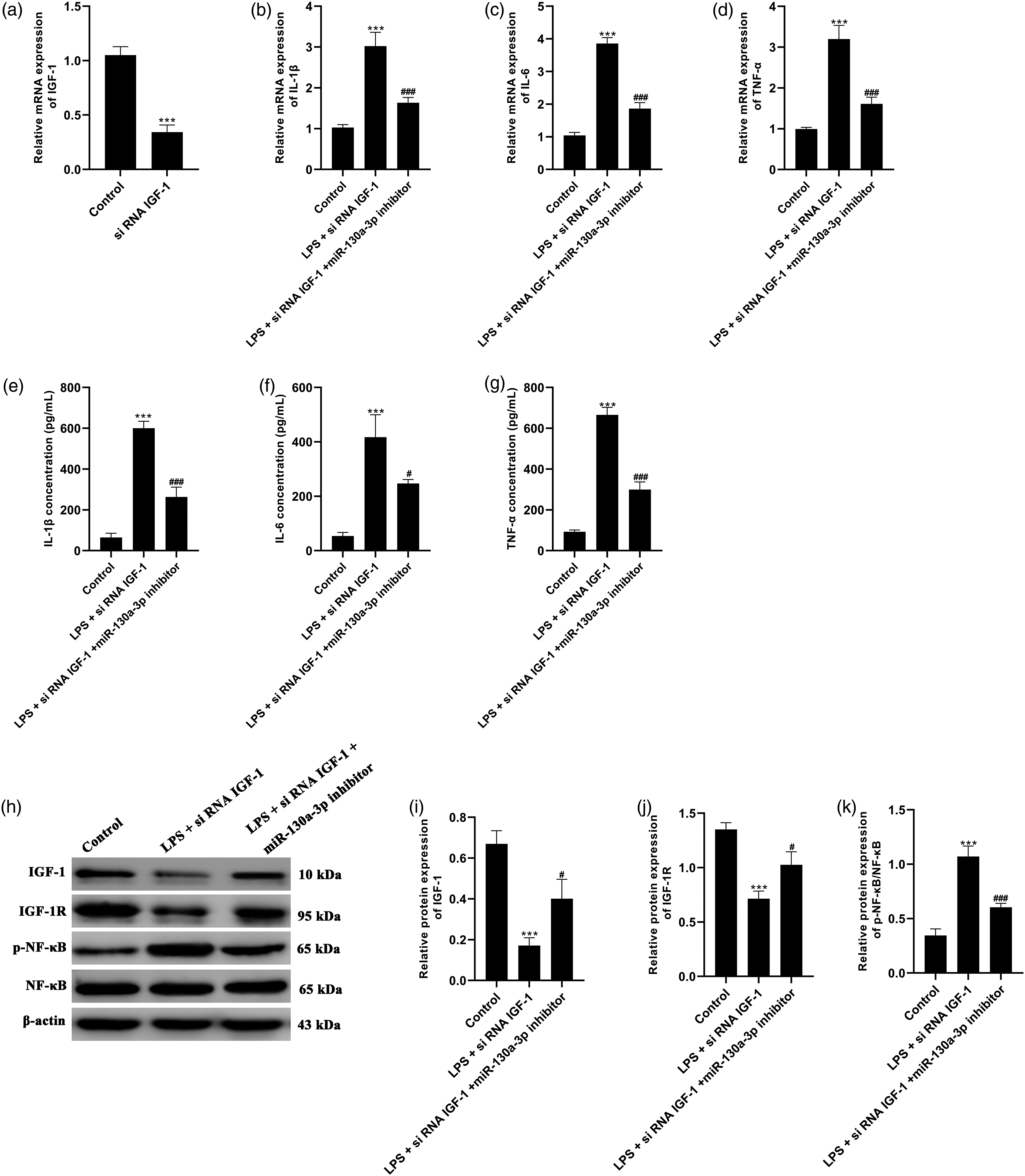
Discussion
Pulmonary inflammatory diseases are associated with increased levels of inflammatory cytokines of lung epithelial cells. 1 We employed LPS-induced A549 cells to mimic inflammatory epithelial cells. The inflammatory cytokines were notably increased by LPS. Meanwhile, CA alleviated LPS-induced inflammatory response. Further studies revealed that CA alleviated LPS-induced inflammatory response through miR-130a-3p/IGF-1pathway.
CA is a polyphenol of a natural plant which has been documented to have multiple health benefits. 20 Accumulating evidence suggests that CA can down-regulate the levels of inflammatory cytokines such as cyclooxygenase-2 (COX-2), iNOS, TNF-α, IL-6, and IL-1β.9,10 Consistent with previous studies, current results showed that CA effectively suppressed the increase of TNF-α, IL-6, and IL-1β stimulated by LPS in A549 cells. Furthermore, miR-130a-3p was elevated in response to LPS and was restored following CA treatment, indicating its role in inflammatory epithelial cells.
Emerging evidence suggests that post-transcriptional regulation of microRNAs may coordinate transcription factors to mediate various biological processes. 21 Despite a panel of microRNAs has been proved to participate in pulmonary inflammatory diseases,13–16 the role of miR-130a-3p in pulmonary inflammatory response remains largely unknown. In recent studies, overexpression of miR-130a-3p inhibited LPS-stimulated osteoarthritis. 22 The miR-130a-3p was found to mediate the expression of macrophage profibrogenic gene in chronic inflammation. 23 Accordingly, we investigated the function of miR-130a-3p in pulmonary inflammatory response, demonstrating that miR-130a-3p expression was highly promoted after LPS stimulated in A549 cells. In addition, suppression of miR-130a-3p enhanced the IGF-1 pathway and repressed pro-inflammatory cytokine levels. Our findings supported that IGF-1 pathway might participate in the regulation of miR-130a-3p on inflammatory response in A549 cells.
IGF-1 belongs to the family of growth factors. Its primary action is regulated through binding to IGF-1R. IGF-1 signaling pathway stimulates growth and development under physiological conditions. 24 Furthermore, IGF-1 signaling also plays a paramount action in immune and inflammatory regulation. 25 It has been found that IGF-1 is involved in mesenchymal stem cell derived conditioned medium alleviating LPS-stimulated lung inflammation. 26 Reduction of IGF-1 is related to increased levels of IL-6, IL-1β, and TNF-α in cystic fibrosis. 27 In an idiopathic pulmonary fibrosis of lung epithelial cells, IGF-1 secretion is increased by inhibition of miR-130b-3p, and it belongs to the same miR-130 family as miR-130a-3p. 28 The current study revealed that IGF-1 pathway was down-regulated in LPS-induced A549 cells and was partially restored by CA treatment. Inhibition of miR-130a-3p markedly compromised LPS-induced repression of IGF-1 pathway. Furthermore, IGF-1 silencing enhanced the phosphorylation of NF-κB induced by LPS. Bioinformatics analysis and dual luciferase reporter assay confirmed that miR-130a-3p interacted with the 3′ un-translated region of IGF-1 and repressed its expression, which was consistent with a previous study. 18 Therefore, we suspected that CA might regulate IGF-1 signaling pathway through miR-130a-3p to alleviate LPS-induced inflammatory response in A549 cells.
Our findings suggested that CA improved the inflammatory response in LPS-stimulated human lung A549 epithelial cells. The miR-130a-3p/IGF-1 pathway might play an important role in mediating the inflammatory response. However, as an in vitro study, it would be very difficult to extrapolate the findings to the in vivo situation. Future studies should examine the effect of CA in animal models and patients with pulmonary inflammatory disease.
Conclusion
In summary, we demonstrated that CA alleviated LPS-induced inflammatory response through miR-130a-3p/IGF-1pathway in A549 cells. These findings furnished new insights into the treatment of pulmonary inflammatory diseases.
Footnotes
Author Contribution
Zheng Yan: Conceptualization, investigation, and writing – original draft. Fan Liu: Investigation. Jun Chen: Investigation. Xiao-Ping Lin: Investigation Qing-Lan Huang: Resources and data curation. Yi Wei: Data curation. Shu-Li Chen: Software. Chun-Yan Wu: Conceptualization and supervision. Zhen Li: Writing – review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from the Fuzhou Science and technology planning project (2020WS0101), the Fuzhou key college of newborn (20191204).
